Oxygen The Good the Bad the Ugly The

Oxygen – The Good, the Bad, & the Ugly The Good - Biological Oxidant O 2 The Bad - Damage to Biological Systems by Partially Reduced Oxygen The Ugly - Oxidation of Pollutants

Oxygen - The Bad • Partially reduced oxygen species in metabolism • Reactive Oxygen Species • Physiologic Damage to Proteins, Fatty Acids, DNA • ROS theory of aging • Antioxidants - Moderation of Damage by ROS 10/29/2020 2
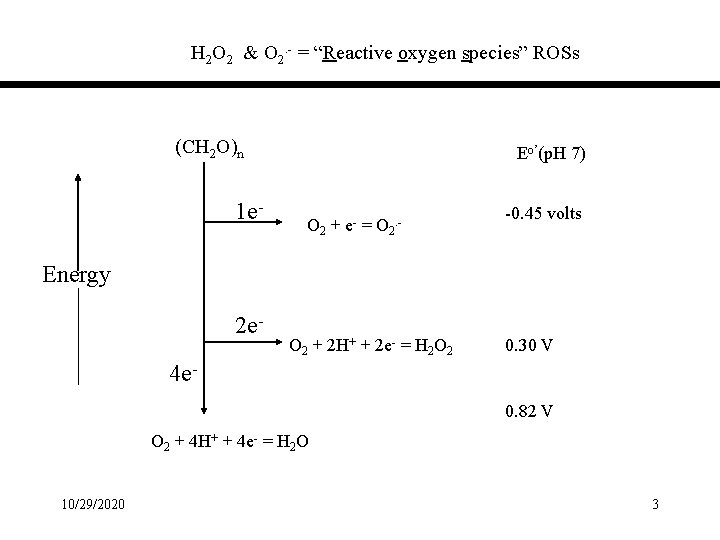
H 2 O 2 & O 2. - = “Reactive oxygen species” ROSs (CH 2 O)n 1 e- Eo’(p. H 7) O 2 + e- = O 2. - -0. 45 volts Energy 2 e- O 2 + 2 H+ + 2 e- = H 2 O 2 0. 30 V 4 e 0. 82 V O 2 + 4 H+ + 4 e- = H 2 O 10/29/2020 3

Dangers of Reactive Oxygen Species *Hydrogen peroxide * Fenton Reaction H 2 O 2 + Fe. II = Fe. III + HO- + HO. + e- = HO- Eo = 1. 8 volts *Superoxide ion * Disproportionation to H 2 O O 2. - + 2 H+ = H 2 O 2 + O 2 * Reducing agent for Fenton rxn. O 2. - + Fe. III(L)n = Fe. II(L)n + O 2 * Reduces Fe 3+(insoluble) to Fe 2+ (soluble) 10/29/2020 4
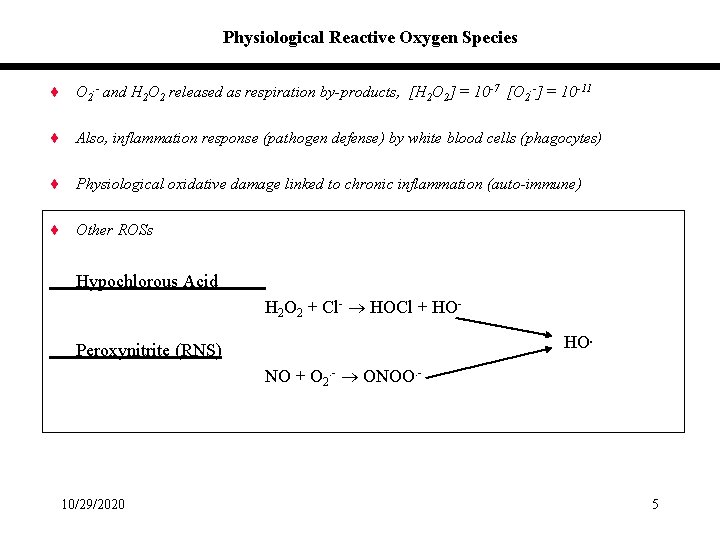
Physiological Reactive Oxygen Species ¨ O 2. - and H 2 O 2 released as respiration by-products, [H 2 O 2] = 10 -7 [O 2. -] = 10 -11 ¨ Also, inflammation response (pathogen defense) by white blood cells (phagocytes) ¨ Physiological oxidative damage linked to chronic inflammation (auto-immune) ¨ Other ROSs Hypochlorous Acid H 2 O 2 + Cl- HOCl + HOHO. Peroxynitrite (RNS) NO + O 2. - ONOO. - 10/29/2020 5

