PACKET 14 Nuclear Chemistry Reference Table N and

PACKET #14: Nuclear Chemistry Reference Table: N and O

Nuclear Stability Some nuclei are stable (don’t change), but some are unstable. An unstable nuclei breaks down spontaneously giving off rays and particles. The ratio of neutrons to protons is what determines whether a nuclei is stable or unstable. Atoms with atomic numbers above 83 have NO stable isotopes

Nuclear Stability n The ratio of neutrons to protons in stable nuclei is between 1: 1 and 1. 5: 1, the higher ratio being associated with larger nuclei that have larger repulsive forces Neutron Proton ARE THESE ISOPTOPES OF HYDROGEN STABLE? 1 H (hydrogen) 2 H (deuteron) 3 H (tritium)

Radioactivity In radioactivity, the nucleus of an unstable isotope or element decays spontaneously and gives off rays and particles. Table O lists the decay particles and rays. MASS CHARGE

Decay Particles & Rays Alpha Decay: The nucleus decays and gives off alpha particles. Table O shows the symbols MASS: +4 CHARGE: +2 Beta Decay (electron): The nucleus decays and gives off beta particles. Table O shows the symbol. MASS: 0 CHARGE: -1

Decay Particles & Rays Positron Decay: The nucleus decays and gives off positron particles. Table O shows the symbols MASS: 0 CHARGE: +1 Gamma Ray: The nucleus decays and gives off gamma rays. Table O shows the symbol. MASS: 0 CHARGE: 0 **HIGHEST PENETRATING POWER**

Separating Alpha, Beta, & Gamma Particles The alpha particle is a positively charged particle. The alpha particle will be attracted to the negative side of the magnet (red) because the red is the negative end. The gamma particle has no charge, so it will not be attracted to either side. The beta particle is negatively charged particle. The beta particle will be attracted to the positive end of the magnet.

Transmutation When the nucleus of an atom decays and one element changes into another element. TWO TYPES: Natural & Artificial Natural Transmutations = Spontaneous Artificial Transmutations = NOT!! An element must be BOMBARDED with decay particles (remember Rutherford? ? )
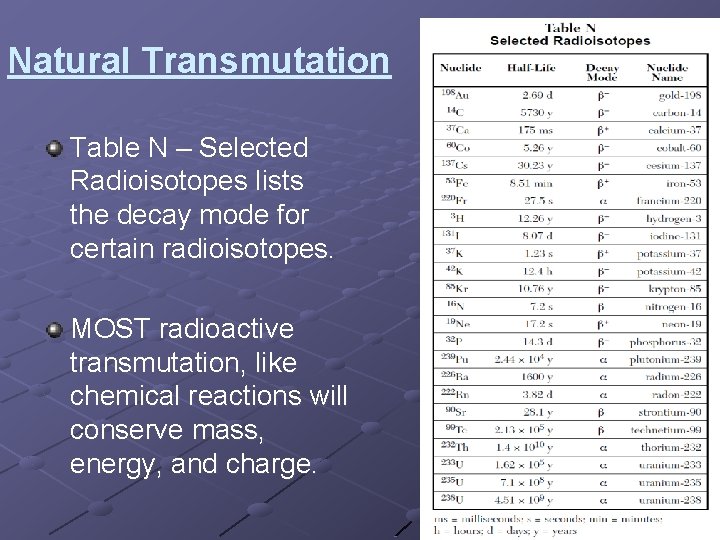
Natural Transmutation Table N – Selected Radioisotopes lists the decay mode for certain radioisotopes. MOST radioactive transmutation, like chemical reactions will conserve mass, energy, and charge.

Natural Transmutation Question: Draw the correct transmutation for the radioactive isotope 220 Fr. What element does it transmutate into? 220 4 He + X Fr 87 2 220 Fr spontaneously decays and gives off alpha particles

Let’s Practice. . . Determine the particle that is represented by X in each equation 1. 230 Th 90 2. 4 He 2 3. 227 Ac 89 4. 4 He 2 → 42 He + X + 2713 Al → X + 10 n → 0 -1 e + X + 147 N → X + 11 H For these transmutation questions, please note that they could ask you to solve for any variable: the initial radioactive isotope, the type of decay, or the transmutated element.

