p 01 AcidBase Eqm 1 New AcidBase Theory
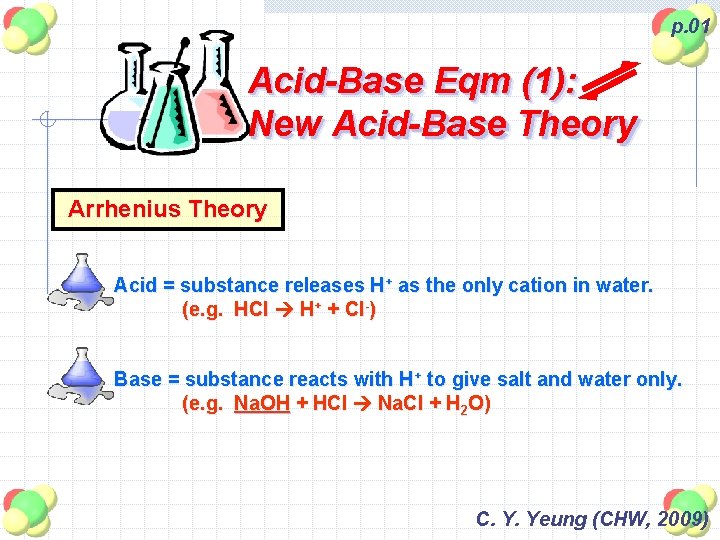
p. 01 Acid-Base Eqm (1): New Acid-Base Theory Arrhenius Theory Acid = substance releases H+ as the only cation in water. (e. g. HCl H+ + Cl-) Base = substance reacts with H+ to give salt and water only. (e. g. Na. OH + HCl Na. Cl + H 2 O) C. Y. Yeung (CHW, 2009)
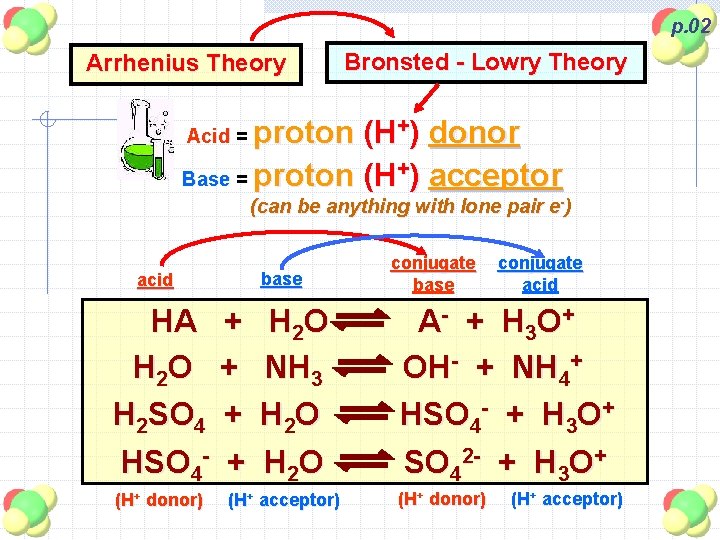
p. 02 Arrhenius Theory Bronsted - Lowry Theory proton (H+) donor Base = proton (H+) acceptor Acid = (can be anything with lone pair e-) base acid HA + H 2 O + H 2 SO 4 + HSO 4 - + (H+ donor) H 2 O NH 3 H 2 O (H+ acceptor) conjugate base conjugate acid A - + H 3 O + OH- + NH 4+ HSO 4 - + H 3 O+ SO 42 - + H 3 O+ (H+ donor) (H+ acceptor)
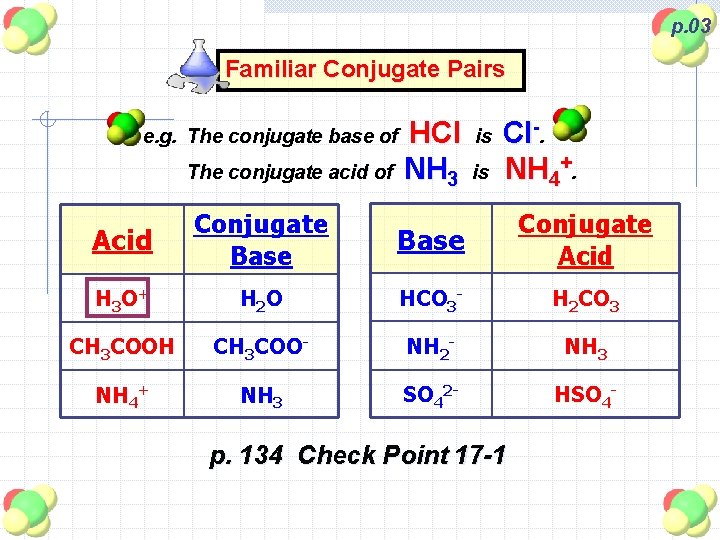
p. 03 Familiar Conjugate Pairs e. g. The conjugate base of The conjugate acid of HCl NH 3 is is Cl-. NH 4+. Acid Conjugate Base Conjugate Acid H 3 O+ H 2 O HCO 3 - H 2 CO 3 CH 3 COOH CH 3 COO- NH 2 - NH 3 NH 4+ NH 3 SO 42 - HSO 4 - p. 134 Check Point 17 -1
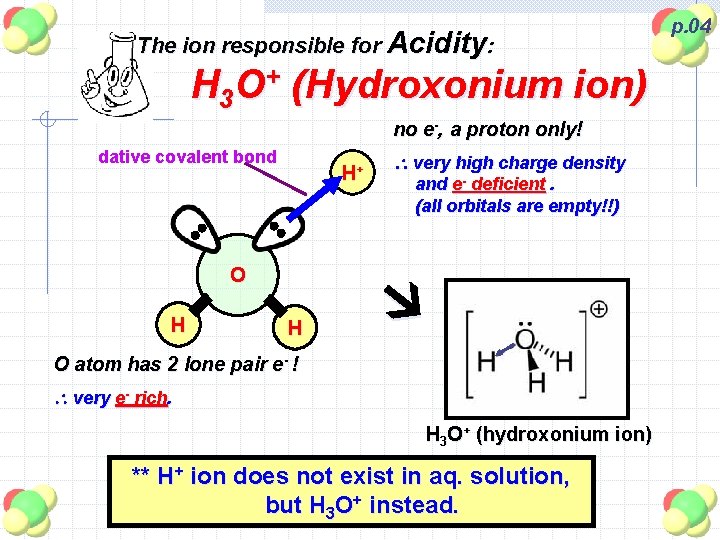
The ion responsible for Acidity: H 3 O+ (Hydroxonium ion) no e-, a proton only! dative covalent bond H+ and e- deficient. (all orbitals are empty!!) O H very high charge density H O atom has 2 lone pair e- ! very e- rich. H 3 O+ (hydroxonium ion) ** H+ ion does not exist in aq. solution, but H 3 O+ instead. p. 04