Hydroxyl Radical Damage to Physiology Results in. . . ¨ Denaturation of lens proteins - cataracts ¨ DNA strand breakage - damage to genes - cancers - mitochondrial dysfunction - aging ¨ Fatty acid cross linking - circulatory diseases ¨ Damage to nervous system - Parkinson’s - Alzheimer’s diseases 10/29/2020 6
Moderation of Physiological ROSs Radical scavenging 10/29/2020 7
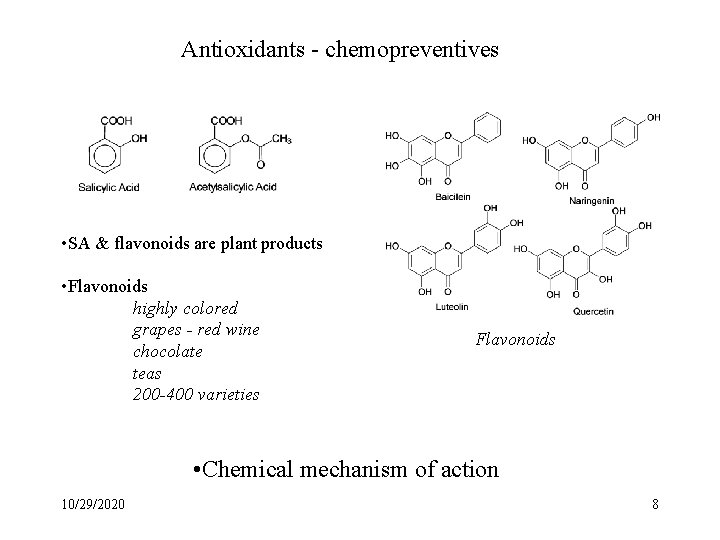
Antioxidants - chemopreventives • SA & flavonoids are plant products • Flavonoids highly colored grapes - red wine chocolate teas 200 -400 varieties Flavonoids • Chemical mechanism of action 10/29/2020 8

Antioxidant Action(s) of Aspirin & Flavonoids 1. Acts a chemopreventive agent flavonoids and aspirin (and aspirin-like drugs) prevent many diseases associated with oxidative damage. Free Radicals in Biology and Medicine 9, (1990) 299. 2. Acetylation mechanism cannot explain anti-inflammatory action salicylic acid = aspirin many flavonoids have anti-inflammatory action * Hypothesis for molecular base action of antioxidant action Chelation of pro-oxidant metal ions………. . . Iron 10/29/2020 9

Metal Chelation Hypothesis * Many transition metals are pro-oxidants Fe Mn Cu V Mo Co per 75 kg human 4. 5 g 1 g 100 mg 15 mg 10 mg 1. 2 mg Biocoordination Chemistry, David E. Fenton Oxford University Press, 1995. * Physiological Systems must control pro-oxidant metals Storage and Transport of Fe. - Redox Deactivation Animals (Humans) - Ferritin, Transferrin Plants & Bacteria - Siderophores 10/29/2020 10
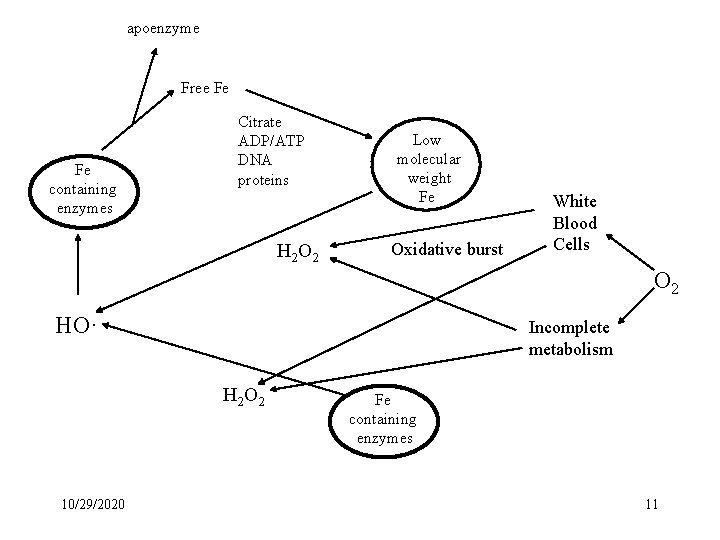
apoenzyme Free Fe Fe containing enzymes Citrate ADP/ATP DNA proteins H 2 O 2 Low molecular weight Fe Oxidative burst White Blood Cells O 2 HO· Incomplete metabolism H 2 O 2 10/29/2020 Fe containing enzymes 11
![Cyclic Voltammetry of Fe. II/III [EDTA] in the Absence and Presence of H 2 Cyclic Voltammetry of Fe. II/III [EDTA] in the Absence and Presence of H 2](http://slidetodoc.com/presentation_image/213314aea82ec6230e38db87d7f2a885/image-12.jpg)
Cyclic Voltammetry of Fe. II/III [EDTA] in the Absence and Presence of H 2 O 2 B 1. 0 u. A A 0. 4 u. A - 0. 7 Potential vs. Ag/Ag. Cl Electrode: Fe. III(EDTA) + e = Fe. II(EDTA) E = - 0. 090 volts SHE Solution: Fe. II(EDTA) + H 2 O 2 = Fe. III(EDTA) + HO- + HO. 10/29/2020 12

