Pancreatic Hormones Antidiabetic Drugs Introduction The pancreas is

Pancreatic Hormones & Antidiabetic Drugs

Introduction • The pancreas is both an exocrine gland that produces the digestive enzymes & an endocrine gland • The islets of Langerhans contain five main cell types. Their hormone products include: • Insulin: the storage and anabolic hormone of the body; • Islet amyloid polypeptide (IAPP, or amylin), modulates appetite, gastric emptying, glucagon and insulin • Glucagon: hyperglycemic factor, mobilizes glycogen stores • Somatostatin: inhibitor of secretory cells • Pancreatic peptide: protein that facilitates digestive processes • Ghrelin: a peptide known to increase pituitary GH release

The endocrine pancreas in the adult human consists of approximately 1 million islets of Langerhans interspersed throughout the pancreatic gland
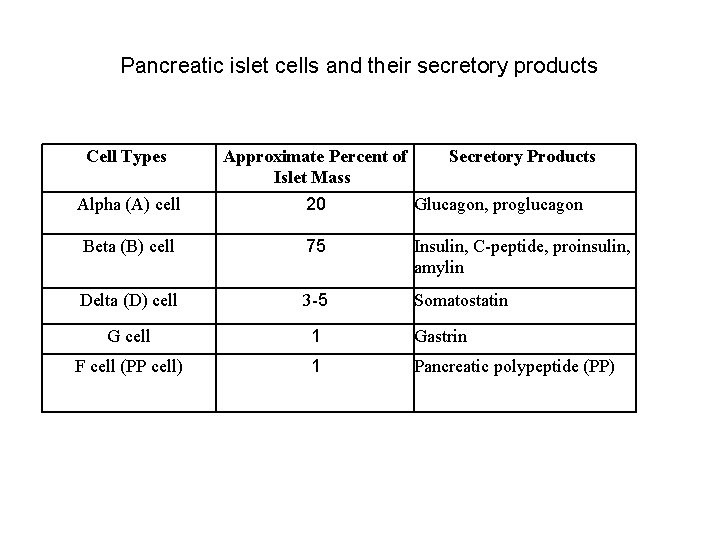
Pancreatic islet cells and their secretory products Cell Types Alpha (A) cell Approximate Percent of Secretory Products Islet Mass 20 Glucagon, proglucagon Beta (B) cell 75 Insulin, C-peptide, proinsulin, amylin Delta (D) cell 3 -5 Somatostatin G cell 1 Gastrin F cell (PP cell) 1 Pancreatic polypeptide (PP)

Diabetes Mellitus • Definition: elevated blood glucose associated with absent or inadequate pancreatic insulin secretion, with or without concurrent impairment of insulin action • It consists of a group of syndromes characterized by hyperglycemia; altered metabolism of lipids, carbohydrates, and proteins; and an increased risk of complications as vascular disease • Hyperglycemia is a common end point for all types of diabetes mellitus and is the parameter that is measured to evaluate and manage the efficacy of diabetes therapy

Diabetes Mellitus • The American diabetic association (ADA) recognizes four clinical classifications of diabetes: – Type 1: Formerly ‘insulin-dependent diabetes’ – Type 2: Formerly ‘non insulin-dependent diabetes’ – Type 3: Other (e. g. genetic defects or medication induced) – Type 4: Gestational diabetes mellitus

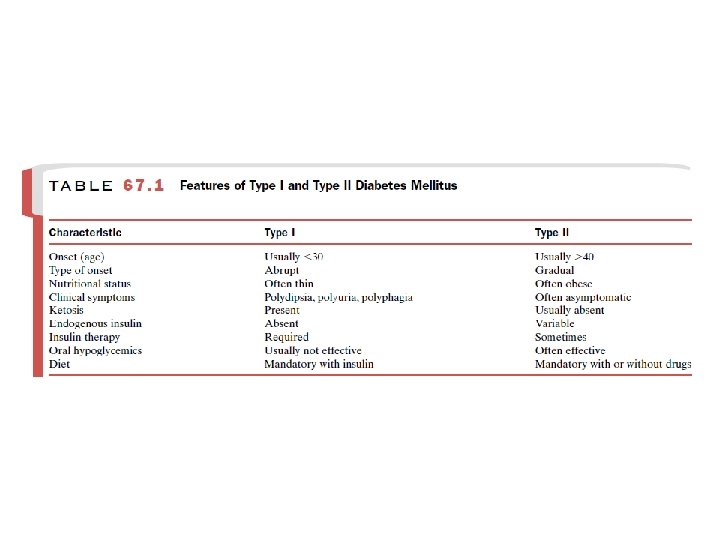
Type 1 Diabetes Mellitus �Type I diabetes mellitus constitutes about 10% of cases of diabetes mellitus �Selective β cell destruction and severe or absolute insulin deficiency �Affects individuals in puberty or early adulthood, but some latent forms can occur later in life �Type 1 diabetes is further subdivided into (type 1 a) immune most common and (type 1 b) idiopathic causes �Susceptibility appears to involve a multifactorial genetic linkage but only 10 -15% of patients have a positive family history

Type 1 Diabetes Mellitus • Antibodies facilitate diagnosis and screening of type 1 DM • Insulin replacement therapy is necessary to sustain life. Interruption of the insulin therapy can be life-threatening and can result in diabetic ketoacidosis (DKA) or death • DKA is caused by insufficient or absent insulin and results from excess release of fatty acids and subsequent formation of toxic levels of ketoacids

Type 2 Diabetes Mellitus • Type II DM is far more common than type 1 • The disease is influenced by genetic factors, aging, & obesity • It is characterized by tissue resistance to the action of insulin combined with a relative deficiency in insulin secretion • In Type 2 diabetes, the pancreas retains some β-cell function, but variable insulin secretion is insufficient to maintain glucose homeostasis and to overcome the resistance • The β-cell mass may become gradually reduced in Type 2 diabetes

Type 2 Diabetes Mellitus �Impaired insulin action affect fat metabolism→↑fatty acids and TG, ↓HDL �Weight reduction, exercise, and dietary modification decrease insulin resistance and correct the hyperglycemia of Type 2 diabetes in some patients. However, most patients are dependent on pharmacologic intervention with oral hypoglycemic agents �Individuals with type 2 diabetes may not require insulin to survive, but 30% or more will benefit from insulin therapy to control the blood glucose

Type 2 Diabetes Mellitus • Dehydration in individuals with untreated or poorly controlled type 2 diabetes can lead to a lifethreatening condition called nonketotic hyperosmolar coma…. dehydration…. . altered mental state →loss of consciousness • This needs urgent care and rehydration
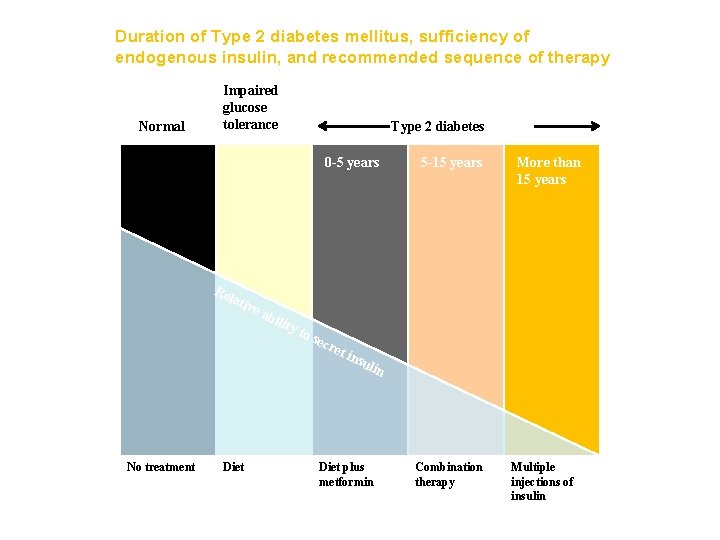
Duration of Type 2 diabetes mellitus, sufficiency of endogenous insulin, and recommended sequence of therapy Normal Impaired glucose tolerance Type 2 diabetes 0 5 years Re lati ve No treatment Diet abi lity to s ecr et i 5 15 years More than 15 years nsu lin Diet plus metformin Combination therapy Multiple injections of insulin

Type 3 Diabetes Mellitus • The type 3 designation refers to multiple other specific causes of an elevated blood glucose: 1) Pancreatectomy 2) Pancreatitis 3) Nonpancreatic diseases (e. g. Cushing’s syndrome & acromegaly) 4) Drug therapy (e. g. anti hypertensive vasodilator diazoxide and corticosteroids)

Type 4 Diabetes Mellitus Gestational diabetes (GDM) �Defined as any abnormality in glucose levels noted for the first time during pregnancy � It is diagnosed in approximately 7% of all pregnancies in the USA �During pregnancy, the placenta and placental hormones create an insulin resistance that is most pronounced in the last trimester �High risk women should be screened at the first prenatal visit. In lower risk women screening is deferred until the 24 th to 28 th week of gestation.

