Overcoming Treatment Challenges in Sarcomas Promising Novel Targeted











- Slides: 32

Overcoming Treatment Challenges in Sarcomas: Promising Novel Targeted Agents George D. Demetri, MD Center for Sarcoma and Bone Oncology Dana-Farber Cancer Institute Ludwig Center at Dana-Farber / Harvard Boston, Massachusetts

Development of Molecularly Targeted Agents for Sarcomas A Model for Personalized Anticancer Drug Development

Searching for Critical Switches in Sarcomas ON OFF Tumor Cell Survival and Growth Tumor Cell Growth Arrest and Cancer Regression

Finding the Critical Switch in the Gastrointestinal Sarcoma known as GIST Gain-of-Function Mutations of c-kit in Human Gastrointestinal Stromal Tumors Seiichi Hirota, Koji Isozaki, Yasuhiro Moriyama, Koji Hashimoto, Toshirou Nishida, Shingo Ishiguro, Kiyoshi Kawano, Masato Hanada, Akihiko Kurata, Masashi Takeda, Ghulam Muhammad Tunio, Yuji Matsuzawa, Yuzuru Kanakura, Yasuhisa Shinomura, Yukihiko Kitamura Science 279: 577 -580, 1998.

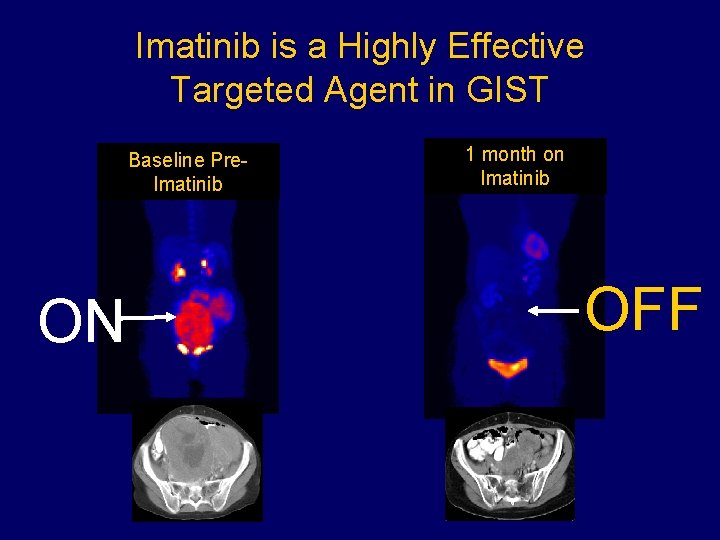
Imatinib is a Highly Effective Targeted Agent in GIST Baseline Pre. Imatinib ON 1 month on Imatinib OFF

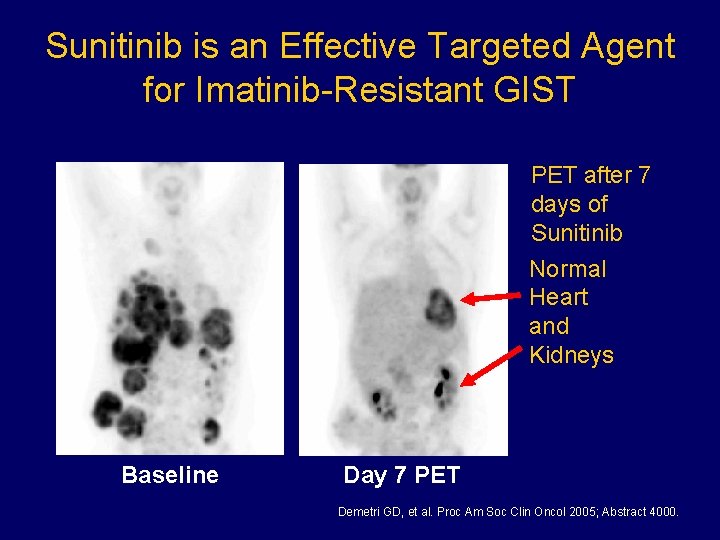
Sunitinib is an Effective Targeted Agent for Imatinib-Resistant GIST PET after 7 days of Sunitinib Normal Heart and Kidneys Baseline Day 7 PET Demetri GD, et al. Proc Am Soc Clin Oncol 2005; Abstract 4000.

