Oxidation and reduction reactions Redox Redox reactions What






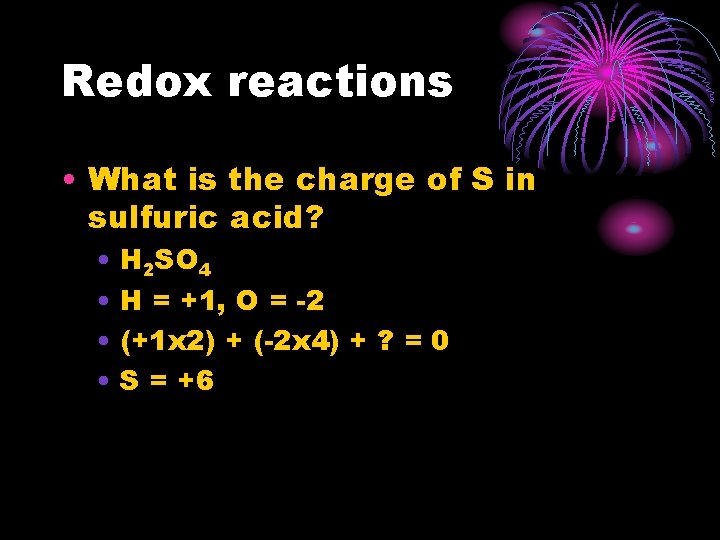






- Slides: 13

Oxidation and reduction reactions (Redox)

Redox reactions • What is a redox reaction? • It is a reaction where one chemical “gains” electrons (reduced) and one “loses” electrons (oxidized). LEO says GER or OIL RIG

Redox reactions • Where have we seen oxidation numbers before? • Oxidation numbers were used to figure ionic compounds.

Redox reactions • How can we use them now? • With the 4 basic rules, we can predict the oxidation number of elements that have different oxidation numbers based on their compound.

Redox reactions • What are the rules? • Fluorine is always -1 • Hydrogen is +1 except in hydride ions • Oxygen is -2 except in hydrogen peroxide • Halogens are -1 except when with a more reactive halogen or oxygen • Alkali metals are +1 • Alkaline earth metals are +2

Redox reactions • Some other things to remember. • Elemental states have a 0 oxidation number. • Oxidation number is equal to ion charge. • The total oxidation number has to add up to the charge of the molecule.
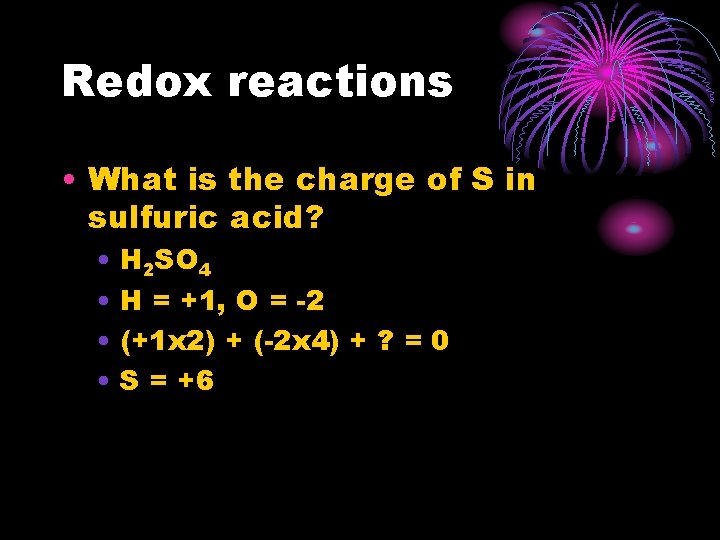
Redox reactions • What is the charge of S in sulfuric acid? • H 2 SO 4 • H = +1, O = -2 • (+1 x 2) + (-2 x 4) + ? = 0 • S = +6

Redox reactions • What is a half-equation? • A half-equation is written with the electrons to show the oxidized or reduced partial equation.

Redox reactions • Some rules for DP half-reactions. • Balance the number of atoms of each element being oxidized or reduced • Add water molecules to balance the oxygen • Add hydrogen ions to balance the hydrogen • Add electrons to balance the electrical charge (multiply by a number that makes the electrons gained = to electrons lost) • Example: exercise 3 on page 167

Redox reactions • How would we make a reactivity series based on group behaviors of oxidizing and reducing agents? • If a substance is a strong reducing agent, its counterpart will be a weak oxidizing agent. (like conjugate acid-base pairs)

Redox reactions • How can we tell strength of oxidation or reduction? • Strength of oxidizing agent rises as electronegativity rises in general. • Strength of reducing agent rises as electronegativity decreases in general.

Redox reactions

Redox reactions • How can you use oxidation numbers to balance an equation? • If you balance oxidation numbers, then just balance the remaining parts. I don’t think this is a really useful tool if you can balance normally.