Palladium Removal from Reaction Mixtures by FixedBed Adsorption

Palladium Removal from Reaction Mixtures by Fixed-Bed Adsorption M. J. Girgis 1, L. E. Kuczynski 2, S. M. Berberena 2, C. A. Boyd 2, P. L. Kubinski 2, M. L. Scherholz 2, D. E. Drinkwater 1, M. Yang 1, X. Shen 1, S. Babiak 1 , S. Farrell 2, R. P. Hesketh 2, and B. G. Lefebvre 2 1 Chemical and Analytical Development, Novartis Pharmaceuticals Corporation, East Hanover, New Jersey 2 Department of Chemical Engineering, Rowan University, Glassboro, New Jersey

Introduction § Metal-containing catalysts used extensively in production of pharmaceutical intermediates and API’s • Organometallic complexes in solution (e. g. , for cross-coupling reactions) • Supported metals (e. g. , hydrogenations) § Specifications for low metal content in drug substance (e. g. , < 10 ppm for Pd for orally administered drugs) efficient separations processes for metals removal needed § Metals removal by adsorption on solids widely used in batch processes • Solid adsorbent added to vessel • After contacting period, adsorbent separated by filtration to give filtrate with low metal content • Tedious vessel cleaning required if adsorbent is comprised of fine particles 2 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
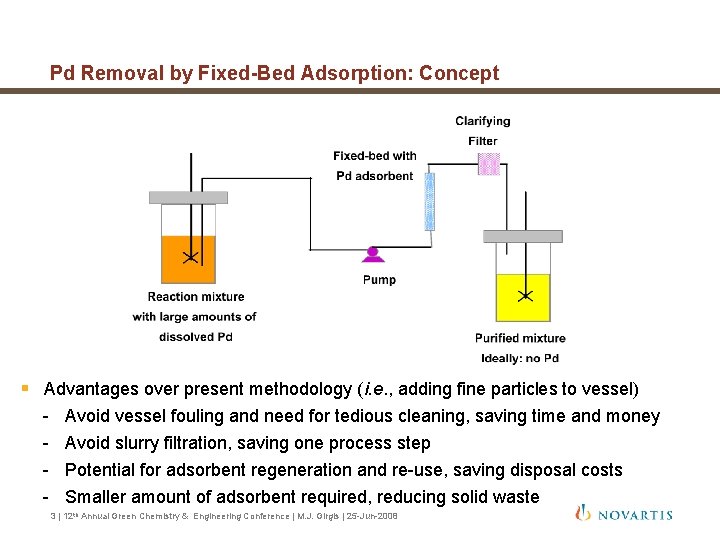
Pd Removal by Fixed-Bed Adsorption: Concept § Advantages over present methodology (i. e. , adding fine particles to vessel) - Avoid vessel fouling and need for tedious cleaning, saving time and money Avoid slurry filtration, saving one process step Potential for adsorbent regeneration and re-use, saving disposal costs Smaller amount of adsorbent required, reducing solid waste 3 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
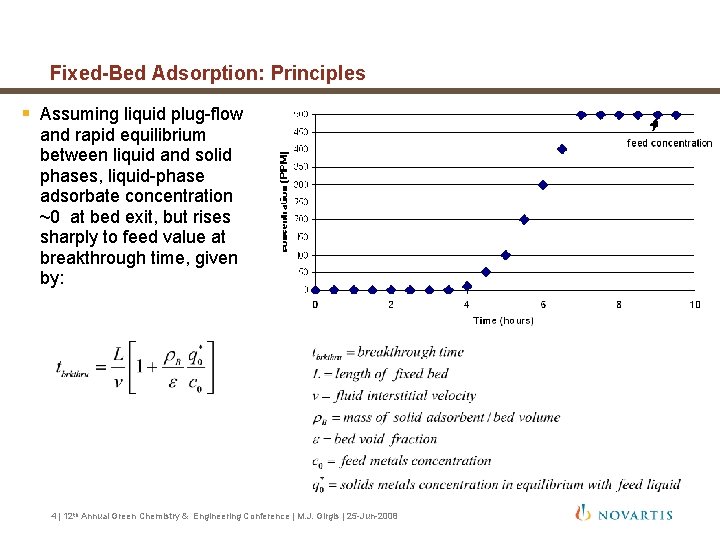
