Oxidation Reduction Regents Review Zns Pb 2 aq

Oxidation Reduction Regents Review
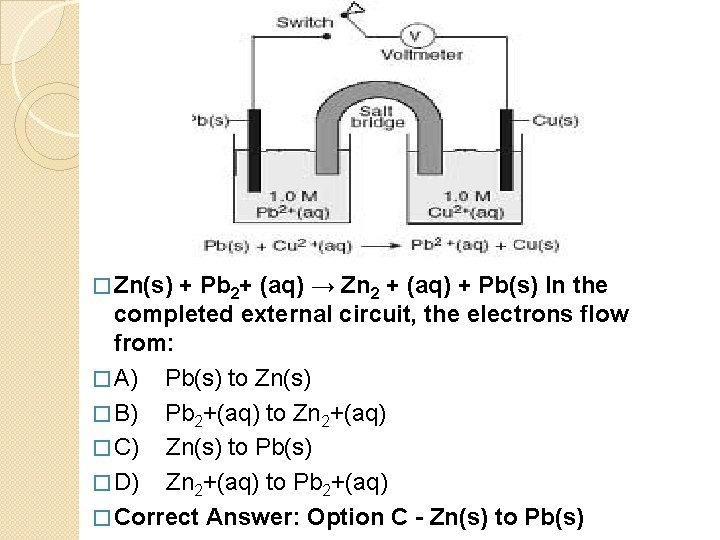
� Zn(s) + Pb 2+ (aq) → Zn 2 + (aq) + Pb(s) In the completed external circuit, the electrons flow from: � A) Pb(s) to Zn(s) � B) Pb 2+(aq) to Zn 2+(aq) � C) Zn(s) to Pb(s) � D) Zn 2+(aq) to Pb 2+(aq) � Correct Answer: Option C - Zn(s) to Pb(s)
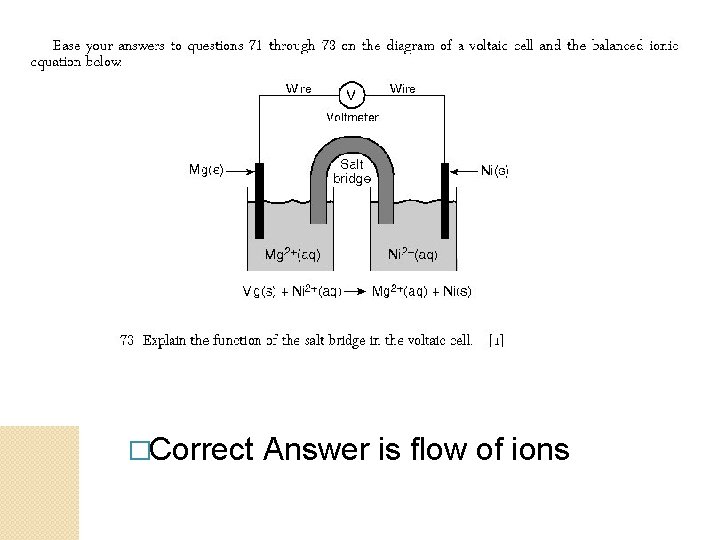
�Correct Answer is flow of ions
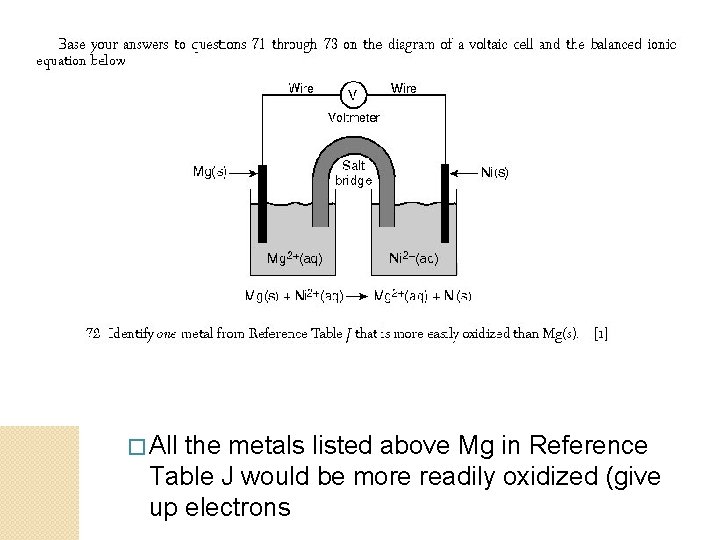
� All the metals listed above Mg in Reference Table J would be more readily oxidized (give up electrons