Figure 2. 3. 2) Moderation of the Fenton reaction characteristic of 0. 25 m. M Fe. IIIATP, 8. 6 m. M H 2 O 2 in 0. 0625 M HEPES buffer p. H 7. 4 by quercetin. The cyclic voltammograms (left) were taken at 20 m. V/s for the various quercetin/iron ratios. The quercetin/Fe. IIIATP ratios (I-VII) are indicated on the x-axis of the righthand graph. The catalytic wave at -0. 400 volts are plotted versus the quercetin/iron ratio in the bottom graph. The current of point VII at – 0. 400 volts is the same as the background current (0. 00 m. M H 2 O 2) 10/29/2020 13
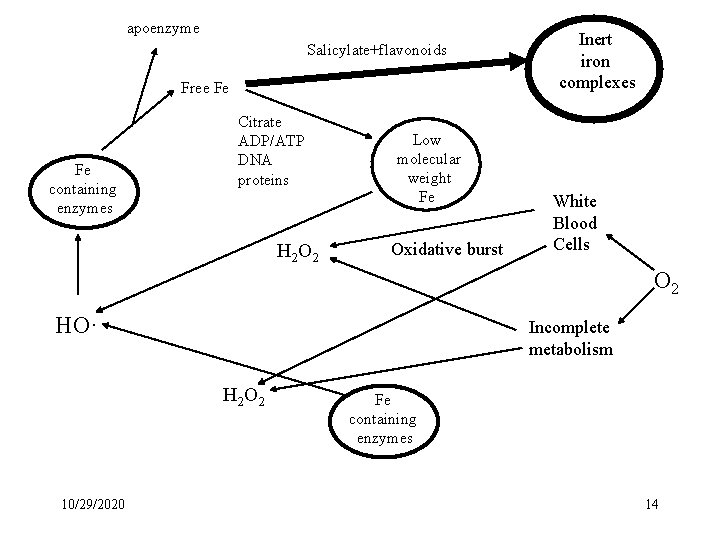
apoenzyme Salicylate+flavonoids Free Fe Fe containing enzymes Citrate ADP/ATP DNA proteins H 2 O 2 Low molecular weight Fe Oxidative burst Inert iron complexes White Blood Cells O 2 HO· Incomplete metabolism H 2 O 2 10/29/2020 Fe containing enzymes 14
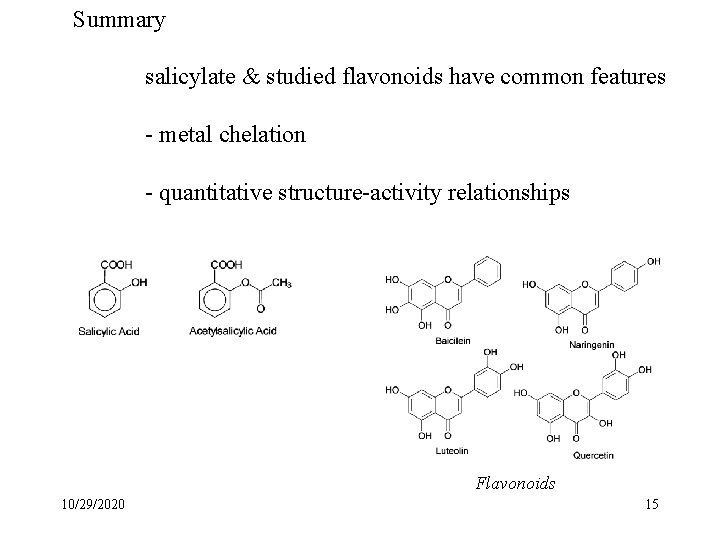
Summary salicylate & studied flavonoids have common features - metal chelation - quantitative structure-activity relationships Flavonoids 10/29/2020 15
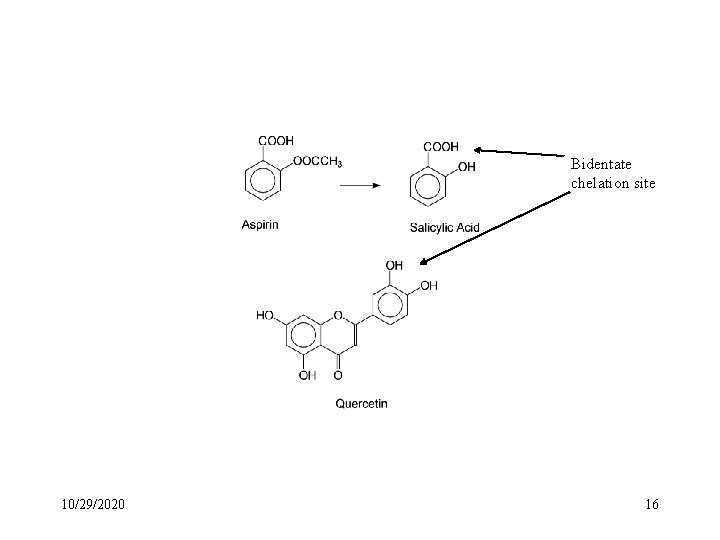
Bidentate chelation site 10/29/2020 16