Artificial Transmutation When the nucleus of an element is first bombarded with high energy particles!! THEN it decays and changes into a new element! **Artificial Transmutations are easy to spot!! If you see an atom AND one of the high energy particles on left side of arrow combining to make a new element on the right side of arrow, then it is an artificial transmutation!! In other words: Artificial Transmutations Have TWO reactants Natural Transmutations Have ONE reactant

Artificial Transmutation EXAMPLE: 27 Al 13 TWO reactants + 42 He 3015 P + 10 n Aluminum absorbs an alpha particle and releases a neutron to form phosphorus Absorption of an alpha particle by aluminum increases the mass by 4 from 27 to 31 and increases the atomic number by 2 from 13 to 15 Loss of a neutron reduces the mass by 1 from 31 to 30 but does not effect the atomic number


Induced Nuclear Reactors All artificial transmutations, are induced by targeting a nucleus and bombarding it with a nuclear particle. Particle Accelerators: A device that uses electric and magnetic fields to provide a bombarded nuclear particle with sufficient energy to induce a desired nuclear reaction

Nuclear Energy There are two types of nuclear reactions that we will discuss: Fission Fusion IN FISSION AND FUSION REACTIONS, THE MASS OF THE PRODUCTS IS SLIGHTLY LESS THAN THE MASS OF THE REACTANTS. SOME MASS IS CONVERTED TO ENERGY

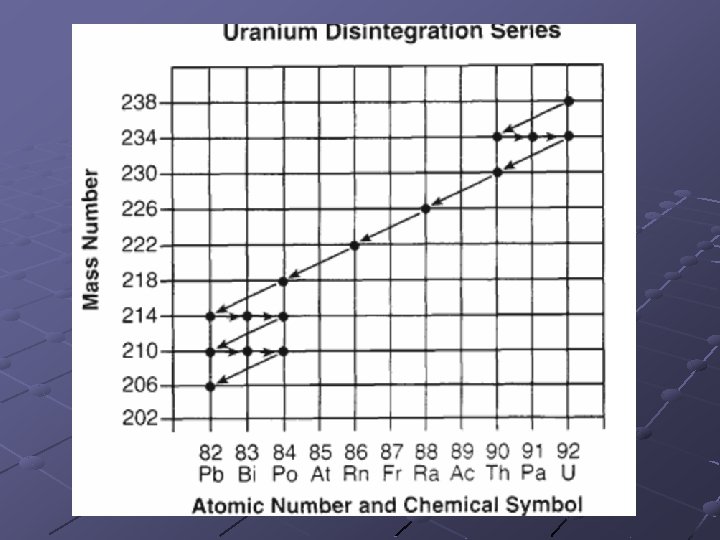
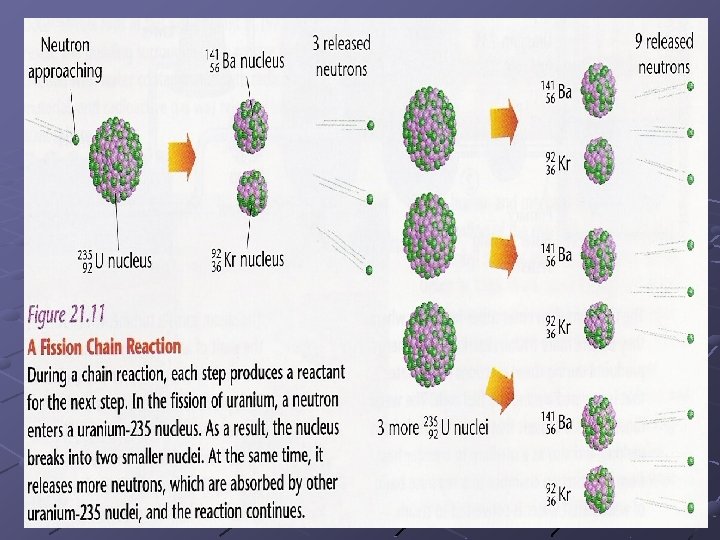
Nuclear Fission One atom absorbs a neutron and splits into two or more pieces, giving off a tremendous amount of energy. 235 U 92 + 1 n 0 → 23692 U 141 Ba 56 + 9236 Kr + 310 n + energy Fission occurs in nuclear reactors and the atomic bomb (chain reaction) FISSION – “SPLIT”

Nuclear Fusion When the two light nuclei unite to form a heavier nucleus. 2 H 1 + 2 H 1 → 4 He 2 This is the type of energy that exists on the SUN, and the HYDROGEN BOMB. Fusion requires extremely high temperature and pressure in order for it to occur. Fusion creates more energy than fission FUSION – “UNITE”