�Correct answer: (2) Zn (s) ® Zn 2+ (aq) + 2 e-

�Correct Answer: (3) at the cathode in an electrolytic cell and the anode in a voltaic cell
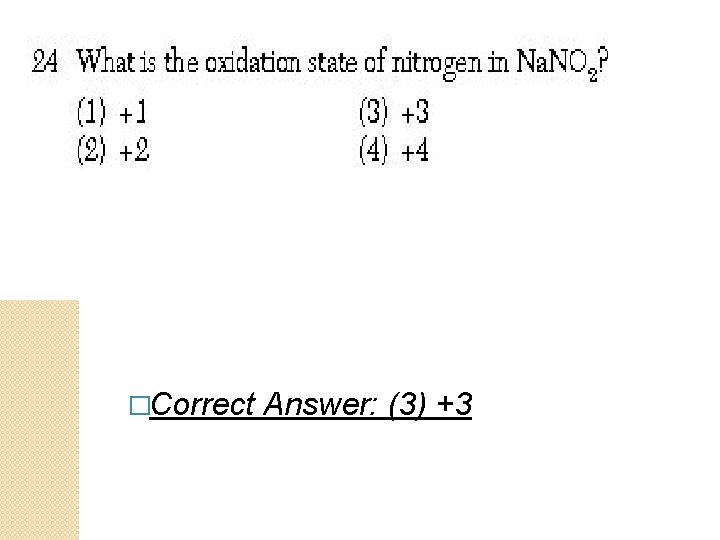
�Correct Answer: (3) +3

� Correct answer: In a voltaic cell, the reaction is spontaneous. In an electrolytic cell, it is nonspontaneous.

� Correct answer: Al+3 + 3 e- → Al

� (1) is the correct answer. Reduction means the gain of an electron, which removes the positive charge from potassium (because an electron is negatively charged).

� (3) the total number of electrons lost equals the number of electrons gained.

Given the redox reaction: Ni + Sn 4+ → Ni 2+ + Sn 2+ Which species has been oxidized? �Ni �Sn 4+ �Ni 2+ �Sn 2+ �Correct Answer is Ni

In the reaction: 2 H 2(g) + O 2(g) → 2 H 2 O(g), the oxidizing agent is �H 2 �O 2 �H− �O 2− �Correct answer is O 2
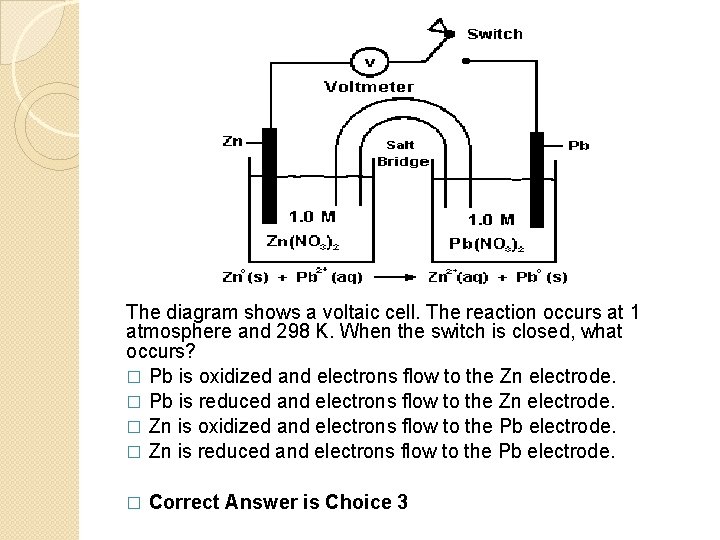
The diagram shows a voltaic cell. The reaction occurs at 1 atmosphere and 298 K. When the switch is closed, what occurs? � Pb is oxidized and electrons flow to the Zn electrode. � Pb is reduced and electrons flow to the Zn electrode. � Zn is oxidized and electrons flow to the Pb electrode. � Zn is reduced and electrons flow to the Pb electrode. � Correct Answer is Choice 3

