OxidationReduction Reactions n Carbonate reactions are acidbase reactions

Oxidation-Reduction Reactions n Carbonate reactions are acid-base reactions: n n Transfer of protons – H+ Other acid-base systems are similar: Sulfuric acid - H 2 SO 4 n Phosphoric acid - H 2 PO 3 n Nitric Acid HNO 3 n

Oxidation-Reduction Reactions n n Redox reactions are analogous, but are transfer of electrons rather than protons Very important class of reactions Solutes may have variable charges n Charge depends on number of valence electrons (valence state) n Valence state controls speciation of solutes & ionic strength, activity coefficients… n

n Examples of primary valence states of some elements C = +4 or -4 n S +6 or -2 n N +5 or +3, also +4, +2 n Fe +3 or +2 n Mn +3 or +2, also +7, +6, +4 n

n Minor elements also have various valence states V, Cr, As, Mo, V, Se, Sb, W, Cu… n All can be hazardous, but depend on complexing, which depends on valence n

n Valence state very important for mobility, as well as absorption and thus toxicity n Fe 3+ (oxidized) is highly insoluble n n Precipitate as Fe-oxide minerals (amorphous Feoxides, magnetite, hematite, goethite, lepidocrocite, limonite) Fe 2+ (reduced) much more soluble – most Fe in solution is +2 valence n Common precipitates are Fe-sulfides (pyrite, marcasite)

Assignment of oxidation state n Valence state of oxygen is always -2 except for peroxides, where it is -1, e. g. : n n H 2 O 2 and Na 2 O 2 Valence state of hydrogen is +1 in all compounds except when bonded with metals where it is -1, e. g. : Na. H n Na. BH 4 n Li. Al. H 4 n

n n Valence state of all other elements are selected to make the compound neutral Certain elements almost always have the same valence state Alkali metals = +1 (left most column) n Alkaline earths = +2 (second column from left) n Halogens = -1 (2 nd column from right) n

Examples n What are the oxidation states of N in NO 3 and NO 2 -? n n 3 O 2 - + Nx = NO 3 N = +5 6 - + x = -1 2 O 2 - + Nx = NO 2 - 4 - + x = -1 N = +3

n What are the oxidation states of S in H 2 S and SO 42 -? n n 2 H+ + Sx = H 2 S S = -2 2+ + x = 0 4 O 2 - + Sx = SO 42 - 8 - + x = -2 S = +6

Oxidation Reactions n Oxidation can be thought of as involving molecular oxygen n 3 Fe 2 O 3 (hematite) All as Fe 3+ 2 Fe 3 O 4 + 1/2 O 2 (magnetite) One as Fe 2+ + two as Fe 3+ High O/Fe ratio Oxidized Lower O/Fe ratio Reduced In this case, the generation of molecular oxygen controls the charge imbalance

n Also possible to write these reactions in terms of electrons: 3 Fe 2 O 3 + 2 H+ + 2 e 2 Fe 3 O 4+ H 2 O n LEO – lose electron oxidation – the Fe 3+ is oxidized n GER – gain electron reduction – the Fe 2+ is reduced n OIL – oxidation is loss n RIG – reduction is gain n

n Generally easiest to consider reactions as transfer of electrons n Many redox reaction do not involve molecular oxygen

