p H HOH Equilibrium of H OH Water
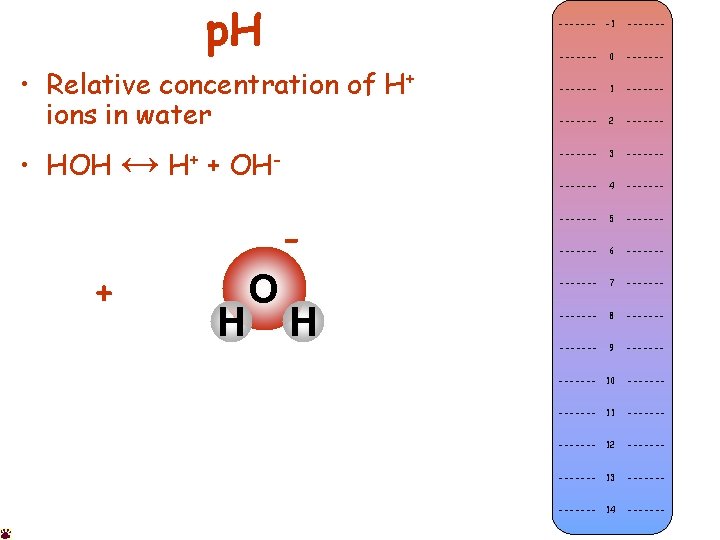
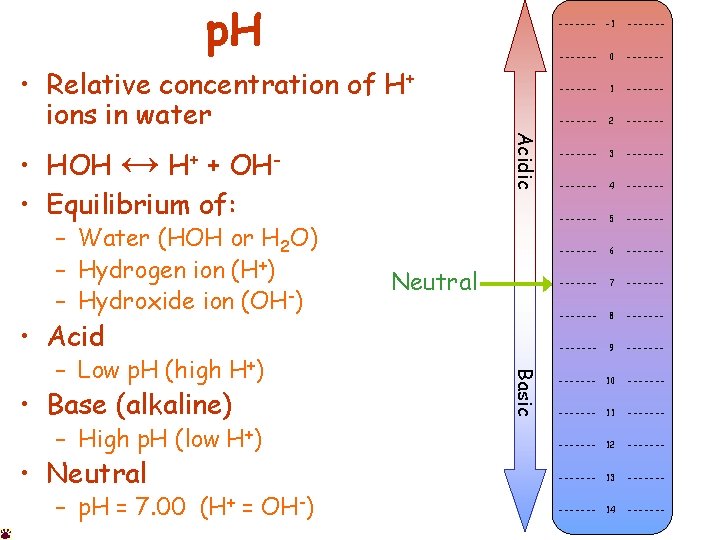
p. H • HOH ↔ + • Equilibrium of: H+ OH- – Water (HOH or H 2 O) – Hydrogen ion (H+) – Hydroxide ion (OH-) Acidic • Relative concentration of H+ ions in water Neutral • Acid • Base (alkaline) – High p. H (low H+) • Neutral – p. H = 7. 00 (H+ = OH-) Basic – Low p. H (high H+) ------- -1 ------- 0 ------- 1 ------- 2 ------- 3 ------- 4 ------- 5 ------- 6 ------- 7 ------- 8 ------- 9 ------- 10 ------- 11 ------- 12 ------- 13 ------- 14 -------
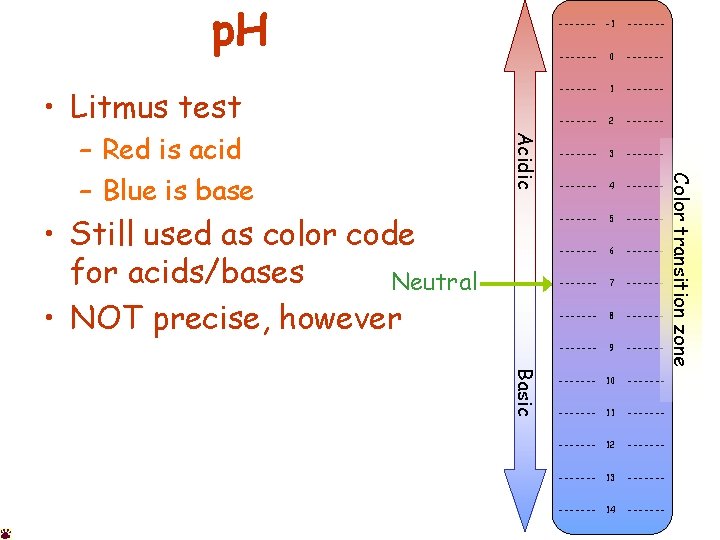
p. H • Litmus test • Still used as color code for acids/bases Neutral • NOT precise, however Basic ------- 0 ------- 1 ------- 2 ------- 3 ------- 4 ------- 5 ------- 6 ------- 7 ------- 8 ------- 9 ------- 10 ------- 11 ------- 12 ------- 13 ------- 14 ------- Color transition zone Acidic – Red is acid – Blue is base ------- -1

p. H • Buffers – Multiple mechanisms – Maximizes safety Neutral Basic • Redundancy Acidic – Chemicals that absorb excess acids and bases added to the solution – Buffered solutions do not change in p. H (very much) when acids or bases are added – Buffers can be used up ------- -1 ------- 0 ------- 1 ------- 2 ------- 3 ------- 4 ------- 5 ------- 6 ------- 7 ------- 8 ------- 9 ------- 10 ------- 11 ------- 12 ------- 13 ------- 14 -------
- Slides: 4