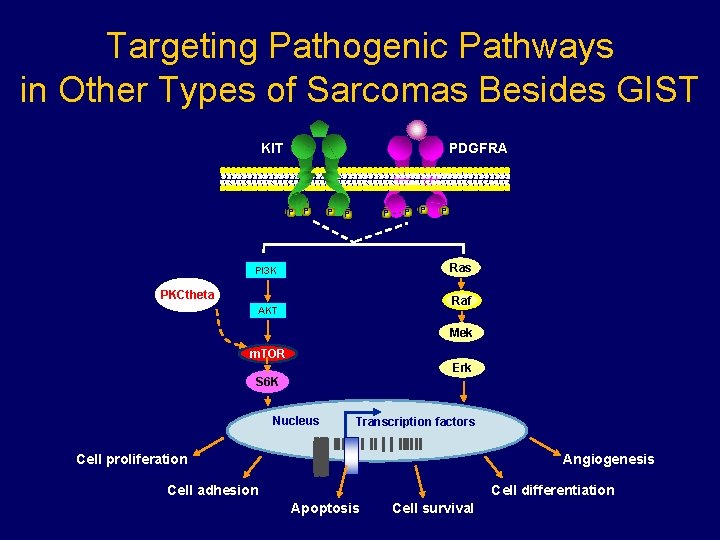
Targeting Pathogenic Pathways in Other Types of Sarcomas Besides GIST KIT PDGFRA P P P P Ras PI 3 K PKCtheta Raf AKT Mek m. TOR Erk S 6 K Nucleus Transcription factors Cell proliferation Angiogenesis Cell adhesion Cell differentiation Apoptosis Cell survival

The Next Successful Application of Kinase Inhibition to Sarcoma Therapy • Dermatofibrosarcoma Protuberans (DFSP) – Balanced translocation - t(17; 22) - leads to uncontrolled production of PDGF ligand – This induces autocrine activation of the wild-type PDGF-receptor system – Imatinib and Sunitinib also block PDGF-receptors

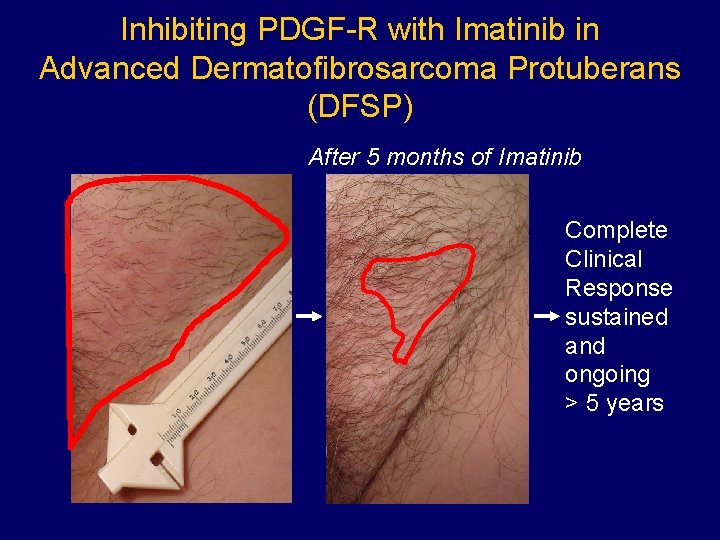
Inhibiting PDGF-R with Imatinib in Advanced Dermatofibrosarcoma Protuberans (DFSP) After 5 months of Imatinib Complete Clinical Response sustained and ongoing > 5 years

