Oxidants in organic synthesis Initiators of freeradical reactions











- Slides: 58


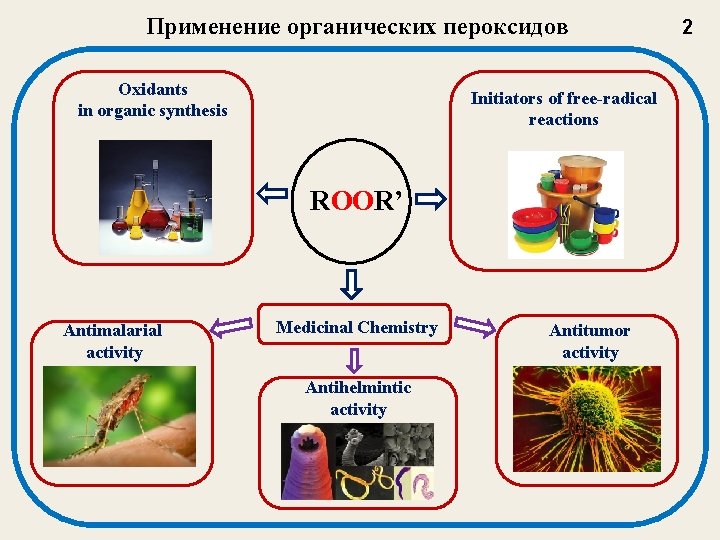
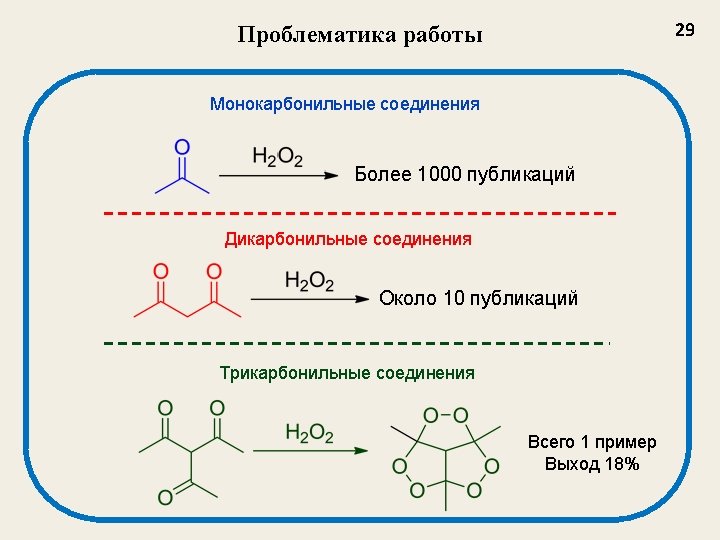
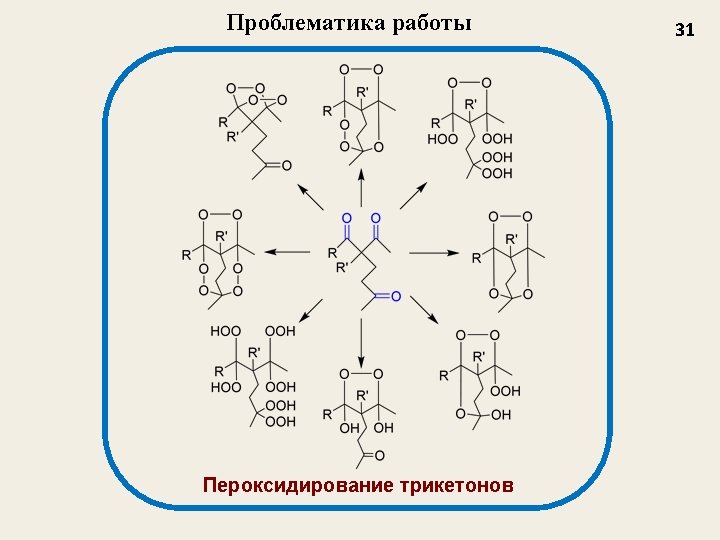
Применение органических пероксидов Oxidants in organic synthesis Initiators of free-radical reactions ROOR’ Antimalarial activity Medicinal Chemistry Antihelmintic activity Antitumor activity 2


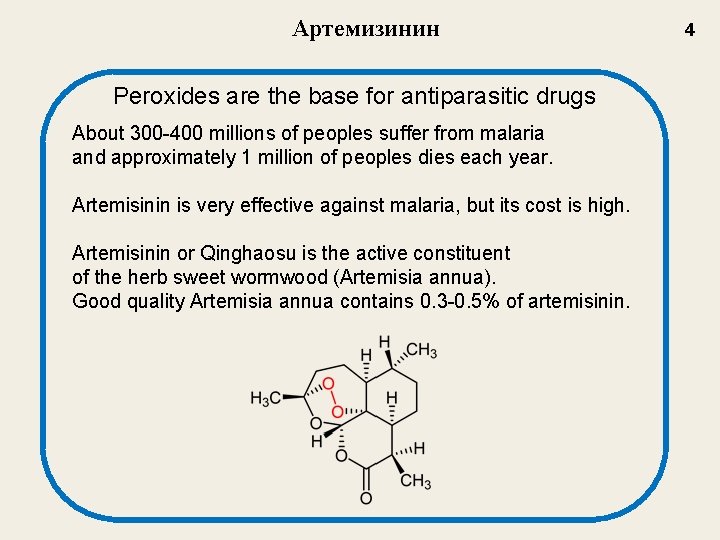
Артемизинин Peroxides are the base for antiparasitic drugs About 300 -400 millions of peoples suffer from malaria and approximately 1 million of peoples dies each year. Artemisinin is very effective against malaria, but its cost is high. Artemisinin or Qinghaosu is the active constituent of the herb sweet wormwood (Artemisia annua). Good quality Artemisia annua contains 0. 3 -0. 5% of artemisinin. 4

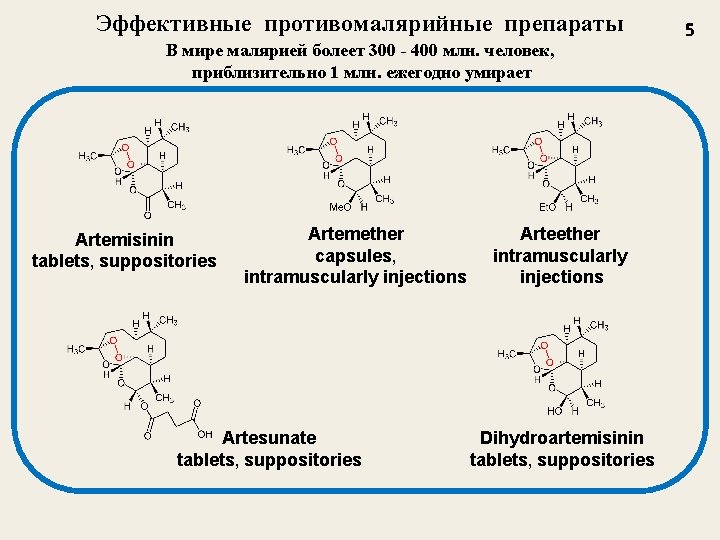
Эффективные противомалярийные препараты В мире малярией болеет 300 - 400 млн. человек, приблизительно 1 млн. ежегодно умирает Artemisinin tablets, suppositories Artemether capsules, intramuscularly injections Artesunate tablets, suppositories Arteether intramuscularly injections Dihydroartemisinin tablets, suppositories 5