In the reaction: Pb + 2 Ag+ → Pb 2+ + 2 Ag, the Ag+ is �reduced, and the oxidation number changes from +1 to 0 �reduced, and the oxidation number changes from +2 to 0 �oxidized, and the oxidation number changes from 0 to +1 �oxidized, and the oxidation number changes from +1 to 0 �Correct Answer is Choice 1

The diagram represents the electroplating of a metal fork with Ag(s). Which part of the electroplating is represented by the fork? � the anode, which is the negative electrode � the cathode, which is the negative electrode � the anode, which is the positive electrode � the cathode, which is the positive electrode � Correct Answer is Choice 2
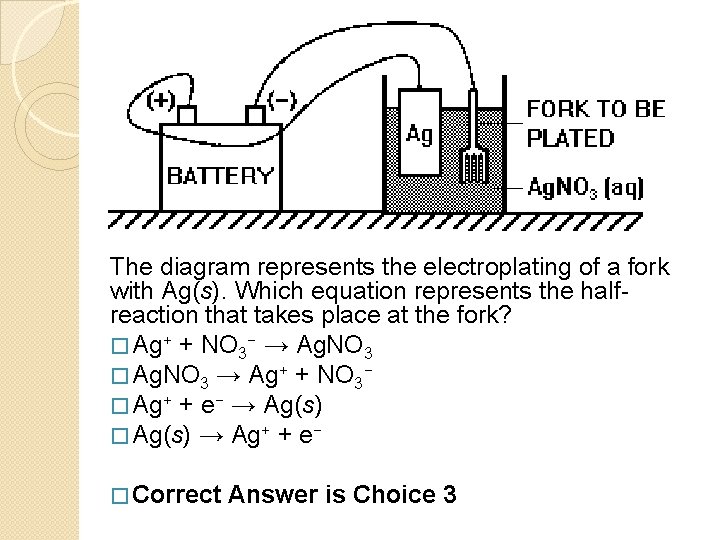
The diagram represents the electroplating of a fork with Ag(s). Which equation represents the halfreaction that takes place at the fork? � Ag+ + NO 3− → Ag. NO 3 � Ag. NO 3 → Ag+ + NO 3− � Ag+ + e− → Ag(s) � Ag(s) → Ag+ + e− � Correct Answer is Choice 3

What is the oxidation number of iodine in Na. IO 3? �+1 �+5 �+3 �+6 �Correct Answer is +5

In the reaction: 2 Mg + O 2 → 2 Mg. O, the magnesium is the �oxidizing agent and is reduced �oxidizing agent and is oxidized �reducing agent and is reduced �reducing agent and is oxidized �Correct Answer is Choice 4
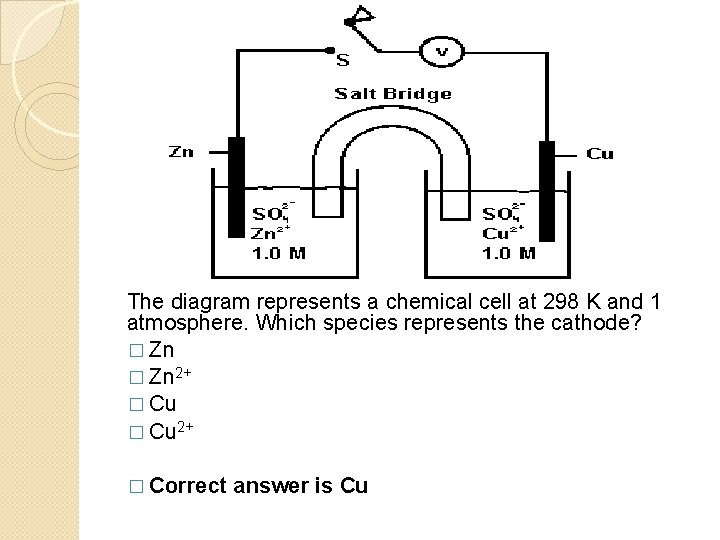
The diagram represents a chemical cell at 298 K and 1 atmosphere. Which species represents the cathode? � Zn 2+ � Cu 2+ � Correct answer is Cu
- Slides: 20