p H A Measure of Acidity and Basicity

p. H – A Measure of Acidity and Basicity • Logarithm: In mathematics, the logarithm of a number is the exponent to which another fixed value, the base, must be raised to produce that number. For example, the logarithm of 1000 to base 10 is 3, because 10 to the power 3 is 1000 (as 1000 = 10 × 10 = 103). for any two real numbers b and x where b is positive and not equal to 1, the logarithm of x to base b, denoted logb(x), is the unique real number y such that by = x. • For example, as 64 = 43, we have log 4(64) = 3. http: //en. wikipedia. org/wiki/Logarithm • Logarithms are significant especially where we are primarily concerned with the exponents to which numbers are raised. • One such application is p. H.
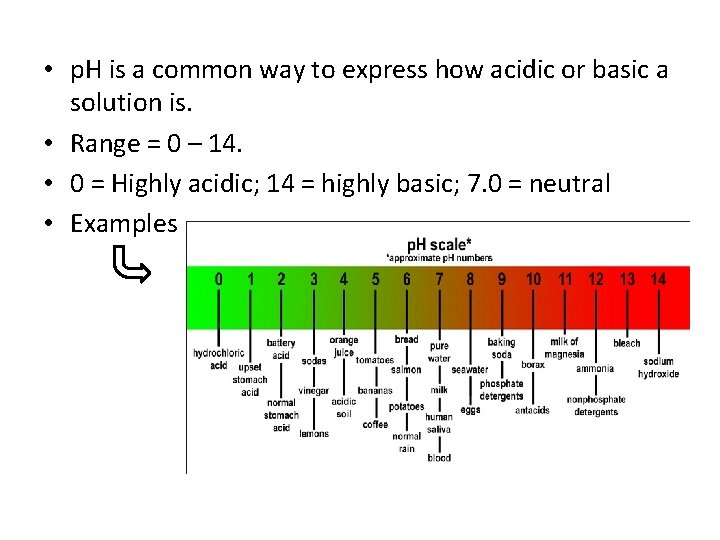
• p. H is a common way to express how acidic or basic a solution is. • Range = 0 – 14. • 0 = Highly acidic; 14 = highly basic; 7. 0 = neutral • Examples
![Equation for calculating p. H: p. H = ‒log [H+], where [H+] = molar Equation for calculating p. H: p. H = ‒log [H+], where [H+] = molar](http://slidetodoc.com/presentation_image_h2/c42fa8c7f2536d0ac24ca0ac0f62bf15/image-3.jpg)
Equation for calculating p. H: p. H = ‒log [H+], where [H+] = molar concentration of hydrogen ion. Example: What is the p. H of a 5. 15 M HCl solution? Since the molarity of the HCl solution = 5. 15, the molarity of the H+ ion also = 5. 15 mol/L: HCl --> H+ + Cl‒ p. H = ‒log [H+] = ‒log 5. 15 = 0. 71. Since HCl is an acid, you would expect a low value for the p. H.

p. OH: The opposite of p. H. • An indication of the concentration of hydroxide ion. • The presence of hydroxide is the feature that causes compounds to be basic (Arrhenius base. ) • p. OH = ‒log [OH‒] • p. OH = 0: Strongly basic; p. OH = 14: Strongly acidic • p. H + p. OH = 14. 0
- Slides: 4