Overview of regulatory capacity and NRA priorities based

Overview of regulatory capacity and NRA priorities based on PRAIS data and NRA survey Murilo Freitas, Regulatory Affairs Specialist PANDRH Secretariat VII Pan American Network for Drug Regulatory Harmonization Conference Ottawa, Canada September 5 -7 th, 2013

OUTLINE • Background • Regulatory challenges of the Americas – PRAIS data: Observatory Tool • PANDRH Survey – Methodology – Results – Conclusions

BACKGROUND • On March 2013, during the Ad Hoc meeting, the group requested the development of a survey that identifies current and future regulatory challenges in the Region of the Americas • On April 2013, the Pan American Network for Drug Regulatory Harmonization (PANDRH) Secretariat designed an electronic survey in response to the request of the Ad Hoc group • The survey is divided in three main sections: – Identification of future national regulatory priorities/challenges for the work of PANDRH – Offers of training and NRAs strengthening capacities – Mechanisms of cooperation in the Pan American Region
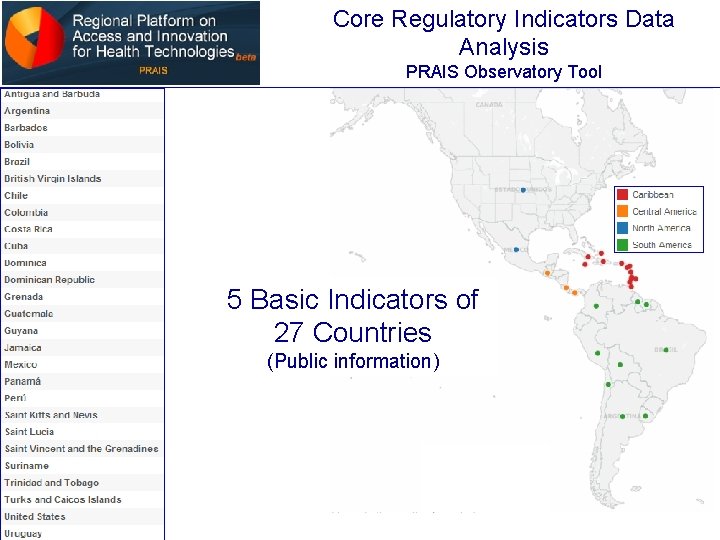
Core Regulatory Indicators Data Analysis PRAIS Observatory Tool 5 Basic Indicators of 27 Countries (Public information)
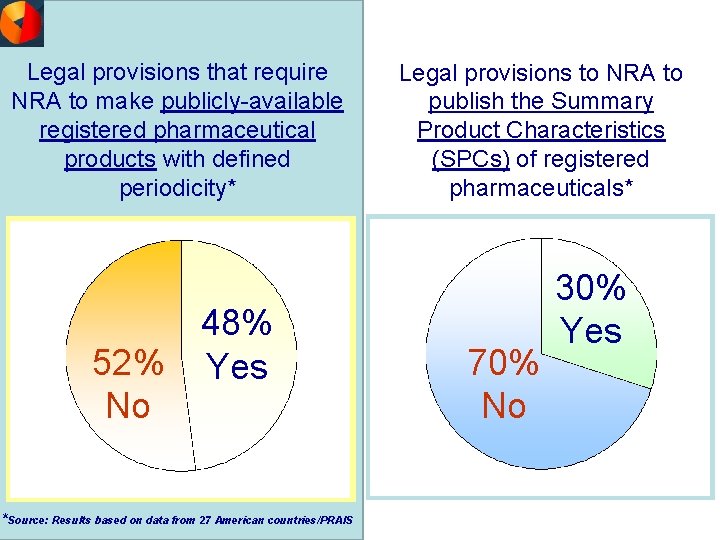
Legal provisions that require NRA to make publicly-available registered pharmaceutical products with defined periodicity* 52% No 48% Yes *Source: Results based on data from 27 American countries/PRAIS Legal provisions to NRA to publish the Summary Product Characteristics (SPCs) of registered pharmaceuticals* 70% No 30% Yes
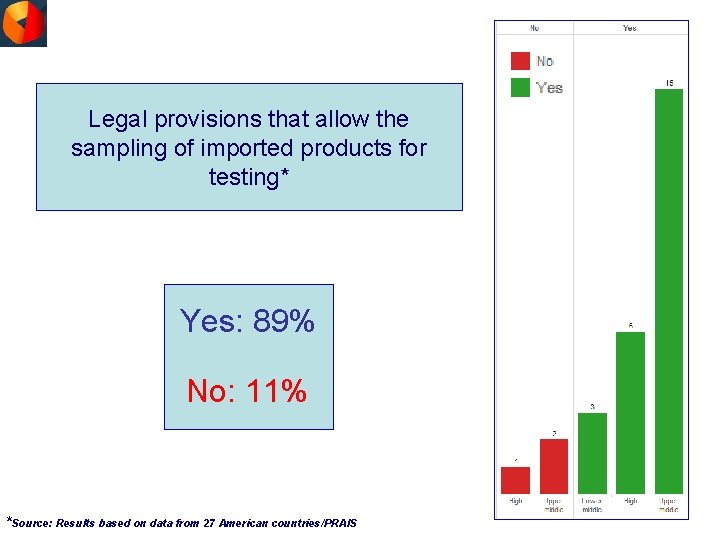
Legal provisions that allow the sampling of imported products for testing* Yes: 89% No: 11% *Source: Results based on data from 27 American countries/PRAIS
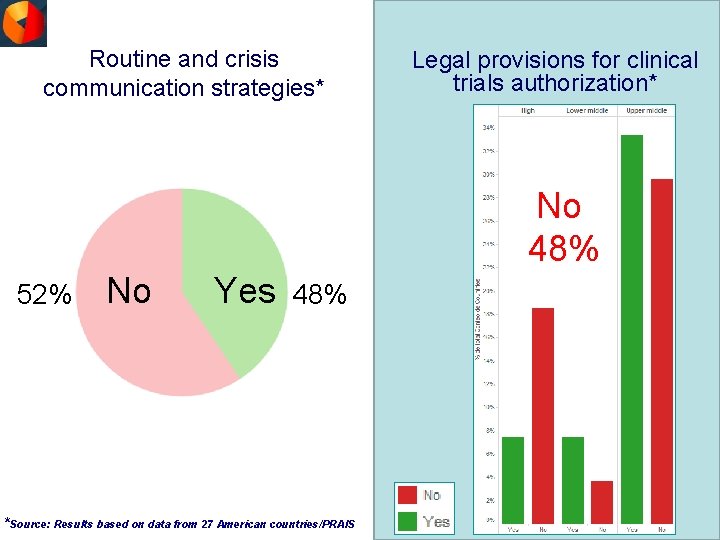
Routine and crisis communication strategies* Legal provisions for clinical trials authorization* No 48% 52% No Yes 48% *Source: Results based on data from 27 American countries/PRAIS

