Overview of electrochemistry 1 Equilibrium electrochemistry no current

Overview of electrochemistry 1 Equilibrium electrochemistry (no current flows) Dynamic electrochemistry (current does flow) Homogeneous Thermodynamics of electrolyte solutions: • electrolytic dissociation • thermodynamics and activities of ions Conductance of electrolytes: • electrolytic conduction • mobilities of ions • Kohlrausch laws Heterogeneous Thermodynamics of electrodes and Galvanic cells: • electrochemical cells • cell and electrode potential • types of electrodes Kinetics of electrode reactions: • exchange current • overpotential • Tafel equation • Butler-Volmer equation • electrolysis • batteries

HOMOGENOUS DYNAMIC ELECTROCHEMISTRY Simultaneous or coupled transport phenomena I. Conductivity of electrolytes (κ, Λmo) • law of the independent migration of ions: • Λ m = ν + λ + + ν –λ – II. Strong electrolytes: • Kohlrausch law: Λm = Λmo – Kc½ III. Weak electrolytes: (α < 1, α = Λm/Λmo) • Ostwald’s dilution law IV. Mobilities of ions, ion transport number

I. Conductivity of electrolytes Ionic conductivity: Ohm’s law is valid: Conductance G is the reciprocal of Rres: as T increases, do does κ (opposite to metals). Solution: κ conductivity: (l: length of cell, A: surface, C: cell constant) Concentration is important, molar conductivity is used: The limiting value of Λm at infinite dilution is Λmo (limiting molar conductivity).

I. Conductivity of electrolytes The limiting value of Λm at infinite dilution is Λmo (limiting molar conductivity). The conductivity of the electrolyte is obtained by adding the conductivities of ions: law of the independent migration of ions: � λ+ and λ-: limiting molar conductivities of (individual) cations and anions � ν+ and ν-: stoichiometric number of the cation and anion
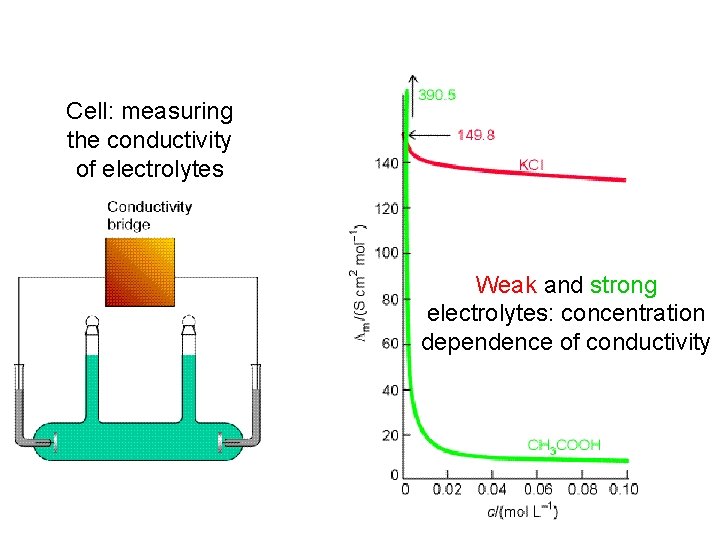
Cell: measuring the conductivity of electrolytes Weak and strong electrolytes: concentration dependence of conductivity
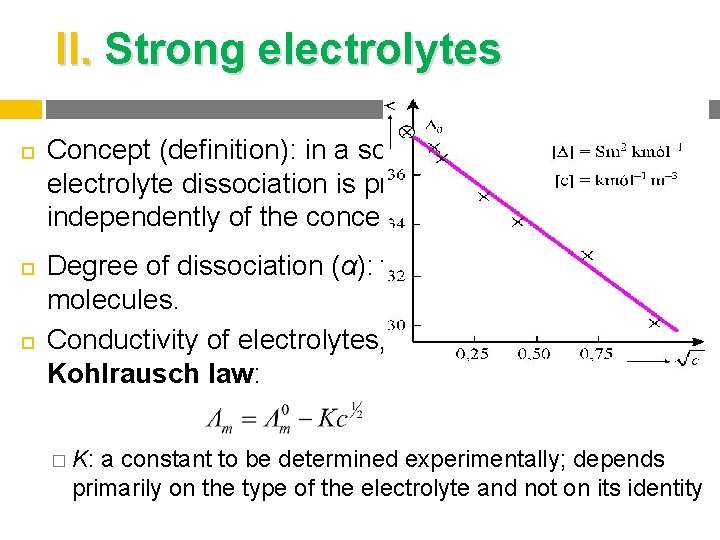
II. Strong electrolytes Concept (definition): in a solution of a strong electrolyte dissociation is practically complete independently of the concentration, so α = 1. Degree of dissociation (α): the ratio of dissociated molecules. Conductivity of electrolytes, Kohlrausch law: � K: a constant to be determined experimentally; depends primarily on the type of the electrolyte and not on its identity
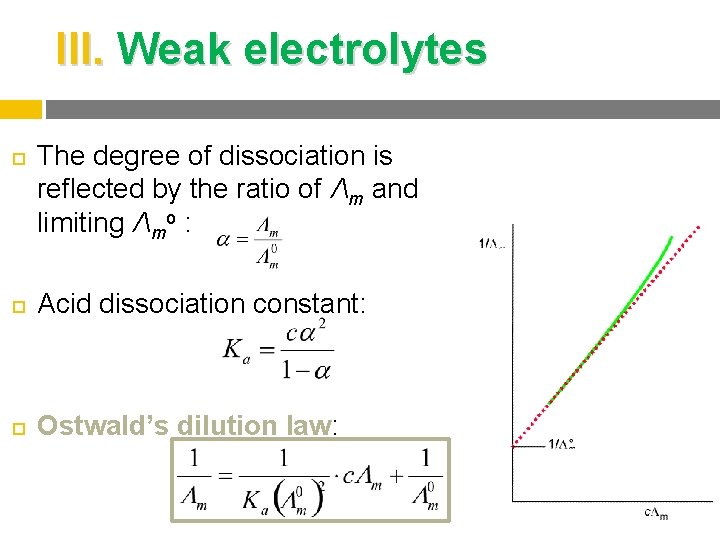
III. Weak electrolytes The degree of dissociation is reflected by the ratio of Λm and limiting Λmo : Acid dissociation constant: Ostwald’s dilution law:

IV. Mobilities of ions drift speed(s) mobilities of ions (u) frictional coefficient(f) mobility (u) and conductivity (λ) transport numbers (t+ and t-) determination methods for transport numbers

IV. Mobilities of ions Molecular view: ions with radius ahydr are accelerated in a medium of viscosity η by an electric force of Fel, but this is balanced by a counter-force of friction Ffric (Stokes law). A stationary state is established with constant drift speed: The drift speed of the ion is the product of electric field ε and ion mobility u. The value of u:
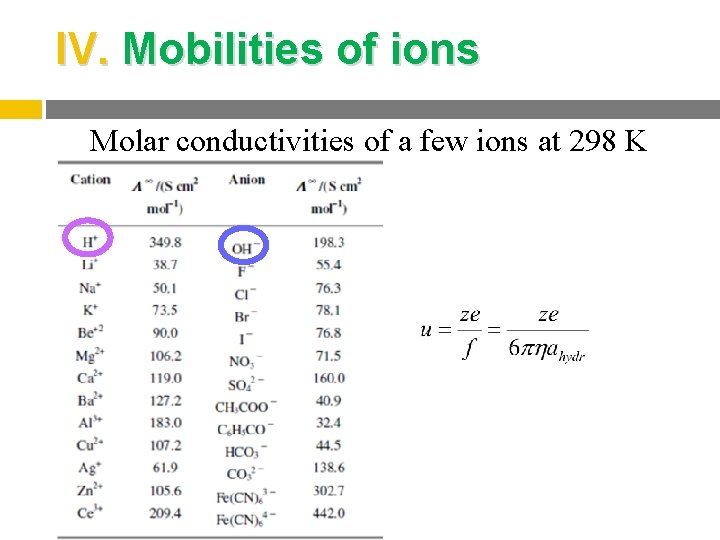
IV. Mobilities of ions Molar conductivities of a few ions at 298 K

Ion transport number Ion transport number: the fraction of current transported by the ion: Obviously: t+ + t- = 1 Determination methods for transport numbers: moving boundary method, � Hittorf’s method (measuring concentration change in electrode compartments), � comparison of cell potentials with and without liquid junction. �

Overview of electrochemistry 12 Equilibrium electrochemistry (no current flows) Dynamic electrochemistry (current does flow) Homogeneous Thermodynamics of electrolyte solutions: • electrolytic dissociation • thermodynamics and activities of ions Conductance of electrolytes: • electrolytic conduction • mobilities of ions • Kohlrausch laws Heterogeneous Thermodynamics of electrodes and Galvanic cells: • electrochemical cells • cell and electrode potential • types of electrodes Kinetics of electrode reactions: • exchange current • overpotential • Tafel equation • Butler-Volmer equation • electrolysis • batteries

13 HETEROGENEOUS DYNAMIC ELECTROCHEMISTRY 1. 2. 3. Observations (current density–overpotential, exchange current(s), Tafel equation, . . . ) Interpretation of the observations (the double layer and its models, Galvani and Volta potentials; kinetics of electrode reactions, Butler– Volmer equation; lower and upper limits of overpotential, polarization, limiting current) Practical electrochemistry (working galvanic cells, accumulators, fuel cells, polarography, voltammetry, electrolysis, corrosion)

Kinetics of electrode processes (heterogeneous dynamic electrochemistry) 14 In equilibrium electrochemistry (thermodynamics of electrolytes, Galvanic cells and electrodes), states of equilibrium are described and the possibility of changes. No current flows in such systems, the Galvanic cell does no useful work, no change occurs on the electrodes. In non-equilibrium electrochemistry [dynamic electrochemistry], dynamic processes are described: the system is not in equilibrium: current flows in the solutions, substances deposit or dissolve on electrodes. In addition to equilibrium electrode potential, overpotential occurs.

Kinetics of electrode processes (heterogeneous dynamic electrochemistry) 15 In dynamic electrochemistry, the voltage and current of spontaneous reactions in Galvanic cells or those of forced reactions in electrolysis are described in a temporal and spatial manner. Understanding of the mechanism of electrode processes is necessary for both electrolysis and Galvanic cells.

Kinetics of electrode processes (heterogeneous dynamic electrochemistry) 16 Macroscopic measurements give important information about the processes on electrode surfaces and also contribute to understanding phenomena occurring at the molecular level.

1. Observations 17 Quantities in dynamic electrochemistry: a) Current-related: The definition of reaction rate in a heterogeneous process: product flux = k[particle] In the two electrode processes: vox = kc[Ox], and vred = ka[Red] Characterization of the on an electrode: j current density (j) = current / surface [A cm-2] [Remark: there is Ohmic resistance in systems like this (it is readily measurable). ]

1. Observations 18 Quantities in dynamic electrochemistry: b) Potential / voltage related: The original equilibrium electrode potentials (E) are not valid in a working Galvanic or electrolytic cell. The differences are formally described as: On electrodes: η overpotential (polarization potential) Galvanic cells: actual cell potential ( < E) [not necessarily constant, depends on the current] Electrolytic cells: η overpotantial ( > E) [set by the experimenter based on the objectives] All of these quantities are measurable – similarly to E.

1. Observations 19 Quantities in dynamic electrochemistry: c) Current density, exchange current(s): Observations show that current density j changes when the overpotential η is changed: increase and decrease, even a sign change are possible. On all electrodes , cathodic jc = Fkc[Ox] And anodic ja = Fka[Red] current densities are. The actual (and measurable) current density j is the difference of these two: if ja > jc, then j > 0, the net current is anodic, if ja < jc, then j < 0, the net current is cathodic, if ja = jc, then j = 0, the net current is zero.
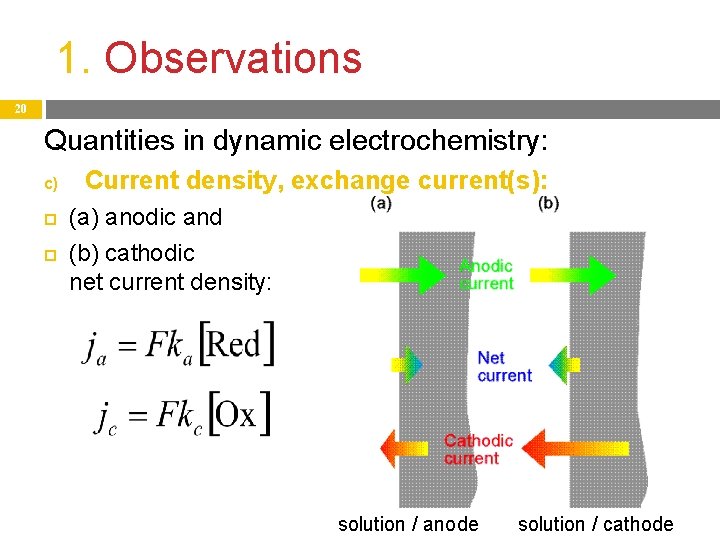
1. Observations 20 Quantities in dynamic electrochemistry: c) Current density, exchange current(s): (a) anodic and (b) cathodic net current density: solution / anode solution / cathode