n Problem is that free electrons are not really defined Reactions that consume “free electrons” represent only half of the reaction n A complementary reaction required to produce a “free electron” n Concept is two “half reactions” n The half reactions simultaneously create and consume electrons, so typically not expressed in the overall reaction n
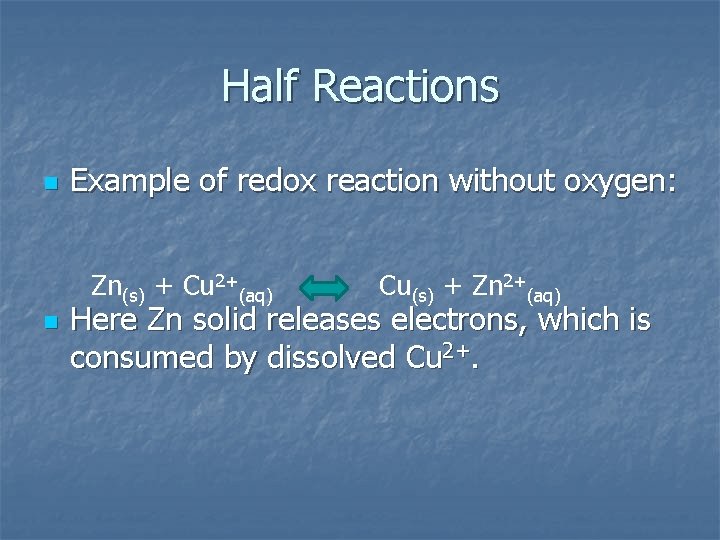
Half Reactions n Example of redox reaction without oxygen: Zn(s) + Cu 2+(aq) n Cu(s) + Zn 2+(aq) Here Zn solid releases electrons, which is consumed by dissolved Cu 2+.
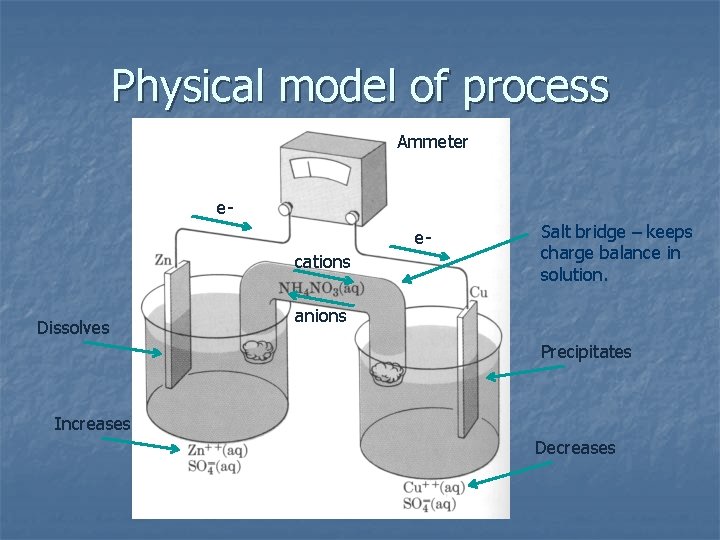
Physical model of process Ammeter eecations Dissolves Salt bridge – keeps charge balance in solution. anions Precipitates Increases Decreases

n Ammeter shows flow of electrons from Zn to Cu: Zn rod dissolves – Zn 2+ increases n Cu rod precipitates – Cu 2+ decreases n

n At the rod, the reactions are: Zn = Zn 2+(aq) + 2 e 2 e- + Cu 2+(aq) = Cu Zn + Cu 2+(aq) = Zn 2+(aq) + Cu Half reactions

n Benefits of using half reactions: Half reactions help balance redox reactions n Used to create framework to compare strengths of oxidizing and reducing agents n

Rules for writing and balancing half reactions 1. 2. 3. 4. Identify species being oxidized and reduced Write separate half reactions for oxidation and reduction Balance reactions using (1) atoms and (2) electrical charge by adding e- or H+ Combine half reactions to form net oxidation-reduction reactions

n Consider reaction H 2 O 2 + I - n I 2 + H 2 O First: ID oxidized and reduced species: Iodine is being oxidized from -1 to 0 charge n Oxygen in peroxide is being reduced to water n IH 2 O 2 I 2 H 2 O

n Second: write oxidation and reduction half reaction (not necessarily balanced): I- I 2 H 2 O

n Third: balance elements (here is oxidation half reaction): 2 I- n I 2 And charge: 2 I- I 2 + 2 e-

n Balance reduction half reaction: n First balance oxygen, then add H+ to balance hydrogen, then add electrons for electrical neutrality: H 2 O 2 2 H 2 O 2 H+ + H 2 O 2 2 H 2 O 2 e- + 2 H+ + H 2 O 2 2 H 2 O