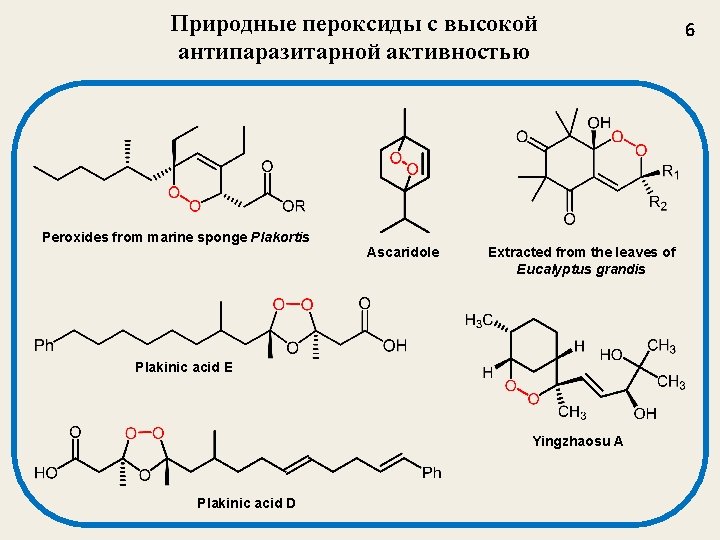
Природные пероксиды с высокой антипаразитарной активностью Peroxides from marine sponge Plakortis Ascaridole Extracted from the leaves of Eucalyptus grandis Plakinic acid E Yingzhaosu A Plakinic acid D 6

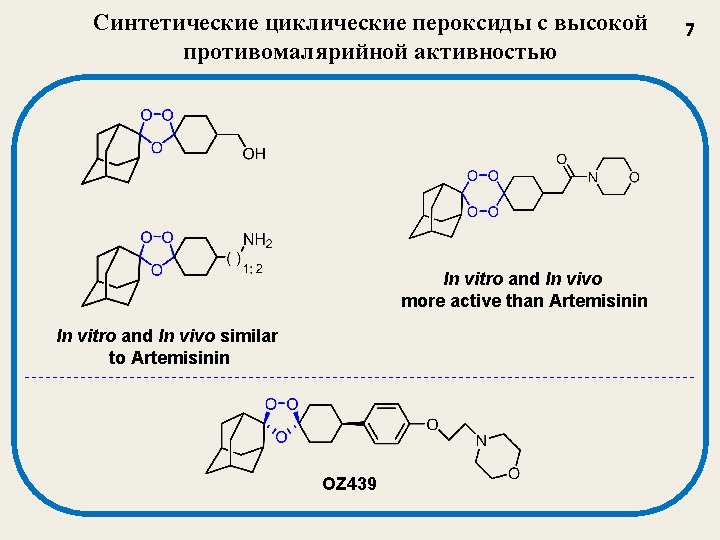
Синтетические циклические пероксиды с высокой противомалярийной активностью In vitro and In vivo more active than Artemisinin In vitro and In vivo similar to Artemisinin OZ 439 7

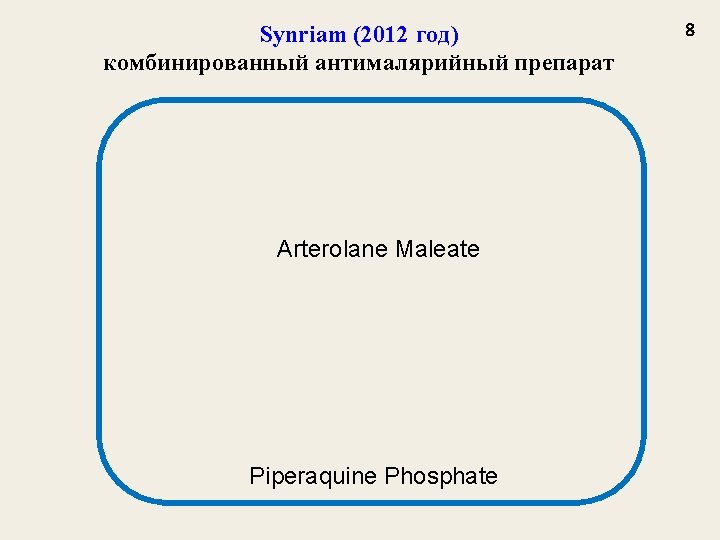
Synriam (2012 год) комбинированный антималярийный препарат Arterolane Maleate Piperaquine Phosphate 8