Fixed-Bed Adsorption: Principles § Assuming liquid plug-flow and rapid equilibrium between liquid and solid phases, liquid-phase adsorbate concentration ~0 at bed exit, but rises sharply to feed value at breakthrough time, given by: 4 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
Objective and Approach § Objective: devise methodology allowing estimation of breakthrough time for fixed-bed adsorption applied to the removal of soluble organopalladium complexes § Approach: use an actual reaction mixture (representative of those encountered in practice) to characterize adsorption and evaluate fixed-bed performance § Heck coupling selected as a representative reaction § Adsorption characterized in presence of other species (e. g. , reaction product, catalyst, solvent, reagents) which could impact adsorption 5 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Chemical Reaction Procedure § Conducted based on procedure developed in-housea, and performed typically on 600 -m. L scale using jacketed vessel with overhead agitation • Charge: 183 g of aldehyde 1 (989 mmol), 1. 11 g of Pd(OAc)2 (5 mmol), 6. 02 g of P(o-Tol)3 (20 mmol), 467 g of CH 3 CN (594 m. L), 189 g of Bu 3 N (1. 02 mmol), and 87. 9 g of olefin 2 (1. 02 mmol) • Heat reaction mixture to 75 °C over 20 min and hold for at least 16 h § Untreated final reaction mixture contains about 560 ppm Pd, isolated solid 3 contains about 2100 ppm Pd suitable reaction for investigating Pd-removal a. Slade, J. and Liu, H. personal communication 6 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Batch Adsorption Experiments Procedure § Apparatus: six constant-temperature small baths, each with independent temperature and agitation control § Vials containing 15 -20 m. L of Heck coupling reaction mixture placed in each bath, with varying amounts of adsorbent (0. 