Ozone and CFCs What is Ozone l Ozone






- Slides: 13

Ozone and CFCs

What is Ozone? l Ozone is an allotrope of oxygen. It’s formula is O 3. l It is naturally present in the atmosphere. l It exists both in the troposphere (lower atmosphere) and the stratosphere.

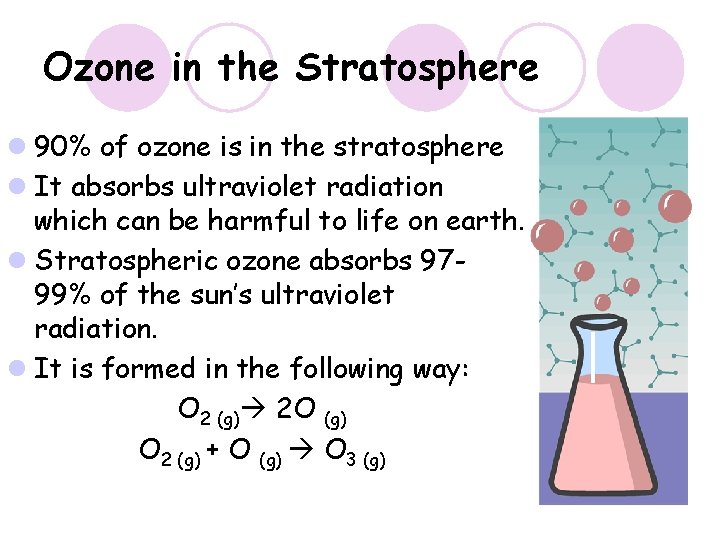
Ozone in the Stratosphere l 90% of ozone is in the stratosphere l It absorbs ultraviolet radiation which can be harmful to life on earth. l Stratospheric ozone absorbs 9799% of the sun’s ultraviolet radiation. l It is formed in the following way: O 2 (g) 2 O (g) O 2 (g) + O (g) O 3 (g)

Ozone in the Troposphere l Ozone is harmful in the troposphere. It is toxic, and harmful to health at low concentrations (0. 1 ppm) l It forms photochemical smog by reacting with exhaust gases from motor vehicles. NO 2 (g) + sunlight NO (g) + O (g) The oxygen radicals then react with O 2, forming ozone. O 2 (g) + O (g) O 3 (g)

Chlorofluorocarbons (CFCs) l Synthetic chemicals l Contain fluorine, chlorine and carbon (no hydrogen) l Have a similar structure to alkanes, with hydrogen replaced by fluorine and chlorine. l E. g. Dichlorodifluoromethane

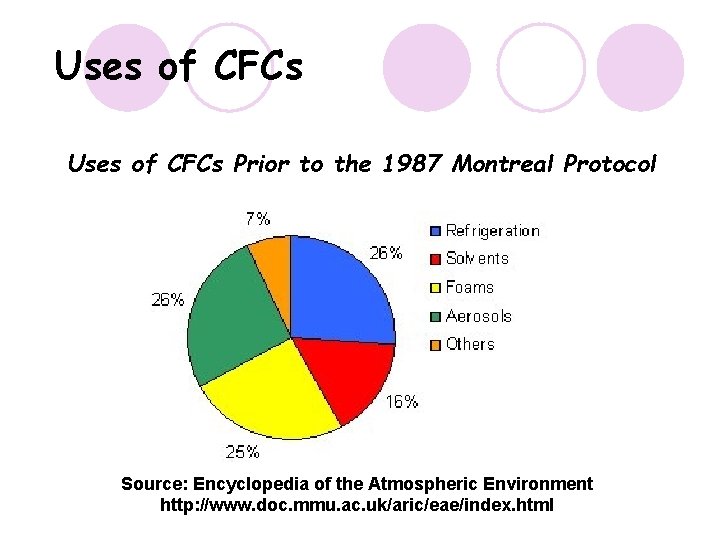
Uses of CFCs Prior to the 1987 Montreal Protocol Source: Encyclopedia of the Atmospheric Environment http: //www. doc. mmu. ac. uk/aric/eae/index. html

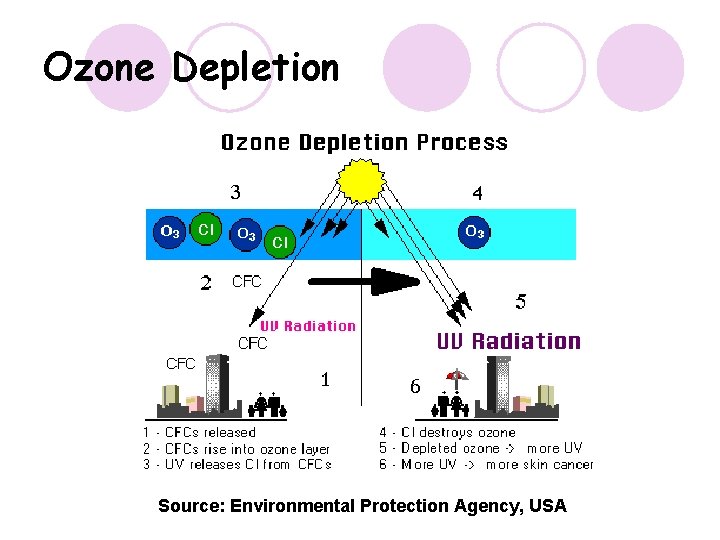
CFCs and Ozone 1 l CFCs are chemically inert and are not broken down at low altitudes. They are also insoluble in water so they are not washed from the atmosphere in rain. l They remain in the troposphere for long periods of time, and diffuse into the stratosphere. l CFCs in the stratosphere cause damage to the protective ozone layer.

