Oxidation of alcohols 1 1 2 1 1
![Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O](https://slidetodoc.com/presentation_image_h2/5818f06156fa57dc48d71e14e52123b2/image-2.jpg)
![Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O](https://slidetodoc.com/presentation_image_h2/5818f06156fa57dc48d71e14e52123b2/image-3.jpg)
![Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O](https://slidetodoc.com/presentation_image_h2/5818f06156fa57dc48d71e14e52123b2/image-4.jpg)


![Reactions Reduction [R] = reducing agent [R] 4 3 1 2 CH 3 - Reactions Reduction [R] = reducing agent [R] 4 3 1 2 CH 3 -](https://slidetodoc.com/presentation_image_h2/5818f06156fa57dc48d71e14e52123b2/image-9.jpg)



- Slides: 15

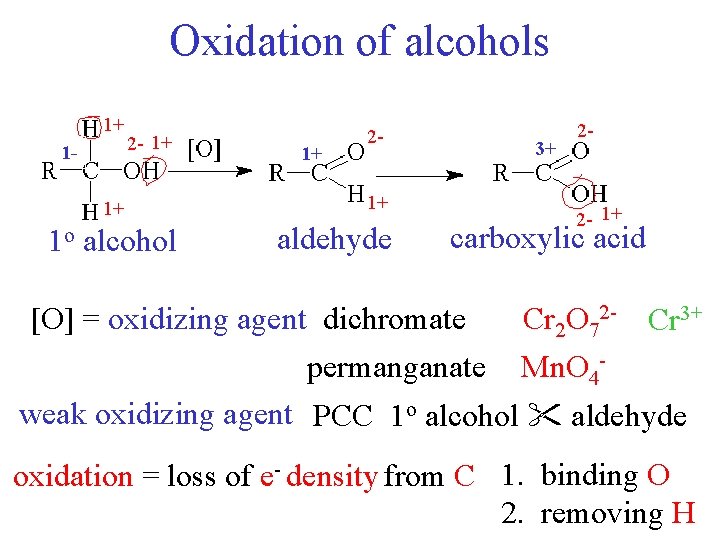
Oxidation of alcohols 1+ 1 - 2 - 1+ 1+ 1 o alcohol 1+ 21+ aldehyde 3+ 2 - 2 - 1+ carboxylic acid [O] = oxidizing agent dichromate Cr 2 O 72 - Cr 3+ permanganate Mn. O 4 weak oxidizing agent PCC 1 o alcohol aldehyde oxidation = loss of e- density from C 1. binding O 2. removing H
![Alcohols oxidation of alcohols O Cr 2 O 72 or Mn O Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O](https://slidetodoc.com/presentation_image_h2/5818f06156fa57dc48d71e14e52123b2/image-2.jpg)
Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O 421 o alcohols [O] aldehyde [O] carboxylic acid [O] + Cr 3+ + Cr 2 O 72 methanol [O] methanal formaldehyde methanoic acid formic acid
![Alcohols oxidation of alcohols O Cr 2 O 72 or Mn O Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O](https://slidetodoc.com/presentation_image_h2/5818f06156fa57dc48d71e14e52123b2/image-3.jpg)
Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O 42 - or PCC 2 o alcohols [O] ketone [O] no reaction
![Alcohols oxidation of alcohols O Cr 2 O 72 or Mn O Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O](https://slidetodoc.com/presentation_image_h2/5818f06156fa57dc48d71e14e52123b2/image-4.jpg)
Alcohols oxidation of alcohols: [O] = Cr 2 O 72 - or Mn. O 42 - or PCC 3 o alcohols [O] no reaction

Aldehydes and Ketones 120 o aldehyde C=O carbonyl group C and O are sp 2 hybridized ketone

Aldehydes and Ketones Nomenclature 1. Longest chain with C=O is parent 2. Suffix is “-al” or “-one” 3. C 1 is always C=O in aldehydes In ketones, C=O is given lowest number 4. C=O has precedence over -OH (called “hydroxy”)