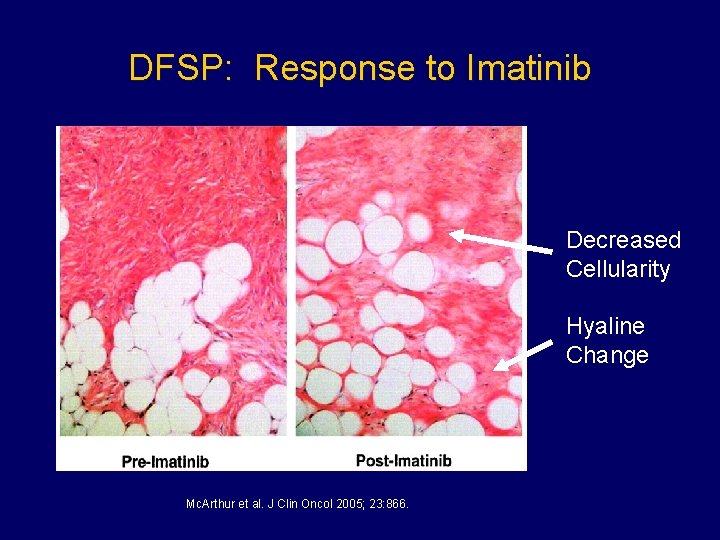
DFSP: Response to Imatinib Decreased Cellularity Hyaline Change Mc. Arthur et al. J Clin Oncol 2005; 23: 866.

Testing Kinase Inhibitors as Therapy for Other Sarcoma Subtypes • Imatinib – Occasional activity in desmoid tumors (SARC) • Sorafenib – Some activity in vascular sarcomas (MSKCC) • Sunitinib – Some activity in vascular sarcomas and desmoplastic small round cell tumor (MSKCC and DFCI) • Dasatinib – SARC trial ongoing

Testing Kinase Inhibitors as Therapy for Other Sarcoma Subtypes • Pazopanib – Oral Tyrosine Kinase Inhibitor with activity against VEGF-R, PDGFR, and KIT, tested by EORTC – Activity demonstrated in several subtypes including leiomyosarcomas and synovial sarcoma – Phase III clinical trial to begin soon with EORTC and other international collaborations

Targeting Pathogenic Pathways in Other Sarcomas KIT PDGFRA P P P P Ras PI 3 K PKCtheta Raf AKT Mek m. TOR Erk S 6 K Nucleus Cell proliferation Transcription factors Angiogenesis Cell adhesion Apoptosis Cell survival Cell differentiation

m. TOR-driven Sarcomas • LAM and PEComa family of tumors – Lymphangioleiomyomatosis (LAM) – Angiomyolipoma (AML) – Perivascular Epitheliod Cell-oma (PEComa) • LAM/AML can be associated with Tuberous Sclerosis Complex or sporadic

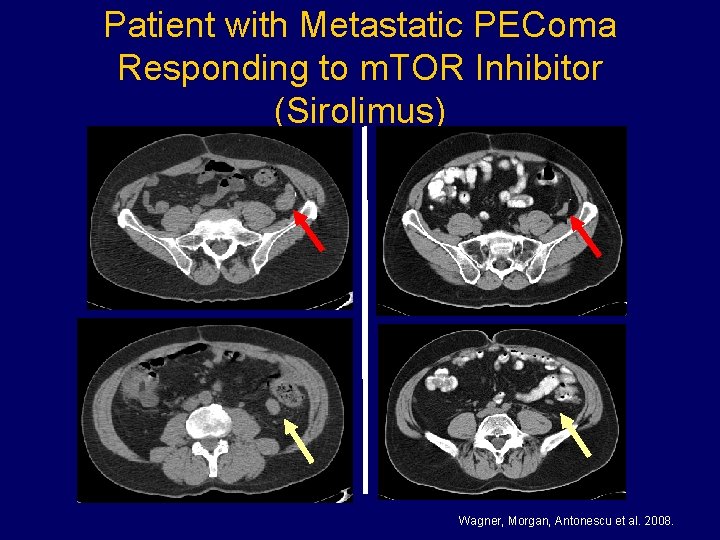
Patient with Metastatic PEComa Responding to m. TOR Inhibitor (Sirolimus) Wagner, Morgan, Antonescu et al. 2008.