Diabetes-Related Complications • Chronic complications are commonly divided into: 1) Microvascular complications: nephropathy and neuropathy retinopathy, 2) Macrovascular complications refer to increased atherosclerosis-related events such as MI and stroke

Diagnosis of Diabetes • American Diabetes Association (ADA) have adopted criteria for the diagnosis of diabetes, based on: 1) Symptoms of diabetes plus random blood glucose concentration ≥ 200 mg/d. L (11. 1 m. M) 2) The fasting blood glucose (FBG) ≥ 126 mg/d. L (7. 0 m. M) (no caloric intake for at least 8 hrs) 3) Two hour plasma glucose ≥ 200 mg/d. L (11. 1 m. M) during an oral glucose tolerance test (OGTT) = 75 g of glucose dissolved in water 4) Level of hemoglobin A 1 c (Hb. A 1 c) ≥ 6. 5%
Insulin & its analogs

Insulin • Insulin is a polypeptide hormone (mwt =5808 Da) • 51 a. a. arranged in two chains (A and B) linked by disulfide bridges; there are species differences in the amino acids of both chains • Proinsulin, a long single chain protein molecule, is processed within the Golgi apparatus of beta cells, packaged into granules, and then hydrolyzed into insulin and a residual connecting segment called C peptide
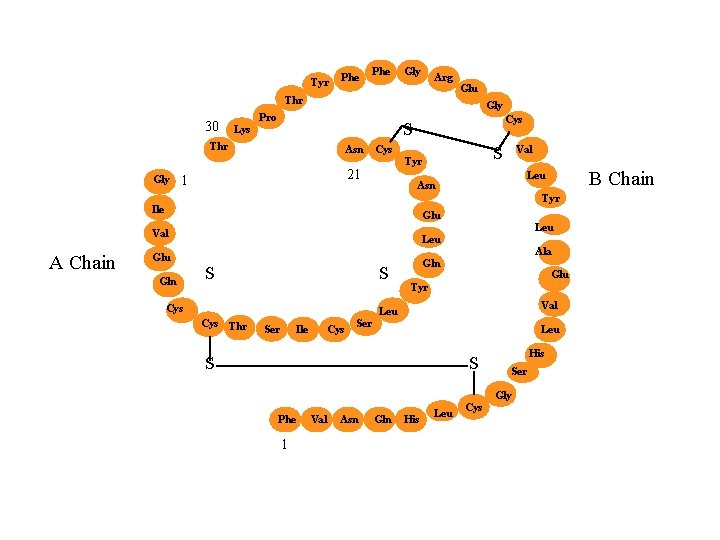
Tyr Phe Gly Arg Thr 30 Lys Gly Pro Cys S Thr Gly Asn Tyr 21 1 S Cys Val B Chain Leu Asn Tyr Ile Glu Leu Val A Chain Glu Gln Leu Ala S S Cys Gln Glu Tyr Val Leu Cys Thr Ser Ile Cys Ser Leu S His S Ser Gly Phe 1 Val Asn Gln His Leu Cys
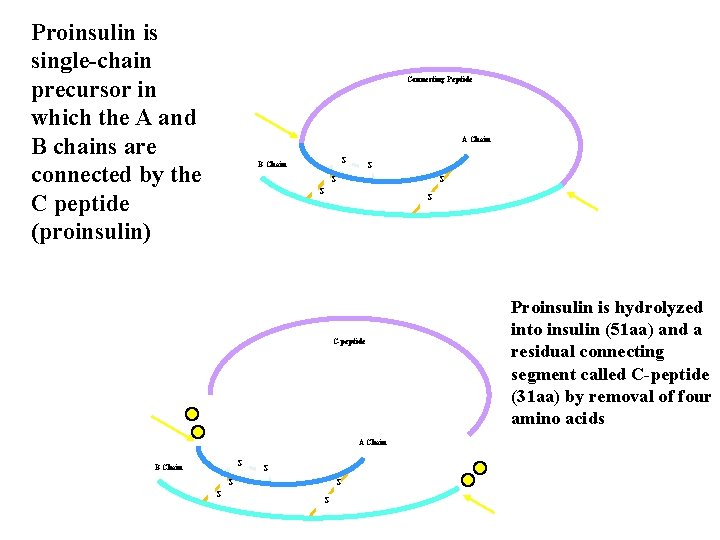
Proinsulin is single chain precursor in which the A and B chains are connected by the C peptide (proinsulin) Connecting Peptide A Chain S B Chain S S S C peptide A Chain S B Chain S S S Proinsulin is hydrolyzed into insulin (51 aa) and a residual connecting segment called C peptide (31 aa) by removal of four amino acids

Insulin Biosynthesis • Insulin and C peptide are secreted in equimolar amounts in response to all insulin stimulants • C peptide has no known physiologic function but serves as a useful index of indicator of endogenous insulin production/ secretion

Granules within the beta cells store the insulin in the form of crystals consisting of two atoms of zinc and six molecules of insulin (hexamer). The entire human pancreas contains up to 8 mg of insulin (28 units per milligram)

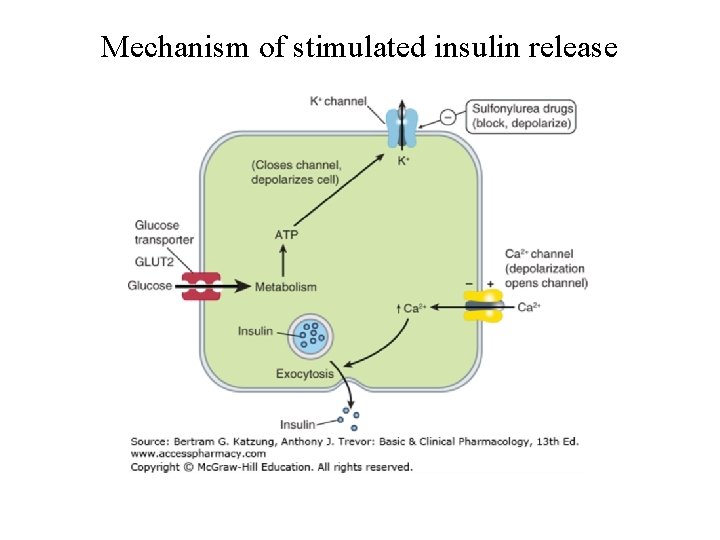
Insulin secretion • Insulin is released from pancreatic β cells at a low basal rate during fasting and at a much higher stimulated rate in response to a variety of stimuli, especially glucose • Other stimulants include: 1. Other sugars (e. g. mannose) 2. Certain amino acids (e. g. leucine, arginine), 3. Hormones (e. g, glucagon-like polypeptide-1 (GLP-1), glucose -dependent insulinotropic polypeptide (GIP), glucagon, cholecystokinin) 4. Vagal (cholinergic) activity • Inhibitory signals include somatostatin, leptin, and chronically elevated glucose and fatty acid levels
Mechanism of stimulated insulin release

Insulin secretion • Basically, the resting β cell is hyperpolarized, and its depolarization leads to the secretion of insulin. A rising plasma glucose concentration initiates a series of events that leads to depolarization • Glucose enters the β cell by facilitated transport, which is mediated by GLUT 2 • Glucose is phosphorylated by glucokinase & enhances adenosine triphosphate (ATP) production • The rise in ATP levels causes a block of K+ channels, leading to membrane depolarization and an influx of Ca 2+, which results in pulsatile insulin exocytosis

Glucose transporters Transporter Tissues Function GLUT 1 All tissues, especially red cells, brain Basal uptake of glucose; transport across the bloodbrain barrier GLUT 2 β cells of pancreas; liver, kidney; gut Regulation of insulin release, other aspects of glucose Homeostasis GLUT 3 Brain, kidney, placenta, other tissues Uptake into neurons, other Tissues GLUT 4 Muscle, adipose Insulin mediated uptake of glucose GLUT 5 Gut, kidney Absorption of fructose
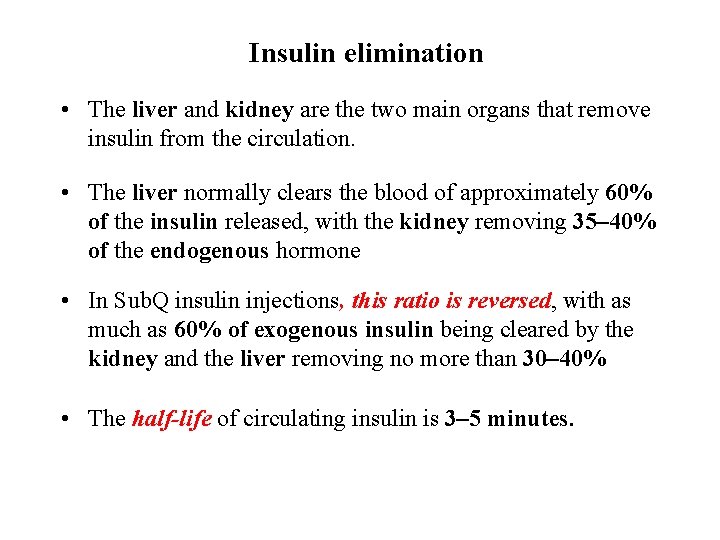
Insulin elimination • The liver and kidney are the two main organs that remove insulin from the circulation. • The liver normally clears the blood of approximately 60% of the insulin released, with the kidney removing 35– 40% of the endogenous hormone • In Sub. Q insulin injections, this ratio is reversed, with as much as 60% of exogenous insulin being cleared by the kidney and the liver removing no more than 30– 40% • The half-life of circulating insulin is 3– 5 minutes.

The Insulin Receptor • Insulin initiates its actions by binding to a cell-surface receptor that is found on the membranes of most tissues • The number of receptors varies from as few as 40 per cell on erythrocytes to 300, 000 per cell on adipocytes and hepatocytes • The insulin receptor is a heterotetrameric tyrosine kinase receptor composed of two α subunits and two β subunits; the subunits are linked by disulfide bonds to form β-α-α-β heterotetramer
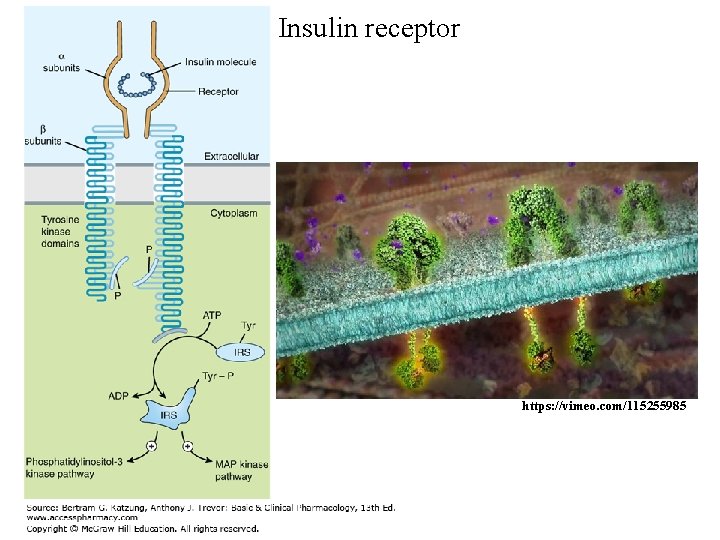
Insulin receptor https: //vimeo. com/115255985
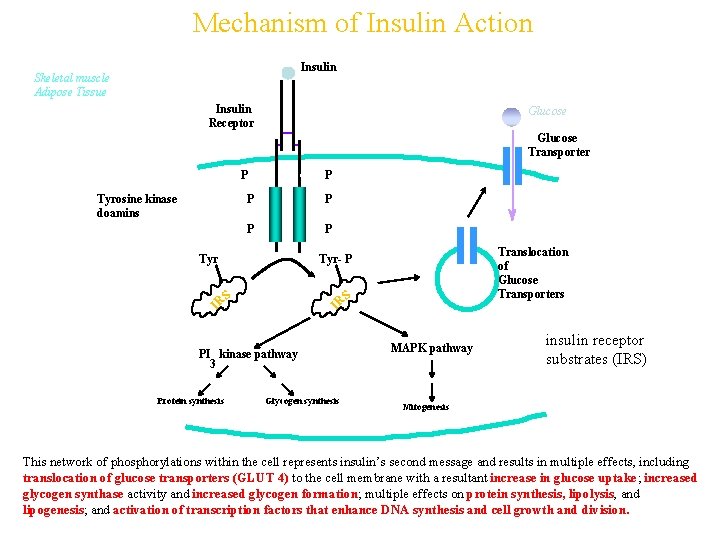
Mechanism of Insulin Action Insulin Skeletal muscle Adipose Tissue Insulin Receptor Glucose Transporter P Tyrosine kinase doamins P P P Tyr S S IR IR PI kinase pathway 3 Protein synthesis Translocation of Glucose Transporters Tyr P Glycogen synthesis MAPK pathway insulin receptor substrates (IRS) Mitogenesis This network of phosphorylations within the cell represents insulin’s second message and results in multiple effects, including translocation of glucose transporters (GLUT 4) to the cell membrane with a resultant increase in glucose uptake; increased glycogen synthase activity and increased glycogen formation; multiple effects on protein synthesis, lipolysis, and lipogenesis; and activation of transcription factors that enhance DNA synthesis and cell growth and division.