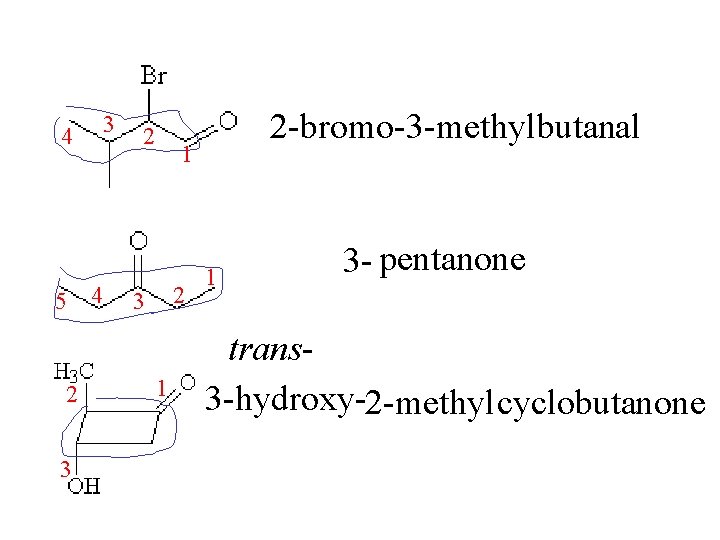
3 4 5 4 2 3 2 2 -bromo-3 -methylbutanal 1 2 3 1 1 3 - pentanone trans 3 -hydroxy-2 -methyl cyclobutanone

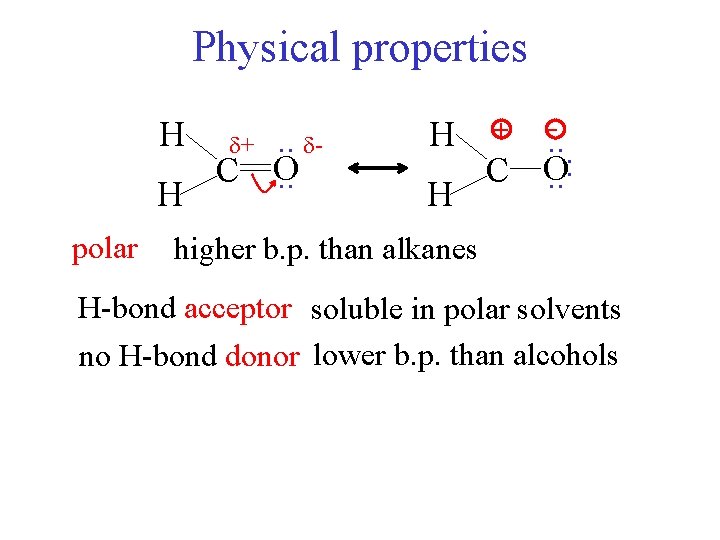
Physical properties H H polar . . C O. . + H H + -. . C O. . : higher b. p. than alkanes H-bond acceptor soluble in polar solvents no H-bond donor lower b. p. than alcohols
![Reactions Reduction R reducing agent R 4 3 1 2 CH 3 Reactions Reduction [R] = reducing agent [R] 4 3 1 2 CH 3 -](https://slidetodoc.com/presentation_image_h2/5818f06156fa57dc48d71e14e52123b2/image-9.jpg)
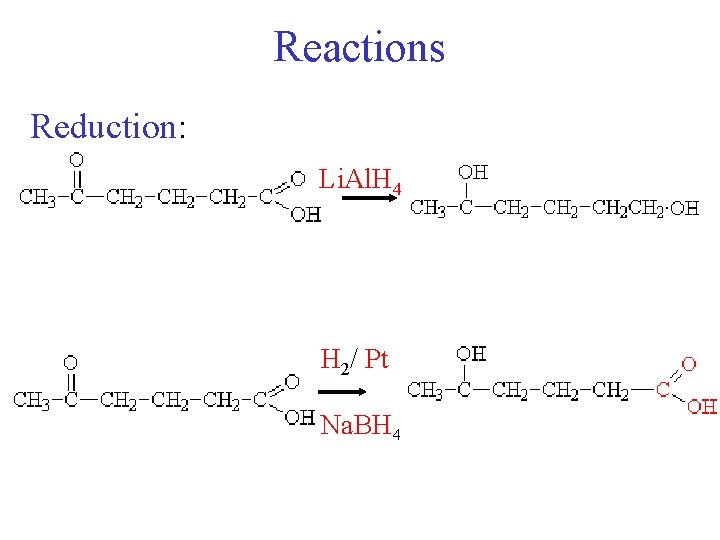
Reactions Reduction [R] = reducing agent [R] 4 3 1 2 CH 3 - CH 2 - CHO butanal [R] 1 3 2 Li. Al. H 4 Na. BH 4 H 2/Pt 4 2 - butanone CH 3 - CH 2 -OH

