OVERVIEW OF RANDOMIZED CONTROLLED TRIALS Basic Workshop on

OVERVIEW OF RANDOMIZED CONTROLLED TRIALS Basic Workshop on Randomized Controlled Trials Pragmatic Randomized Controlled Trials in Health Care - Practihc Basic Workshop - Overview

OBJECTIVES Describing: • What is a randomized controlled trial (RCT) • The reasons for conducting RCTs • The main components of an RCT • The main types of clinical trials Practihc Basic Workshop - Overview

CLINICAL TRIAL A prospective study that compares the effects of at least two different interventions (experimental vs. control) in human subjects (Friedman, 1998) • Prospective: follow-up of subjects from a defined moment of their condition • Interventions: – The researcher decides which interventions is allocated to each participant subject – Experimental: prophylactic, diagnostic, therapeutic – Control: against which the experimental is compared – Both are administered in standardized form to modify an outcome in participants Practihc Basic Workshop - Overview
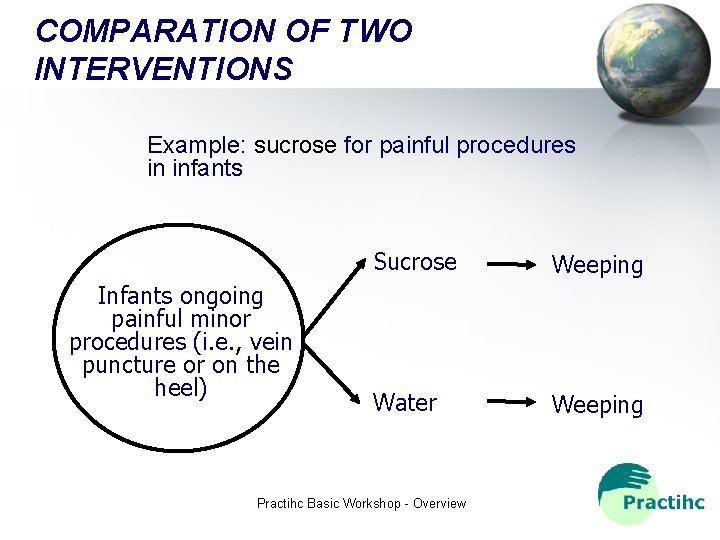
COMPARATION OF TWO INTERVENTIONS Example: sucrose for painful procedures in infants Infants ongoing painful minor procedures (i. e. , vein puncture or on the heel) Sucrose Weeping Water Weeping Practihc Basic Workshop - Overview

CLINICAL TRIAL The best method to determine: • If an intervention really has a presumed effect • The direction and the size of such effect • It separates: – The signal: true effect of the intervention – The noise: other factors, different to the intervention, that may have similar effects Practihc Basic Workshop - Overview

CLINICAL TRIAL Example: sucrose vs. water to reduce pain in infants ongoing venous or heel punctures • Signal: pain difference between both interventions • Noise: other factors: – – – Puncture site Experience of person performing the procedure Infant´s gestational age Presence of other disease (neurological impairment) Concurrent use of drugs (sedatives, analgesics) Practihc Basic Workshop - Overview

¿HOW ARE THE NOISE – SIGNAL ISOLATED? Objective: making the groups submitted to both interventions as similar as possible in all aspects, except by the interventions Infants ongoing painful minor procedures (i. e. , vein puncture or on the heel)) Sucrose Weeping Water Weeping Practihc Basic Workshop - Overview

RESEARCH DESIGNS 1. Experimental studies 2. Observational Studies 2. 1. Descriptive – Case Reports – Case Series 2. 2. Analytic – Cross sectional and Ecologic – Cohort – Case Control Practihc Basic Workshop - Overview

REASONS WHY A CONTROL GROUP IS NEEDED 1. The outcome of interventions or diseases are not predictable 2. Hawthorne effect 3. Regression to the media 4. Placebo effect Practihc Basic Workshop - Overview