1. Observations 21 Quantities in dynamic electrochemistry: d) Relationship between the overpotential and current density, the Tafel equation: At small overpotential, current density increases linearly with the overpotential: j = j 0 f η (f = F/RT) At intermediate overpotential, the relationship is exponential (logarithmic). This is the observed Tafel equation: , At large overpotential, current density reaches an upper limit, this is the limiting current. If η < 0, then j < 0: ln(−j) = ln j 0 – α f η
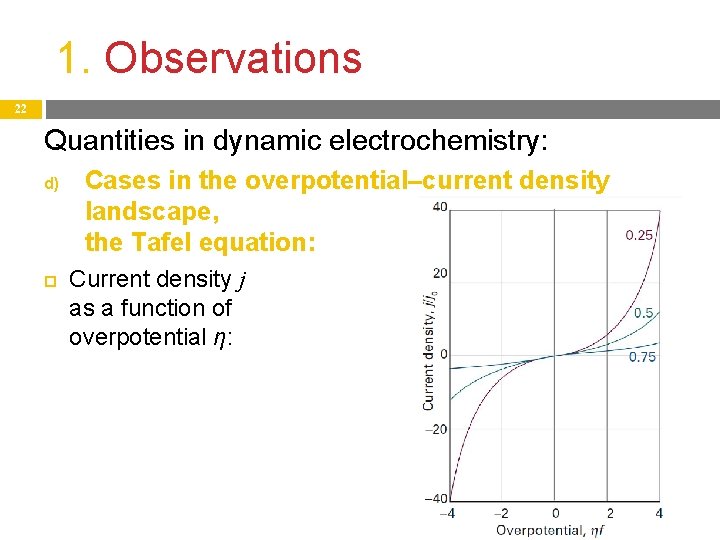
1. Observations 22 Quantities in dynamic electrochemistry: d) Cases in the overpotential–current density landscape, the Tafel equation: Current density j as a function of overpotential η:

2. Interpretation of observations 23 To interpret the observations, we need: an understanding of the structure of the surface boundary between the electrode and the electrolyte: a description of the electric double layer and the Nernst adsorption layer. the mechanisms of the rate limiting step: the activation free energy Δ#G of the charge transfer step and its possible dependence on the overpotential η. to clarify the role of diffusion and activation. Recognizing the relationship between η and Δ#G is the core issue in the kinetic description of electrode processes.

2. Interpretation of observations 24 a) The structure of the electrode /electrolyte boundary: In the bulk solution, the flow of the solute toward (and from) the surface is caused by (fast) convection (stirring). On a solid-liquid boundary, there is always a highly adhesive liquid layer (not only for electrolytes and not only on electrodes) : this is the Nernst δ diffusion layer. In this layer, transport of matter is only possible by (slow) diffusion, so a concentration gradient forms in it. There is no specific structure in it. Its width depends on the intensity of stirring or rotation – 10 -3 -10 -2 mm. [On a rotating disc electrode: δ = D 1/3 η 1/6 ω-1/2 ].
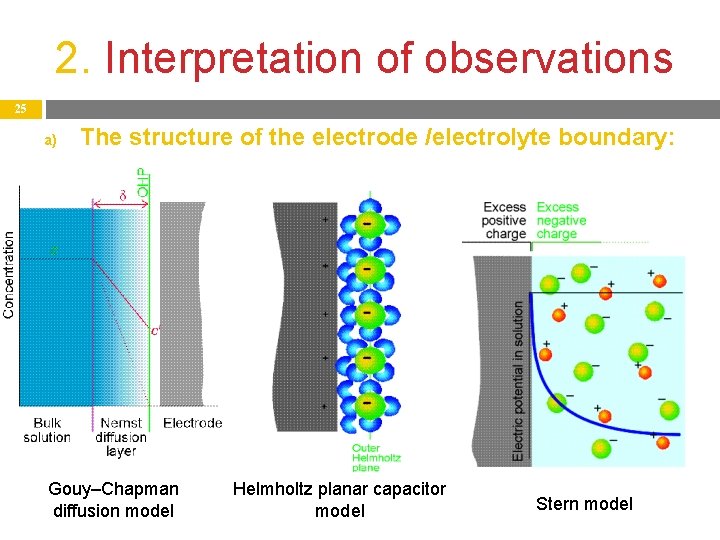
2. Interpretation of observations 25 a) The structure of the electrode /electrolyte boundary: Gouy–Chapman diffusion model Helmholtz planar capacitor model Stern model
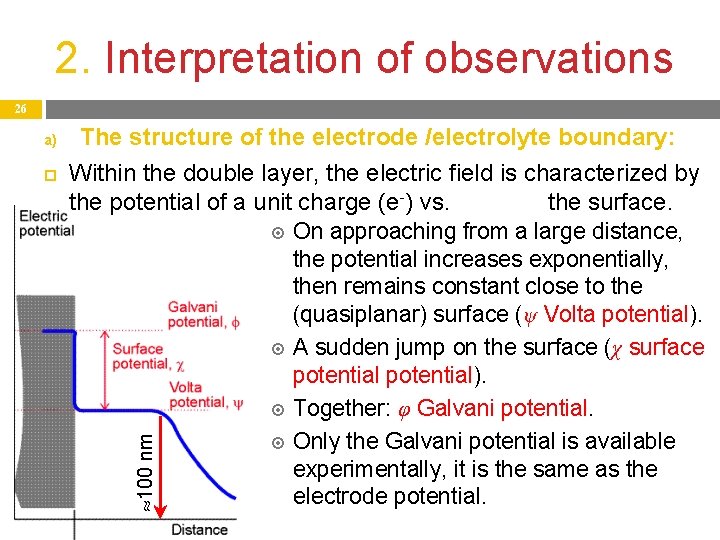
2. Interpretation of observations 26 The structure of the electrode /electrolyte boundary: Within the double layer, the electric field is characterized by the potential of a unit charge (e-) vs. the surface. On approaching from a large distance, the potential increases exponentially, then remains constant close to the (quasiplanar) surface (ψ Volta potential). A sudden jump on the surface (χ surface potential). Together: φ Galvani potential. Only the Galvani potential is available experimentally, it is the same as the electrode potential. 100 nm a)