Quantitative Structure-Activity Relationships (QSAR) for Salicylates and Derivatives (Anti-inflammatory action) Rule 1. Substitution on either the carboxyl or the phenolic hydroxyl groups affect activity. Rule 2. Placing the phenolic hydroxyl group meta or para to the carboxyl group abolishes activity. Rule 3. Substitution of halogen atoms on the aromatic ring enhances potency. Rule 4. Substitution of aromatic rings meta to the carboxyl and para to the phenolic hydroxyl groups increases anti-inflammatory activity. Rules 1 and 2 may imply metal chelation as an important action. 10/29/2020 17
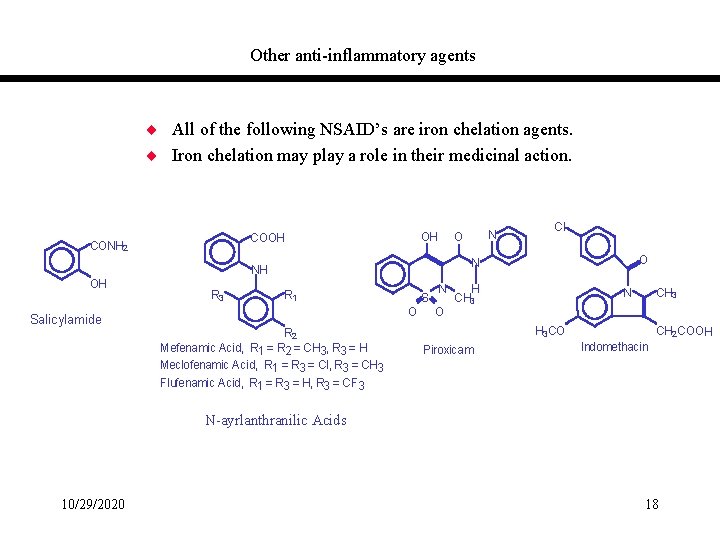
Other anti-inflammatory agents ¨ All of the following NSAID’s are iron chelation agents. ¨ Iron chelation may play a role in their medicinal action. OH CONH 2 Salicylamide R 3 Cl O N NH OH N O R 1 S O R 2 Mefenamic Acid, R 1 = R 2 = CH 3, R 3 = H Meclofenamic Acid, R 1 = R 3 = Cl, R 3 = CH 3 Flufenamic Acid, R 1 = R 3 = H, R 3 = CF 3 N O H CH 3 N CH 3 H 3 CO Piroxicam CH 2 COOH Indomethacin N-ayrlanthranilic Acids 10/29/2020 18
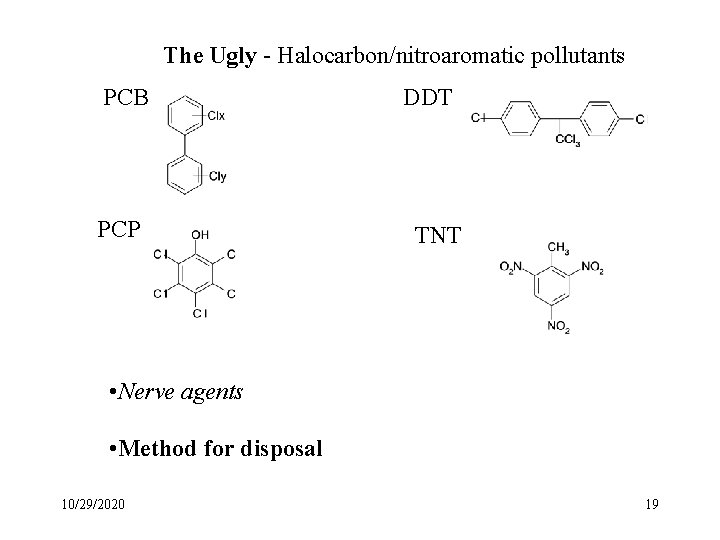
The Ugly - Halocarbon/nitroaromatic pollutants PCB PCP DDT TNT • Nerve agents • Method for disposal 10/29/2020 19
Present method of disposal - Incineration > 1200 0 C - Cx. Hy. Clz + O 2 = CO 2 + H 2 O + HCl (unbalanced) Problems: - Costs (incineration and transport) - PCDFs PCDs - NIMBY 10/29/2020 20