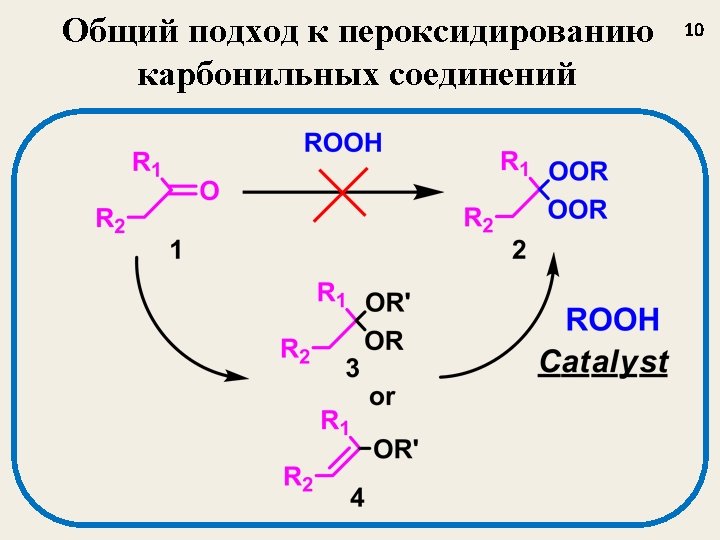
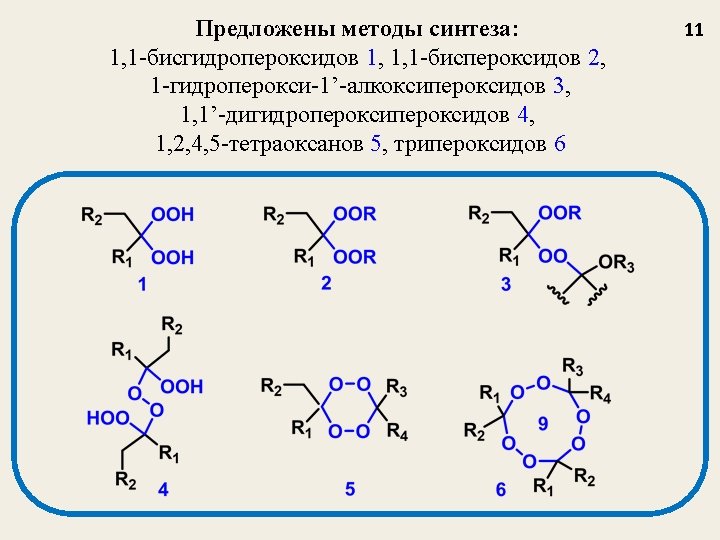
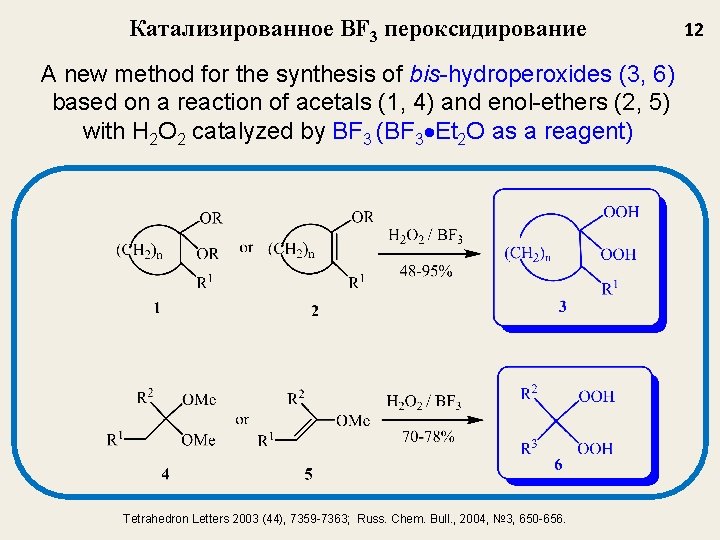
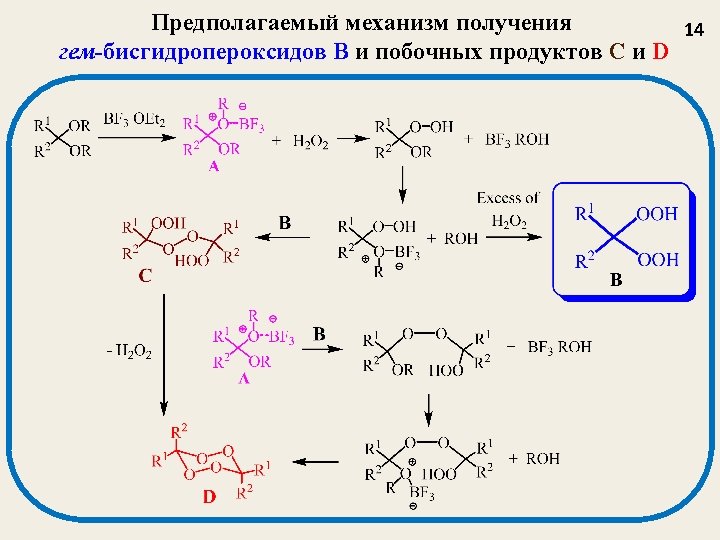
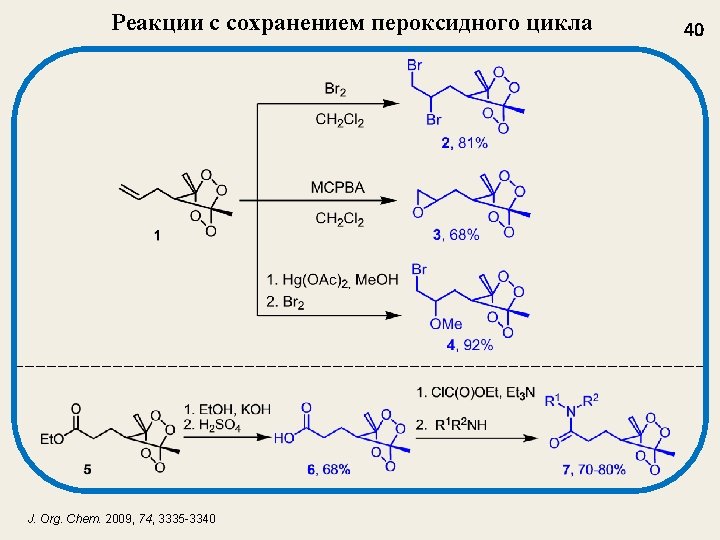
Катализированное BF 3 пероксидирование A new method for the synthesis of bis-hydroperoxides (3, 6) based on a reaction of acetals (1, 4) and enol-ethers (2, 5) with H 2 O 2 catalyzed by BF 3 (BF 3 Et 2 O as a reagent) Tetrahedron Letters 2003 (44), 7359 -7363; Russ. Chem. Bull. , 2004, № 3, 650 -656. 12

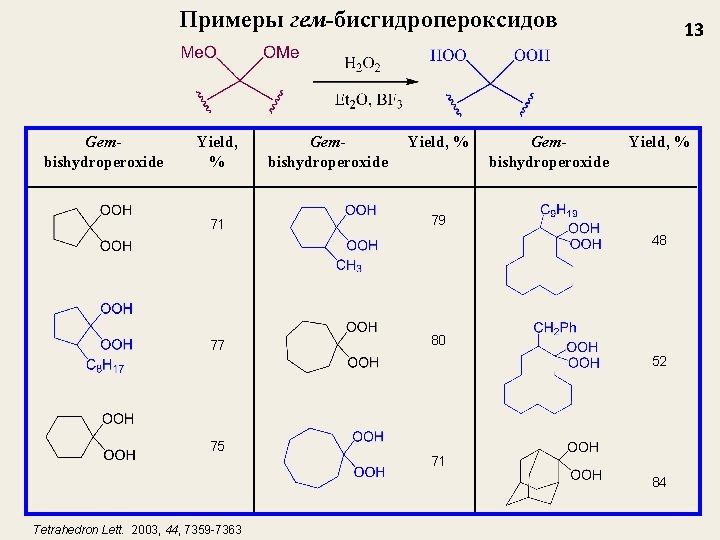
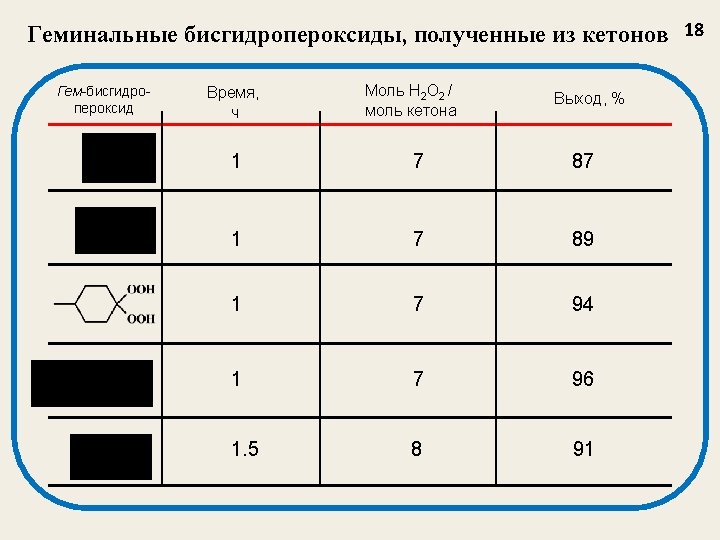
Примеры гем-бисгидропероксидов Gembishydroperoxide Yield, % 71 79 77 80 75 Gembishydroperoxide 13 Yield, % 48 52 71 84 Tetrahedron Lett. 2003, 44, 7359 -7363