NON PREDICTABLE RESULTS • A control group is not required when the outcome after a given exposure is completely predictable ( all or nothing): – Beheading causes dead – A new treatment cures a few rabies cases – Antibiotics reduced pneumonia mortality • However: – No intervention has 100% efficacy – Many diseases may recover spontaneously (without treatment) Practihc Basic Workshop - Overview
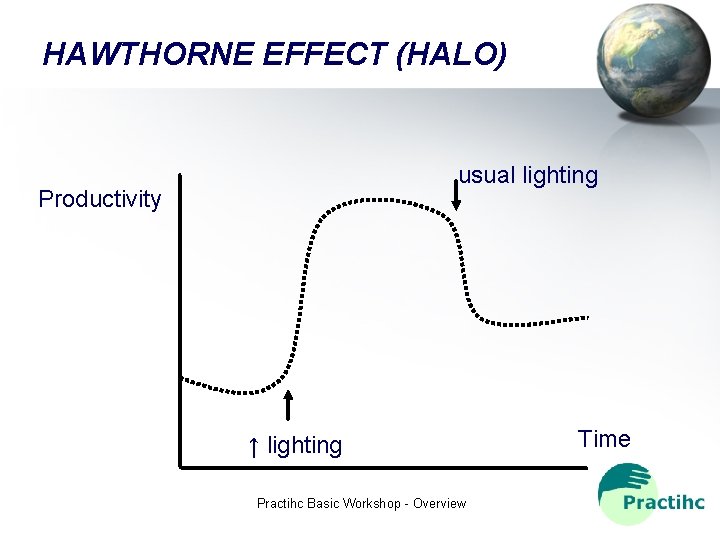
HAWTHORNE EFFECT (HALO) usual lighting Productivity ↑ lighting Practihc Basic Workshop - Overview Time
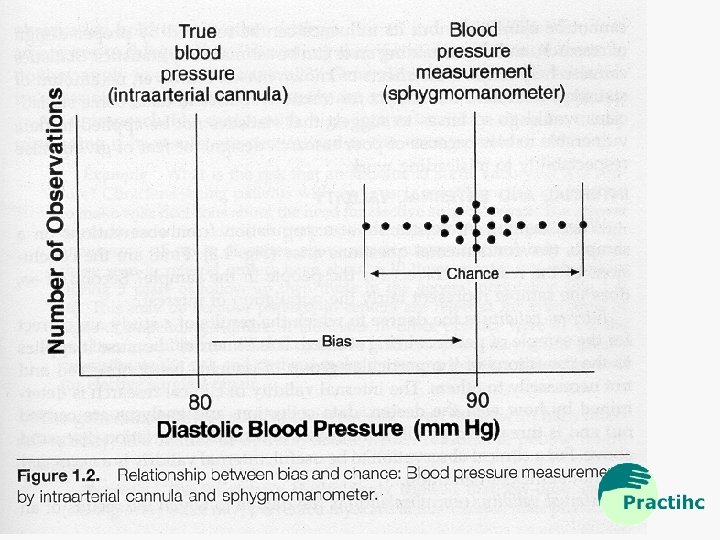
Practihc Basic Workshop - Overview
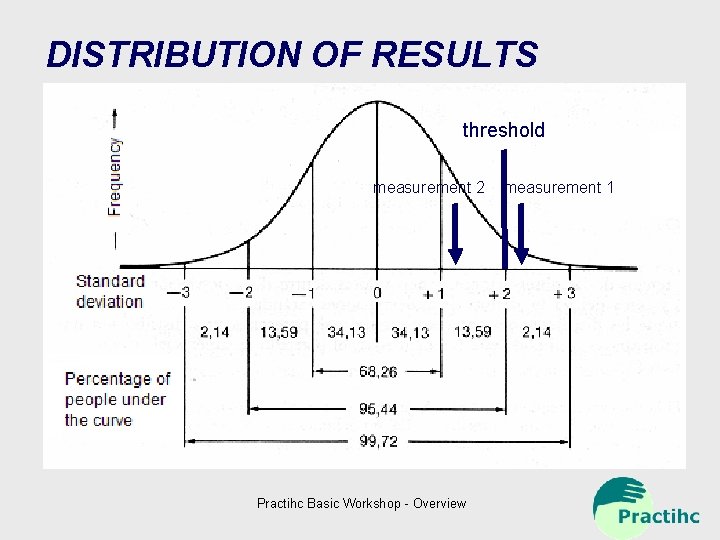
DISTRIBUTION OF RESULTS threshold measurement 2 Practihc Basic Workshop - Overview measurement 1