025 -2 g) added to each vial § Slurries agitated with magnetic stir bars overnight to allow equilibration, with vials sealed to minimize solvent loss (material balance closure typically > 95%) § Solids filtered, filtrates analyzed for Pd content using ICP/OES (Robertson Microanalytical Laboratories, Madison, NJ), solid-phase Pd content calculated by difference § Three adsorbents investigated: (a) P 1400 activated carbon (PICA), (b) Smopex 110, comprising thiourea grafted polyolefin fiber (Johnson Matthey) and (c) Quadra. Pure TU, consisting of thiourea bound to 0. 5 -mm resin beads (Reaxa Ltd. ) 7 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
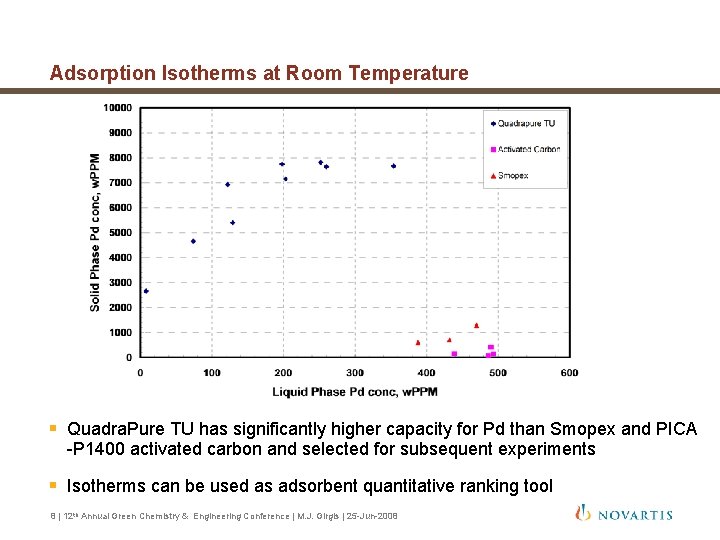
Adsorption Isotherms at Room Temperature § Quadra. Pure TU has significantly higher capacity for Pd than Smopex and PICA -P 1400 activated carbon and selected for subsequent experiments § Isotherms can be used as adsorbent quantitative ranking tool 8 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
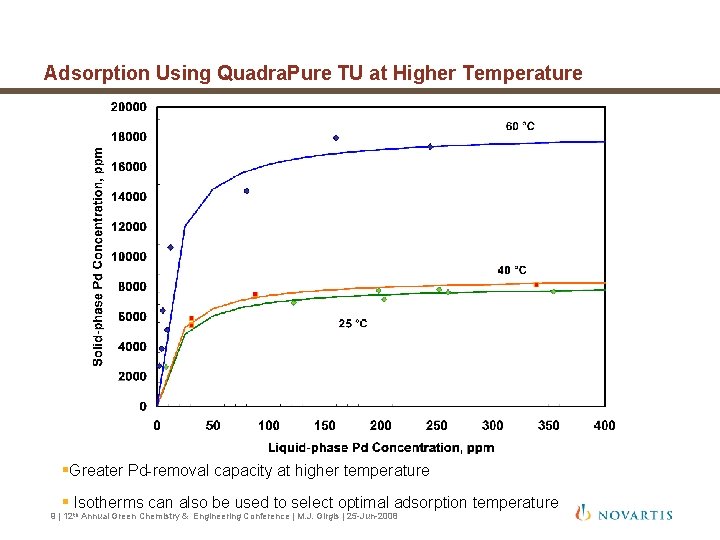
Adsorption Using Quadra. Pure TU at Higher Temperature §Greater Pd-removal capacity at higher temperature § Isotherms can also be used to select optimal adsorption temperature 9 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
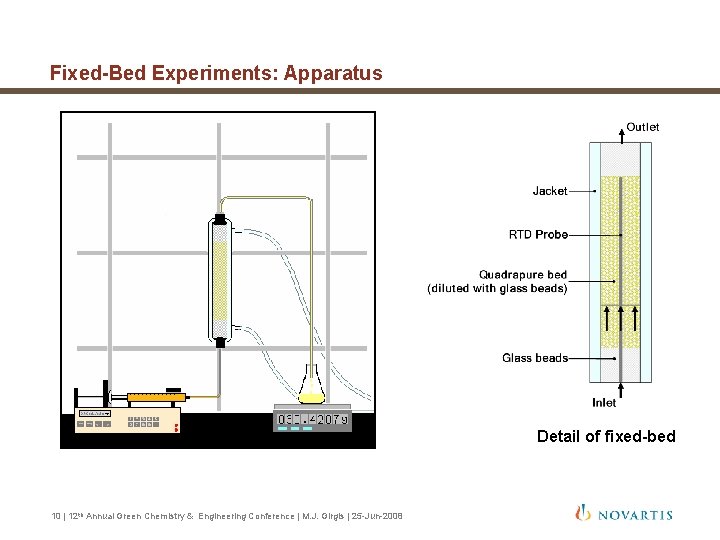
Fixed-Bed Experiments: Apparatus Detail of fixed-bed 10 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

HPLC Method for Pd Detection § Sodium diethyldithiocarbamate (Na-DEDTC) forms complexes with Pd species in solution with good UV absorption § Structure of complex unknown, but may involve coordination of 1 Pd cation with 2 DEDTC anions § Sample preparation: • 0. 1 m. L of reaction mixture • 0. 1 m. L of Na-DEDTC solution • 0. 