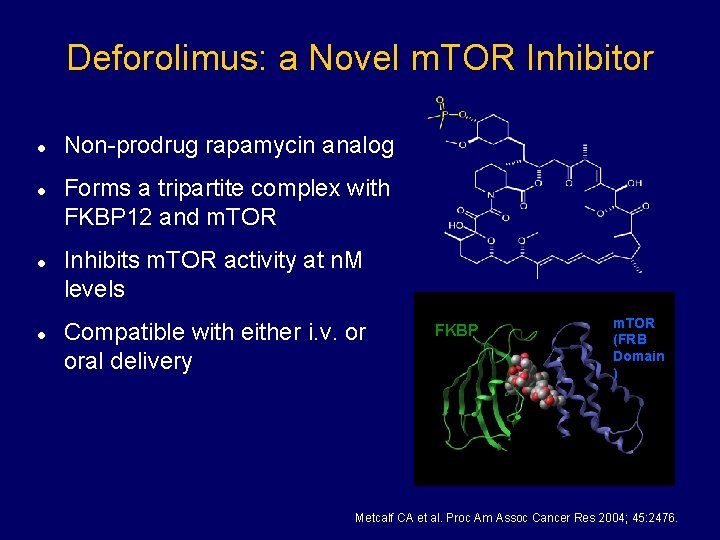
Deforolimus: a Novel m. TOR Inhibitor l l Non-prodrug rapamycin analog Forms a tripartite complex with FKBP 12 and m. TOR Inhibits m. TOR activity at n. M levels Compatible with either i. v. or oral delivery FKBP m. TOR (FRB Domain ) Metcalf CA et al. Proc Am Assoc Cancer Res 2004; 45: 2476.

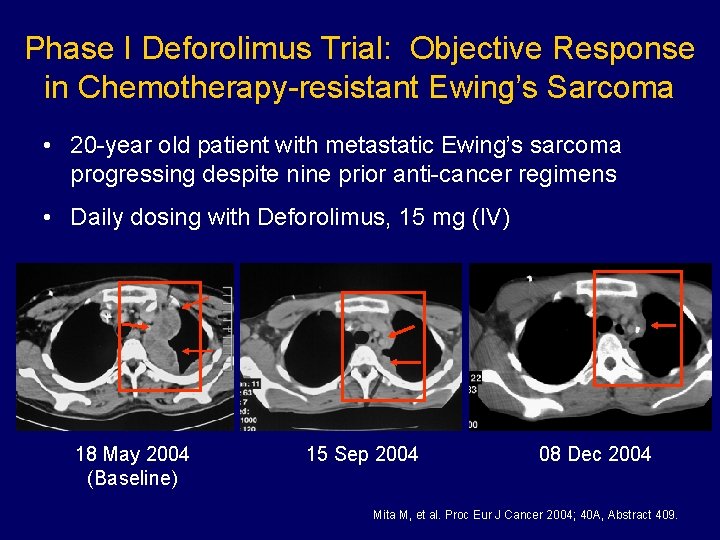
Phase I Deforolimus Trial: Objective Response in Chemotherapy-resistant Ewing’s Sarcoma • 20 -year old patient with metastatic Ewing’s sarcoma progressing despite nine prior anti-cancer regimens • Daily dosing with Deforolimus, 15 mg (IV) 18 May 2004 (Baseline) 15 Sep 2004 08 Dec 2004 Mita M, et al. Proc Eur J Cancer 2004; 40 A, Abstract 409.