2. Interpretation of observations 27 b) 1. 2. Kinetics of electrode processes, the Butler–Volmer equation: If in the electrode reaction Ox + e- = Red (cathodic reduction), the electrode becomes more positive (is polarized), the activation free energy of the electrode process increases: In a Red – e- = Ox oxidation (anodic) process, the effect of polarization: With these two modified values of activation free energy, the net current density is obtained, which connects j and η. This is called the Butler–Volmer equation:
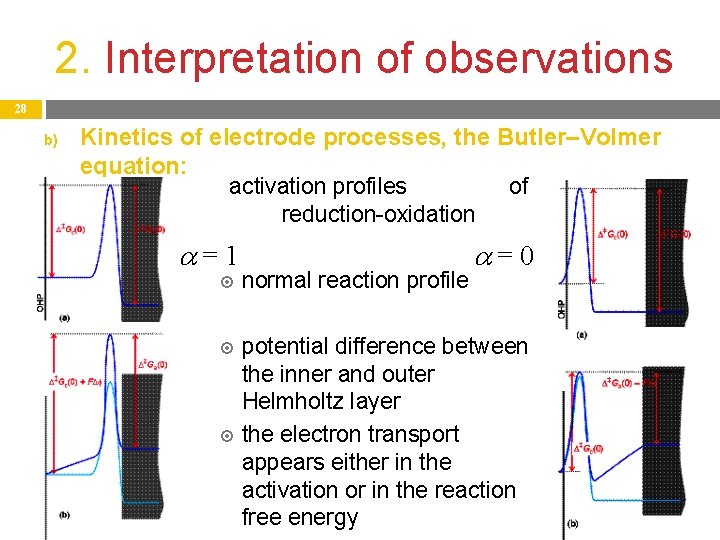
2. Interpretation of observations 28 b) Kinetics of electrode processes, the Butler–Volmer equation: activation profiles of reduction-oxidation =1 normal reaction profile =0 potential difference between the inner and outer Helmholtz layer the electron transport appears either in the activation or in the reaction free energy

2. Interpretation of observations 29 b) Kinetics of electrode processes, the Butler–Volmer equation: Without any overpotential (η = 0), the formula gives the exchange current density j 0 arising from the equilibrium cell potential Ecell without current: j 0 = j a = – j c Exchange current density j 0 is not measurable directly, but can be extrapolated based on the Tafel equation (η → 0). The introduction of j 0 gives a simpler from for the Butler– Volmer equation:

2. Interpretation of observations 30 c) Limits of the overpotential: The lower limit of overpotential: � At low overpotentials of η << 0. 01 V, i. e. fη << 1, series expansion (ex = 1 + x +…) gives: j = j 0 [1 + (1 – α) fη + …– 1 – (– αfη) –…] ≈ j 0 fη � Ohm’ law is valid in this regime, and – in agreement with the observations – a linear relationship is valid between j and η.

2. Interpretation of observations 31 c) Limits of the overpotential: Upper limit of the overpotential: � At η > 0. 12 V (intermediate overpotential), the second term in the Butler–Volmer equation becomes negligible, so j = j 0 e(1−α)fη, i. e. : ln j = ln j 0 + (1–α)fη. � At η < – 0. 12 V (intermediate cathodic overpotential), the first term becomes negligible, so j=– j 0 e –αfη, i. e. : ln (–j) = ln j 0 – αfη. These are the same as the Tafel equation. The transfer coefficient α and exchange current density j 0 can be determined in this way.

2. Interpretation of observations 32 d) Limiting current: At high overpotentials, the Nernst diffusion layer quickly becomes depleted because of the fast charge transfer (metal deposition). A further increase in η cannot increase the current density any more, as diffusion across the Nernst layer becomes the limiting step. This is characterized by the jlim limiting current density. Its value only depends on on the concentration gradient dc/dx, diffusion constant D, and layer thickness δ:

PROCESSES AT SOLID SURFACES – HETEROGENEOUS REACTIONS

HETEROGENEOUS REACTIONS • (So far, we were talking about homogeneous reactions in g and l phases. ) • Heterogeneous reactions occur at the surface between two phases. • The change can be physical or chemical, • may involve nucleation or may be nucleation-free, • may be catalytic or non-catalytic. • Combinations: s-l, s-g, s-s, l-l (here are just examples, details will be discussed in colloid chemistry): • melting, physical dissolution, adsorption (clarification) • leaching, surface treatment (oxidation, phosphating) • electrode reactions (later) • corrosion • precipitation, crystallization, etc. 34

SURFACE PHENOMENA - APPLICATION EXAMPLES • Heterogeneous (contact) catalysis (gas/solid) is very important in the chemical industry (> 90%) [kinetics] • Methods of separation techniques [equilibrium]: • chromatography (column, paper, thin layer, gas, liquid: many versions), • ion exchangers (natural [zeolites, etc. ] and artificial resins), • carbo medicinalis, soil tillage and wastewater treatment - lots of applications. • Electrolysis, galvanic cells. 35

OVERVIEW: I. Surface a) concept, nature; b) formation, growth, size; c) composition, structure. Measuring methods. II. Adsorption a) characteristics: physisorption and chemisorption b) extent of adsorption: equilibrium adsorption isotherms: Langmuir, BET and other equations c) rate: kinetics of adsorption, desorption, and surface diffusion III. Catalytic activity of surfaces a) heterogeneous catalysis b) adsorption and catalysis. Langmuir-Hinshelwood and Eley. Rideal mechanisms. 36