Effects of insulin on its target • The important target tissues for regulation of glucose homeostasis by insulin are liver, muscle, and fat • Some effects of insulin occur within seconds or minutes, including the activation of glucose transport systems • Other effects, such as those on protein synthesis and gene transcription, may take a few hours • Effects of insulin on cell proliferation and differentiation may take days

• Effect of insulin on liver: • • • Reversal of catabolic features of insulin deficiency: Inhibits glycogenolysis Inhibits conversion of F. As and amino acids to keto acids Inhibits conversion of amino acids to glucose Anabolic action Promotes glucose storage as glycogen (induces glucokinase and glycogen synthase, inhibits glycogen phosphorylase) • Increases TGs synthesis and VLDL formation

�Effect of insulin on muscle: � Increased protein synthesis � Increases amino acid transport � Increases ribosomal protein synthesis � Increased glycogen synthesis � Increases glucose transport � Induces glycogen synthase and inhibits glycogen phosphorylase �Effect of insulin on adipose tissue: � Increased TGs storage � Lipoprotein lipase is induced and activated by insulin to hydrolyze triglycerides from lipoproteins � Glucose transport into cell provides glycerol phosphate to permit esterification of fatty acids supplied by lipoprotein transport � Intracellular lipase is inhibited by insulin
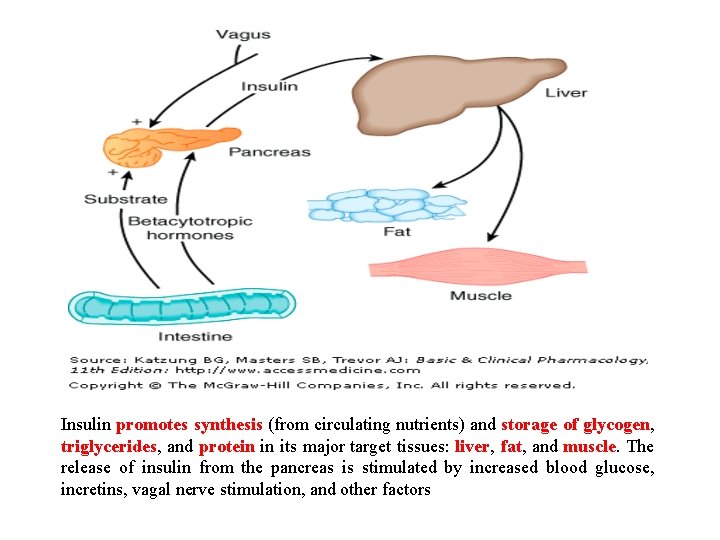
Insulin promotes synthesis (from circulating nutrients) and storage of glycogen, triglycerides, and protein in its major target tissues: liver, fat, and muscle. The release of insulin from the pancreas is stimulated by increased blood glucose, incretins, vagal nerve stimulation, and other factors
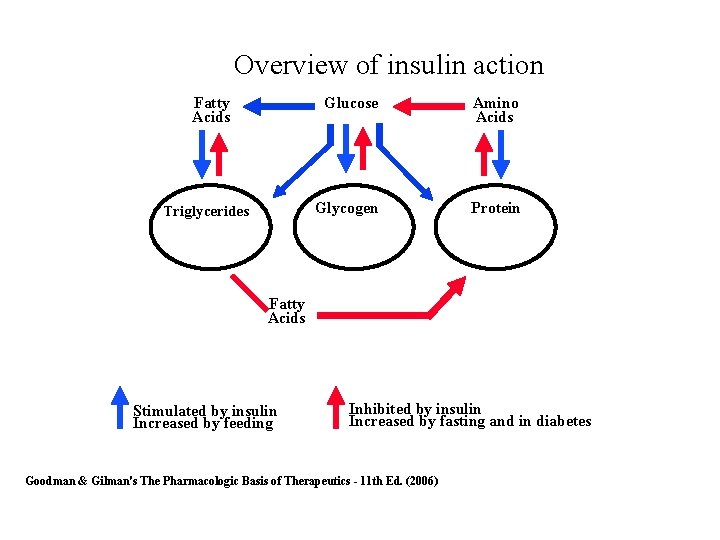
Overview of insulin action Fatty Acids Triglycerides Adipose Tissue Glucose Amino Acids Glycogen Protein Liver Muscle Fatty Acids Stimulated by insulin Increased by feeding Inhibited by insulin Increased by fasting and in diabetes Goodman & Gilman's The Pharmacologic Basis of Therapeutics 11 th Ed. (2006)

Characteristics of Available Insulin Preparations • Commercial insulin preparations differ in a number of ways, such as differences in the recombinant DNA production techniques, a. a. sequence, concentration, solubility, and the time of onset and duration of their biologic action • Preparations of insulin can be classified according to their duration of action into rapid, short, intermediate, and long acting and by their species of origin human or porcine

Characteristics of Available Insulin Preparations • Human insulin, produced by recombinant technology (e. g. Humulin, Novolin) has largely replaced insulins derived from beef and pork • Modifications of the a. a sequence of human insulin have produced insulins with different PK properties • Insulin is administered Sub. Q by conventional disposable needles and syringes. • Primary treatment for all type 1 DM patients, some type 2 DM and for patients with postpancreatectomy diabetes or gestational diabetes (GDM)

Characteristics of Available Insulin Preparations • Long-term treatment relies predominantly on Sub. Q injections in the abdomen, buttock, anterior thigh, or dorsal arm • Goals of therapy: • replicate normal physiologic insulin secretion (replacing the background or basal overnight, fasting, and between meal) • as well as bolus or prandial (mealtime) insulin
Insulin administration sites

Characteristics of Available Insulin Preparations • For therapeutic purposes, doses and concentrations of insulin are expressed in units • The unit is defined on the basis of weight, and present insulin standards used for assay purposes contain 28 units per 1 milligram • Insulin strengths (supplied in solution or suspension): – 100 units/m. L (U 100) – 500 units/m. L (U 500) - only available in regular insulin

Characteristics of Available Insulin Preparations • Four principal types of injected insulins are available: 1. Rapid acting with very fast onset and short duration 2. Short acting with rapid onset of action 3. Intermediate acting 4. Long acting with slow onset of action ü N. B: All insulins should be refrigerated and brought to room temperature just before injection
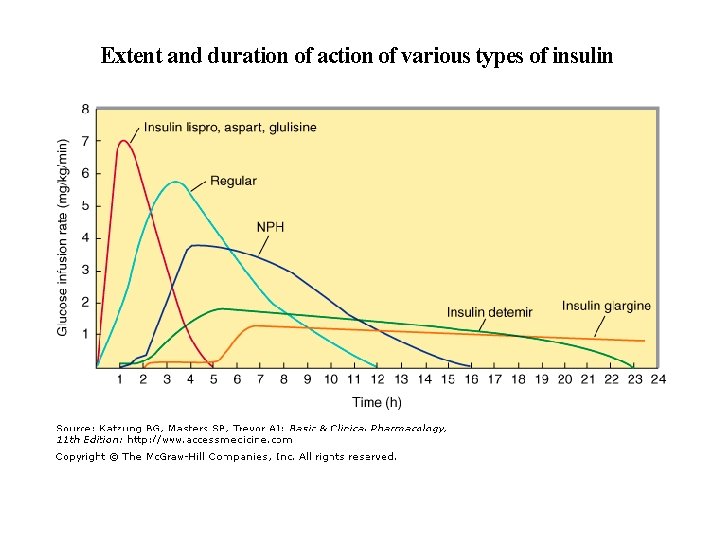
Extent and duration of action of various types of insulin

1. Rapid-acting insulin • Analogs: insulin LISPRO, insulin ASPART, and insulin GLULISINE • Clear solutions at neutral p. H and contain small amounts of zinc to improve their stability and shelf life • Contain amino acid modifications that promote rapid entry into the circulation from subcutaneous tissue • Rapid onset of action (5 -10 min). after administration. The duration of action is 3 -5 hours

1. Rapid-acting insulin • Permit more physiologic prandial insulin replacement • …. . allowing insulin to be taken immediately before the meal • Advantages: ü low risk of postmeal hypoglycemia ü more flexible treatment regimens ü lowest variability of absorption (approxiamtely 5%)