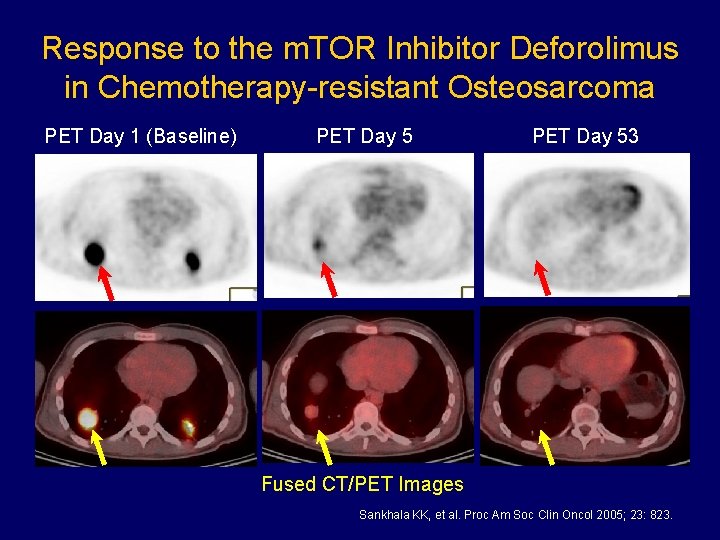
Response to the m. TOR Inhibitor Deforolimus in Chemotherapy-resistant Osteosarcoma PET Day 1 (Baseline) PET Day 53 Fused CT/PET Images Sankhala KK, et al. Proc Am Soc Clin Oncol 2005; 23: 823.

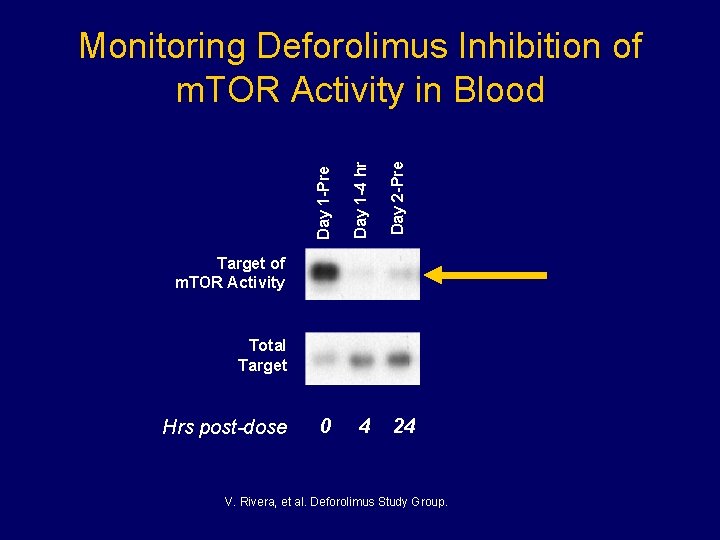
Day 1 -4 hr 0 4 Day 2 -Pre Day 1 -Pre Monitoring Deforolimus Inhibition of m. TOR Activity in Blood Target of m. TOR Activity Total Target Hrs post-dose 24 V. Rivera, et al. Deforolimus Study Group.

Phase II Deforolimus Trial: Promising Progression-Free Survival (PFS) 6 -Month Rate Median (wks) Bone Sarcoma 25% 16 Soft-tissue Sarcomas 24% 15 • Leiomyosarcoma 22% 16 • Liposarcoma 30% 16 • Other soft-tissue sarcomas 23% 15 24% 15 Sarcoma Subtype Overall PFS • Benefits observed across all sarcoma subtypes

Historical Context for Promising Disease Control with Deforolimus in Sarcomas Progression-free Survival (PFS) Compared with Historical Data from EORTC Historical Data (EORTC) Deforolimus Inactive Agents Active Agents 158 234 146 Median PFS 15 wks 7 wks 8 wks 3 -month PFS 58% 21% 39% 6 -month PFS 24% 8% 14% No. of Patients

Phase II Activity of Deforolimus in Sarcomas • Promising activity of Deforolimus in patients with a broad range of advanced soft-tissue and bone sarcomas – Achieved primary endpoint of ≥ 25% Clinical Benefit Response in each histologic subgroup of advanced sarcomas • No significant differences among the 4 sub-groups – 29% overall CBR rate despite low incidence of tumor shrinkage – PFS more than double that of historical control (EORTC) • 24% 6 -month PFS rate (vs. 8%) • 15 -week median PFS (vs. 7 weeks) • Well-tolerated with manageable side-effects • A solid foundation upon which a definitive phase III trial has been developed to test the activity of deforolimus