I. SURFACE a) the concept of surface, its nature • macroscopic experience – geometric size is unrealistic (Na. Cl versus chalk) • today: defined in an atomic-molecular dimension • the surface of crystals is examined and used (amorphous materials can also be studied) • crystals consist of atoms, ions or molecules: atomic, ionic, molecular lattices • crystals are composed of unit cells, based on which there are 7 crystal systems, • the geometric structure of the surface, the distances of the atoms, etc. are important. 37
I. SURFACE a) the concept of surface, its nature • The position of the atoms is varies a lot: on a flat crystal surface: like a tray of oranges in a zeolite type material 38
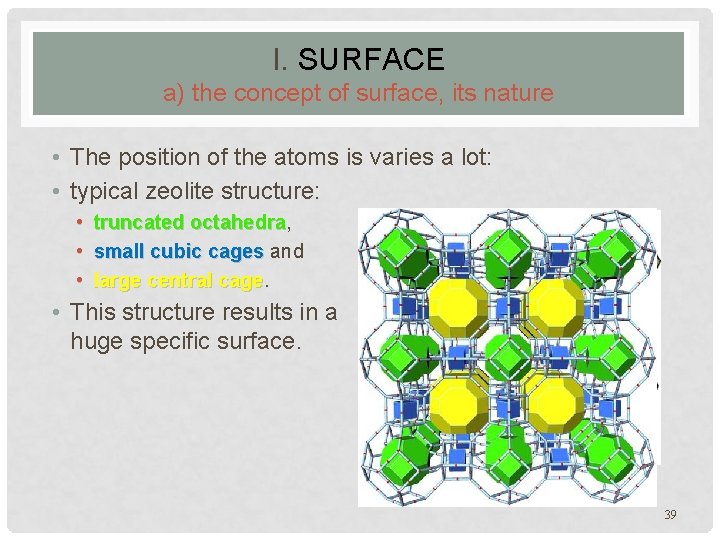
I. SURFACE a) the concept of surface, its nature • The position of the atoms is varies a lot: • typical zeolite structure: • truncated octahedra, octahedra • small cubic cages and • large central cage • This structure results in a huge specific surface. in a zeolite type material 39

I. SURFACE b) formation and growth of the surface • The surface is formed from the crystal nuclei which has a size of only some atoms (molecules). It can grow from a melt, a solution or a gas. • Forms (can be grown) a: • perfect single crystal, • but general (and even more important) are the real crystals with defects (often polycrystallic materials). • It can be made by grinding (breaking the crystal). • [About the determination of the surface area, see later. ] 40

I. SURFACE b) formation and growth of the surface • Key concept: dislocation (deviation from perfect). • It is so important that it is often induced artificially (e. g. a perfect Ag. Br crystal is not photo-sensitive). • The difference between the flat layers, the edge, and the peak is obvious: the environment of the atoms / ions is different, resulting in a different potential energy. • Types of defects (often mixed): • geometric and chemical defects, • which can be located • on the surface or inside the bulk crystal. 41

I. SURFACE b) formation and growth of the surface • Geometric defects: on the surface or inside the crystal bulk • Some types of surface geometry defects: • (terrace – not a defect!) • step • kink • peak • adatom 42
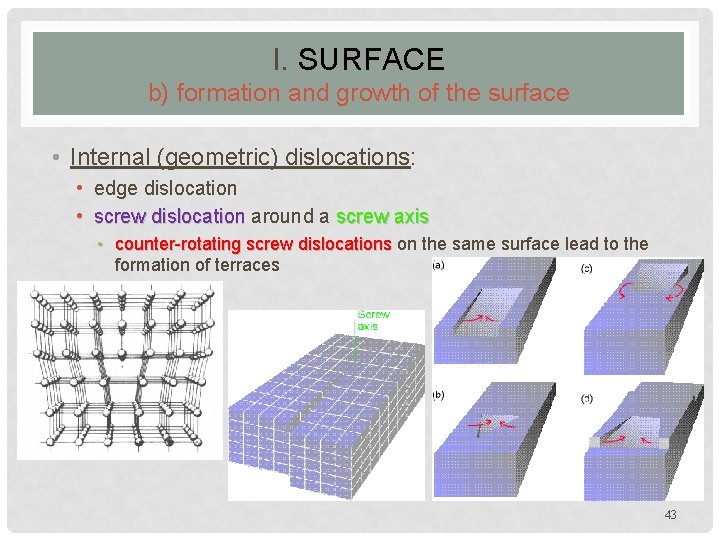
I. SURFACE b) formation and growth of the surface • Internal (geometric) dislocations: • edge dislocation • screw dislocation around a screw axis • counter-rotating screw dislocations on the same surface lead to the formation of terraces 43

I. SURFACE b) formation and growth of the surface • Chemical defects: some deviation in composition • simple hole = point defect = vacancy: an atom/ion is missing from the lattice position • interstitial is an atom/ion that occupies a place outside the normal lattice position (results in lattice distortion around the defect) • e. g. for photoemulsions: precipitating Ag+ with a lot of Br−and traces of I − • e. g. semiconductors are made with artificial impurities: they are diffused, infiltrated, etc. into the lattice (solid state physics) 44

I. SURFACE c) composition and structure of the surface • The fresh surface of the solid material is "immediately" covered by gas molecules – by fast collisions. • This can be calculated from the kinetic theory of gases: • In air (M ~ 29 g/mol, 1 atm, 25 C): ZW = 3× 1027 m− 2 s− 1. • Since there are 1019 atom/m 2, each surface lattice atoms has 108 collisions/s, • that is, the fresh crystal lattice surface is practically immediately covered by the gas molecules (they are 45 adsorbed on the surface).