PLACEBO EFFECT • Effect (usually, but not always, positive) attributed to the expectation that a therapy will have an effect. • The effect is due to the power of suggestion • Placebo: a inert medication or procedure Last JM. A Dictionary of Epidemiology. 1983. New York, Oxford University Press Practihc Basic Workshop - Overview
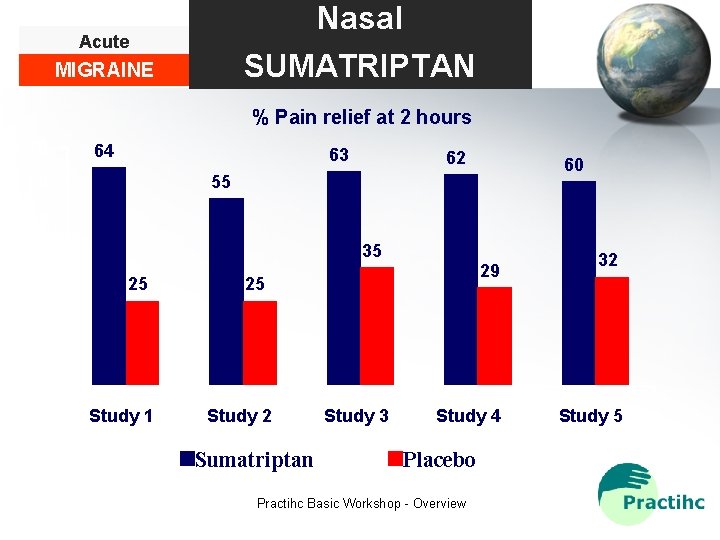
Nasal SUMATRIPTAN Acute MIGRAINE % Pain relief at 2 hours 70 64 63 62 60 55 60 50 35 40 30 25 29 25 32 20 10 0 Study 1 Study 2 Sumatriptan Study 3 Study 4 Placebo Practihc Basic Workshop - Overview Study 5
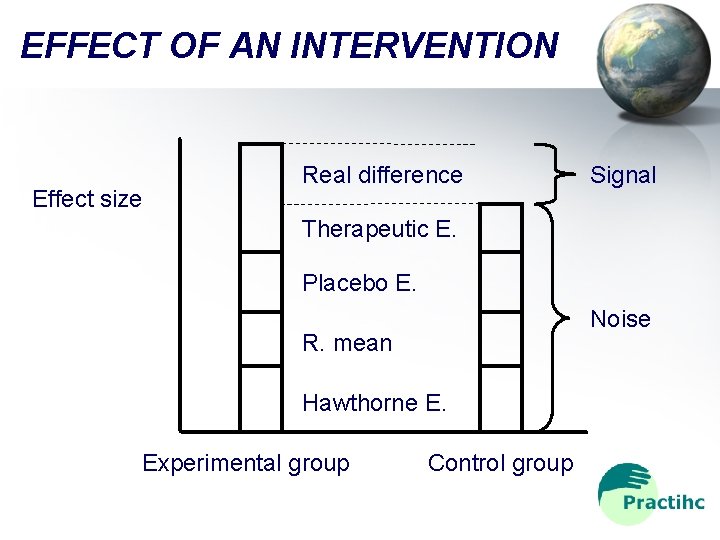
EFFECT OF AN INTERVENTION Effect size Real difference Signal Therapeutic E. Placebo E. Noise R. mean Hawthorne E. Experimental group Control group
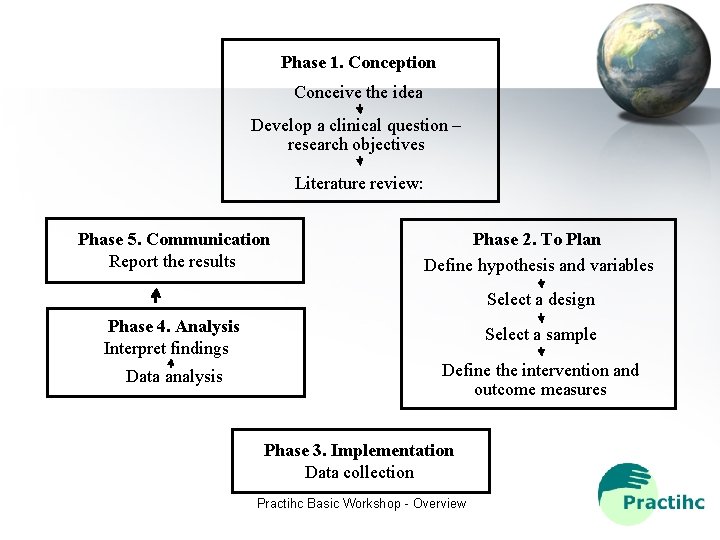
Phase 1. Conception Conceive the idea Develop a clinical question – research objectives Literature review: Phase 5. Communication Report the results Phase 2. To Plan Define hypothesis and variables Select a design Phase 4. Analysis Interpret findings Select a sample Data analysis Define the intervention and outcome measures Phase 3. Implementation Data collection Practihc Basic Workshop - Overview