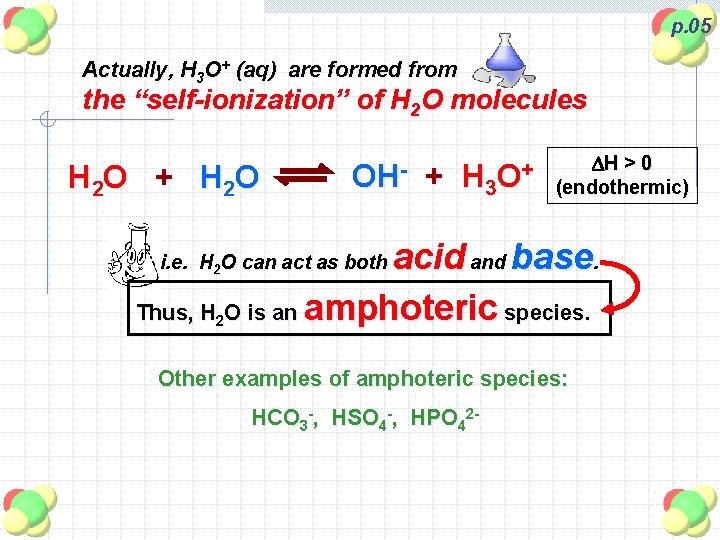
p. 05 Actually, H 3 O+ (aq) are formed from the “self-ionization” of H 2 O molecules H 2 O + H 2 O OH- + DH > 0 + H 3 O (endothermic) acid and base. Thus, H 2 O is an amphoteric species. i. e. H 2 O can act as both Other examples of amphoteric species: HCO 3 -, HSO 4 -, HPO 42 -
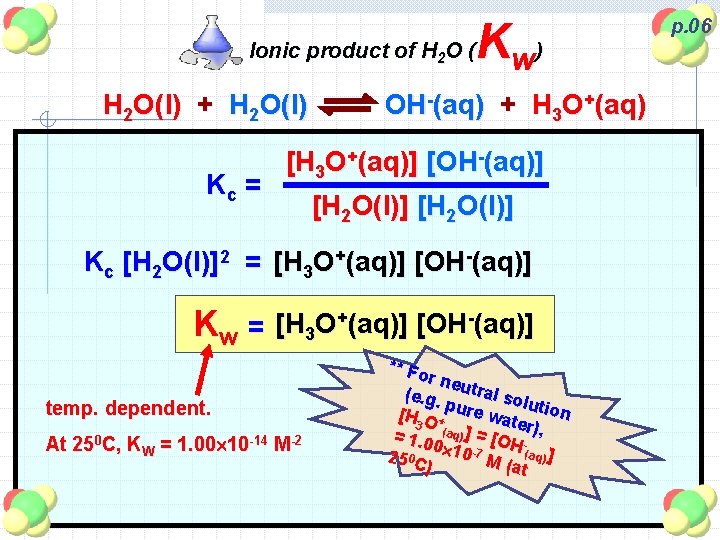
Kw ) Ionic product of H 2 O ( H 2 O(l) + H 2 O(l) Kc = OH-(aq) + H 3 O+(aq) [H 3 O+(aq)] [OH-(aq)] [H 2 O(l)] Kc [H 2 O(l)]2 = [H 3 O+(aq)] [OH-(aq)] Kw = [H 3 O+(aq)] [OH-(aq)] temp. dependent. At 250 C, KW = 1. 00 10 -14 M-2 ** Fo r neu tral s (e. g. olu p [H O + ure wat tion er 3 = 1. 0 (aq) ] = [OH - ), 0 25 0 C 10 -7 M (aq) ] (at ) p. 06
![For all aq. solutions, [H 3 O+] [OH-] = 1. 00 10 -14 mol For all aq. solutions, [H 3 O+] [OH-] = 1. 00 10 -14 mol](http://slidetodoc.com/presentation_image_h2/6ba7d63323fb78b6f69f2577b4918e73/image-7.jpg)
For all aq. solutions, [H 3 O+] [OH-] = 1. 00 10 -14 mol 2 dm-6 If [H 3 O+] = [OH-] : NEUTRAL SOLUTION If [H 3 O+] > [OH-] : ACIDIC SOLUTION If [H 3 O+] < [OH-] : BASIC SOLUTION p. H = – log [H 3 O+] p. OH = – log [OH-] e. g. If [H 3 O+] of solution A is 2. 51 10 -4 M, then at 298 K, p. H = – log (2. 51 10 -4) = 3. 60, p. OH = 14 – 3. 60 = 10. 4 p. 07