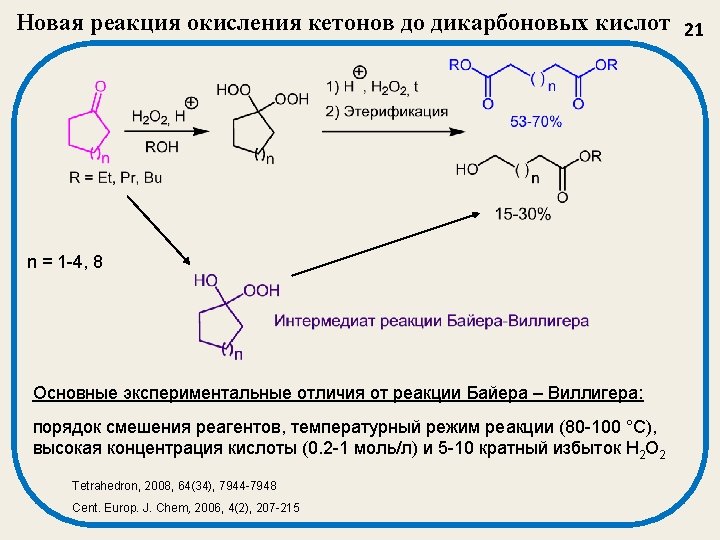
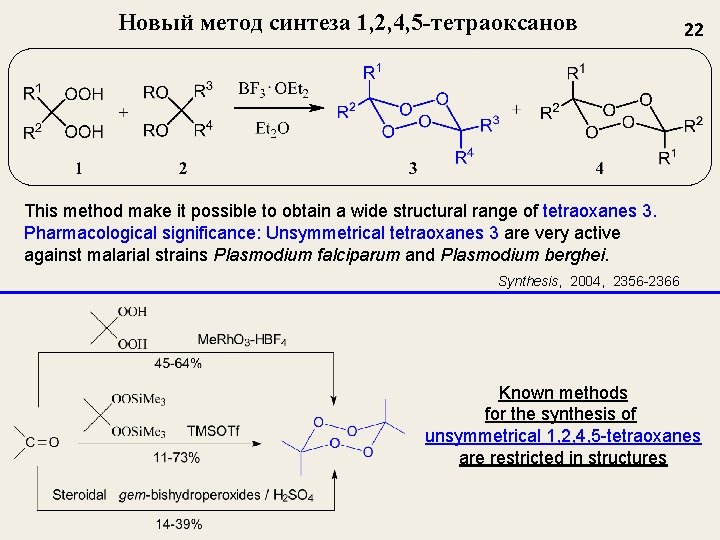
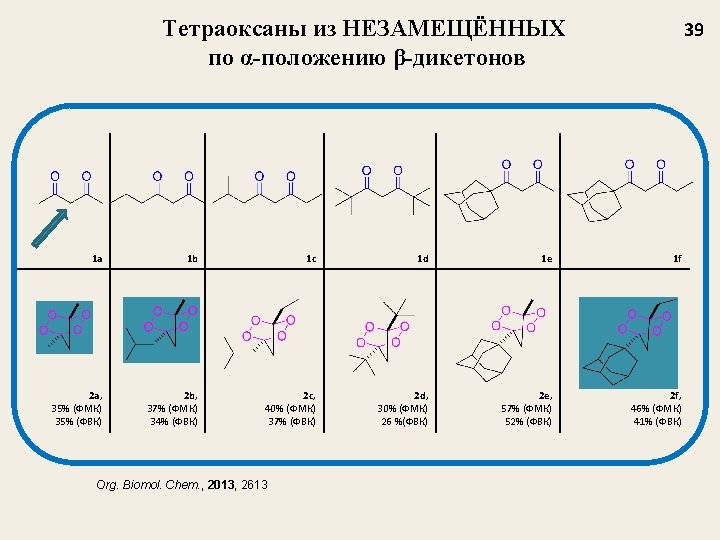
Новый метод синтеза 1, 2, 4, 5 -тетраоксанов 22 This method make it possible to obtain a wide structural range of tetraoxanes 3. Pharmacological significance: Unsymmetrical tetraoxanes 3 are very active against malarial strains Plasmodium falciparum and Plasmodium berghei. Synthesis, 2004, 2356 -2366 Known methods for the synthesis of unsymmetrical 1, 2, 4, 5 -tetraoxanes are restricted in structures

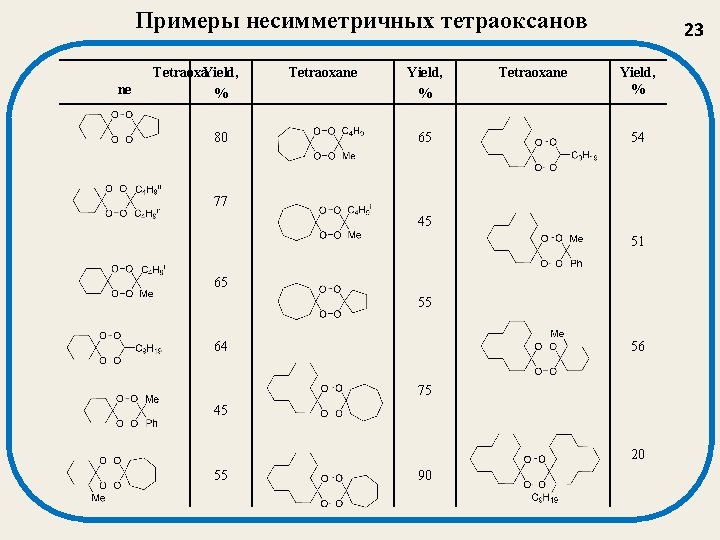
Примеры несимметричных тетраоксанов ne Tetraoxa. Yield, % 80 Tetraoxane Yield, % 65 Tetraoxane 23 Yield, % 54 77 45 51 65 55 64 56 75 45 20 55 90