II. ADSORPTION a) characteristics • Adsorption is a quantitative surplus on the surface and at the same time – by some kind of interaction – causes a "quality" (structural, energetic) change. • CO adsorbed on (111) platinum surface: a) at low coverage, the original lattice structure remains b) at higher coverage, a metastable metal lattice develops [Absorption is enrichment (surplus) in another phase, not on the surface. ] 46

II. ADSORPTION a) characteristics • Definitions of adsorption: • adsorbent: the adsorbing solid (with large surface) • adsorbate: the substrate that adsorbs on the surface • Desorption: a process opposite to adsorption; the removal of the adsorbate from the surface (adsorbent). • Quantitative characteristics or extent of adsorption is the fractional coverage: • Θ changes during the adsorption process and reaches a state of equilibrium, a value depending on the conditions (substances, p, T). Θ = V/Vmax (Vmax for 1 layer) • Rate of adsorption: change of Θ in time. 47

II. ADSORPTION a) characteristics Adsorption is characterized by the nature and strength of the surface bonding. Based on this, there are: physisorption • small Δad. Hθ [k. J/mol] < 0 • long range • multilayer • non-specific • no change in molecular structure • mobile molecules • van der Waals interaction chemisorption • • • large Δad. Hθ <<0 short range monolayer specific change in molecular structure • localized (bonded) molecules • chemical [covalent] bond 48

II. ADSORPTION a) characteristics • Schematic representation of the two types of adsorption (the figures refer to a similar phenomenon, but also reflect the essence of physi- and chemisorption). • Physisorption(P) means greater distance from the surface and less bond energy, • the adsorbate, which is closer to the surface and more strongly bound to it, is characteristic of chemisorption (C). 49

II. ADSORPTION b) extent of adsorption • Reversible equilibrium process (reverse direction: desorption): a dynamic equilibrium between the gas phase adsorbent and the adsorbate. • The extent of equilibrium (extent of adsorption) depends on: • the chemical identity of the surface and the gas, • the pressure p and • the temperature T. • Equilibrium description: Adsorption isotherm (how Θ changes with pressure at constant temperature) • Langmuir-, BET-, Temkin-, Freudlich isotherms • Irreversible adsorption: the equilibrium is shifted right!? 50

II. ADSORPTION b) extent of adsorption • Dependence of equilibrium on gas pressure. • Derivation of the Langmuir adsorption isotherm: (This applies to monolayer chemisorption - multilayer physisorption is described by the BET isotherm. ) • It is based on four assumptions: • adsorption is a reversible equilibrium process, • it cannot proceed beyond monolayer coverage, • all sites of the surface are equivalent, • The ability of a molecule to adsorb at a given site is independent of the occupation of neighboring sites (i. e. there are no interactions between the adsorbed molecules). • These assumptions are often valid entirely, but sometimes 51 only partially.

