Oxidation Numbers Redox Reactions Ox always change during

Oxidation Numbers & Redox Reactions Ox always change during redox reactions: Increase Ox = Oxidation Decrease Ox = Reduction It’s a redox reaction if: or: element → compound → element S 8(s) + 8 O 2(g) → 8 SO 2(g) Ox(S) : 0 → +4 Ox(O) : 0 → -2 (oxidized: lost e- ; increased Ox) (reduced: gained e- ; decreased Ox)

Oxidation Numbers & Redox Reactions A more complex example: Cu(s)+4 H+(aq)+2 NO 3 -(aq) → Cu 2+(aq)+2 NO 2(g)+2 H 2 O(ℓ) 0 +1 +5 -2 +2 Cu is oxidized (Ox ↑; loss of e-). H is unchanged. O is unchanged. N is reduced (Ox ↓; gain of e-). +4 -2 +1 -2

Oxidation Numbers & Redox Reactions Exchange reactions are not redox – no change in oxidation state occurs. +2 +1 -1 +1 +2 -1 Pb(NO 3)2(aq) + 2 Kl(aq) 2 KNO 3(aq) + Pbl 2(s) NO 3 -: Ox(O) = -2; Ox(N) = +5
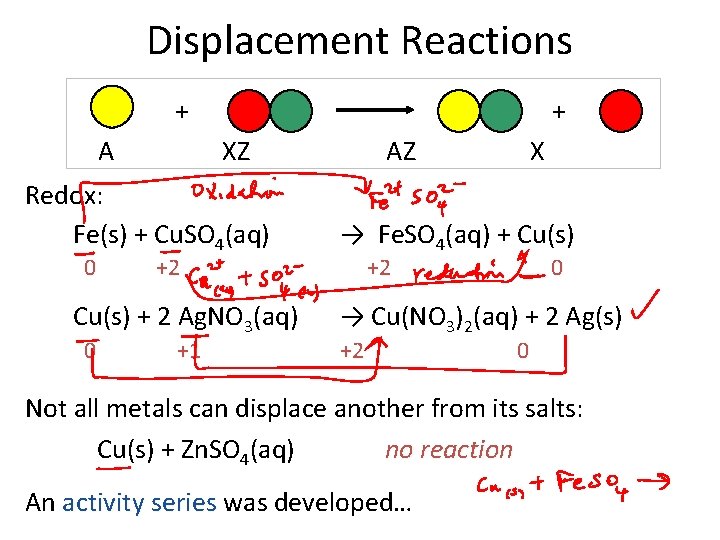
Displacement Reactions + A + XZ Redox: Fe(s) + Cu. SO 4(aq) 0 +2 Cu(s) + 2 Ag. NO 3(aq) 0 +1 AZ X → Fe. SO 4(aq) + Cu(s) +2 0 → Cu(NO 3)2(aq) + 2 Ag(s) +2 0 Not all metals can displace another from its salts: Cu(s) + Zn. SO 4(aq) no reaction An activity series was developed…
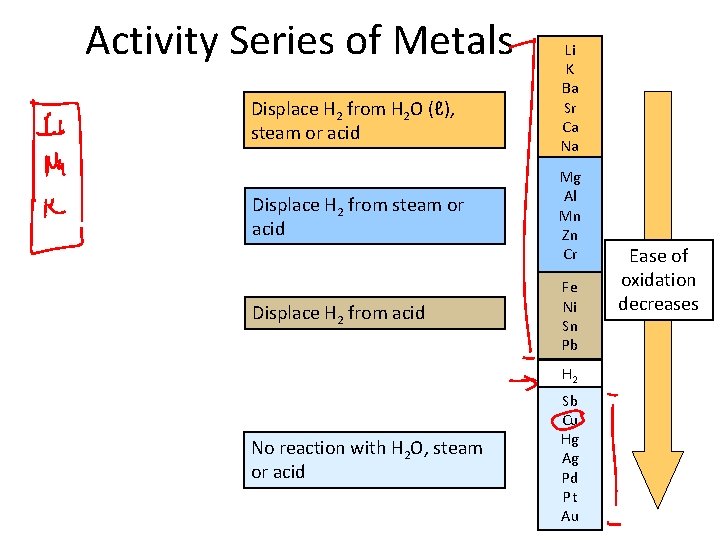
Activity Series of Metals Displace H 2 from H 2 O (ℓ), steam or acid Displace H 2 from acid Li K Ba Sr Ca Na Mg Al Mn Zn Cr Fe Ni Sn Pb H 2 No reaction with H 2 O, steam or acid Sb Cu Hg Ag Pd Pt Au Ease of oxidation decreases