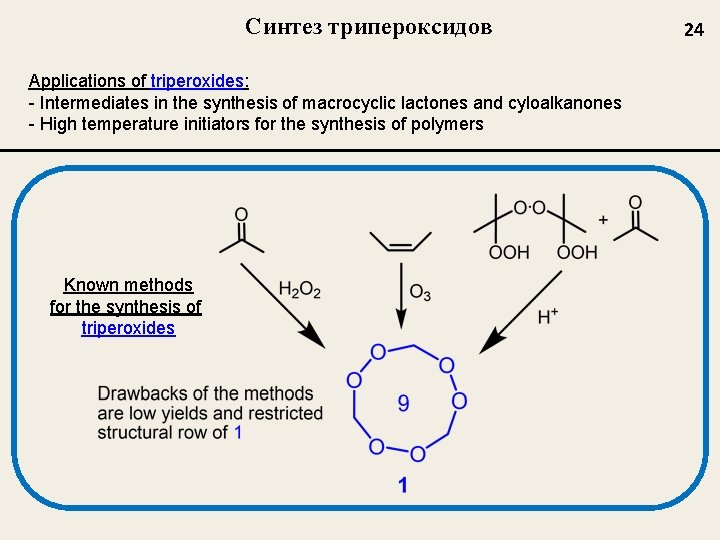
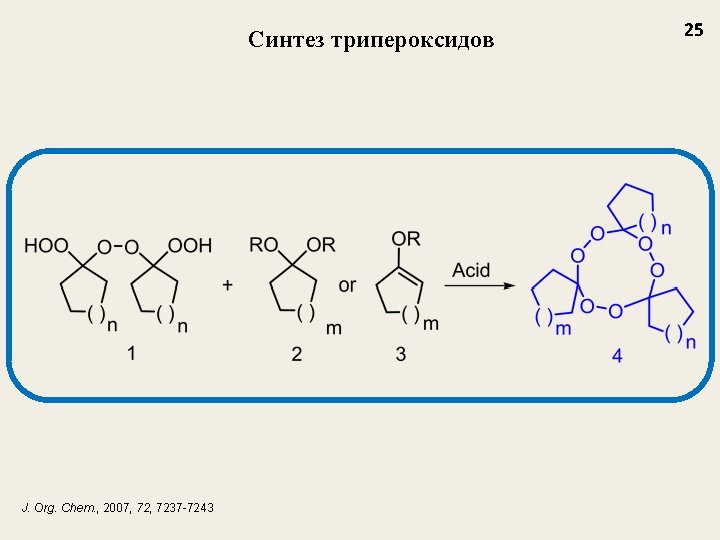
Синтез трипероксидов Applications of triperoxides: - Intermediates in the synthesis of macrocyclic lactones and cyloalkanones - High temperature initiators for the synthesis of polymers Known methods for the synthesis of triperoxides 24


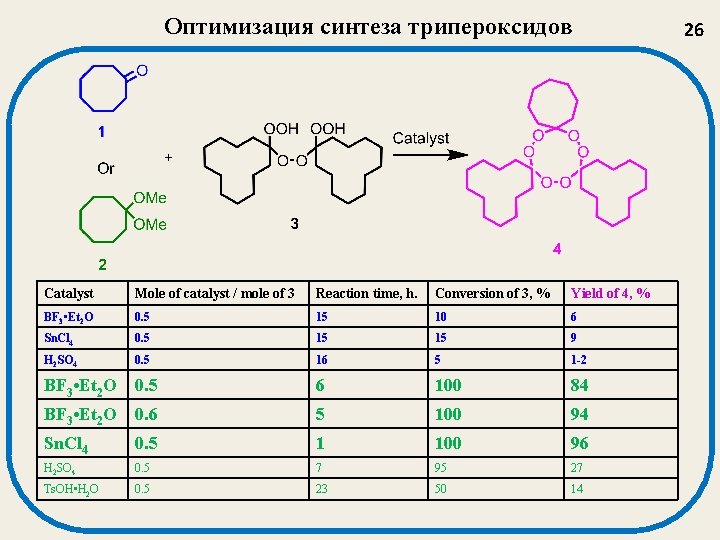
Оптимизация синтеза трипероксидов Catalyst Mole of catalyst / mole of 3 Reaction time, h. Conversion of 3, % Yield of 4, % BF 3 • Et 2 O 0. 5 15 10 6 Sn. Cl 4 0. 5 15 15 9 H 2 SO 4 0. 5 16 5 1 -2 BF 3 • Et 2 O 0. 5 6 100 84 BF 3 • Et 2 O 0. 6 5 100 94 Sn. Cl 4 0. 5 1 100 96 H 2 SO 4 0. 5 7 95 27 Ts. OH • H 2 O 0. 5 23 50 14 26

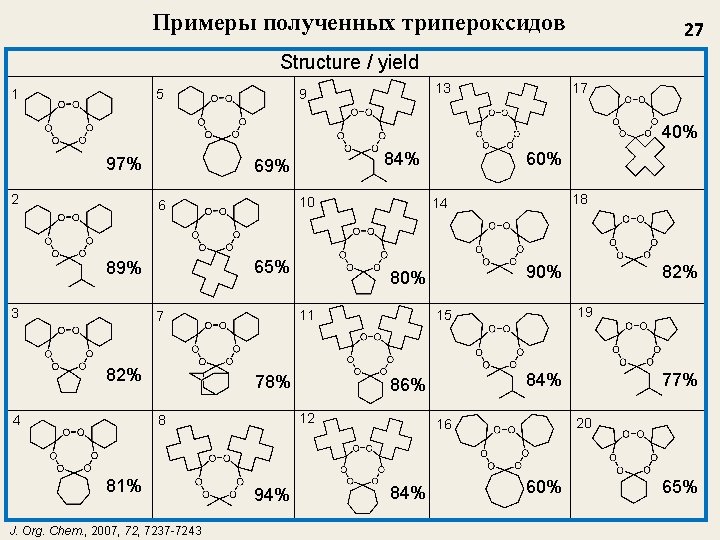
Примеры полученных трипероксидов 27 Structure / yield 1 5 17 13 9 40% 97% 2 10 6 65% 89% 3 82% 81% J. Org. Chem. , 2007, 7237 -7243 84% 86% 77% 20 16 84% 82% 19 15 12 94% 18 90% 80% 78% 8 60% 14 11 7 4 84% 69% 60% 65%