Preferences • Inexpensive • Safe • Transportable • Environmentally Benign • Acceptable to NIMBY 10/29/2020 21

Ideal Characteristics for Pollutant Destruction Reactions -Complete mineralization, i. e. CO 2 - Mild reaction conditions, i. e. room temperature & pressure - Inexpensive reagents Other investigators: -UV radiation - Ti. O 2 -Sonochemical -H 2 O 2 oxidation -Electrochemical oxidation - reductions What about air? 10/29/2020 22

O 2 is kinetically stable -Requires “activation”, partially reduced oxygen. -Scheme: O 2(air) + 2 H+(aq) + 2 e- = H 2 O 2 Chemical reducing agent -Zero-valent iron, Fe(0) Inexpensive Spent agent, Fe 2+/Fe 3+ environmentally benign. Chelated forms, capable oxidizing agents 10/29/2020 23
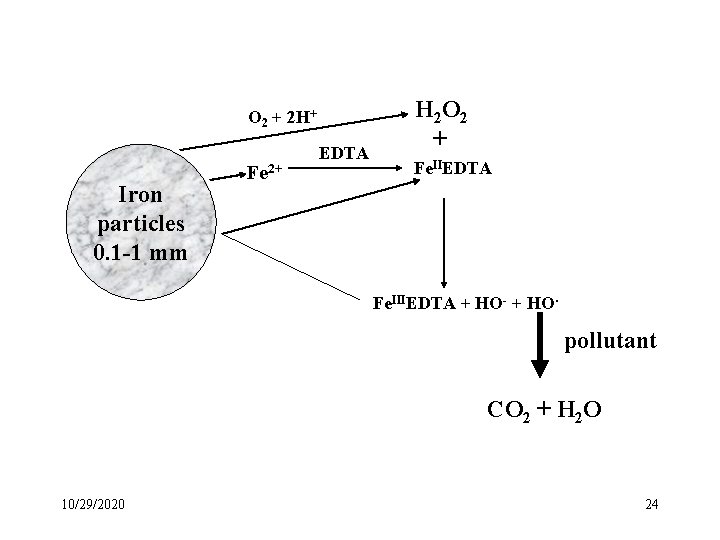
O 2 + 2 H+ Iron particles 0. 1 -1 mm Fe 2+ EDTA H 2 O 2 + Fe. IIEDTA Fe. IIIEDTA + HO- + HO. pollutant CO 2 + H 2 O 10/29/2020 24

Summary - 80% CO 2 yields for chlorinated phenols, complete destruction of PCB, PCP, DDT, nitrophenols - First known example of room temperature & pressure activation of O 2 to a form that can oxidized organics to CO 2. - Inexpensive reagents, air, iron particles, water, EDTA. -PCDFs, PCDs not detected (to the D. L. GC-ECD, 1 ppb) -Unspecialized reactors, transportable technology. 10/29/2020 25

Other Interests……. . . Chemical Mechanisms of O 2 Evolution from Plants Chemical Analyses of PCB, PCP, & DDT Electrochemical Detectors Lead-Acid Battery 10/29/2020 26
- Slides: 26