n Fourth: combine two half reactions to get net reactions: 2 I 2 e- + 2 H+ + H 2 O 2 2 H+ + 2 I- + H 2 O 2 I 2 + 2 e 2 H 2 O + I 2 Flow of electrons – Oxygen is electron acceptor, reduced; I- is electron donor, oxidized
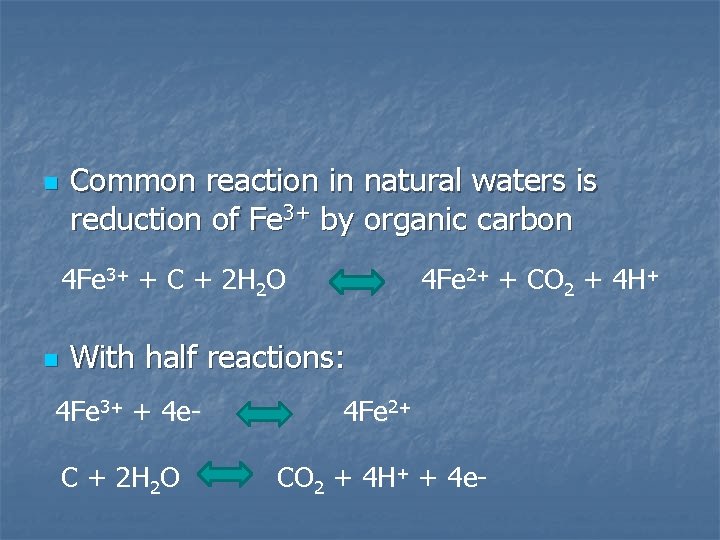
n Common reaction in natural waters is reduction of Fe 3+ by organic carbon 4 Fe 3+ + C + 2 H 2 O n 4 Fe 2+ + CO 2 + 4 H+ With half reactions: 4 Fe 3+ + 4 e- 4 Fe 2+ C + 2 H 2 O CO 2 + 4 H+ + 4 e-

n From thermodynamic conventions, its impossible to consider a single half reaction n There is no thermodynamic data for e- Practically, half reactions are defined relative to a standard The standard is the “Standard Hydrogen Electrode (SHE)”

SHE n n Platinum electrode in solution containing H 2 gas at P = 1 Atm. Assign arbitrary values to quantities that can’t be measured Difference in electrical potential between metal electrode and solution is zero n DGfº of H+ = 0 n DGfº of e- = 0 n n Similar to assumption that DGfºelements = 0
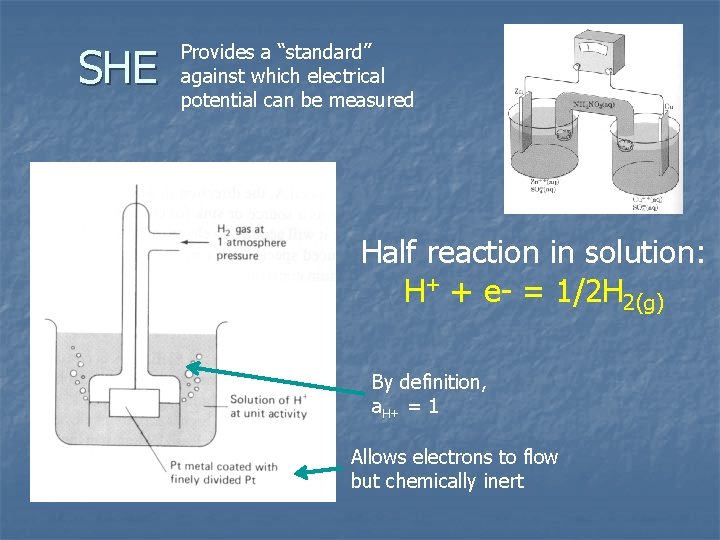
SHE Provides a “standard” against which electrical potential can be measured Half reaction in solution: H+ + e- = 1/2 H 2(g) By definition, a. H+ = 1 Allows electrons to flow but chemically inert
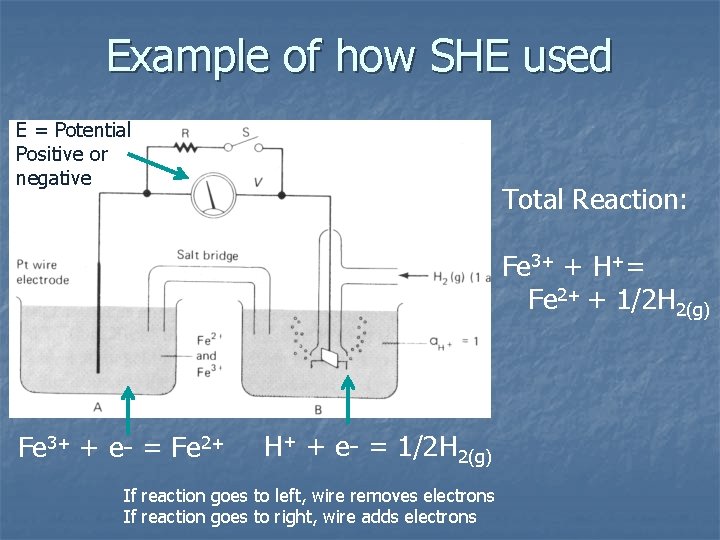
Example of how SHE used E = Potential Positive or negative Total Reaction: Fe 3+ + H+= Fe 2+ + 1/2 H 2(g) Fe 3+ + e- = Fe 2+ H+ + e- = 1/2 H 2(g) If reaction goes to left, wire removes electrons If reaction goes to right, wire adds electrons