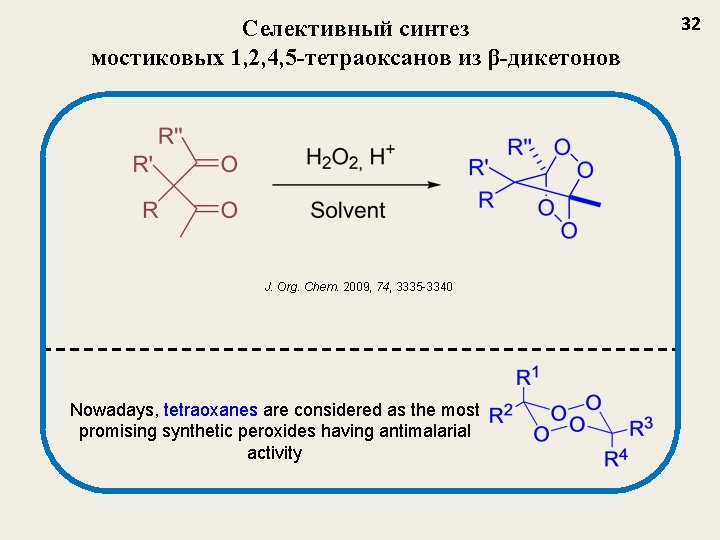
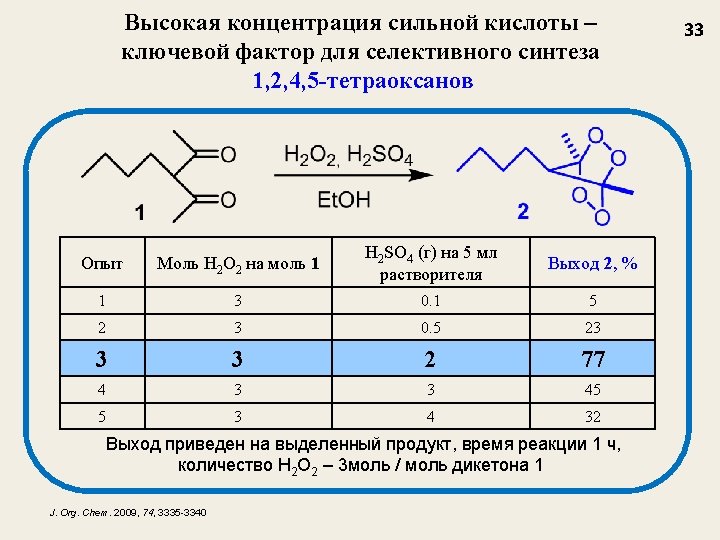
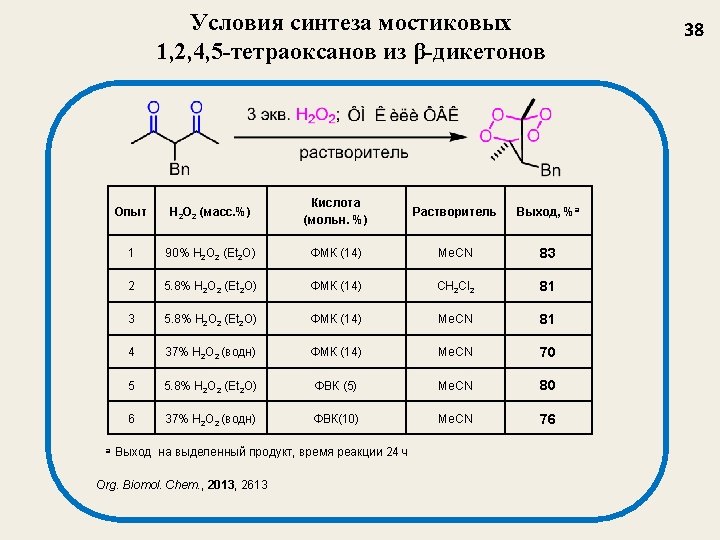
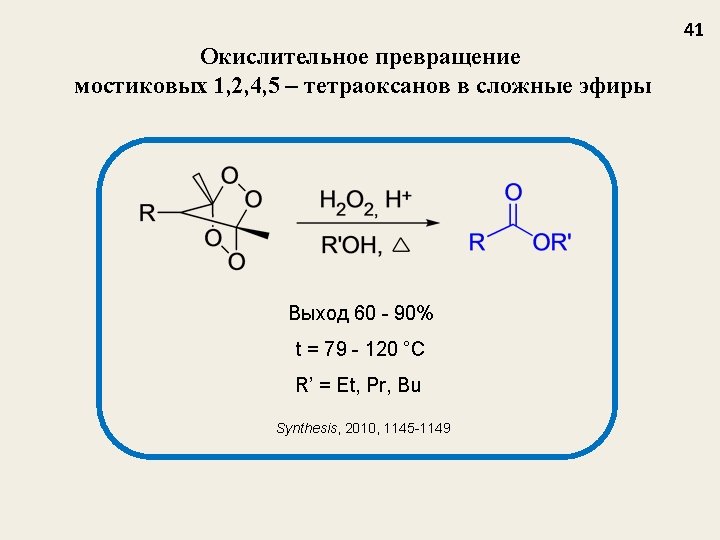
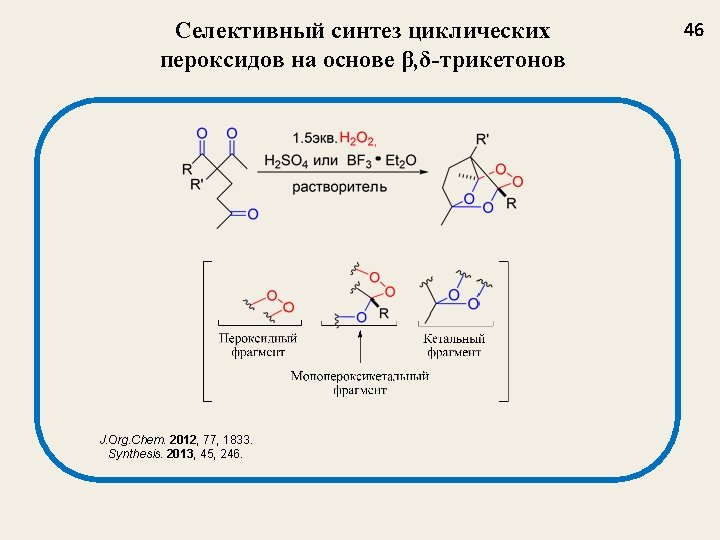
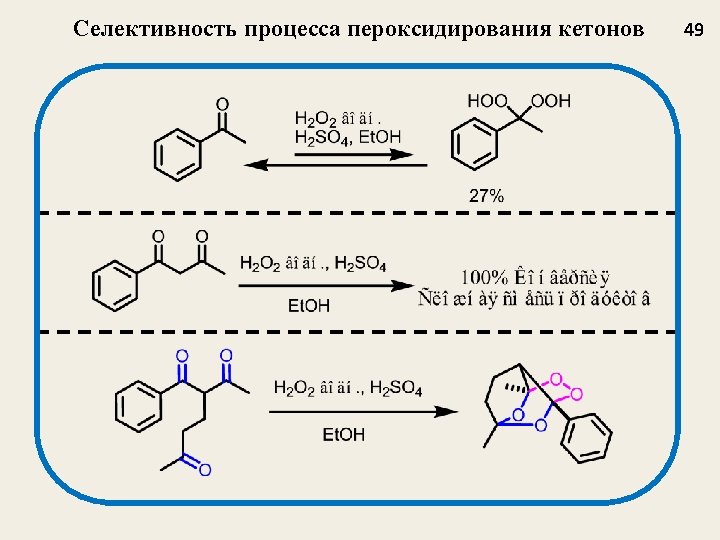
Селективный синтез мостиковых 1, 2, 4, 5 -тетраоксанов из β-дикетонов J. Org. Chem. 2009, 74, 3335 -3340 Nowadays, tetraoxanes are considered as the most promising synthetic peroxides having antimalarial activity 32



