Ozone Depletion Topic 5 Atmosphere A S S









- Slides: 14

Ozone Depletion Topic 5 Atmosphere A. S: S – W Depletion of Stratospheric Ozone

The Role of Ozone • Stratospheric ozone shields earth against biologically harmful Ultraviolet radiation (up to 99%). • UV rays are absorbed during the formation and destruction of ozone (O 3) from oxygen (O 2).

How is ozone formed? • Ozone, under natural conditions, is constantly being consumed and created by a cyclic photolytic reaction. What does this mean? • Oxygen (O 2) is continuously being converted to ozone (O 3) and back again to O 2. • Reaction is driven by UV rays • Net Reaction: 2 O 3 + UV 3 O 2 • It is in steady state equilibrium

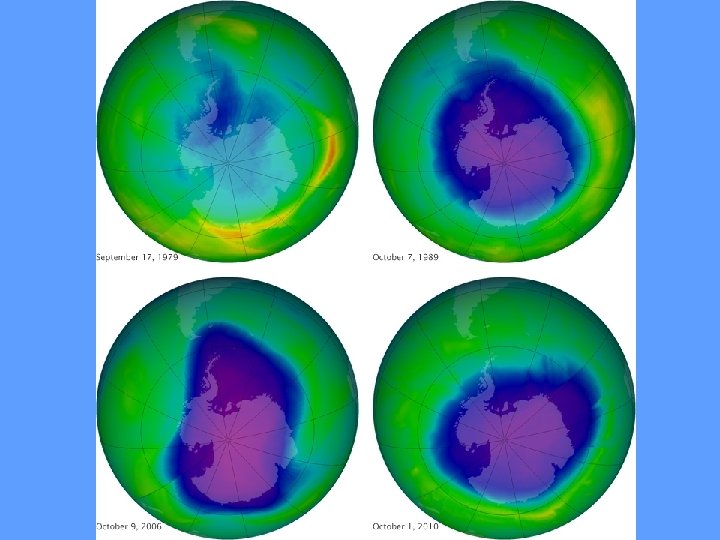
Depletion of Ozone • The natural process of ozone formation is slowing down due to gases that we are putting into the atmosphere! • Halogenated organic gases like Chlorofluorocarbons (CFCs) and molecules with chlorine, flourine and bromine, and nitrous oxide (N 2 O). • CFCs are used as aerosol propellants, gas blown plastics like Styrofoam and plastic bottle shapes, pesticides, flame retardants, and as refrigerants. • These gases cause ozone in the stratosphere to be destroyed faster than it is formed.