PANDRH Survey Current and future regulatory challenges in the Americas

METHODOLOGY • Development of survey questions based on the identification of general regulatory and regional aspects (by PANDRH Secretariat) • Study type: Ecological/ transversal (unit- cluster of countries) • PANDRH Survey development and dissemination: Electronic platform/tool (11 multiple choice, open-ended and close-ended questions) • Follow up: E-mail and phone calls • Phases – Phase 1 - Internal validation: Testing electronic tool and survey content – Phase 2 - External validation: Pilot study in 5 countries • Countries were selected based on geographical location and language • Participants: High level NRA and International Affairs (if available) representatives – Phase 3 - Launch of survey (June 2013) • Survey sent to remaining 33 countries (English and Spanish) • Graphs and map show data received until August 28, 2013
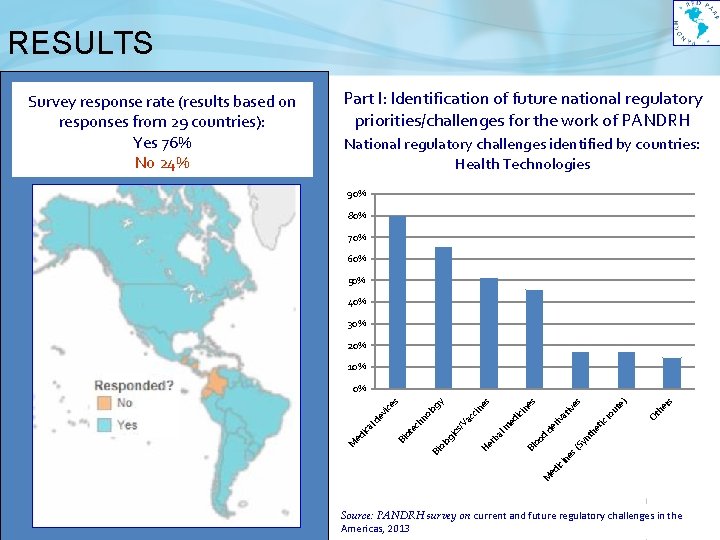
RESULTS Part I: Identification of future national regulatory priorities/challenges for the work of PANDRH National regulatory challenges identified by countries: Health Technologies 90% 80% 70% 60% 50% 40% 30% 20% 10% s er th ro tic he de nt d M ed ici ne s( Sy oo Bl O e) ut ive riv at ed ici He rb al m ac c ics /V og ol Bi s s ne es in og ol ec hn ot Bi ed ica ld ev ice s y 0% M Survey response rate (results based on responses from 29 countries): Yes 76% No 24% Source: PANDRH survey on current and future regulatory challenges in the Americas, 2013
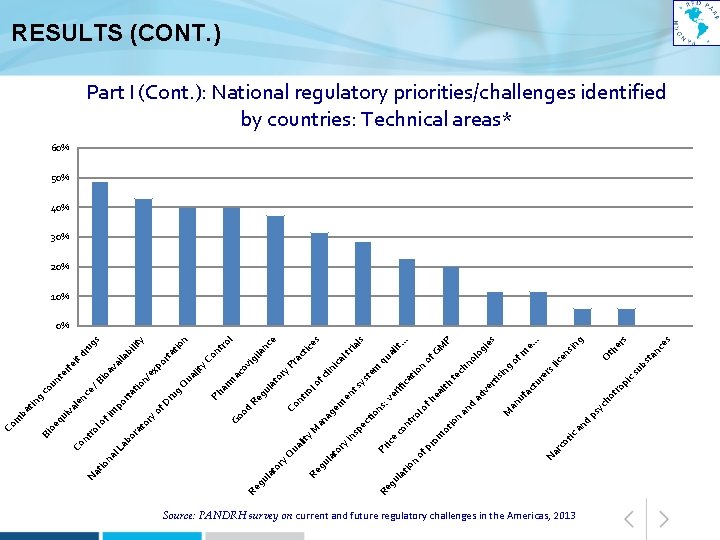