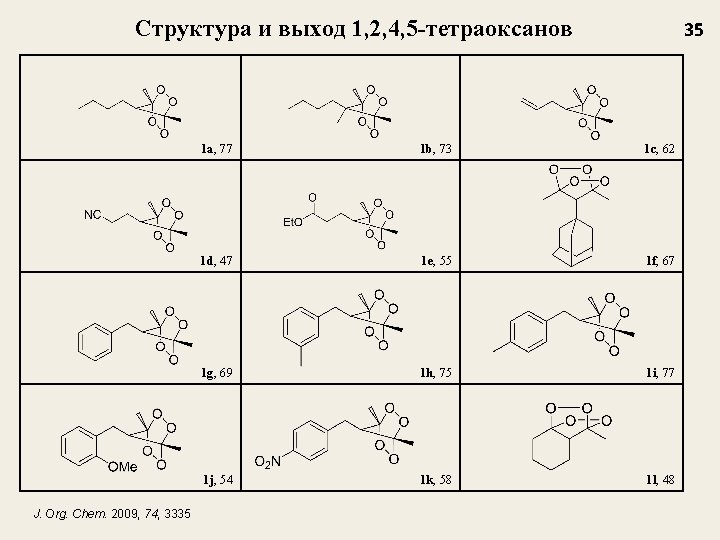
Структура и выход 1, 2, 4, 5 -тетраоксанов J. Org. Chem. 2009, 74, 3335 35 1 a, 77 1 b, 73 1 c, 62 1 d, 47 1 e, 55 1 f, 67 1 g, 69 1 h, 75 1 i, 77 1 j, 54 1 k, 58 1 l, 48













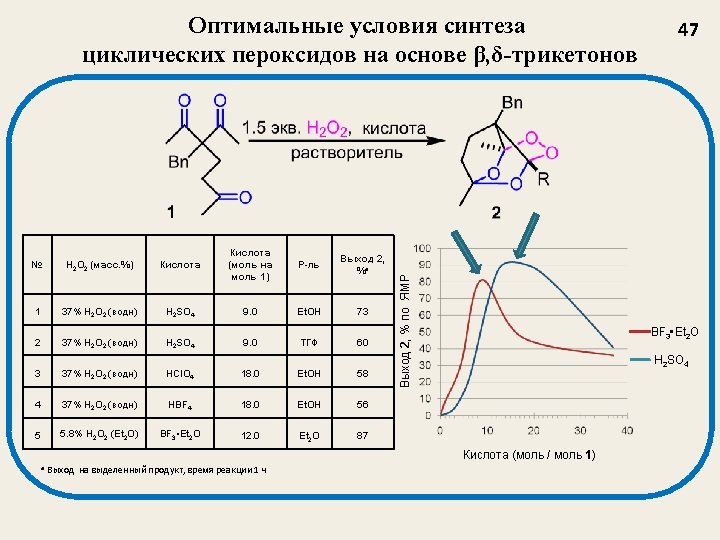
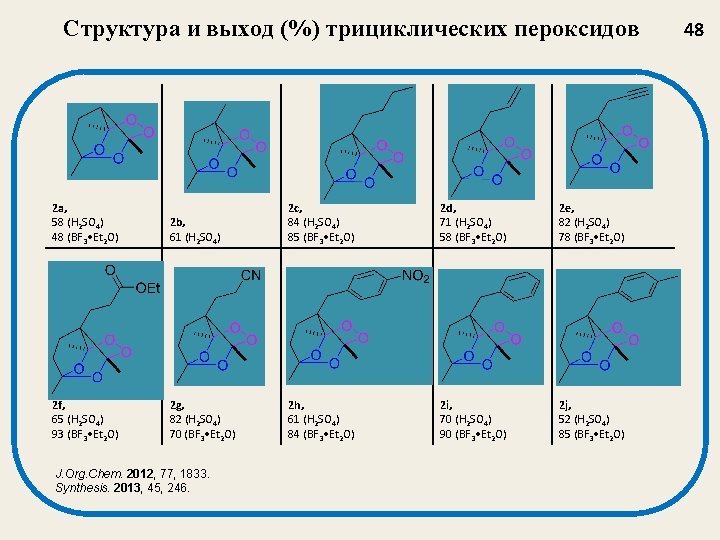
Структура и выход (%) трициклических пероксидов 2 a, 58 (H 2 SO 4) 48 (BF 3 • Et 2 O) 2 f, 65 (H 2 SO 4) 93 (BF 3 • Et 2 O) 2 b, 61 (H 2 SO 4) 2 c, 84 (H 2 SO 4) 85 (BF 3 • Et 2 O) 2 d, 71 (H 2 SO 4) 58 (BF 3 • Et 2 O) 2 e, 82 (H 2 SO 4) 78 (BF 3 • Et 2 O) 2 g, 82 (H 2 SO 4) 70 (BF 3 • Et 2 O) 2 h, 61 (H 2 SO 4) 84 (BF 3 • Et 2 O) 2 i, 70 (H 2 SO 4) 90 (BF 3 • Et 2 O) 2 j, 52 (H 2 SO 4) 85 (BF 3 • Et 2 O) J. Org. Chem. 2012, 77, 1833. Synthesis. 2013, 45, 246. 48