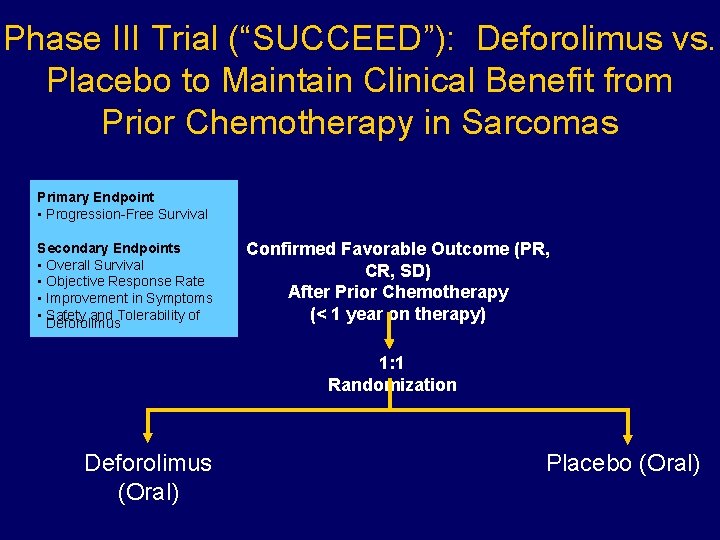
Phase III Trial (“SUCCEED”): Deforolimus vs. Placebo to Maintain Clinical Benefit from Prior Chemotherapy in Sarcomas Primary Endpoint • Progression-Free Survival Secondary Endpoints • Overall Survival • Objective Response Rate • Improvement in Symptoms • Safety and Tolerability of Deforolimus Confirmed Favorable Outcome (PR, CR, SD) After Prior Chemotherapy (< 1 year on therapy) 1: 1 Randomization Deforolimus (Oral) Placebo (Oral)

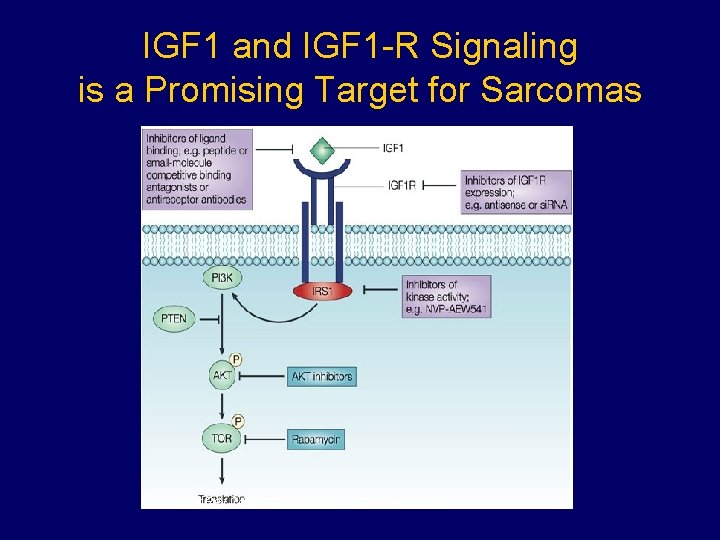
IGF 1 and IGF 1 -R Signaling is a Promising Target for Sarcomas Nat Rev Cancer © 2004 Nature Publishing Group.