2. Short-acting insulin • Regular insulin: soluble crystalline zinc insulin (recombinant DNA techniques) • Regular insulin molecules self-aggregate to form dimers that stabilize around zinc ions to create insulin hexamers • Onset appear within 30 min, peak action between 2 -3 hrs, and the effect lasts 5 -8 hrs • Administered several minutes (30 -45 mins) before a meal

2. Short-acting insulin • primarily used to supplement intermediate- & longacting insulin preparations • Only one to be administered i. v. (DKA, after surgery or during acute infections) • The delayed absorption, dose dependent duration of action, and variability of absorption (~ 25%) of regular human insulin results in a mismatching of insulin availability with need, and its use is declining

3. Intermediate-acting insulin • Formulated to dissolve more gradually administered Sub. Q; longer durations of action when • Neutral Protamine Hagedorn (NPH) or Isophane insulin: turbid suspension of insulin with appropriate amounts of protamine……. after Sub. Q inj. , proteolytic tissue enzymes degrade the protamine to permit absorption of insulin • NPH insulin has an onset of approximately 2 -5 hours and duration of 4 -12 hours

3. Intermediate-acting insulins • Used for basal control usually mixed with regular, lispro, aspart, or glulisine insulin and given two to four times daily for insulin replacement • The action of NPH is highly unpredictable, and its variability of absorption is over 50% • The clinical use of NPH is waning because of its adverse PK combined with the availability of longacting insulin analogs that have a more predictable and physiologic action

4. Long-Acting Insulins: A. Insulin Glargine Lantus® • Insulin glargine is a soluble, peakless (ie, having a broad plasma conc. plateau) insulin analog that provides reproducible, convenient, background insulin replacement • The a. a sequence created an analog soluble in an acidic solution (p. H 4. 0) but precipitates in the more neutral body p. H after Sub. Q injection • slow onset of action (1 -1. 5 hours), achieves a maximum effect after 4 -6 hours which is maintained for 11 -24 hours or longer

A. Insulin Glargine • Usually given once daily, some individuals benefit from split (twice a day) dosing • Should not be mixed with other insulins. Use separate syringes to minimize the risk of contamination and subsequent loss of efficacy

4. Long-Acting Insulins: B. Insulin Detemir (Levemir®) • It has a fatty acid side chain which its prolongs the availability (higher self-aggregation in subcutaneous tissue and reversible albumin binding) • Has the most reproducible effect and its use is associated with less hypoglycemia than NPH insulin. • Insulin detemir has a dose dependent onset of action of 1 2 hours and duration of action of more than 24 hours • Given twice daily and is associated with less hypoglycemia
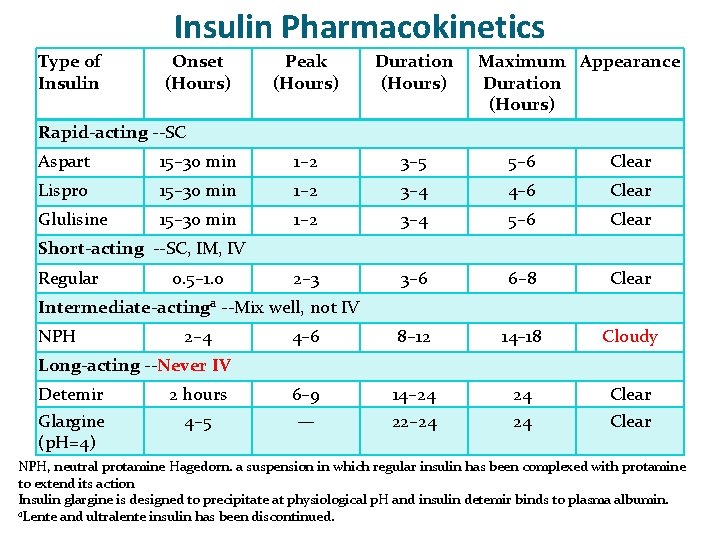
Insulin Pharmacokinetics Type of Insulin Onset (Hours) Peak (Hours) Duration (Hours) Maximum Appearance Duration (Hours) Rapid-acting --SC Aspart 15– 30 min 1– 2 3– 5 5– 6 Clear Lispro 15– 30 min 1– 2 3– 4 4– 6 Clear Glulisine 15– 30 min 1– 2 3– 4 5– 6 Clear 2– 3 3– 6 6– 8 Clear 4– 6 8– 12 14– 18 Cloudy 6– 9 — 14– 24 22– 24 24 24 Clear Short-acting --SC, IM, IV Regular 0. 5– 1. 0 Intermediate-actinga --Mix well, not IV NPH 2– 4 Long-acting --Never IV Detemir Glargine (p. H=4) 2 hours 4– 5 NPH, neutral protamine Hagedorn. a suspension in which regular insulin has been complexed with protamine to extend its action Insulin glargine is designed to precipitate at physiological p. H and insulin detemir binds to plasma albumin. a. Lente and ultralente insulin has been discontinued.

September 2015! FDA update. . Tresiba® Insulin degludec is an ultralong-acting basal insulin analogue. It is administered via SC once daily. It has a duration of action that lasts up to 42 hours (compared to 18 to 26 hours of insulin glargine and insulin detemir) Onset is 60 -90 minutes, It is available in 2 strengths: 100 and 200 units/ml.

Tresiba®
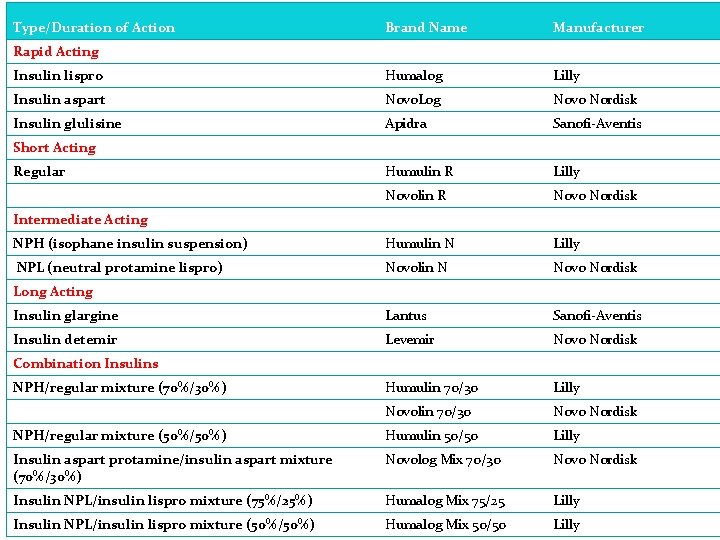
Type/Duration of Action Brand Name Manufacturer Insulin lispro Humalog Lilly Insulin aspart Novo. Log Novo Nordisk Insulin glulisine Apidra Sanofi-Aventis Humulin R Lilly Novolin R Novo Nordisk NPH (isophane insulin suspension) Humulin N Lilly NPL (neutral protamine lispro) Novolin N Novo Nordisk Insulin glargine Lantus Sanofi-Aventis Insulin detemir Levemir Novo Nordisk Humulin 70/30 Lilly Novolin 70/30 Novo Nordisk NPH/regular mixture (50%/50%) Humulin 50/50 Lilly Insulin aspart protamine/insulin aspart mixture (70%/30%) Novolog Mix 70/30 Novo Nordisk Insulin NPL/insulin lispro mixture (75%/25%) Humalog Mix 75/25 Lilly Insulin NPL/insulin lispro mixture (50%/50%) Humalog Mix 50/50 Lilly Rapid Acting Short Acting Regular Intermediate Acting Long Acting Combination Insulins NPH/regular mixture (70%/30%)

5. Mixtures of insulins • Insulin lispro, aspart, and glulisine can be acutely mixed (ie, just before injection) with NPH insulin without affecting their rapid absorption • Premixed combinations of neutral protamine aspart (NPA) with aspart and neutral protamine lispro (NPL) with lispro have been developed • Fixed-ratio mixtures exist: 50%/50% and 75%/25% NPL/insulin lispro and 70%/30% NPA/insulin aspart • Benefits: reduced errors, improved dosing accuracy as well as the convenience of using a single vial

5. Mixtures of insulins • Insulin glargine and detemir must be given as separate injections. They are NOT miscible acutely or in a premixed preparation with any other insulin formulation • These combination products regimens for all diabetics…. . not suitable
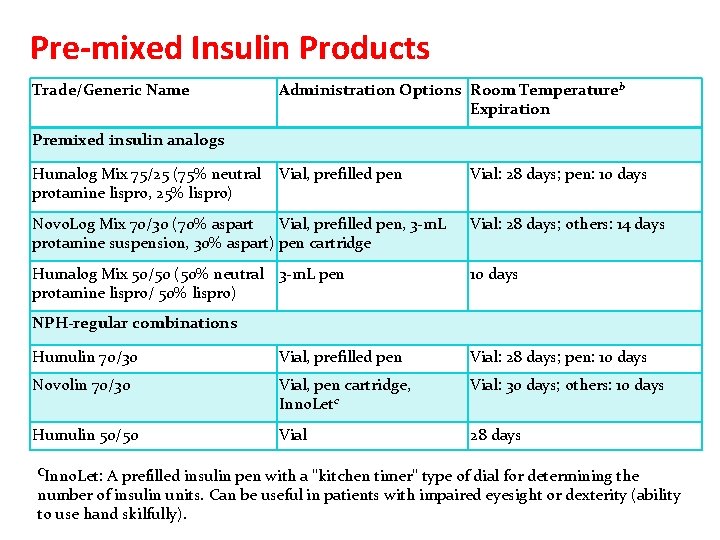
Pre-mixed Insulin Products Trade/Generic Name Administration Options Room Temperatureb Expiration Premixed insulin analogs Humalog Mix 75/25 (75% neutral protamine lispro, 25% lispro) Vial, prefilled pen Vial: 28 days; pen: 10 days Novo. Log Mix 70/30 (70% aspart Vial, prefilled pen, 3 -m. L protamine suspension, 30% aspart) pen cartridge Vial: 28 days; others: 14 days Humalog Mix 50/50 (50% neutral 3 -m. L pen protamine lispro/ 50% lispro) 10 days NPH-regular combinations Humulin 70/30 Vial, prefilled pen Vial: 28 days; pen: 10 days Novolin 70/30 Vial, pen cartridge, Inno. Letc Vial: 30 days; others: 10 days Humulin 50/50 Vial 28 days CInno. Let: A prefilled insulin pen with a "kitchen timer" type of dial for determining the number of insulin units. Can be useful in patients with impaired eyesight or dexterity (ability to use hand skilfully).
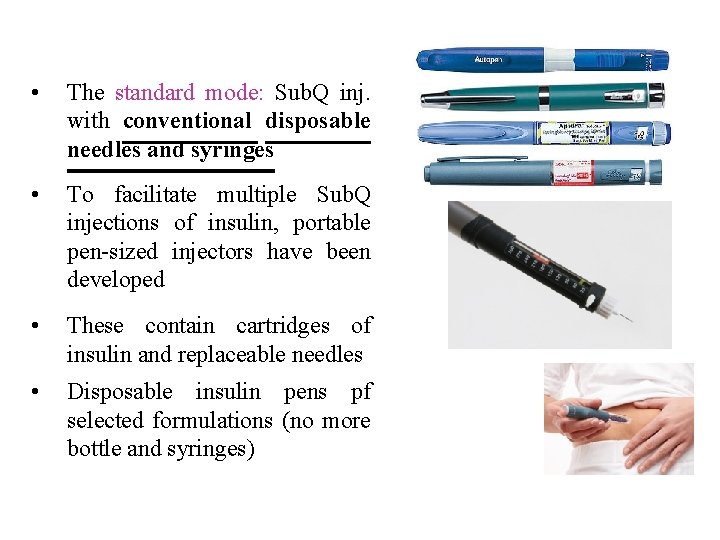
• The standard mode: Sub. Q inj. with conventional disposable needles and syringes • To facilitate multiple Sub. Q injections of insulin, portable pen-sized injectors have been developed • These contain cartridges of insulin and replaceable needles • Disposable insulin pens pf selected formulations (no more bottle and syringes)