Displacement Reactions Powerful reducing agents at the top. Higher elements displace lower ones: Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Mg(s) + 2 HCl(aq) → Mg. Cl 2(aq) + H 2(g) Metals at the bottom are unreactive. • Coinage metals • Their ions are powerful oxidizing agents. Li K : : Na Mg : Zn Cr Fe : Pb H 2 Sb Cu : Ag : Au

Displacement Reactions

Displacement Reactions 2 Na(s) + 2 H 2 O(ℓ) → 2 Na. OH(aq) + H 2(g)
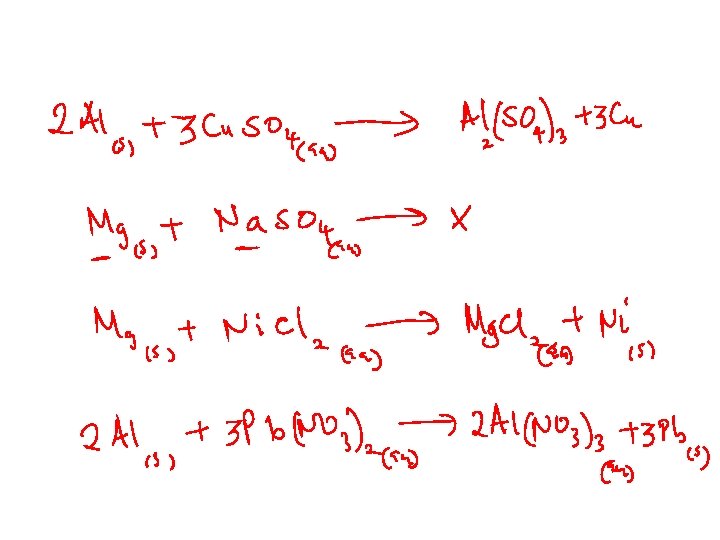
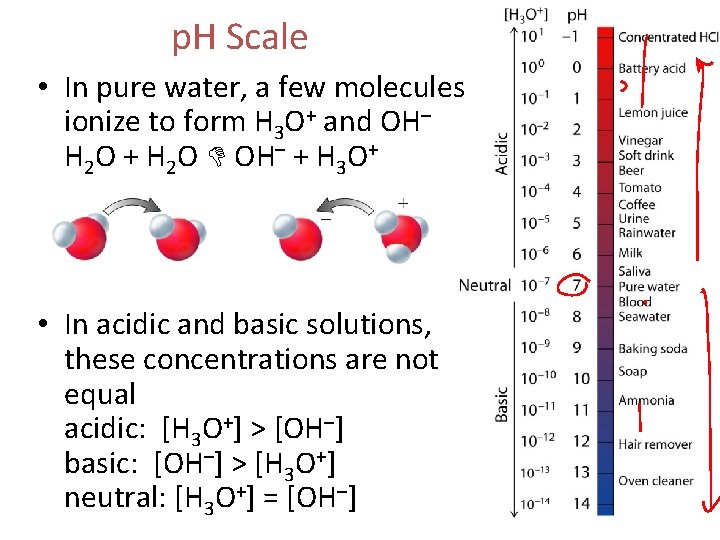
p. H Scale • In pure water, a few molecules ionize to form H 3 O+ and OH– H 2 O + H 2 O OH– + H 3 O+ • In acidic and basic solutions, these concentrations are not equal acidic: [H 3 O+] > [OH–] basic: [OH–] > [H 3 O+] neutral: [H 3 O+] = [OH–]
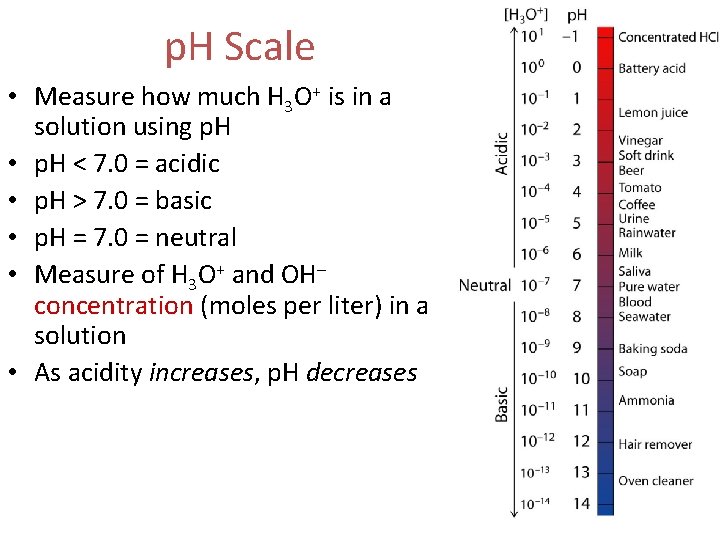