Comparing Nuclear vs. Chemical Reaction Nuclear reactions release more energy than chemical reactions. RISKS & BENEFITS OF NUCLEAR REACTIONS: Benefits: provide energy (produces the energy of the sun). Risks: -Wastes from are very radioactive, must be stored for more than 100, 000 years without leaking into the ground (long half-lives). -Accidents can cause radioactive spills (mutation/death).
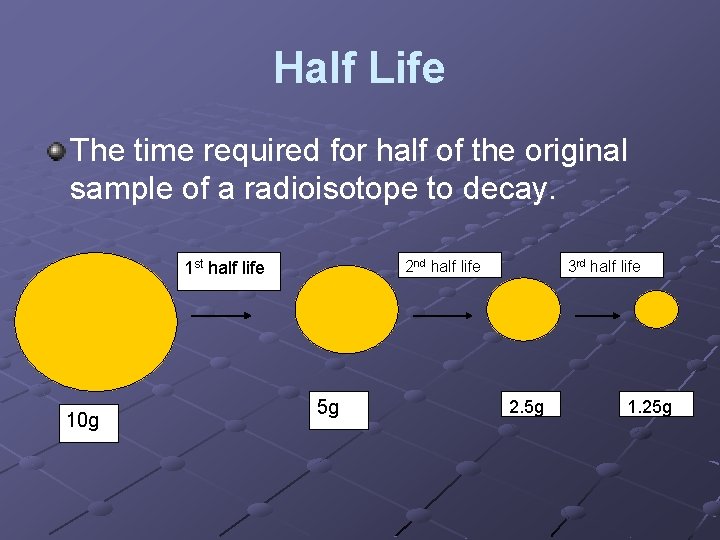
Half Life The time required for half of the original sample of a radioisotope to decay. 2 nd half life 1 st half life 10 g 5 g 3 rd half life 2. 5 g 1. 25 g

NOTHING CAN CHANGE THE HALF LIFE OF A RADIOISOTOPE!

Half Life Half life problems of all types are best solved by setting up a layout that shows the number of half lives, the mass, the time elapsed, and the fraction left. (any of these 4 variables can be the unknown) The one “equation” that you need to know is: Fraction remaining = (1/2)# of half-lives EVERYTHIONG CAN BE SOLVED BY KNOWING THE NUMBER OF HALF LIVES THAT OCCUR!!
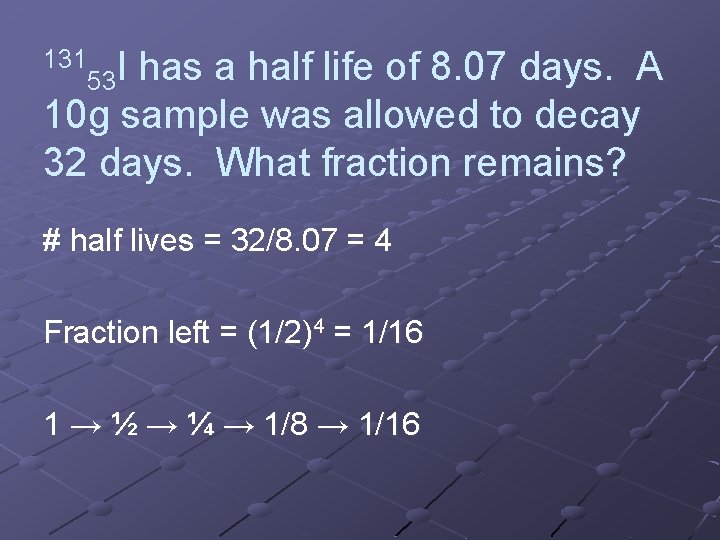
131 I 53 has a half life of 8. 07 days. A 10 g sample was allowed to decay 32 days. What fraction remains? # half lives = 32/8. 07 = 4 Fraction left = (1/2)4 = 1/16 1 → ½ → ¼ → 1/8 → 1/16
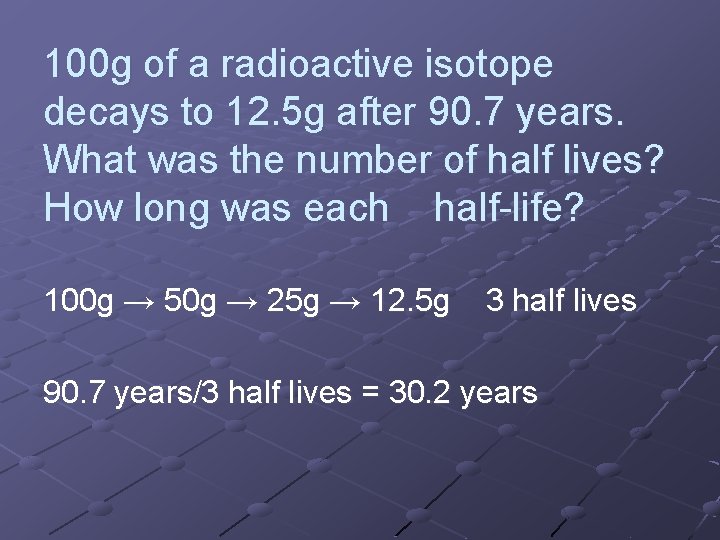
100 g of a radioactive isotope decays to 12. 5 g after 90. 7 years. What was the number of half lives? How long was each half-life? 100 g → 50 g → 25 g → 12. 5 g 3 half lives 90. 7 years/3 half lives = 30. 2 years