Continuous Sub. Q Insulin Infusion Device • Insulin pumps or CSII are small, portable devices worn externally that deliver a continuous supply of insulin Sub. Q through a hypodermic needle based on blood glucose selfmonitoring results • CSII devices avoid the need for multiple daily injections and provide flexibility in the scheduling of patients daily activities • Programmable pumps deliver a constant 24 hour basal rate, and adjustments in the rate of delivery can be made to accommodate changes in insulin requirements (eg. before meals or exercise)
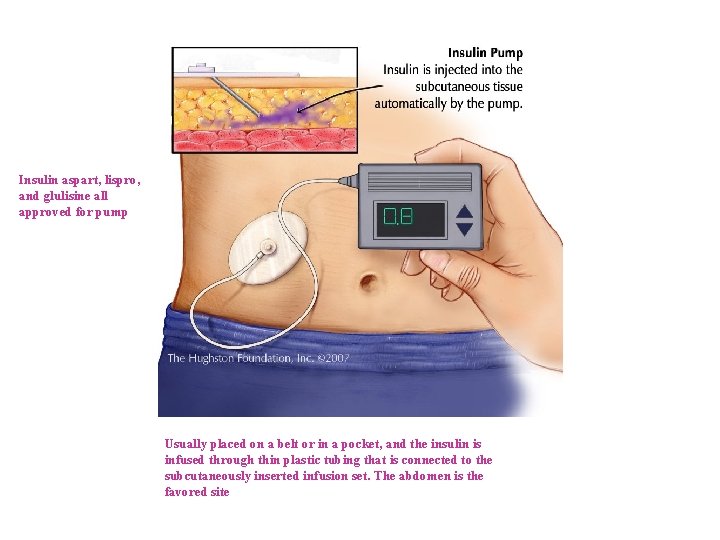
Insulin aspart, lispro, and glulisine all approved for pump Usually placed on a belt or in a pocket, and the insulin is infused through thin plastic tubing that is connected to the subcutaneously inserted infusion set. The abdomen is the favored site

Inhaled Insulin (Afrezza®) (June 2014) • Dry powder formulation of recombinant regular insulin (technosphere insulin) approved for use in adults • After inhalation peak levels reached in 12 15 min. and decline to baseline in 3 hours • Faster in onset and shorter in duration than Sub. Q insulin • In trials, inhaled insulin was as effective in lowering glucose • Most common ADEs was cough, monitor pulmonary function • C/I in smokers and patients with COPD

Insulin Treatment of Special Circumstances • Diabetic Ketoacidosis (DKA): life threatening medical emergency caused by inadequate or absent insulin replacement…. occurs in people with type 1 diabetes and infrequently in those with type 2 diabetes • S&S: N, V, abdominal pain, deep slow breathing, change in mental status, elevated blood and urinary ketones and glucose, and low blood p. H • Tx. aggressive i. v hydration (normal saline) and i. v insulin therapy (regular insulin) and maintenance of potassium and other electrolyte levels

Insulin Treatment of Special Circumstances • Hyperosmolar Hyperglycemic Syndrome (HHS): diagnosed in persons with type 2 DM and characterized by profound hyperglycemia and dehydration • The diagnostic hallmarks: altered mental status and even seizures, high plasma glucose, and high serum osmolality; patients are not acidotic • Tx. aggressive rehydration and restoration of glucose and electrolyte homeostasis; Low-dose insulin therapy may be required

Adverse reactions 1. Hypoglycemia (most common complication of insulin therapy) • Causes: a) b) c) d) Inadequate carbohydrate consumption Unusual physical exertion Inappropriately large dose of insulin Mismatch between the time of peak delivery of insulin and food intake

Adverse reactions 1. Hypoglycemic manifestation: • Result from autonomic hyperactivity—sympathetic counterregulatory hormones (epinephrine): tachycardia, palpitations, sweating, tremulousness and parasympathetic nausea, hunger • Neuroglycopenic effects produced by the reduced brain sugar: altered mental status – May progress to convulsions and coma if untreated

Adverse reactions 1. Hypoglycemia • All the manifestations of hypoglycemia are relieved by glucose administration a) Oral CHOs: dextrose tablets, glucose gel, or any sugarcontaining beverage or food may be given b) If unconsciousness or stupor: 20– 50 m. L of 50% glucose solution IV infused over a period of 2– 3 minutes c) If IV glucose not available, SC or IM glucagon may restore consciousness within 15 minutes d) If stuporous and glucagon not available, small amounts of honey or syrup can be inserted into the buccal pouch…oral feeding is C/I in unconscious patients

Adverse reactions 2. Immunopathology of insulin therapy A. Insulin allergy: • Ig. E-mediated local cutaneous reactions in which local or systemic urticaria results from histamine release from tissue mast cells. . . Antihistamines B. Immune insulin resistance: • Low titer of circulating Ig. G anti-insulin antibodies that neutralize the action of insulin…. . Glucocorticoids ü Human insulin preparations should be used

Adverse reactions 3. Lipodystrophy at Injection Sites A. Atrophy of subcutaneous fatty tissue at the site of insulin injection (lipoatrophy) • almost never seen ever since the development of human and analog insulin preparations of neutral p. H B. Hypertrophy: Enlargement of subcutaneous fat depots due to the lipogenic action of insulin if injected repeatedly at the same site • May be corrected by avoiding the specific injection site or by liposuction

Increased Cancer Risk • An increased risk of cancer attributed to insulin resistance and hyperinsulinemia has been reported in individuals with insulin resistance, prediabetes, and type 2 diabetes. • Treatment with insulin and sulfonylureas (which increase circulating insulin levels), but not metformin possibly exacerbates that risk. • These epidemiologic observations are preliminary and have not changed prescribing guidelines. 72

Antidiabetic Agents other than insulin

Overview • Also known as oral hypoglycemic agents (some are injectables) • These agents are useful in the treatment of patients who have Type 2 diabetes but who cannot be managed by diet or weight loss and exercise • Patients with long-standing type 2 DM may require a combination of hypoglycemic drugs with or without insulin to control their hyperglycemia • Oral hypoglycemic agents should not be given to patients with Type 1 DM

Overview 1. Insulin secretagogues (sulfonylureas, meglitinides, Dphenylalanine derivatives) 2. Insulin sensitizers lower glucose levels by acting on liver, muscle, & adipose tissue (biguanides & thiazolidinediones) 3. α glucosidase inhibitors (inh. GI abs. of glucose) 4. Incretin based therapies (glucagon-like peptide-1 agonists, dipeptidyl peptidase-4 inhibitors) 5. Inhibition glucose reabs. from kidney (Na-gluc. cotransporter inhibitors [SGLTs] / Flozins) 6. Amylin analog (Pramlinitide)

1. Insulin Secretagogue: Sulfonylurea • In the presence of viable pancreatic β-cells, sulfonylureas directly enhance the release of endogenous insulin, thereby reducing blood glucose levels • Ineffective for management of type I and severe type II DM since the number of viable β-cells in these forms of diabetes is too small • They are conventionally divided into first generation and second generation agents, which differ primarily in their potency and adverse effects