p. H Scale • Measure how much H 3 O+ is in a solution using p. H • p. H < 7. 0 = acidic • p. H > 7. 0 = basic • p. H = 7. 0 = neutral • Measure of H 3 O+ and OH– concentration (moles per liter) in a solution • As acidity increases, p. H decreases
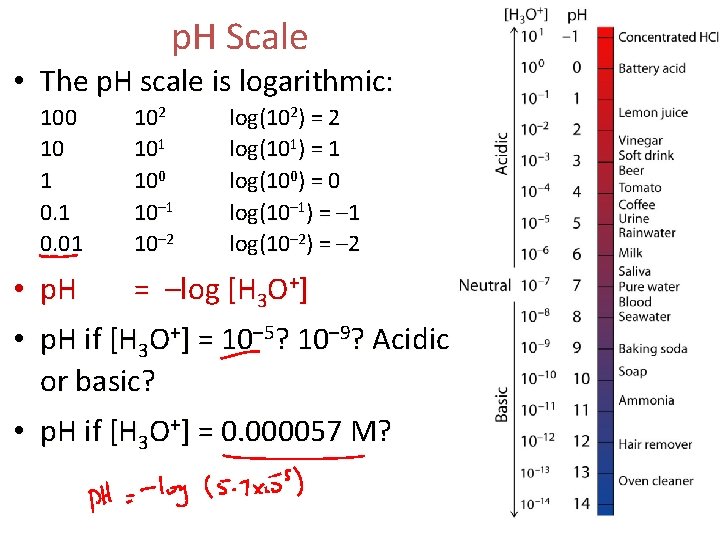
p. H Scale • The p. H scale is logarithmic: 100 10 1 0. 01 102 101 100 10– 1 10– 2 log(102) = 2 log(101) = 1 log(100) = 0 log(10– 1) = – 1 log(10– 2) = – 2 • p. H = –log [H 3 O+] • p. H if [H 3 O+] = 10– 5? 10– 9? Acidic or basic? • p. H if [H 3 O+] = 0. 000057 M?
![Finding [H 3 O+] from p. H [H 3 O+] = 10 -p. H Finding [H 3 O+] from p. H [H 3 O+] = 10 -p. H](http://slidetodoc.com/presentation_image/33f8655cb612b1e91ae441176da5d42c/image-13.jpg)
Finding [H 3 O+] from p. H [H 3 O+] = 10 -p. H What is [H 3 O+] if p. H = 8. 9?

p. H: Quantitative Measure of Acidity • Acidity is related to concentration of H+ (or H 3 O +) • p. H = -log[H 3 O+]
- Slides: 14