A radioisotope has a half life of 10 days. 1 g remains after 40 days. What was the initial amount (original amount) of the radioisotope? Half life = 10 days Time = 40 days 4 half lives passed 1 → 2 → 4 → 8 → 16 g

Uses of Radioactive Isotopes 1. Tracers: Used to trace the path of chemical reactions in living organisms (Carbon-14). 2. Medical Uses: Used in medical diagnosis and treatment of diseases! Isotopes with very short half-lives are used for this because they decay very quickly! Examples: Technetium -99 : Used to pinpoint brain tumors! Iodine -131: Used to diagnose and treat thyroid disorders! Radium & Cobalt-60: Used in cancer treatment! 3. Food Storage: Radiation kills bacteria, yeast, and mold in food, therefore permitting food to be stored for longer periods of time!

Uses of Radioactive Isotopes 4. Radioactive Dating: n Geologic Dating: Based on half-life! Certain radioactive elements occur naturally in ROCK and so they are used to find the AGE of certain rocks! Examples: URANIUM-238 & LEAD-206 n Dating Living Materials: Can help determine the age of wood, bone, animal skin, etc… Example: CARBON -14 & CARBON-12 5. Nuclear Power: Nuclear reactors are used for ELECTRICAL ENERGY!! 6. Indiustrial Measurement: Used to measure thickness of certain metals. Example: Alpha, Beta, and Gamma particles!

Packet #14 – Nuclear Chemistry

















Review Questions 1) The stability of an isotope is based on the ratio of A) electrons and protons B) neutrons and electrons C) neutrons and protons D) atomic mass and atomic number 2) What is the neutron-to-proton ratio in a stable atom of carbon-12? A) 1: 2 B) 12: 1 C) 2: 1 D) 1: 1
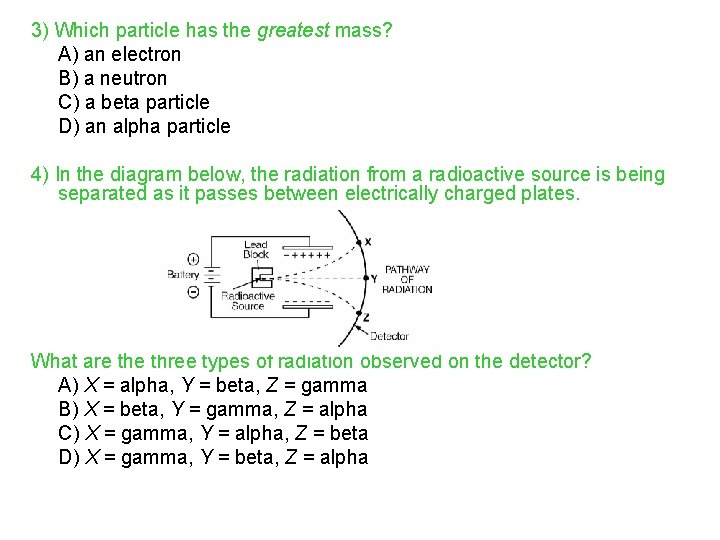
3) Which particle has the greatest mass? A) an electron B) a neutron C) a beta particle D) an alpha particle 4) In the diagram below, the radiation from a radioactive source is being separated as it passes between electrically charged plates. What are three types of radiation observed on the detector? A) X = alpha, Y = beta, Z = gamma B) X = beta, Y = gamma, Z = alpha C) X = gamma, Y = alpha, Z = beta D) X = gamma, Y = beta, Z = alpha