ic an d ot yc Source: PANDRH survey on current and future regulatory challenges in the Americas, 2013 es an c rs th e O st ub cs pi ro ho t g sin . . e. m es gi P GM en of lic g er s ur ct fa of . . lit. qu a no lo ch te rti sin ve ad an u M d an ps io n ot ea lth n m te ys s ice lt ria ls ca cli ni ica tio rif ve ts en fh lo nt ro s: io n ct pe co om pr Na rc of ice Pr or yi ns of l e nc ila ig ra ct or y. P ul at ro l nt Re g Co od ac ov on tro y. C n tio rta y s ug dr ila bi lit po ua lit Q ar m Ph g ru of D ex n/ tio rta ag em an y. M ua lit Q ul at io n Re g or y la t ul at Re g gu Re Go ry at o or po av a io /B it rfe te un co ce g le n ui va lo fi m ro nt l. L ab na tio Na Co oe q Bi tin ba m Co RESULTS (CONT. ) Part I (Cont. ): National regulatory priorities/challenges identified by countries: Technical areas* 60% 50% 40% 30% 20% 10% 0%
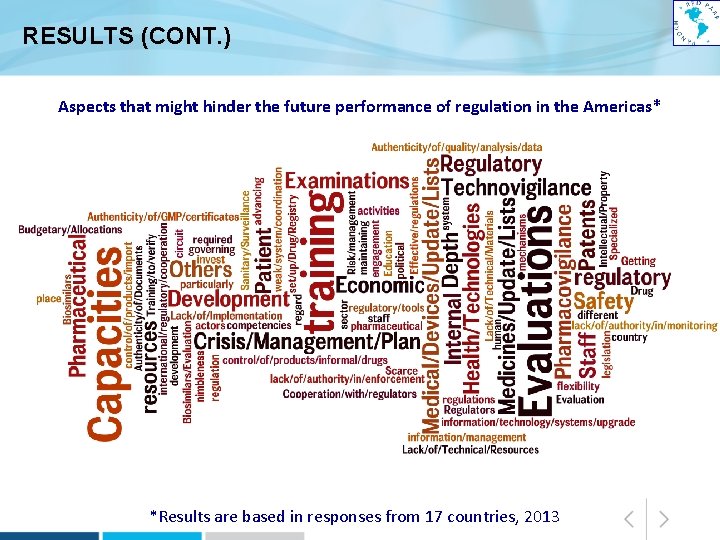
RESULTS (CONT. ) Aspects that might hinder the future performance of regulation in the Americas* *Results are based in responses from 17 countries, 2013
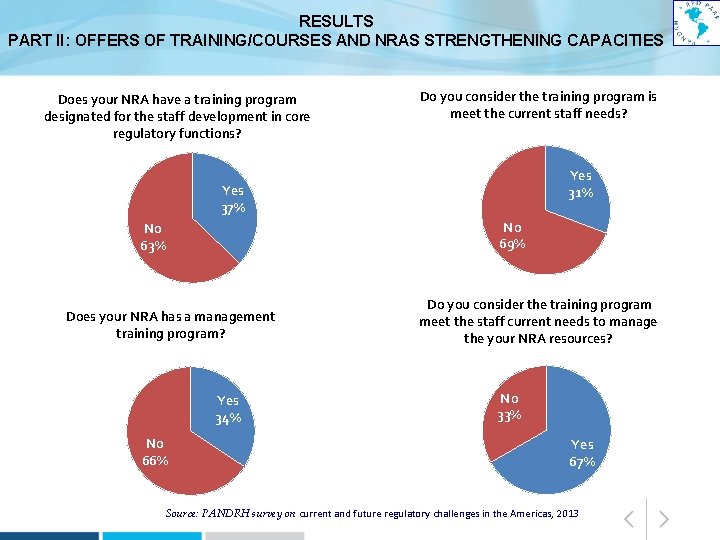
RESULTS PART II: OFFERS OF TRAINING/COURSES AND NRAS STRENGTHENING CAPACITIES Does your NRA have a training program designated for the staff development in core regulatory functions? Do you consider the training program is meet the current staff needs? Yes 31% Yes 37% No 69% No 63% Does your NRA has a management training program? Yes 34% No 66% Do you consider the training program meet the staff current needs to manage the your NRA resources? No 33% Yes 67% Source: PANDRH survey on current and future regulatory challenges in the Americas, 2013