Reactions Reduction: Li. Al. H 4 H 2/ Pt Na. BH 4

Alkanes Cn. H 2 n+2 conformations (Newman) optical isomers LDF cyclic Cn. H 2 n geometric isomers combustion halogenation

Alkyl halides Nucleophilic substitution 1 o SN 2 transition state inversion of stereocenter 3 o SN 1 carbocation racemic mixture 2 o ?

Alkenes sp 2 Cn. H 2 n geometric isomers Addition reactions H–X H – OH H–H Br – Br polymerization

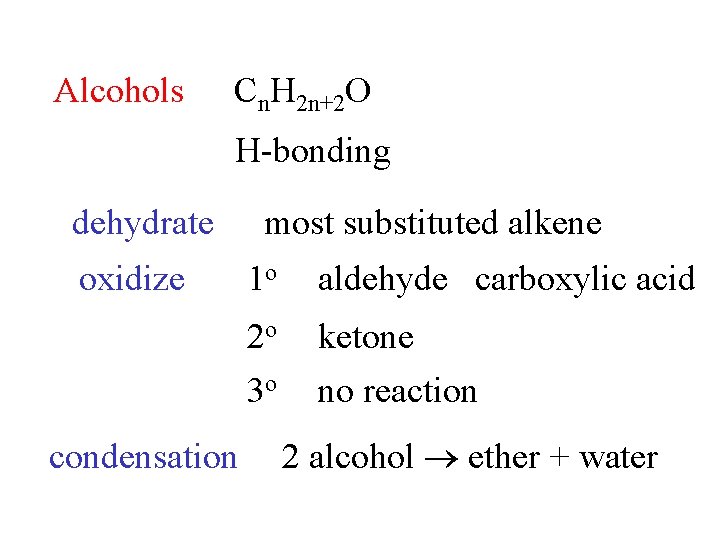
Alcohols Cn. H 2 n+2 O H-bonding dehydrate oxidize condensation most substituted alkene 1 o aldehyde carboxylic acid 2 o 3 o ketone no reaction 2 alcohol ether + water

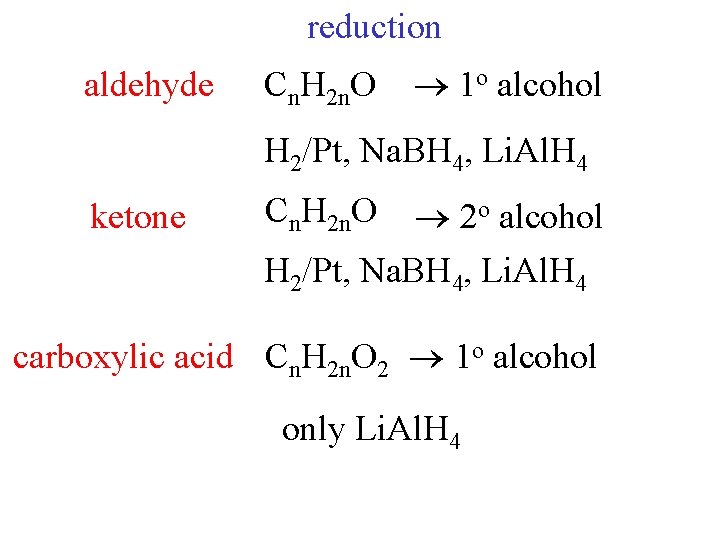
reduction aldehyde Cn. H 2 n. O 1 o alcohol H 2/Pt, Na. BH 4, Li. Al. H 4 ketone Cn. H 2 n. O 2 o alcohol H 2/Pt, Na. BH 4, Li. Al. H 4 carboxylic acid Cn. H 2 n. O 2 1 o alcohol only Li. Al. H 4