TRIAL PHASES 1. Clinical research question 2. Type of trial 3. Selecting trial population – – – Inclusion/exclusion criteria Sampling Sample size 4. Assignment – – Stratification Randomized vs. non-randomized Practihc Basic Workshop - Overview

TRIAL PHASES 5. Intervention – Experimental – Control (other, placebo, nothing) 2. Follow up and measurement – Baseline (at entry) – At the end of follow-up (open, blinded) – Confounders 3. Analysis of results – Comparability of groups – Outcomes – Confounders Practihc Basic Workshop - Overview
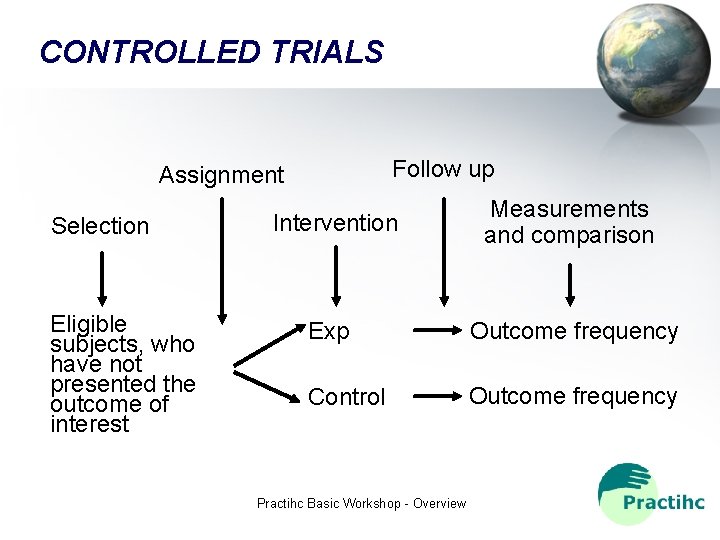
CONTROLLED TRIALS Follow up Assignment Selection Eligible subjects, who have not presented the outcome of interest Intervention Measurements and comparison Exp Outcome frequency Control Outcome frequency Practihc Basic Workshop - Overview
Practihc Basic Workshop - Overview
Practihc Basic Workshop - Overview

