Oxidation of Ni30Cr according to the Wagner Model












- Slides: 19

Oxidation of Ni-30%Cr according to the Wagner Model Thibaut DUBÉDAT tdubedat@messel. emse. fr Tutor : Krzysztof WOLSKI 1

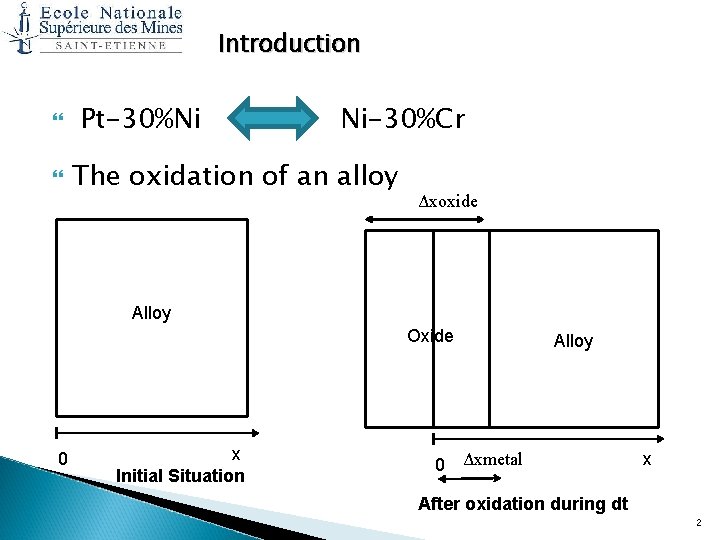
Introduction Pt-30%Ni Ni-30%Cr The oxidation of an alloy Δxoxide Alloy Oxide 0 x Initial Situation 0 Alloy Δxmetal x After oxidation during dt 2

Plan I) Presentation of the Wagner model described for Ni-Pt II) Analysis of the results obtained by the Wagner model on Ni-30%Pt III) My experimental study on Ni-30%Cr Conclusion 3

What is the Wagner model ? • Theoretical analysis of the diffusion processes. Published in 1952, under the title : Theoretical Analysis of the Diffusion Processes Determining the Oxidation Rate of Alloys, by Carl Wagner. C. Wagner, “Theoretical Analysis of the Diffusion Process Determining the Oxidation Rate of Alloys”, Journal of the Electrochemical Society, 99 [10] (1952) 369 -380. 4

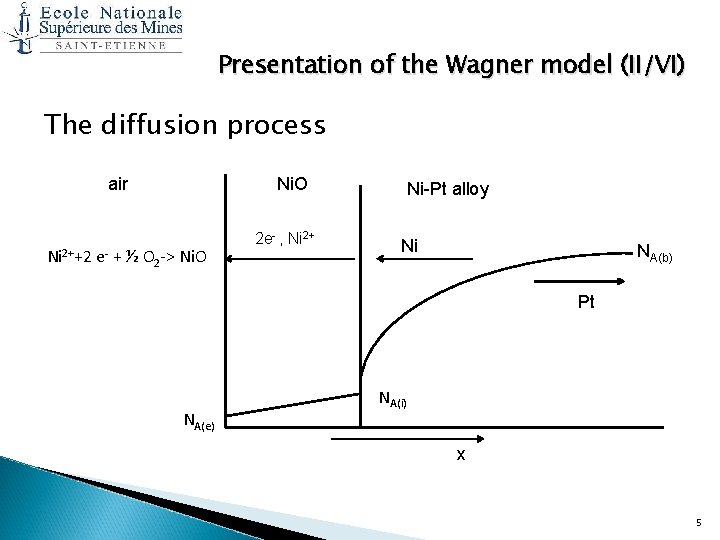
Presentation of the Wagner model (II/VI) The diffusion process air Ni. O Ni 2++2 e- + ½ O 2 -> Ni. O 2 e- , Ni 2+ Ni-Pt alloy Ni NA(b) Pt NA(e) NA(i) x 5

Presentation of the Wagner model (III/VI) The main assumptions of the Wagner model : In the alloy Ø Ø Migration of nickel ions takes place by jumping of nickel ions from normal lattice sites to adjacent vacant sites. No variation of the interdiffusion coefficient D In the oxide Ø Ø Thermodynamic equilibrium in the oxide scale The oxidation rate follows a parabolic law : 6

Presentation of the Wagner model (IV/VI) The Flux of metal ions : Equilibrium condition for the reaction 2 Ni (alloy) + O 2 (gaz) = 2 Ni. O : with a. Ni. O=1 and a. Ni=NA (NA(e)is « equilibrium mole fraction « for a given ambient partial pressure, at the oxide-air interface. ) 7