p. 08 However, as KW is temperature dependent, p. H also changes with temp. !! Therefore, “p. H of water = 7” is only true at 298 K!! If temp. increases to 313 K, KW becomes 2. 92 10 -14 M-2. at 313 K, [H 3 O+] = [OH-] = (2. 92 10 -14)1/2 = 1. 71 10 -7 M p. H = -log (1. 71 10 -7) = 6. 77 It is neutral !! (at 313 K)

p. 09 For your reference …. Kw & Temperature T , DH>0 eqm. shifts FW, Kw p. 140 Check Point 17 -3

p. 10 Since the p. H falls as temperature increases, does it mean that water becomes more acidic at higher temperature? NO!! [H 3 O+] = [OH-], neutral! p. H drops due to the increase of Kw at high temperature. p. H of an impure water sample at 1000 C = 7. 0 i. e. the sample is acidic / neutral / alkaline. Neutral p. H of water at 1000 C is 6. 14! p. H of an impure water sample at 00 C = 7. 0 i. e. the sample is acidic / neutral / alkaline. Neutral p. H of water at 00 C is 7. 48! p. 140 Check Point 17 -3

p. 11 1994 HKALE Paper 2 Q. 3(b) At 323 K, the p. H of pure water is less than 7. 0. Explain. The dissociation of H 2 O is an endothermic process. H 2 O(l) + H 2 O(l) OH-(aq) + H 3 O+(aq) Increase in temperature, the equilibrium shifts to the right (forward) and hence [H 3 O+] increases. In pure water, [H 3 O+] at 323 K is higher than that at 298 K, therefore p. H of pure water at 323 K < 7.

p. 12 Assignment Study all examples in p. 138 - 139 p. 152 Q. 1 – 2 [due date: 25/3(Wed)] Next …. Calculation of Ka and Kb. Strength of Acid and Base.
- Slides: 12