Sulphonylurea Mechanism of action 1. Insulin Release from Pancreatic Beta Cells • Sulfonylureas bind to a 140 k. Da high affinity sulfonylurea receptor (SUR 1)…. . associated with an inward rectifier ATPsensitive potassium channel • Binding of a sulfonylurea inhibits the efflux of potassium ions through the channel and results in depolarization • Depolarization opens a voltage gated calcium channel and results in the release of preformed insulin
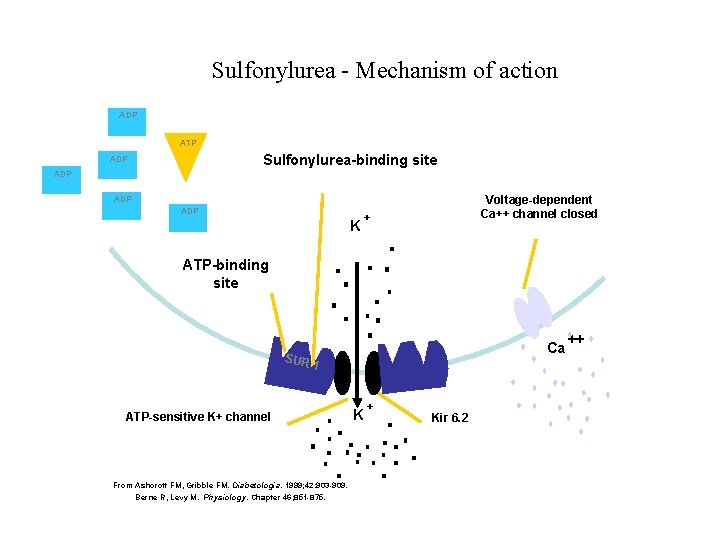
Sulfonylurea - Mechanism of action ADP ATP Sulfonylurea-binding site ADP Voltage-dependent Ca++ channel closed ADP K + ATP-binding site SUR Ca 1 ATP-sensitive K+ channel From Ashcroft FM, Gribble FM. Diabetologia. 1999; 42: 903 -909. Berne R, Levy M. Physiology. Chapter 46; 851 -875. K + Kir 6. 2 ++
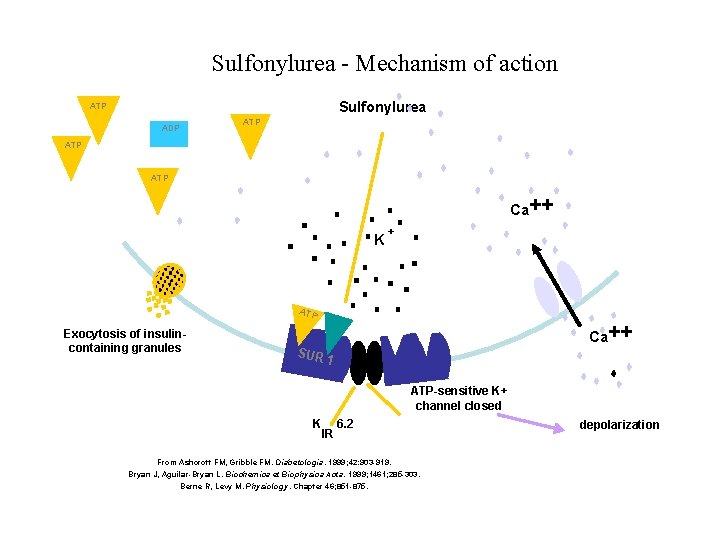
Sulfonylurea - Mechanism of action Sulfonylurea ATP ADP ATP Ca++ K + ATP Exocytosis of insulincontaining granules Ca++ SUR 1 ATP-sensitive K+ channel closed K IR 6. 2 From Ashcroft FM, Gribble FM. Diabetologia. 1999; 42: 903 -919. Bryan J, Aguilar-Bryan L. Biochemica et Biophysica Acta. 1999; 1461; 285 -303. Berne R, Levy M. Physiology. Chapter 46; 851 -875. depolarization

Sulfonylurea- Pharmacokinetics • Absorbed from the GIT following oral administration. All bind strongly to plasma albumin • All the sulfonylureas (except acetohexamide) are metabolized by the liver, and the metabolites are excreted in part in the urine…. caution in patients with either renal or hepatic insufficiency • Most sulfonylureas cross the placenta and enter breast milk; as a result, use of sulfonylureas is C/I in pregnancy and in breast feeding
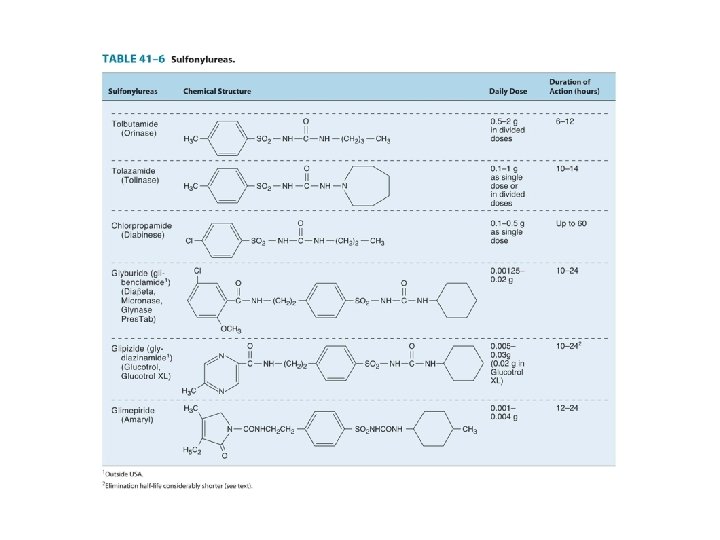

Sulfonylurea- First generation sulfonylurea • Agents: ACETOXAMIDE, CHLORPROPAMIDE, TOLAZAMIDE, & TOLBUTAMIDE • Are not frequently used in the management b/c of their: 1) Delay in time of onset 2) Occasional long duration of action 3) Side effects 4) Potential drug-drug interactions

Sulfonylurea- First generation sulfonylurea 1. Tolbutamide: • Short acting (6 -10 hrs) and rapidly metabolized by liver: relatively safe in elderly and in patients with renal impairment…. rare prolonged hypoglycemia! • Administered in divided doses before the meals

Sulfonylurea- First generation sulfonylurea 2. Chlorpropamide: • Relative slow onset of action, maximal hypoglycemic potential reached after 1 or 2 weeks. Slowly metabolized by the liver (T 1/2 ~32 hrs) • Common prolonged hypoglycemic reactions in elderly patients…. . C/I in this group • Other ADEs: ü hyperemic flush after alcohol ingestion in genetically predisposed patients (disulfiram reaction) ü hyponatremia due to its effect on vasopressin secretion and action.

Sulfonylurea- First generation sulfonylurea 3. Tolazamide: • It is comparable to chlorpropamide in potency but has a shorter duration of action • Tolazamide is more slowly absorbed than the other sulfonylureas, and its effect on blood glucose does not appear for several hours • Tolazamide is metabolized to several active compounds 4. Acetohexamide is no longer available in the United States

Sulfonylurea- Second generation sulfonylurea • Agents: GLYBURIDE, GLIPIZIDE, GLICLAZIDE & GLIMEPIRIDE • Fewer ADE and drug interactions…. . prescribed more • 100 200 times more potent than tolbutamide • Used with caution in patients with CV disease or in elderly patients with risk of dangerous hypoglycemia • Biologic effects evident for 12 -24 hrs……can be administered once daily

Sulfonylurea- Second generation sulfonylurea 1. Glyburide: ……. . GLIBENCLIMIDE • Completely metabolized in the liver to two weakly active metabolites…. can cause hypoglycemia • C/I if hepatic impairment or renal insufficiency 2. Glipizide: • Shortest T 1/2 (2– 4 hrs)…less likely to produce hypoglycemia • Absorption delayed when taken with food…. . should be ingested 30 min. before breakfast • Metabolized in the liver to inactive products (90%), 10% excreted unchanged in urine v C/I in patients with significant hepatic, preferable in the elderly

Sulfonylurea- Second generation sulfonylurea 3. Glimepiride: • Most potent sulfonylurea (daily dose of 1 mg is effective)) • Long duration of effect allowing once-daily dosing • It is completely metabolized by the liver to inactive products 4. Gliclazide (not available in the United States)

Sulfonylureas- Adverse reactions • Infrequent (4% of patients taking 1 st-generation drugs and less in patients receiving 2 nd-generation agents) • The commonest adverse effect is hypoglycemia, which can be severe and prolonged. Its incidence is related to the potency and duration of action of the agent • The highest incidence occurring with chlorpropamide and glyburide and the lowest with tolbutamide

Sulfonylureas- Adverse reactions 2. Weight gain: stimulate appetite (probably via their effects on insulin secretion and blood glucose). This is a major concern in obese diabetic patients 3. Chlorpropamide: may cause alcohol induced flushing and hyponatremia by potentiating the effects of ADH on the renal collecting duct 4. Others: N, V, agranulocytosis, aplastic and hemolytic anemias, generalized hypersensitivity reactions, and dermatological reactions
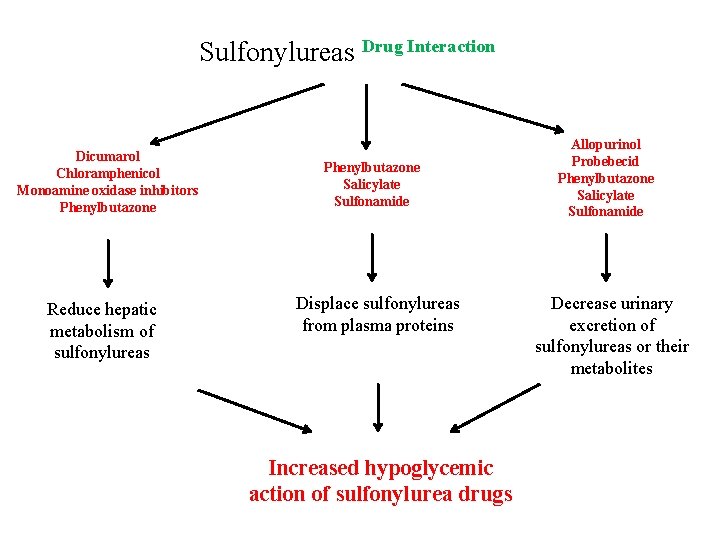
Sulfonylureas Drug Interaction Dicumarol Chloramphenicol Monoamine oxidase inhibitors Phenylbutazone Reduce hepatic metabolism of sulfonylureas Phenylbutazone Salicylate Sulfonamide Displace sulfonylureas from plasma proteins Increased hypoglycemic action of sulfonylurea drugs Allopurinol Probebecid Phenylbutazone Salicylate Sulfonamide Decrease urinary excretion of sulfonylureas or their metabolites

Short acting Insulin Secretagogue: Meglitinides • Agent: REPAGLINIDE • Similar mechanism to sulfonylureas • Less potent than sulfonylureas (with the exception of tolbutamide) • Faster onset…. . indicated for controlling postprandial glucose excursions…. . taken up to 30 min before each meal • Short duration of action……safer than long‐acting sulfonylurea (less hypoglycemia) and less weight • Hepatically cleared by CYP 3 A 4…. safe in patients with renal impairment