TYPE OF TRIALS • • Conventional Cross-Over Equivalence Factorial Assignment by groups (cluster) Sequential Individual (N = 1) Practihc Basic Workshop - Overview
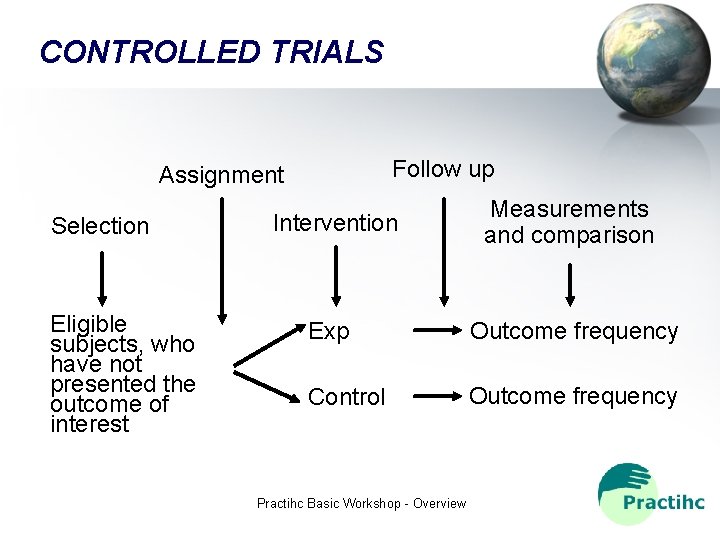
CONTROLLED TRIALS Follow up Assignment Selection Eligible subjects, who have not presented the outcome of interest Intervention Measurements and comparison Exp Outcome frequency Control Outcome frequency Practihc Basic Workshop - Overview
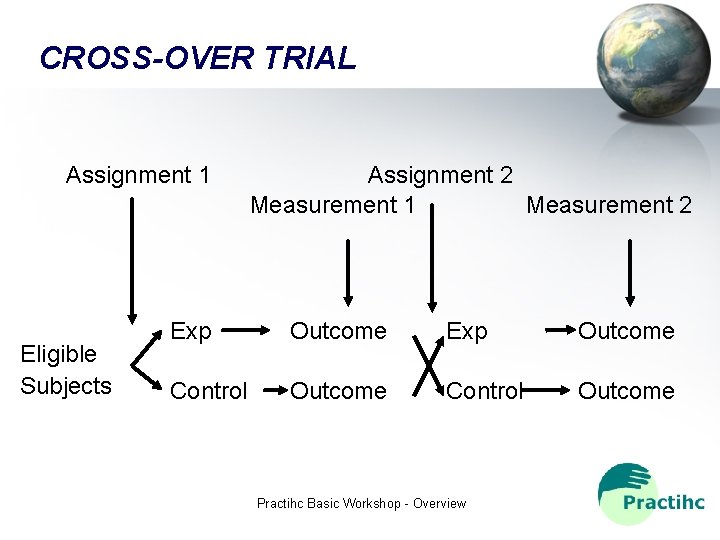
CROSS-OVER TRIAL Assignment 1 Eligible Subjects Assignment 2 Measurement 1 Measurement 2 Exp Outcome Control Outcome Practihc Basic Workshop - Overview

EQUIVALENCE TRIALS • • Conventional treatment produces good results The new treatment has some advantages different to the efficacy: – Cheaper – Safer – More convenient or comfortable • • The new treatment would be of choice even if it is as effective as the conventional Requires special planning: if it is not possible to demonstrate differences between two treatments, it does not mean they are the same. Practihc Basic Workshop - Overview

CONCLUSSIONS • The randomized controlled trial is the best design to assess the effect of an experimental intervention • It compares the experimental intervention with a control in subject groups whose only difference is the intervention received • Requires careful planning and conduction to avoid the interference of bias and chance (noise) Practihc Basic Workshop - Overview
- Slides: 27