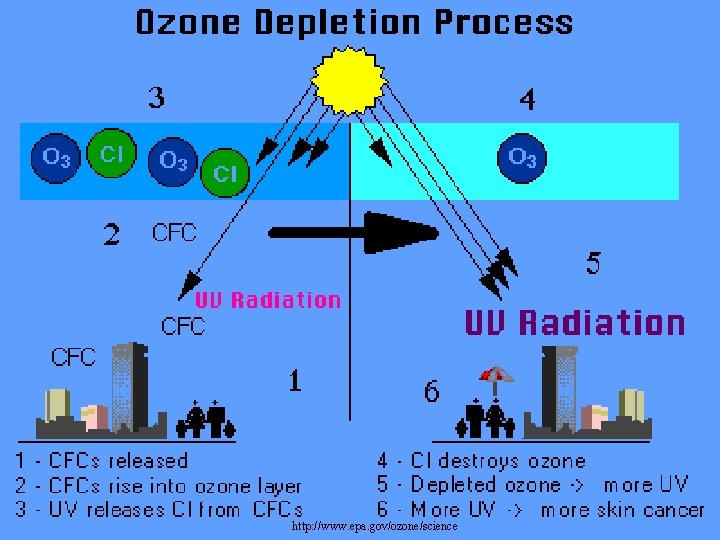
http: //www. epa. gov/ozone/science

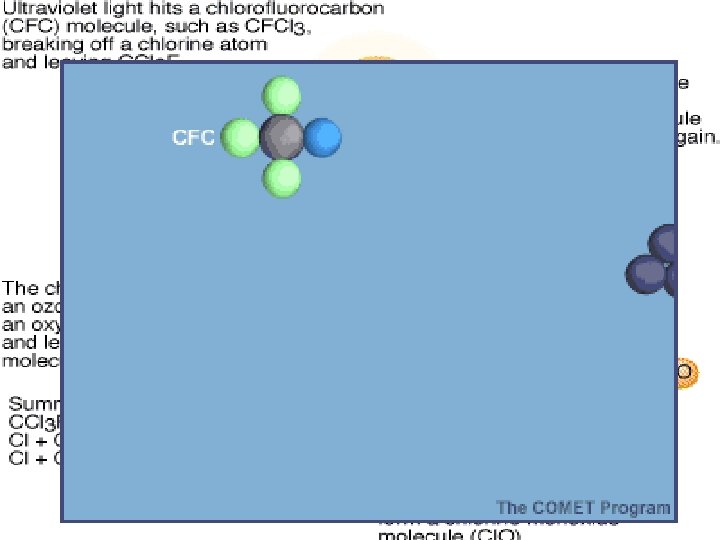
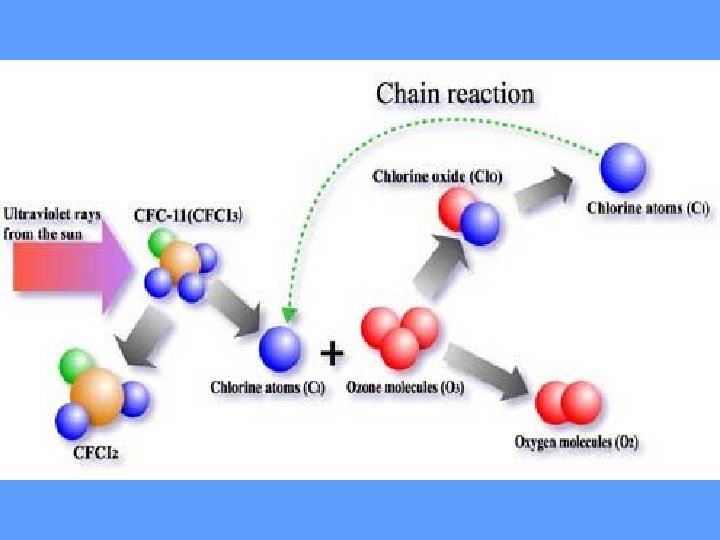
How Does Depletion Occur? • CFCs are stable in the troposphere and were thought to be heavy enough to stay in there, however, they move from troposphere into stratosphere • UV breaks off chlorine molecule (Cl) from CFC • Cl acts as a catalyst to break down ozone (O 3) – catalyst – promotes a chemical reaction without itself being used up in the reaction. – shifts equilibrium of oxygen / ozone reaction. – more O 3 destroyed than made.




Effects of UV radiation • Damage DNA, causes many problems, including cancer. • Increase in skin cancer & cataracts. • Lower crop yield • Decline in forest productivity • Reduction of surface dwelling phytoplankton, which would therefore affect who? – This would impact ocean productivity and oxygen production. • Damage & degrade materials paints & plastics


Methods of reducing the manufacture and release of ozone depleting substances 1. Use of CFC Alternatives: • Replace CFCs with: • HFCs (hydrofluorocarbons) - alternative to CFCs don’t contain chlorine. – Contain fluorine (1000 times less efficient in UV Reactions) • HCFC - replace a Cl atom with H atom which enables it to be broken down in troposphere. – Problem: if it gets to stratosphere it will deplete ozone • **The problem with most of these replacements is that they are greenhouse gases!** 2. Recycle old refrigerators to allow for the proper capture and disposal of the CFCs inside.

Role of national & world organizations in reducing the emissions of ozone depleting substances National: • EPA made amendments to 1967 clean air act and is responsible for monitoring air quality standards for hazardous substances. International: • UNEP has had a key role in providing information, and creating and evaluating international agreements, for the protection of stratospheric ozone. • An illegal market for ozone-depleting substances persists and requires consistent monitoring.

Montreal Protocol “Perhaps the single most successful international agreement to date has been the Montreal Protocol. ” -Kofi Annan, Former Secretary General of the United Nations • UNEP agreement originally signed in 1987; amended in 1990 & 1992. • The treaty provided a timetable on which the production of ozone depleting substances must be phased out and eventually eliminated. • Provided aid for developing nations to do so as well. • If international agreement is adhered to, ozone layer expected to recover by 2050. IMPACTS: Atmospheric concentration of CFCs has leveled off and/or decreased. Concentration of HFCs (an alternative) has increased substantially. Currently there are plans for a complete worldwide phase out by 2030.