Short-acting Insulin Secretagogue: D-Phenylalanine Derivatives • NATEGLINIDE…newest agent • Stimulates a rapid and transient release of insulin • Dose up to 30 min before each meal…. reduce postprandial glycemic elevations • The incidence of hypoglycemia may be the lowest of all the secretagogues • Like repaglinide, it is approved for both monotherapy and in combination with metformin or TZDs • Can be used in patients with renal impairment or erderly
2. Insulin sensitizers • They lower blood glucose by improving target-cell response to insulin without increasing pancreatic insulin secretion • Their effects do not depend upon functional islet cells and generally do not cause hypoglycemia • Two classes of oral agents improve insulin action A. Biguanides B. Thiazolidinediones

1. Biguanides • Metformin (Glucophage®), the only agent available • Phenformin was withdrawn during the 1970 s because of an association with lactic acidosis • “euglycemic” agents…hypoglycemia is rare • Not bound to plasma proteins, not metabolized and excreted unchanged by the kidneys • only oral antidiabetic agent proven to reduce mortality risk

1. Biguanides- Mechanism of action • Not fully understood! • Activate the enzyme AMP-activated protein kinase (AMPK) and reduce hepatic glucose production. • AMPK is activated by phosphorylation when cellular energy stores are reduced (i. e. , low ATP) • hepatic glucose production, glucose uptake and block gluconeogenesis • insulin sensitivity…. blood glucose levels • Also slows intestinal absorption of sugars and reduce hyperlipidemia: apparent 4 -6 weeks of use

1. Biguanide- Clinical uses • Metformin is recommended as the first line treatment for this condition to treat type 2 DM and is (most commonly used oral agent) • Effective as monotherapy and in combination with nearly every otherapy • Cause weight loss, does not cause hypoglycemia • Metformin therapy decreases the risk of macrovascular as well as microvascular disease (otherapies only modified microvascular morbidity) • Reduce TG, LDL, and increase HDL

1. Biguanide- Clinical uses • Metformin prevents the new onset of type 2 DM in middleaged, obese persons with impaired glucose tolerance and fasting hyperglycemia • It does not prevent diabetes in older, leaner prediabetics • Metformin has been used as a treatment for infertility in women with the polycystic ovarian syndrome (PCOS): it improve ovulation and menstrual cyclicity and reduce circulating androgens and hirsutism • Epidemiologic studies suggest that metformin may reduce the risk of some cancers

1. Biguanide- Adverse reactions 1. GIT: anorexia, N, V, D, abdominal discomfort • Dose-related, tend to occur at the onset of therapy, and often transient……increase the dose slowly and take it with meals 2. Interfere with calcium-dependent intestinal absorption of vitamin B 12 during chronic therapy…increase intake of calcium

1. Biguanide- Lactic acidosis • As a consequence of metformin's blockade of gluconeogenesis, the drug may impair the hepatic metabolism of lactic acid • Lactic acidosis may occur in conditions of tissue hypoxia (increased production of lactic acid) and in renal failure (decreased clearance of metformin) • C/I in patients with kidney, liver, or cardiorespiratory insufficiency; alcoholism • Radiocontrast administration cause AKI…discontinue metformin temporarily

2. Thiazolidinediones (Tzds; Glitazones) • Agents: PIOglitazone and ROSIglitazone • Troglitazone was withdrawn after a number of deaths due to reported liver failure • They all act to decrease insulin resistance and enhance insulin action in target tissues

Thiazolidinediones- Mechanism of action • They are ligands for peroxisome proliferator-activated receptor-γ (PPARγ)…. . nuclear receptor found in muscle, fat, and liver • These drugs bind to PPARγ, which activates insulin responsive genes that regulate lipid and glucose metabolism, insulin signal transduction, adipocyte and other tissue differentiation • Thiazolidinediones increase expression of GLUT 1 and GLUT 4…. . decreased free fatty acid levels, decreased hepatic glucose output

Thiazolidinediones- Mechanism of action • Long term therapy with Tzds is associated with a drop in TG levels and a slight rise in high-density lipoprotein (HDL) and low-density lipoprotein (LDL) cholesterol values. . . more significant with pioglitazone

Thiazolidinediones- Clinical uses • Tzds are euglycemics and efficacious in ~70% of new users. The overall response is similar to sulfonylurea and biguanide monotherapy • Because their mechanism of action involves gene regulation, Tzds have a slow onset and offset of activity (over weeks or even months) • Tzds are approved as a monotherapy and in combination with metformin, sulfonylureas, and insulin for the treatment of type 2 diabetes

Thiazolidinediones- Adverse reactions • anemia, weight gain, fluid retention, plasma volume expansion, which lead to peripheral edema • Some of the weight gain is fluid retention but there is also an increase in total fat mass • Edema and weight gain: more likely to occur when combined with insulin or secretagogues • Both drugs increase the risk of heart failure • Liver dysfunction (rare): monitor transaminase enzymes • Loss of bone mineral density……increase fracture risk in women…. due to decreased osteoblast formation • Increase risk of bladder cancer (pioglitazone) • Hypoglycemia rare with Tzds monotherapy

α Glucosidase Inhibitors • Acarbose and miglitol are competitive inhibitors of the αglucosidases (glucoamylase, α amylase and sucrase) in the intestinal brush border • This slows the absorption of carbohydrates; reduce postmeal glucose excursions in both normal and diabetic subjects • α-Glucosidase inhibitors do not stimulate insulin release, nor do they increase insulin action in target tissues. • Thus, as monotherapy, they do not cause hypoglycemia

α Glucosidase Inhibitors • They are approved for type 2 diabetes as monotherapy and in combination with other agents • Taken 3 times daily at start of the meal • Reduce postprandial hyperglycemia…used in both DM type 1 and 2 • Very little is absorbedo of acarbose • In contrast, miglitol has structural similarity to glucose and is absorbed.

α Glucosidase Inhibitors ADEs • Dose related flatulence, diarrhea, and abdominal pain…result from the appearance of undigested CHO in the colon that is then fermented into short-chain fatty acids, releasing gas (infrequently prescribed in USA)…. diminish with ongoing use • Acarbose may elevate serum aminotransferase • Patients with inflammatory bowel disease, colonic ulceration, or intestinal obstruction should not use these drugs

α Glucosidase Inhibitors ADEs • Hypoglycemia not a problem in monotherpay but may occur if combined to sulfonylurea or insulin • If hypoglycemia occurs? ? Administer glucose (dextrose) NOT sucrose (table sugar) whose breakdown is blocked!!! • Excreted by the kidneys! Need dose adjustment

Incretin based therapies “gut derived hormones that stimulate insulin secretion with nutrient ingestion” In ● cre ● tin Intestine Secretion Insulin

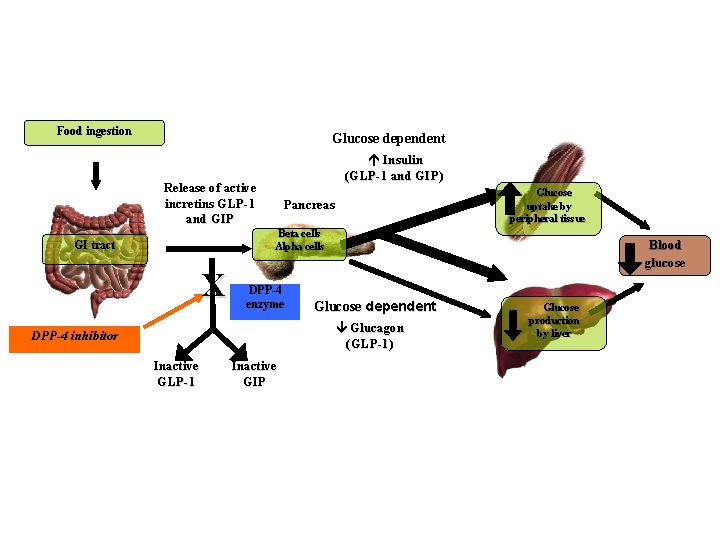
Incretin based therapies • An oral glucose load provokes a higher insulin response compared with an equivalent dose of glucose given intravenously • The oral glucose causes a release of gut insulinotropic hormones (“incretins”): glucagon like peptide 1 (GLP 1) and glucose dependent insulinotropic peptide (GIP)…. . amplify the glucose induced insulin secretion • When GLP 1 is infused in patients with type 2 diabetes, it stimulates insulin release and lowers glucose levels
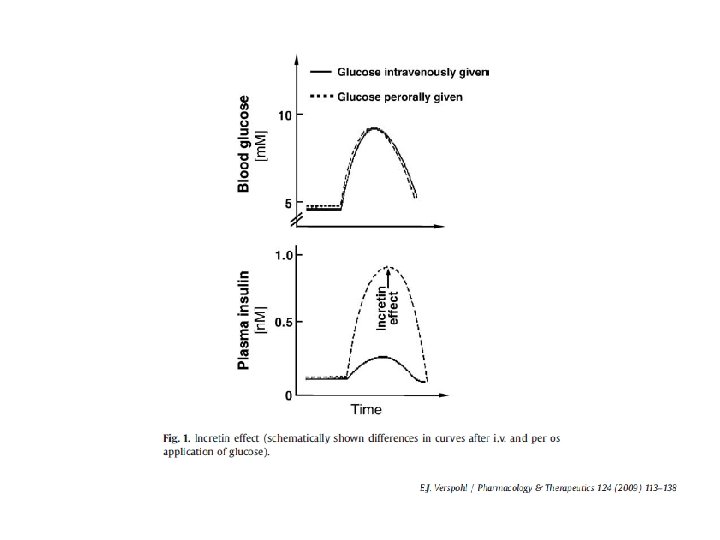
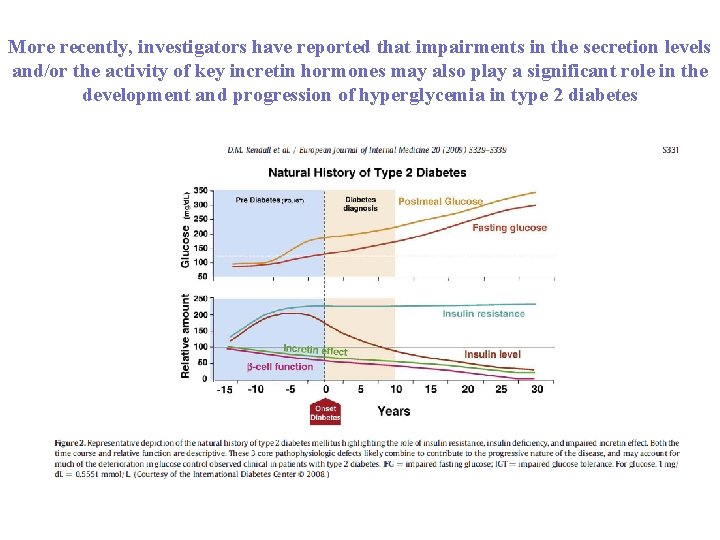
More recently, investigators have reported that impairments in the secretion levels and/or the activity of key incretin hormones may also play a significant role in the development and progression of hyperglycemia in type 2 diabetes