Presentation of the Wagner model (IV/VI) The equilibrium of flux of nickel atoms in the interface alloy-oxide gives us : (1) The Fick’s second law : (2) We define : (3) 8

Presentation of the Wagner model (V/VI) By (1), we have : So, I define : The equation (2) become : So, with erf the error function : 9

Presentation of the Wagner model (V/VI) With : (NA(b) = 0. 3 = « bulk mole fraction » ) we find : (4) For NA(i)= 0. 22 and NA(b) = 0. 3, α=0. 99 and γ= 100 : 10

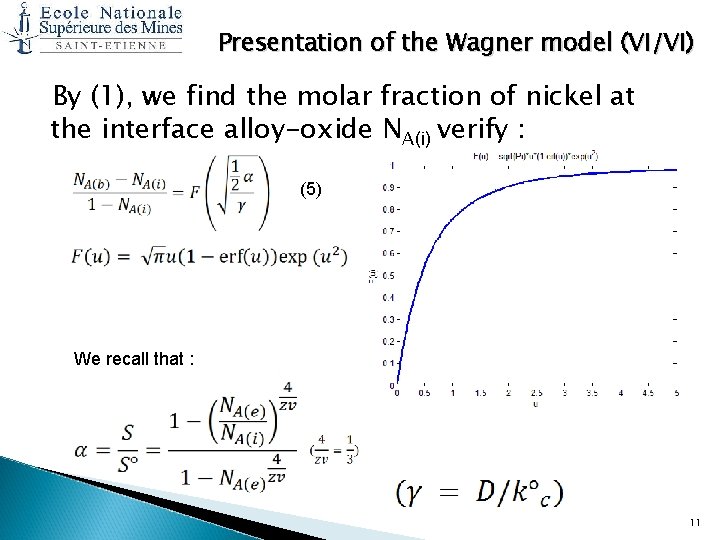
Presentation of the Wagner model (VI/VI) By (1), we find the molar fraction of nickel at the interface alloy-oxide NA(i) verify : (5) We recall that : 11

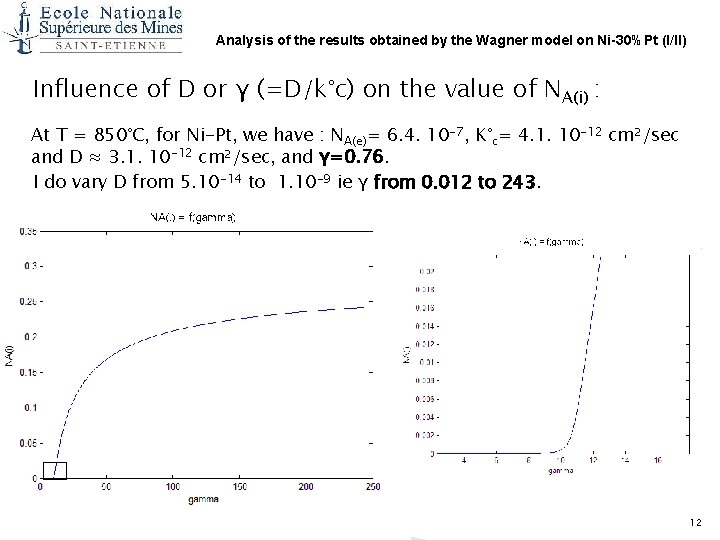
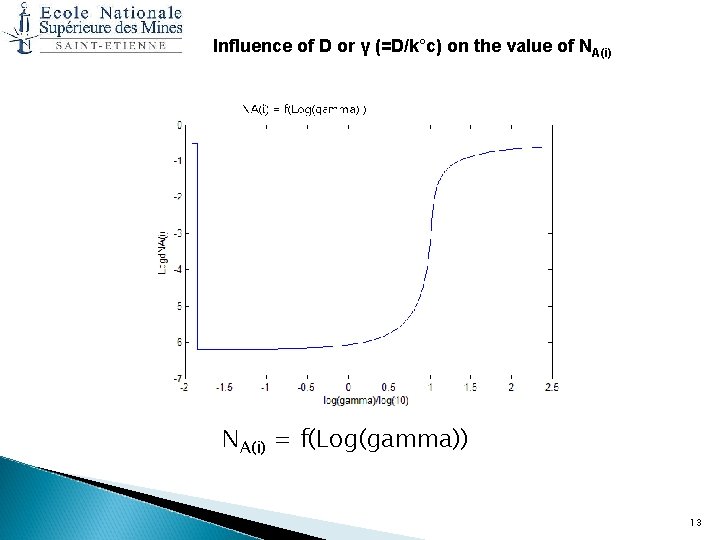
Analysis of the results obtained by the Wagner model on Ni-30%Pt (I/II) Influence of D or γ (=D/k°c) on the value of NA(i) : At T = 850°C, for Ni-Pt, we have : NA(e)= 6. 4. 10 -7, K°c= 4. 1. 10 -12 cm²/sec and D ≈ 3. 1. 10 -12 cm²/sec, and γ=0. 76. I do vary D from 5. 10 -14 to 1. 10 -9 ie γ from 0. 012 to 243. 12