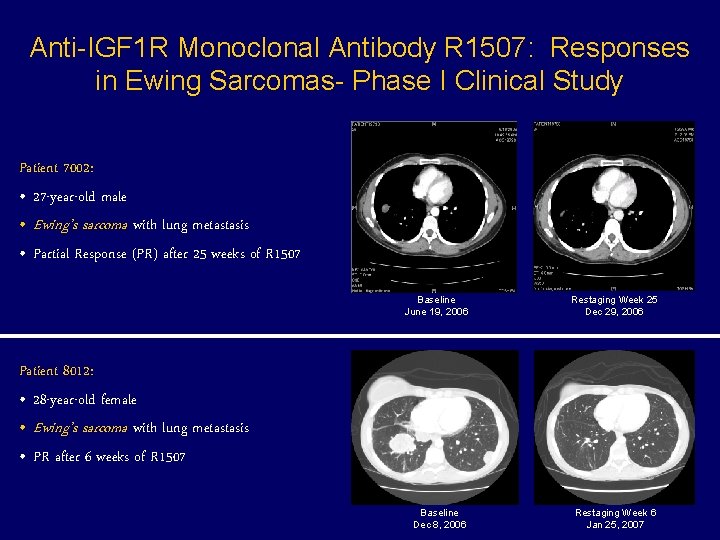
Anti-IGF 1 R Monoclonal Antibody R 1507: Responses in Ewing Sarcomas- Phase I Clinical Study Patient 7002: • 27 -year-old male • Ewing’s sarcoma with lung metastasis • Partial Response (PR) after 25 weeks of R 1507 Baseline June 19, 2006 Restaging Week 25 Dec 29, 2006 Baseline Dec 8, 2006 Restaging Week 6 Jan 25, 2007 Patient 8012: • 28 -year-old female • Ewing’s sarcoma with lung metastasis • PR after 6 weeks of R 1507

Targeting Hsp 90 in Sarcomas Preferential targeting to cancer Cancer cell Hsp 90 is different from Hsp 90 in normal cells Specific chaperone function: stabilization of oncogenes in key cell signaling pathways

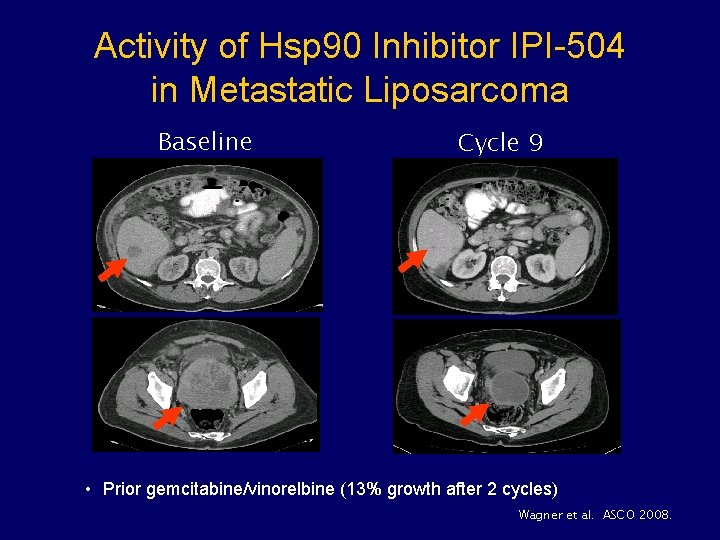
Activity of Hsp 90 Inhibitor IPI-504 in Metastatic Liposarcoma Baseline Cycle 9 • Prior gemcitabine/vinorelbine (13% growth after 2 cycles) Wagner et al. ASCO 2008.

