Oxyanions Hydrates Acids and Bases Lesson Outline Naming

Oxyanions, Hydrates, Acids and Bases

Lesson Outline Naming Hydrates Naming Oxyanions Naming Acids and Bases

Hydrates A number of ionic compounds called hydrates produce water when they decompose upon heating. The number of water molecules is added in the formula copper (II) sulfate pentahydrate Cu. SO 4 5 H 2 O. Prefixes are used to indicate the number of water molecules in a hydrated compound Hydrates Worksheet

Oxyanions Elements that can form more than one oxyanion (polyatomic ions that contain oxygen), each containing a different number of oxygen atoms. Cl. O 4 - Cl. O 3 - Cl. O 2 - Cl. O- In a family of oxyanions, the charge remains the same; only the number of oxygen atom varies

Oxyanions the most common of an element’s oxyanions has a name with the form (root)ate. Relationship General Name Example formula One more oxygen atom than (root)ate perchlorate Cl. O 4 - Most Common (root)ate chlorate Cl. O 3 - One less oxygen than (root)ate (root)ite chlorite Cl. O 2 - Two less oxygen than (root)ate hypo(root)ite hypochlorite Cl. O- Oxyanions Worksheet

Compounds Containing Hydrogen When Hydrogen is the least electronegative atom it is known as an acid HCl Hydrochloric Acid When Hydrogen is not the least electronegative atom it acts as an anion Na. H Sodium Hydride

Acids Steps to Naming Acids 1. temporarily name it as an ionic compound HCl hydrogen chloride. 2. use the rules below to name it properly. hydrochloric acid Compound Name as an acid Hydrogen ------ide ex. Hydrogen chloride becomes Hydro----ic acid ex. Hydrochloric acid Hydrogen -----ate ex. Hydrogen nitrate becomes -----ic acid nitric acid Hydrogen ----ite ex. Hydrogen nitrite becomes ------ous acid nitrous acid

Organic Acids containing carbon and hydrogen (usually the H is at the end of the formula) CH 3 COOH acetic acid

Bases compounds that form hydroxide ions(OH) when dissolved in water Bases are simply named like ionic compounds Na. OH Sodium Hydroxide Acids Worksheet
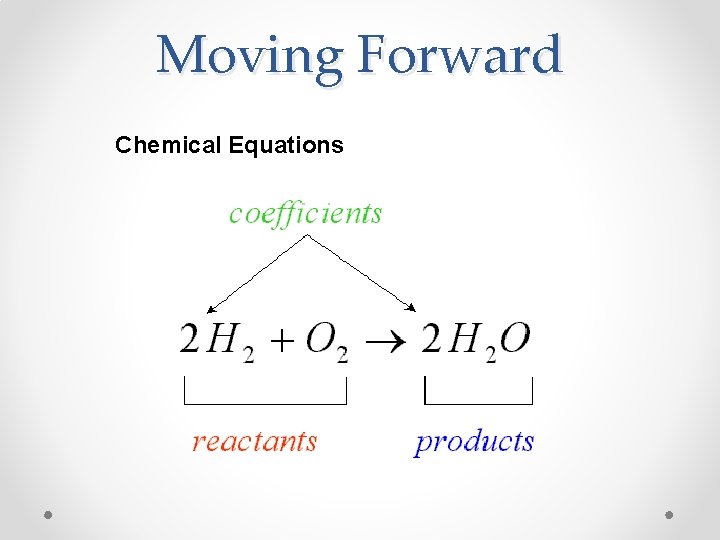
Moving Forward Chemical Equations
- Slides: 10