Influence of D or γ (=D/k°c) on the value of NA(i) = f(Log(gamma)) 13

My experimental study on Ni-30%Cr (I/III) • • • . . . Analysis of the oxidation at 950°C of three samples oxidized during 1 h, 10 h and 100 h and quenched in the liquid nitrogen before observation Observation in Metallography Observation of the concentration profile in the SEM by EDX Observation of the concentration profile by Auger Spectroscopy 14

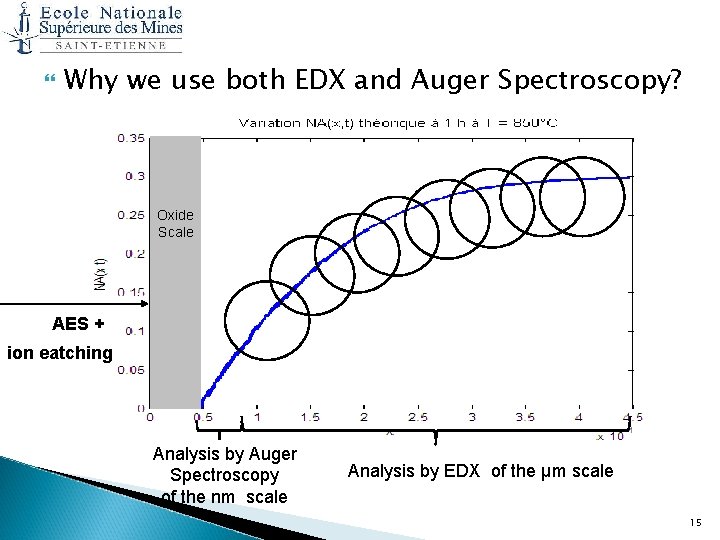
Why we use both EDX and Auger Spectroscopy? Oxide Scale AES + ion eatching Analysis by Auger Spectroscopy of the nm scale Analysis by EDX of the μm scale 15

First results obtained by Metallography Sample oxidized during 1 h x 100 Sample oxidized during 10 h x 100 16

Conclusion With the Wagner model, it is possible to obtain the concentration profile of the Chromium in the alloy. NA(i) strongly depends on the value of diffusion coefficient! My future study is aimed to validate (or not) the utilization of Wagner model to describe the concentration profile of the Chromium in the alloy. 17

Thank you for your attention 18

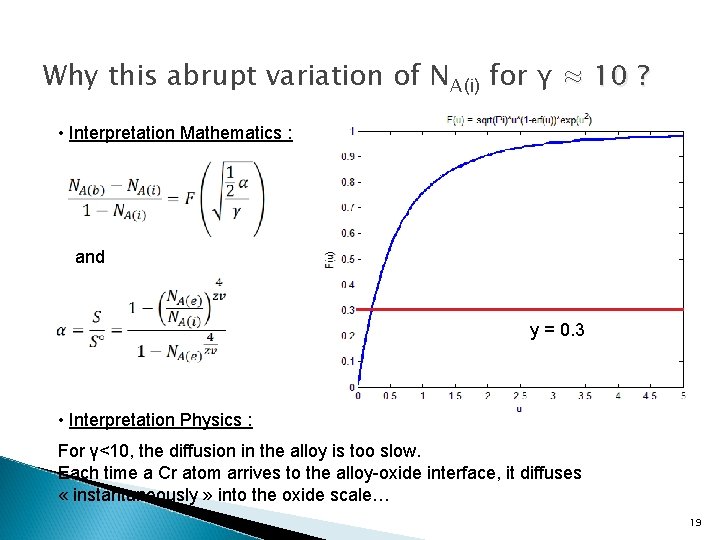
Why this abrupt variation of NA(i) for γ ≈ 10 ? • Interpretation Mathematics : and y = 0. 3 • Interpretation Physics : For γ<10, the diffusion in the alloy is too slow. Each time a Cr atom arrives to the alloy-oxide interface, it diffuses « instantaneously » into the oxide scale… 19