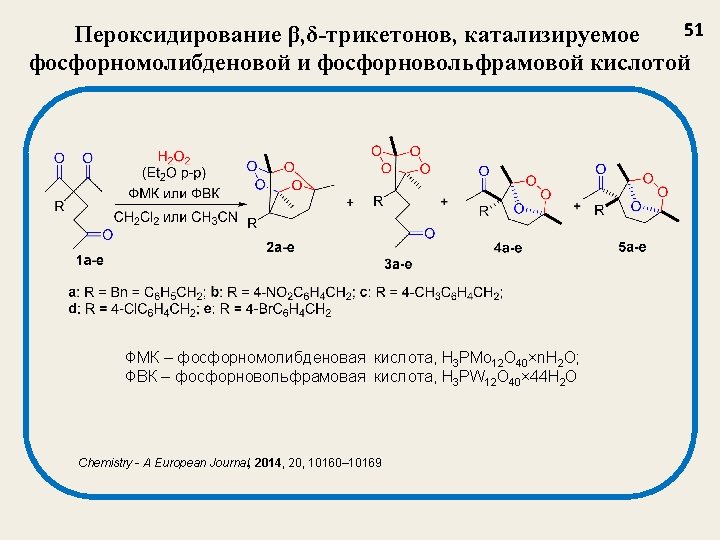
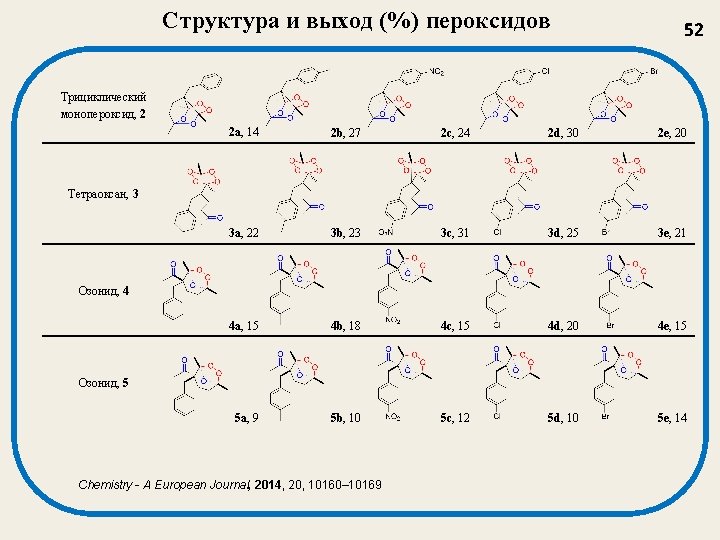
Структура и выход (%) пероксидов 52 Трициклический монопероксид, 2 2 a, 14 2 b, 27 2 c, 24 2 d, 30 2 e, 20 3 a, 22 3 b, 23 3 c, 31 3 d, 25 3 e, 21 4 a, 15 4 b, 18 4 c, 15 4 d, 20 4 e, 15 5 a, 9 5 b, 10 5 c, 12 5 d, 10 5 e, 14 Тетраоксан, 3 Озонид, 4 Озонид, 5 Chemistry - A European Journal, 2014, 20, 10160– 10169



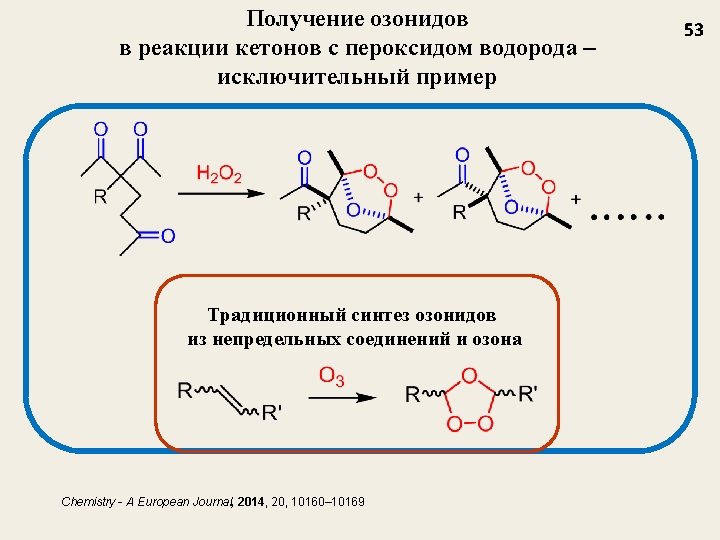
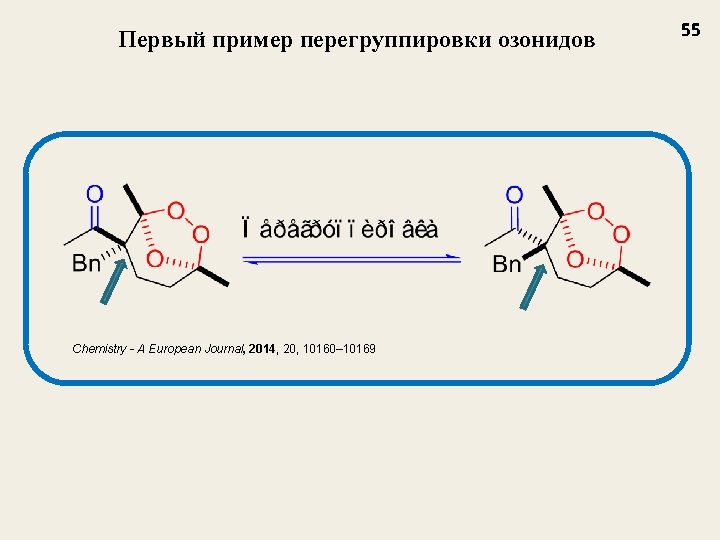
Первый пример перегруппировки озонидов Chemistry - A European Journal, 2014, 20, 10160– 10169 55

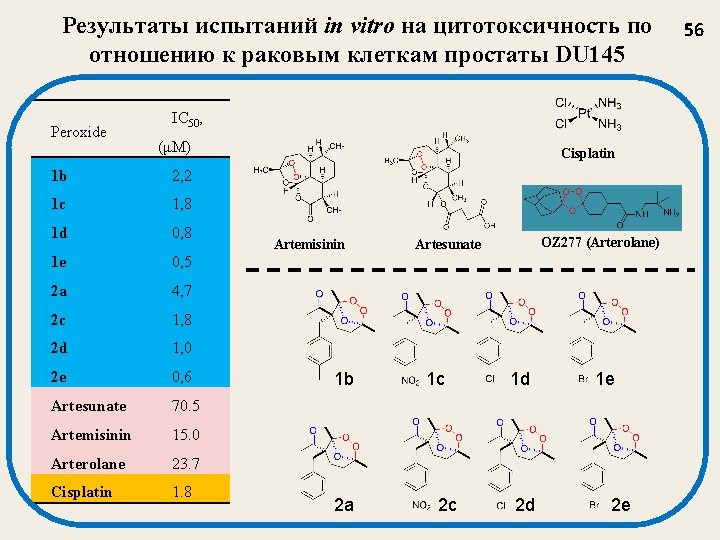
Результаты испытаний in vitro на цитотоксичность по отношению к раковым клеткам простаты DU 145 Peroxide IC 50, (μM) 1 b 2, 2 1 c 1, 8 1 d 0, 8 1 e 0, 5 2 a 4, 7 2 c 1, 8 2 d 1, 0 2 e 0, 6 Artesunate 70. 5 Artemisinin 15. 0 Arterolane 23. 7 Cisplatin 1. 8 Cisplatin Artemisinin 1 b 2 a OZ 277 (Arterolane) Artesunate 1 c 2 c 1 d 2 d 1 e 2 e 56