II. ADSORPTION b) extent of adsorption • The dynamic equilibrium is created by the adsorption (ka) and desorption (kd) processes: A(g) + M(s, surface) ⇌ AM(s, surface) • Rate of adsorption ( uncovered surface): [N = total number of sites) • Rate of desorption ( covered surface): • In equilibrium, the two rates are equal, so: where 52
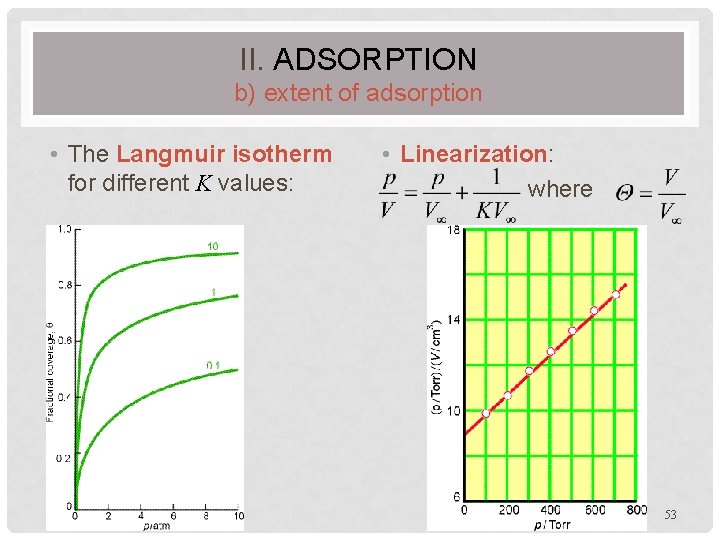
II. ADSORPTION b) extent of adsorption • The Langmuir isotherm for different K values: • Linearization: where 53
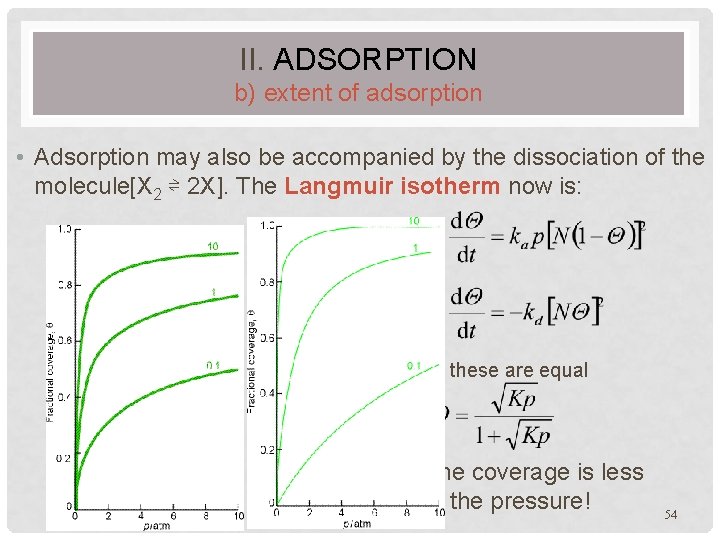
II. ADSORPTION b) extent of adsorption • Adsorption may also be accompanied by the dissociation of the molecule[X 2 ⇌ 2 X]. The Langmuir isotherm now is: • adsorption: • desorption: • In equilibrium, these are equal • In this case, the coverage is less dependent on the pressure! 54
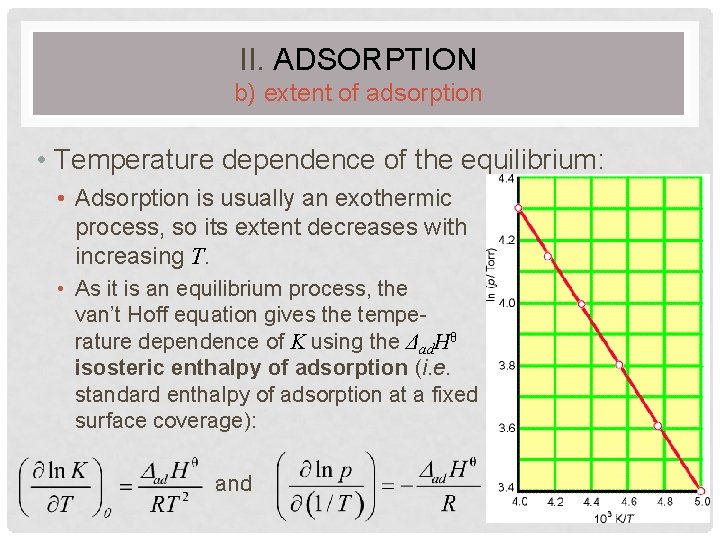
II. ADSORPTION b) extent of adsorption • Temperature dependence of the equilibrium: • Adsorption is usually an exothermic process, so its extent decreases with increasing T. • As it is an equilibrium process, the van’t Hoff equation gives the temperature dependence of K using the Δad. Hθ isosteric enthalpy of adsorption (i. e. standard enthalpy of adsorption at a fixed surface coverage): and 55
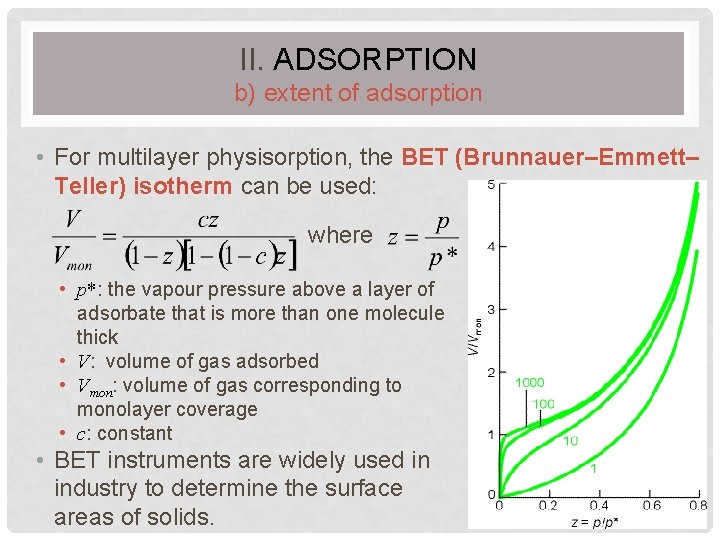
II. ADSORPTION b) extent of adsorption • For multilayer physisorption, the BET (Brunnauer–Emmett– Teller) isotherm can be used: where • p*: the vapour pressure above a layer of adsorbate that is more than one molecule thick • V: volume of gas adsorbed • Vmon: volume of gas corresponding to monolayer coverage • c: constant • BET instruments are widely used in industry to determine the surface areas of solids. 56
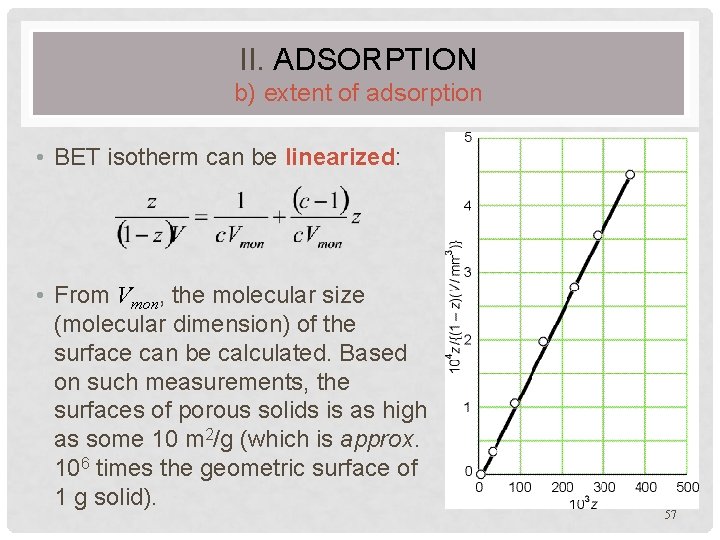
II. ADSORPTION b) extent of adsorption • BET isotherm can be linearized: • From Vmon, the molecular size (molecular dimension) of the surface can be calculated. Based on such measurements, the surfaces of porous solids is as high as some 10 m 2/g (which is approx. 106 times the geometric surface of 1 g solid). 57

II. ADSORPTION c) rates of surface processes • (only the rate of adsorption, desorption and mobility on surfaces – not heterogeneous catalysis jet) • A key datum is the sticking probability (s): • the numerator can be measured by observing the rate of change of pressure, • the denominator can be calculated from the kinetic theory of gases. • Mechanism: the arrival and the collision is followed by diffusion on the surface, and then binding. • Desorption is a kinetically first-order process that requires 58 activation. Its examination is very important.

III. CATALYTIC ACTIVITY OF SURFACES • Heterogeneous catalysis – this is a chemical change i. e. reaction! • Catalysts (as discussed earlier): • • increase the reaction rate (forward and backward), do not influence the equilibrium, open a new reaction path with lower energy of activation, are not involved in the stoichiometry. • Heterogeneous catalysis: usually the catalyst is in a phase other than the reactant (s) and product (s). • Heterogeneous processes can also be described by „normal” rate equations. • The study of the molecular mechanism is also important. 59
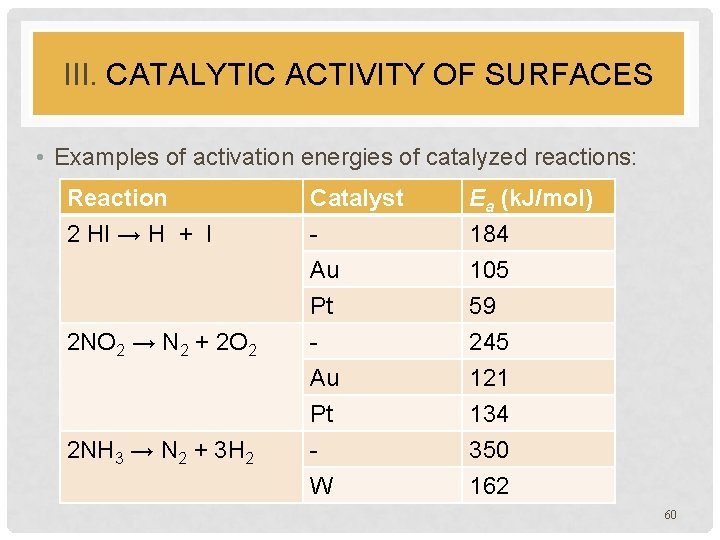
III. CATALYTIC ACTIVITY OF SURFACES • Examples of activation energies of catalyzed reactions: Reaction 2 HI → H + I Catalyst Au Pt Ea (k. J/mol) 184 105 59 2 NO 2 → N 2 + 2 O 2 Au Pt W 245 121 134 350 162 2 NH 3 → N 2 + 3 H 2 60