CFCs and Ozone 2 l CFCs are broken down by UV radiation in the stratosphere releasing halogen atoms, such as chlorine, which can react to break up ozone molecules. CCl 3 F (g) + UV radiation CCl 2 F (g) + Cl (g) + O 3 Cl. O (g) + O 2 (g) Cl. O then reacts with oxygen radicals Cl. O (g) + O (g) O 2 (g) + Cl (g) l Since the chlorine atom is not used up in the reaction it can continue breaking up ozone molecules indefinitely. l This removes ozone from the stratosphere, meaning it cannot perform it’s purpose of absorbing harmful UV rays as effectively.

Ozone Depletion Source: Environmental Protection Agency, USA

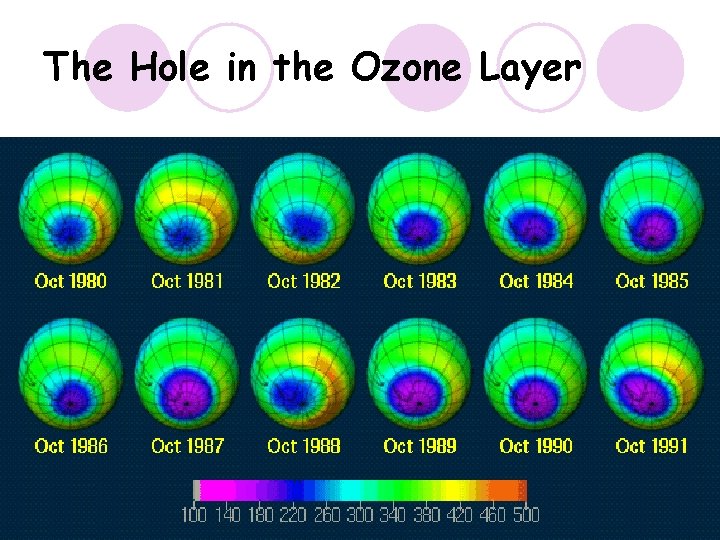
The Hole in the Ozone Layer

Effects of Ozone Depletion l Global Warming l Increase in levels of skin cancer, cataracts, immune system impairment. l Impact on crops and agriculture l Impact on marine food cycle

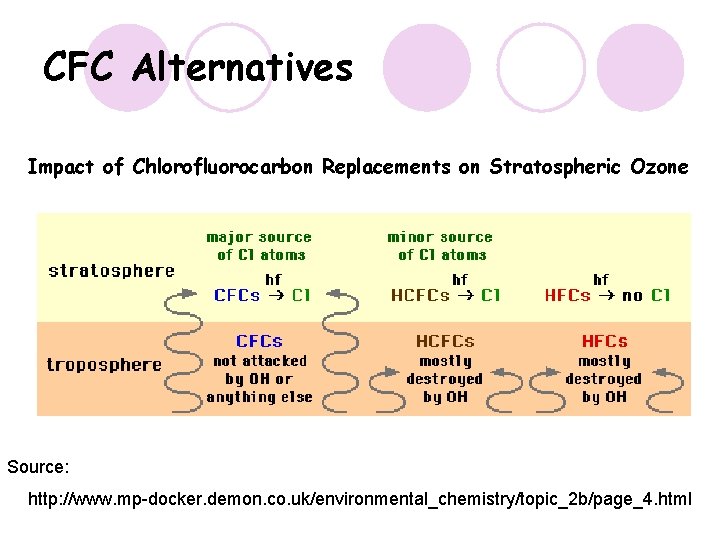
CFC Alternatives Impact of Chlorofluorocarbon Replacements on Stratospheric Ozone Source: http: //www. mp-docker. demon. co. uk/environmental_chemistry/topic_2 b/page_4. html

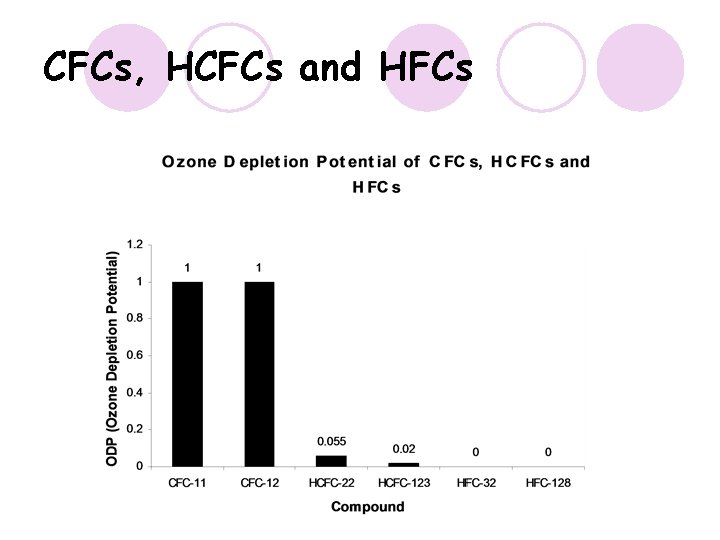
CFCs, HCFCs and HFCs