5 m. L of acetonitrile 11 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Initial Runs: Fixed-Bed Adsorption Experiments with Quadra. Pure TU § Vendor recommendations (Hinchliffe et al. , Org. Proc. Res. Dev. , 11(3), 2007, 477) flow rate of 4 -6 column volumes/h § More conservative value of 1 column volume/h used in initial runs § Vendor-specified resin capacity of 20 mg Pd/g resin and 570 ppm Pd feed content resin mass used (2. 4 g) should process 107 m. L of reaction mixture, giving tbrkthru = 5. 9 h at flow rate employed (0. 3 cm 3/min) 12 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
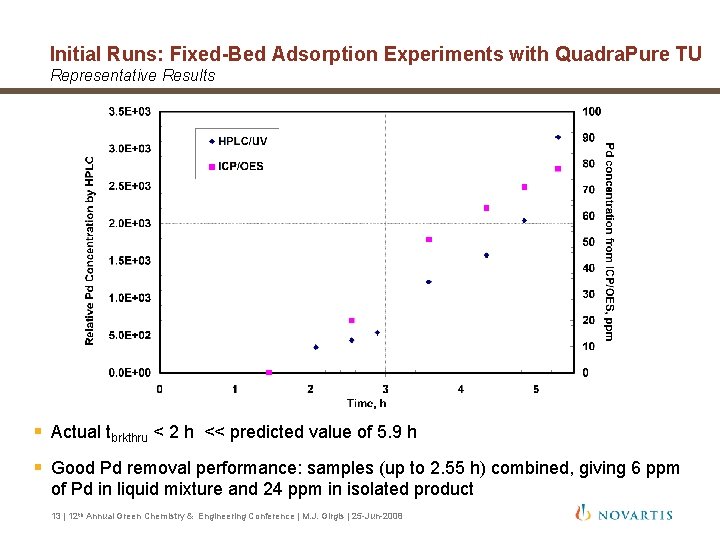
Initial Runs: Fixed-Bed Adsorption Experiments with Quadra. Pure TU Representative Results § Actual tbrkthru < 2 h << predicted value of 5. 9 h § Good Pd removal performance: samples (up to 2. 55 h) combined, giving 6 ppm of Pd in liquid mixture and 24 ppm in isolated product 13 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
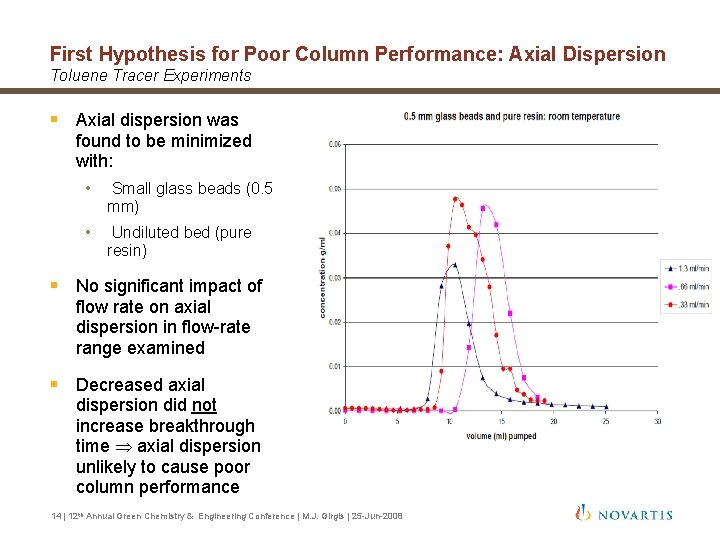
First Hypothesis for Poor Column Performance: Axial Dispersion Toluene Tracer Experiments § Axial dispersion was found to be minimized with: • Small glass beads (0. 5 mm) • Undiluted bed (pure resin) § No significant impact of flow rate on axial dispersion in flow-rate range examined § Decreased axial dispersion did not increase breakthrough time axial dispersion unlikely to cause poor column performance 14 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
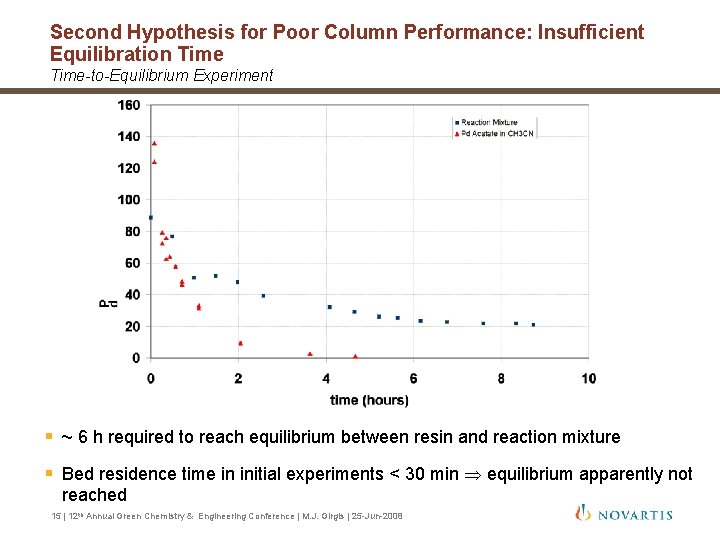
Second Hypothesis for Poor Column Performance: Insufficient Equilibration Time-to-Equilibrium Experiment § ~ 6 h required to reach equilibrium between resin and reaction mixture § Bed residence time in initial experiments < 30 min equilibrium apparently not reached 15 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Adsorption Experiment with 2 -h reaction time Design Procedure = 6. 91 cm 3/h Inputs tres 2 h Mres 17. 3 g r. B 0. 501 g/cm 3 e 0. 400 A 1. 69 cm 2 c 0 570 ppm q 0 * 18, 000 ppm = 20. 5 cm = 10. 2 cm/h = 4. 24 x 10 -3 cm/h = 80. 3 h Volume processed at 16 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008 = 560 cm 3