III. CATALYTIC ACTIVITY OF SURFACES • The mechanism: mechanism the reaction obviously happens on the solid surface (contact catalysis). Two cases: § both reactants adsorb on the surface, the reaction takes place, then the product(s) leave the surface, § only one of the reactants is adsorbed and the other reacts with this, then the product(s) leave. • Meanwhile, the structure of the solid surface is often transformed – at least temporarily. • A general theory used for heterogeneous catalysis is not yet known, many approaches are known instead. These can be (very) good, accurate for a given case, but not generalizable for all cases. 61

III. CATALYTIC ACTIVITY OF SURFACES • Langmuir–Hinshelwood mechanism: • both reactants (A and B) are chemisorbed • stoichiometry: A + B P • kinetics v = k Θ A ΘB • If the Langmuir isotherm is valid (often): • It gives the mathematical form of the rate equation – which can be experimentally determined and then compared with the assumed one: 62

III. CATALYTIC ACTIVITY OF SURFACES • Eley–Rideal mechanism: • a reaction between an adsorbed (A) and a gas-phase molecule (B) : • stoichiometry: A + B P • kinetics: v = k ΘA p. B • If the Langmuir isotherm is valid for A (often): • The Langmuir–Hinshelwood mechanism is much more common, but there also examples of the Eley–Rideal mechanism (e. g. H 2(gas) + D 2(ad)). 63
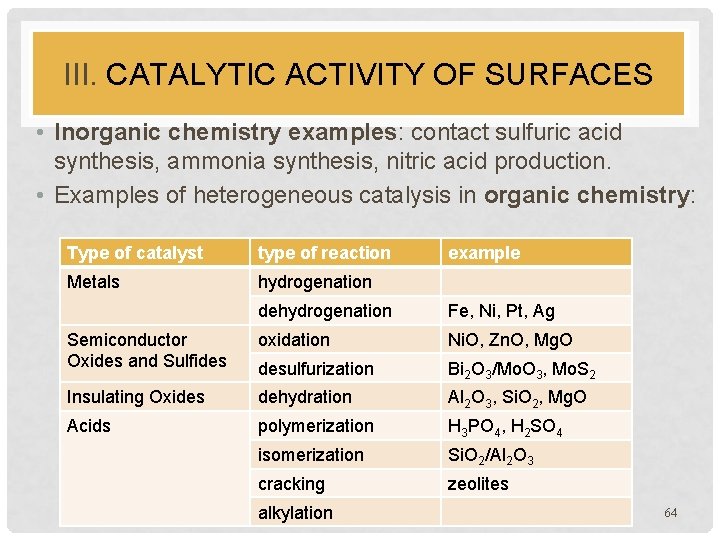
III. CATALYTIC ACTIVITY OF SURFACES • Inorganic chemistry examples: contact sulfuric acid synthesis, ammonia synthesis, nitric acid production. • Examples of heterogeneous catalysis in organic chemistry: Type of catalyst type of reaction Metals hydrogenation example dehydrogenation Fe, Ni, Pt, Ag Semiconductor Oxides and Sulfides oxidation Ni. O, Zn. O, Mg. O desulfurization Bi 2 O 3/Mo. O 3, Mo. S 2 Insulating Oxides dehydration Al 2 O 3, Si. O 2, Mg. O Acids polymerization H 3 PO 4, H 2 SO 4 isomerization Si. O 2/Al 2 O 3 cracking zeolites alkylation 64
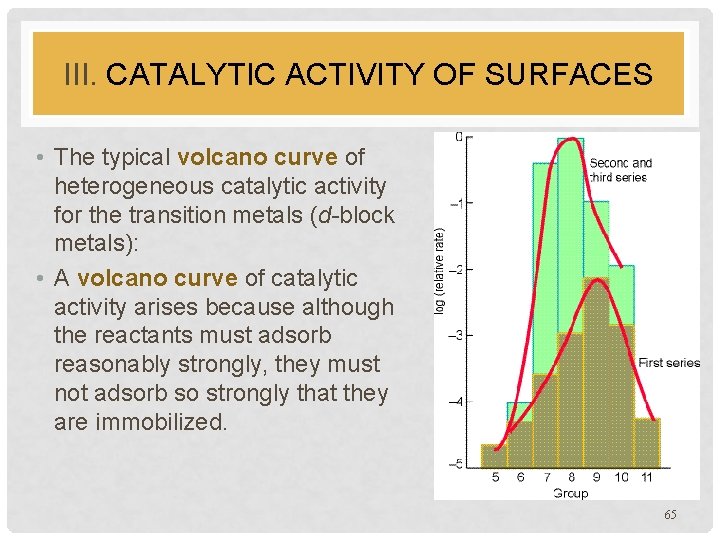
III. CATALYTIC ACTIVITY OF SURFACES • The typical volcano curve of heterogeneous catalytic activity for the transition metals (d-block metals): • A volcano curve of catalytic activity arises because although the reactants must adsorb reasonably strongly, they must not adsorb so strongly that they are immobilized. 65

III. CATALYTIC ACTIVITY OF SURFACES • The practice of heterogeneous (contact) catalysis: • Usually supported catalysts are used (many catalysts are very expensive and a high specific surface can be obtained with the porous support), • they work in open flow reactors, • co-catalysts accelerate the rate even further. • The catalysts are occasionally regenerated because of the presence of inhibitors and even catalyst poisons, • therefore it is important to pre-purify the reactants. • The catalyst often not only accelerates, but also controls (metasubstitution instead of ortho-; how „far” does C-oxidation go (COH, C=O, COOH)). • The radical mechanisms are frequent (strong chemisorption causes homolytic bond breaking). 66
- Slides: 66