RESULTS PART II: OFFERS OF TRAINING/COURSES AND NRA STRENGTHENING CAPACITIES Comment/suggestions related to training offers and the NRAs current needs* *Results are based in responses from 5 countries, 2013
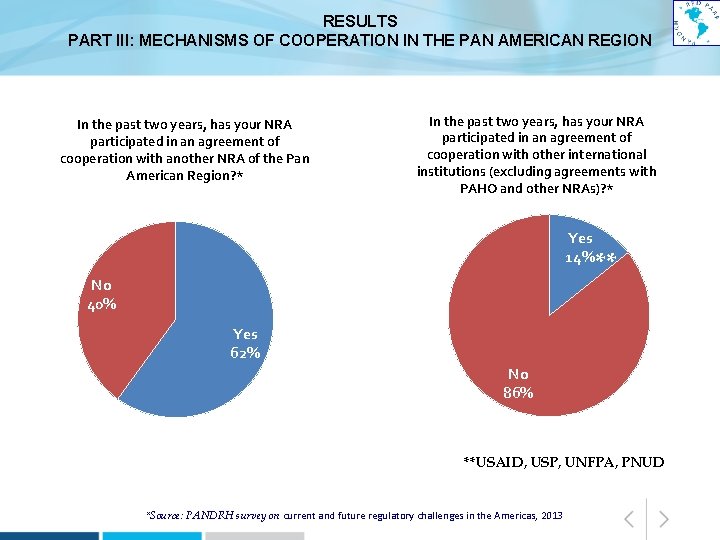
RESULTS PART III: MECHANISMS OF COOPERATION IN THE PAN AMERICAN REGION In the past two years, has your NRA participated in an agreement of cooperation with another NRA of the Pan American Region? * In the past two years, has your NRA participated in an agreement of cooperation with other international institutions (excluding agreements with PAHO and other NRAs)? * Yes 14%** ** No 40% Yes 62% No 86% **USAID, USP, UNFPA, PNUD *Source: PANDRH survey on current and future regulatory challenges in the Americas, 2013

CONCLUSIONS • Future regulatory priorities/challenges for products and functions*: Medical Devices Medicines Counterfeit combat Biotechnologics BE/BD Vaccines Import/export control Herbal Medicines National Lab Quality Control Blood derivatives Pharmacovigilance • Most countries do not have staff training program and those that do have a training program, do not satisfy NRA professionals´ need • Aspects to improve future NRA performance: • • Regulatory public information availability; Capacities/Training/Evaluation; Crisis management plan; and Verification of the authenticity of regulatory official documents. • Current needs: e. g Distance learning *Study limitations: questions do not allow to distinguish countries’ interests/needs from Regional needs

THANK YOU!
- Slides: 17