n n In cell A, platinum wire is inert – transfers electrons to or from solution only. If wire has no source of electrons n n Define the potential as “activity of electrons” = aen n Pt wire develops an electrical potential – “tendency” for electrons to enter or leave solution Not a true activity, really a “tendency” Define pe = -logae-, similar to p. H

n In Cell A solution, Fe is both oxidized and reduced n n Fe 2+ and Fe 3+ Reaction is: Fe 3+ + e- = Fe 2+ If reaction goes to left, Fe 2+ gives up en If reaction goes to right, Fe 3+ acquires en If no source or sink of e-, (switch open), volt meter measures the potential (tendency) n

n Since we have a reaction Fe 3+ + e- = Fe 2+ n can write an equilibrium constant Keq = a. Fe 2+ a. Fe 3+ ae-

n Rearranged: ae-= Keq-1 n n a. Fe 2+ a. Fe 3+ ae- is proportional to the ratio of activity of the reduced species to activity of oxidized species ae- is electrical potential (in volts) caused by ratio of reduced to oxidized species

n Consider half cell B: H+ + e- = 1/2 H 2(g) n n Direction of reaction depends on tendency for wire to gain or lose electrons Equilibrium constant KSHE = PH 21/2 a. H+ ae-

n Switch closed – electrons flow from one half cell to the other Electrons flow from the side with the highest activity of electrons to side with lowest activities Flow of electrons n Overall reaction: n Fe 3+ + 1/2 H 2(g) =Fe 2+ + H+ n Direction of reaction depends on which half cell has highest activity of electrons

n Switch open: No longer transfer of electrons n Now simply potential (E) generated at Pt wire n By convention, potential of SHE (ESHE) = O n Potential called Eh, i. e. E (electromotive force) measured relative to SHE (thus the “h”) n Eh > or < O depends on whether ae- is > or < that of SHE n

n Convention Eh > 0 if ae- of the half cell < SHE n I. e. if electrons flow from the SHE to the fluid n For thermodynamics: Fe 3+ + 1/2 H 2(g) =Fe 2+ + H+ n Is equivalent to: n Fe 3+ + e- =Fe 2+

n Expressions for activities of electrons: Eh or pe n pe = [F/(2. 303 RT)]*Eh n @ 25ºC, pe = 16. 9 Eh; Eh = 0. 059 pe n n n F = Faraday’s constant = 96, 485 coul/mol Coulomb = charge /electron = quantity of electricity transferred by 1 Amp in 1 second.
- Slides: 38