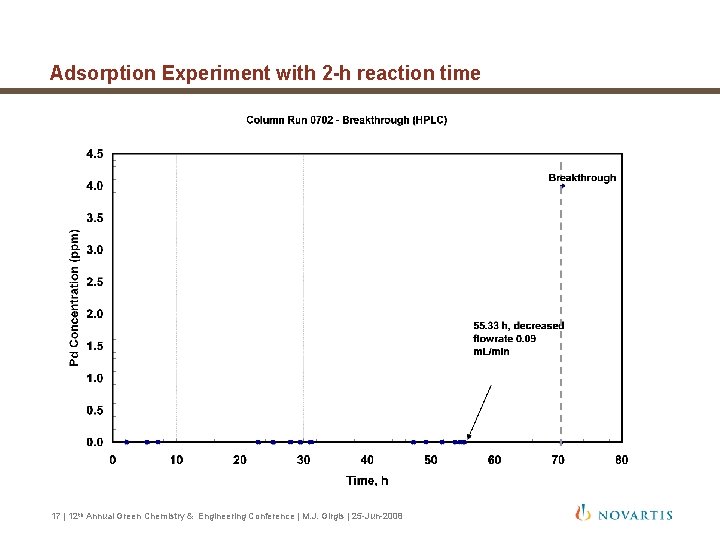
Adsorption Experiment with 2 -h reaction time 17 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
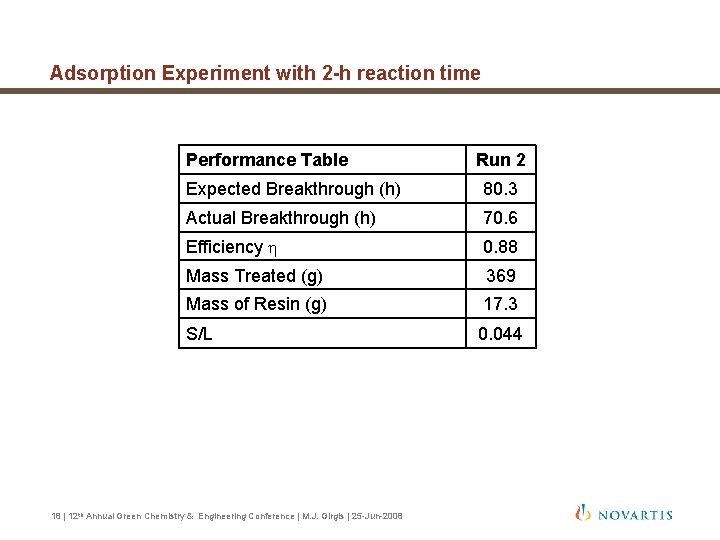
Adsorption Experiment with 2 -h reaction time Performance Table Run 2 Expected Breakthrough (h) 80. 3 Actual Breakthrough (h) 70. 6 Efficiency h 0. 88 Mass Treated (g) 369 Mass of Resin (g) 17. 3 S/L 0. 044 18 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Adsorption Experiment with 2 -h reaction time Product Recovery Crop 1 Mass (g) Crop 1 Concentration (ppm) Crop 2 Mass (g) Run 2 56. 2 3 9. 44 Crop 2 Concentration (ppm) 9 Weight Average Concentration (ppm) 3. 86 Yield (%) 67. 9 19 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
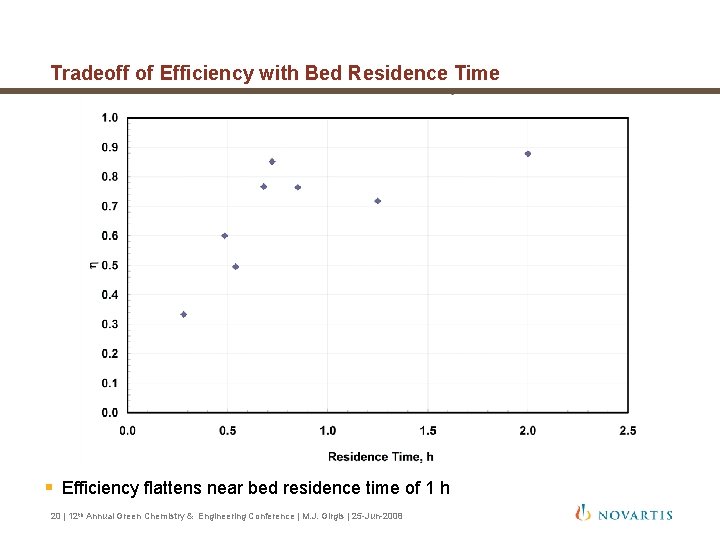
Tradeoff of Efficiency with Bed Residence Time § Efficiency flattens near bed residence time of 1 h 20 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Design Procedure § Breakthrough time given by modified equation: • Efficiency h determined experimentally § Equation above implies: § If same breakthrough time is desired on large and small scales, large-scale column operating parameters are given as follows to process a volume V of solution: 21 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
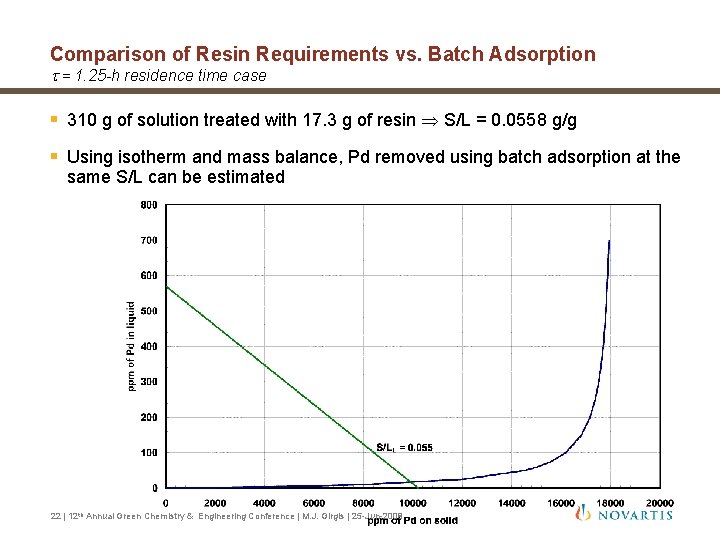
Comparison of Resin Requirements vs. Batch Adsorption t = 1. 25 -h residence time case § 310 g of solution treated with 17. 3 g of resin S/L = 0. 0558 g/g § Using isotherm and mass balance, Pd removed using batch adsorption at the same S/L can be estimated 22 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Comparison of Resin Requirements vs. Batch Adsorption t = 1. 