Incretin based therapies • In addition to promote insulin secretion in a glucose dependent manner GLP-1 also: • Suppresses glucagon secretion, • Slows gastric emptying • Promotes satiety • Circulating GLP-1 is rapidly (1 to 2 minutes) degraded by the dipeptidyl peptidase 4 enzyme (DPP 4) and eliminated renally • The native peptide therefore cannot be used therapeutically!!
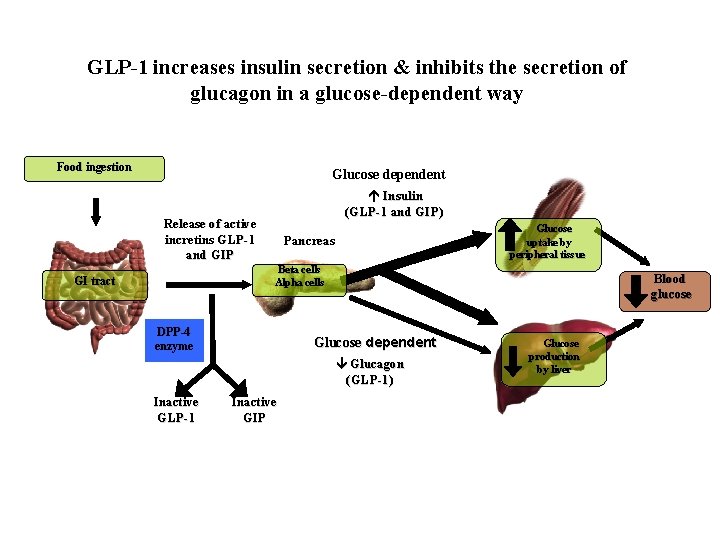
GLP 1 increases insulin secretion & inhibits the secretion of glucagon in a glucose dependent way Food ingestion Glucose dependent Insulin (GLP 1 and GIP) Release of active incretins GLP 1 and GIP Pancreas Glucose uptake by peripheral tissue Beta cells Alpha cells GI tract DPP 4 enzyme Blood glucose Glucose dependent Glucagon (GLP 1) Inactive GLP 1 Inactive GIP Glucose production by liver

Incretin based therapies • Two different approaches can be used: 1. Develop stable analogs of GLP-1 not subject to the same enzymatic degradation or renal clearance • Four GLP-1 receptor agonists (Sub. Q inj. ), exenatide, liraglutide, albiglutide, and dulaglutide 2. Develop inhibitors of DPP-4 and prolong the action of endogenously released GLP-1 and GIP. • Four oral DPP-4 inhibitors (P. O), sitagliptin, saxagliptin, linagliptin, and alogliptin, vildagliptin (EU)

Exenatide • A synthetic analog of glucagon-like-peptide 1 (GLP-1) • The first incretin mimetic to become available for the treatment of DM • It is approved as a SC injectable, adjunctive therapy in persons with type 2 DM treated with metformin or metformin plus sulfonylureas who still have suboptimal glycemic control • Injected Sub. Q within 60 minutes before breakfast and dinner.

Exenatide • Risk of hypoglycemia if sulfonylurea…. decrease the dose combined to • Undergoes glomerular filtration…not approved for patients with decreased GFR of less than 30 m. L/min • Major ADE: nausea (about 44% of users), vomiting, diarrhea…. decrease with ongoing exenatide usage • Box warning…. increase risk of medullary thyroid cancers (MTC) ……. . C/I if personal or family Hx. of cancer

Other GLP 1 analogues • Liraglutide: T 1/2 12 hrs permitting once-daily dosing • increase risk of medullary thyroid cancers • Albiglutide: administered once weekly Sub. Q • Dulaglutide: administered weekly subcutaneously • most frequent SE of these agents: N, D and V • All GLP 1 receptor agonists may increase the risk of pancreatitis…. . seek immediate medical care if persistent severe abdominal pain

Sitagliptin, Saxagliptin, Linagliptin Vildagliptin • Inhibitors of dipeptidyl peptidase 4 (DPP 4), the enzyme that degrades incretin and other GLP-1 -like molecules • Approved as a monotherapy in the U. S. and as an add on therapy to metformin, sulfonylureas, and thiazolidinediones • Orally bioavailable
Food ingestion Glucose dependent Insulin (GLP 1 and GIP) Release of active incretins GLP 1 and GIP Pancreas Glucose uptake by peripheral tissue Beta cells Alpha cells GI tract X DPP 4 enzyme Blood glucose Glucose dependent Glucagon (GLP 1) DPP-4 inhibitor Inactive GLP 1 Inactive GIP Glucose production by liver

Sitagliptin, Saxagliptin, Linagliptin Vildagliptin • ADEs: • Nasopharyngitis, upper respiratory infections, and headaches • Hypoglycemia can occur if combined with secretagogues or insulin • Risk of acute pancreatitis, severe allergic an hypersensitivity reactions (immediately discontinue if they occur)

SODIUM-GLUCOSE CO-TRANSPORTER 2 (SGLT 2) INHIBITORS • Glucose is freely filtered and reabsorbed in the proximal tubules by the action of sodium-glucose transporters (SGLTs)…. accounts for 90% reabs. • …. . its inhibition causes glycosuria and lowers glucose levels in patients with type 2 DM • Three SGLT 2 inhibitors CANAGLIFLOZIN , DAPAGLIFLOZIN , EMPAGLIFLOZIN approved for clinical use

SODIUM-GLUCOSE CO-TRANSPORTER 2 (SGLT 2) INHIBITORS • Used alone or in combination with other oral agents or insulin • This treatment is associated with a very low risk for hypoglycemia and with a certain degree of weight loss • The efficacy of the SGLT 2 inhibitors is reduced in CKD • C/I in patients with decreased GFR • Main ADEs: increased incidence of genital infections and urinary tract infections (affect 8– 9% of patients)

Amylin analogs • Amylin (islet amyloid polypeptide (IAPP)): 37 a. a peptide present in insulin secretory granules and secreted with insulin. • amylin: insulin ratio 1: 10 • Physiologically acts as a negative feedback on insulin secretion • At pharmacologic doses acts centrally and: ü reduces glucagon secretion, ü slows gastric emptying by a vagally mediated mechanism ü centrally decreases appetite

Pramlintide……analog of amylin • Modulates postprandial glucose levels and is approved for preprandial use in persons with type 1 and type 2 DM… • Adjunctive therapy for patients using insulin • Administered Sub. Q immediately before eating (in addition to insulin) in those who are unable to achieve their target postprandial blood sugars • Pramlintide should always be injected separately; cannot be mixed with insulin

Pramlintide • Major ADEs: hypoglycemia and GI symptoms including N, V, and anorexia • Because of the risk of hypoglycemia, concurrent rapidor short-acting mealtime insulin doses should be decreased by 50% or more • Pramlintide is C/I in patients with diabetic gastroparesis (delayed stomach emptying) or a Hx. of hypoglycemic unawareness

Other hypoglycemic agents • Colesevelam hydrochloride: bile acid sequestrant and cholesterol-lowering drug • Approved antihyperglycemic therapy for type 2 DM patients taking other medications or prediabetes • Bromocriptine, the dopamine agonist, in randomized placebo-controlled studies lowered Hb. A 1 c

COMBINATION THERAPY IN TYPE 2 DIABETES • Multiple medications may be required to achieve glycemic control • Unless C/I, medical therapy should be initiated with metformin • If clinical failure occurs a second agent is added • Efficacy of agent, hypoglycemic risk, effect on weight, side effects, and cost should be considered!

Glugagon

Glugagon • Is a single chain polypeptide of 29 amino acids synthesized in alpha cells of pancreas • Glucagon is extensively degraded in the liver, kidney, plasma and at its tissue receptor sites. T 1/2 ~3 6 min. • Glucagon interacts with a GPCR on the plasma membrane of target cells that signals through Gs

Glugagon Pharmacologic Effects Metabolic effect • Activation of glucagon receptors on liver cells increases c. AMP……catabolism of stored glycogen and increases gluconeogenesis and ketogenesis • Glucagon infusion results in elevated blood glucose at the expense of stored hepatic glycogen

Glugagon Pharmacologic Effects Cardiac Effects • Glucagon has a potent inotropic and chronotropic effect on the heart, mediated by the c. AMP mechanism…. effect similar to beta adrenergic agonists! Effects on Smooth Muscle • Large doses of glucagon relaxation of the intestine produce profound

Glugagon Clinical uses • Severe hypoglycemia (IV, IM, SC): glucagon is used for the emergency Tx. of severe hypoglycemia in patients with type 1 DM when unconsciousness precludes oral feeding & IV glucose is not possible • Radiology of the bowel: glucagon has been used extensively in radiology as an aid to x-ray visualization of the bowel because of its ability to relax the intestine • β Adrenoceptor Blocker Overdose: glucagon is sometimes useful for reversing the cardiac effects of an overdose of β-blocking agents

Glugagon ADEs • Transient nausea and occasional vomiting can result from IV glucagon administration • These are generally mild, and glucagon is relatively free of severe adverse reactions

Moderate to Severe Hypoglycemia • Glucagon can be injected SC or IM into the shoulder or anterior thigh • the recommended dose is 1 mg for adult patients • contacts should be taught how to mix, draw up, and administer glucagon during emergency situations • Kits with prefilled syringes containing 1 mg glucagon are available • Patients given glucagon should be positioned so that their face is turned toward the floor to prevent aspiration in the event of vomiting. • As soon as the patient awakens (10– 25 minutes), he or she should be fed.
- Slides: 136