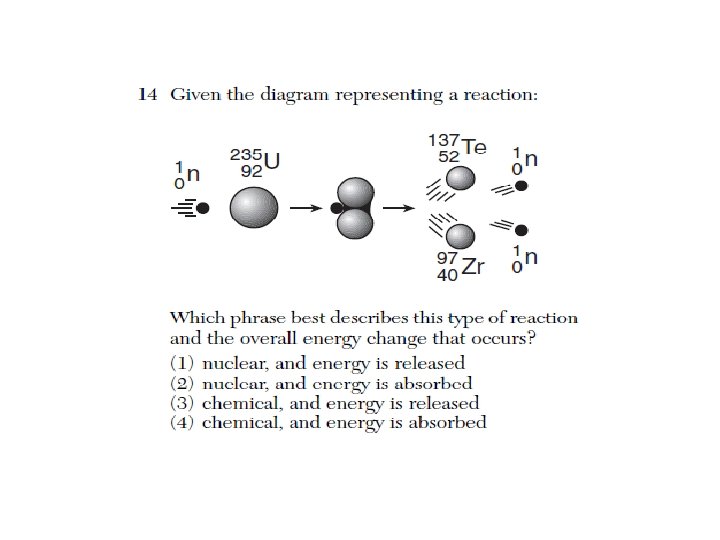
5) According to the Selected Radioisotopes chemistry reference table, which is a decay product of Fe-53? A) Cr-49 B) Mn-53 C) Co-53 D) Ni-47 6) Given the equation: What particle is represented by the letter X? A) beta particle B) positron C) neutron D) alpha particle 7) What is represented by X in the equation below? X 40 18 Ar + 0 -1 e
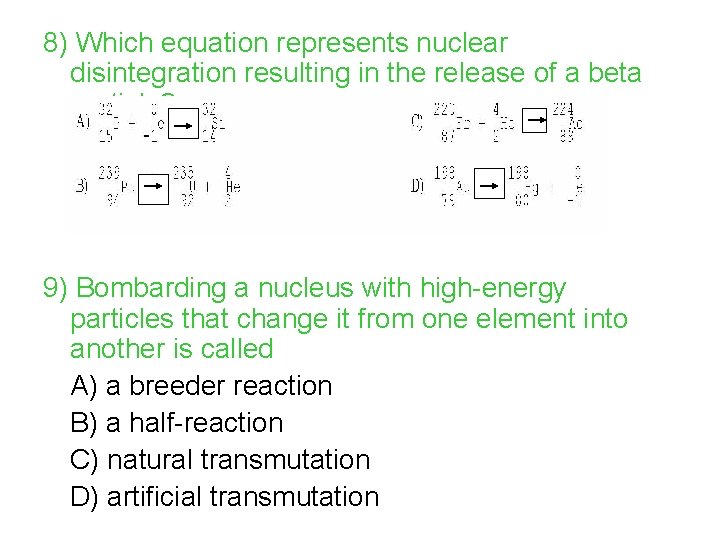
8) Which equation represents nuclear disintegration resulting in the release of a beta particle? 9) Bombarding a nucleus with high-energy particles that change it from one element into another is called A) a breeder reaction B) a half-reaction C) natural transmutation D) artificial transmutation

10) Which nuclear equation represents artificial transmutation? 11) What type of reaction does the following equation represent? A) fission B) fusion C) alpha decay D) beta decay

12) What type of reaction is represented by the equation below? A) natural transmutation B) fusion C) fission D) thermonuclear 13) An original sample of a radioisotope had a mass of 10 grams. After 2 days, 5 grams of the radioisotope remains unchanged. What is the halflife of the radioisotope? A) 4 days B) 2 days C) 5 days D) 1 day

14) What is the number of hours required for potassium-42 to undergo 3 half-life periods? A) 6. 2 hours B) 12. 4 hours C) 24. 8 hours D) 37. 2 hours 15) A radioactive isotope has a half-life of 10 years. What fraction of the original mass will remain unchanged after 50 years?
- Slides: 55