25 -h residence time case § Single batch adsorption stage at S/L = 0. 0558 • 15 ppm of Pd for the liquid-phase palladium concentration (estimated: 55 ppm for solid) § To obtain a Pd content of 7 ppm in the isolated solid (or 1. 9 ppm in the liquidphase reaction mixture) with a single batch adsorption stage • S/L value of 0. 24 required • Corresponds to 4. 3 times resin required in the fixed-bed with 1. 25 -h residence time and 72% efficiency 23 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Conclusions § Adsorption of soluble palladium species characterized quantitatively by determining adsorption isotherms for three adsorbents § Adsorption isotherms used to select best adsorbent (Quadra. Pure TU) and optimal adsorption temperature (60 °C) § Short breakthrough times in initial fixed-bed experiments attributed to insufficient approach to equilibrium § Experiments with bed residence times giving closer approach to equilibrium gave bed performance > 70% of theoretical degree of palladium removal based on the assumption of local fluid-solid equilibrium § Design methodology developed in which actual tbrkthru estimated from experimental determination of bed efficiency vs. residence time § To obtain the same extent of palladium removal, fixed-bed adsorption requires <25% the resin amount relative to a single stage of batch adsorption 24 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008

Acknowledgments § Dr. Joel Slade, Dr. Hui Liu, and Mr. Mark Davis for consultations and assistance with the Heck coupling reaction § Dr. Mahavir Prashad for advice on adsorbent and reaction selection § Mr. Lee Alden for assistance with design and construction of experimental apparatus Drs. Thomas Blacklock and Oljan Repič for management support 25 | 12 th Annual Green Chemistry & Engineering Conference | M. J. Girgis | 25 -Jun-2008
- Slides: 25