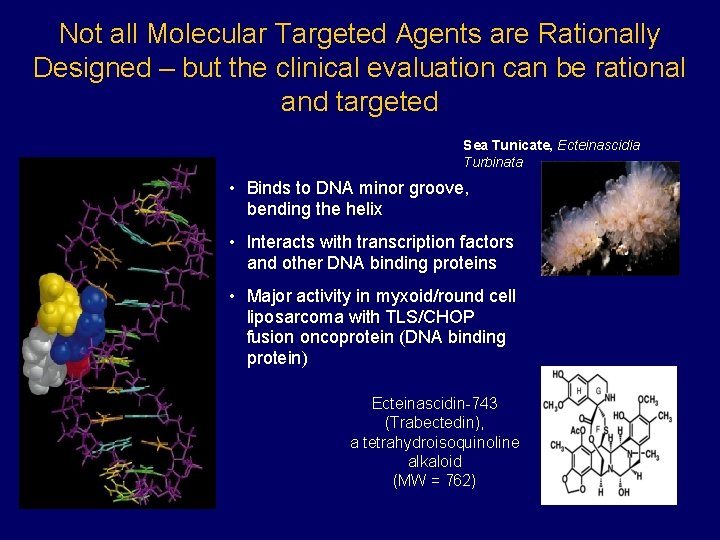
Not all Molecular Targeted Agents are Rationally Designed – but the clinical evaluation can be rational and targeted Sea Tunicate, Ecteinascidia Turbinata • Binds to DNA minor groove, bending the helix • Interacts with transcription factors and other DNA binding proteins • Major activity in myxoid/round cell liposarcoma with TLS/CHOP fusion oncoprotein (DNA binding protein) Ecteinascidin-743 (Trabectedin), a tetrahydroisoquinoline alkaloid (MW = 762)

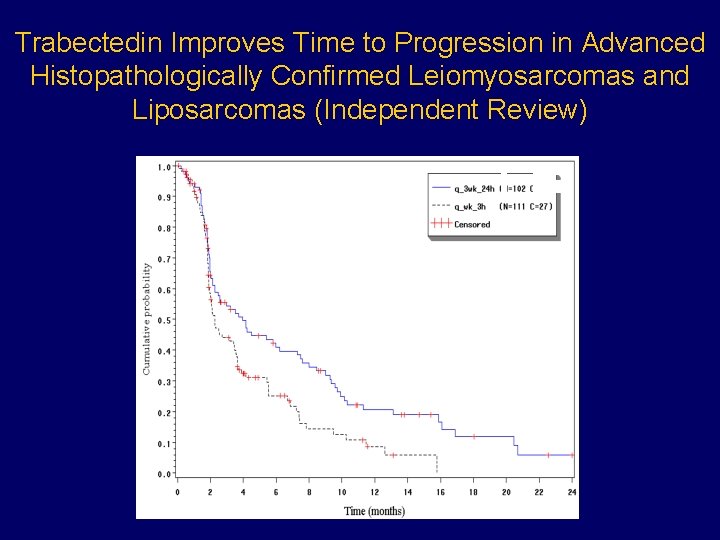
Trabectedin Improves Time to Progression in Advanced Histopathologically Confirmed Leiomyosarcomas and Liposarcomas (Independent Review) p=0. 0076 HR: 0. 647

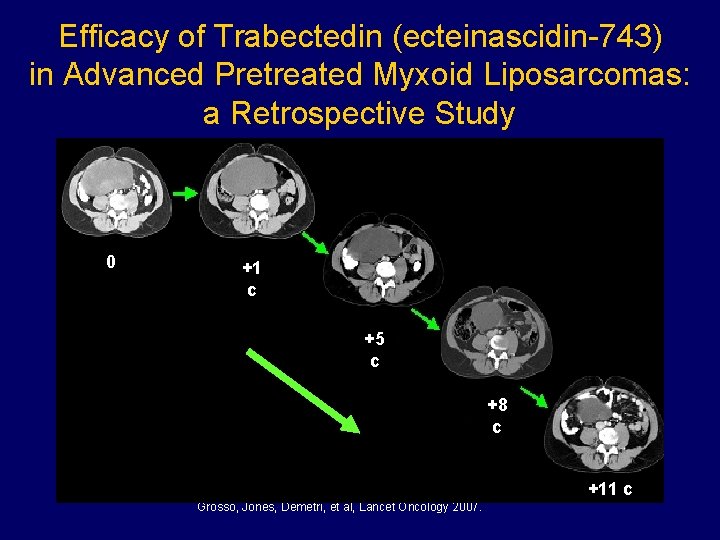
Efficacy of Trabectedin (ecteinascidin-743) in Advanced Pretreated Myxoid Liposarcomas: a Retrospective Study 0 +1 c +5 c +8 c +11 c Grosso, Jones, Demetri, et al, Lancet Oncology 2007.

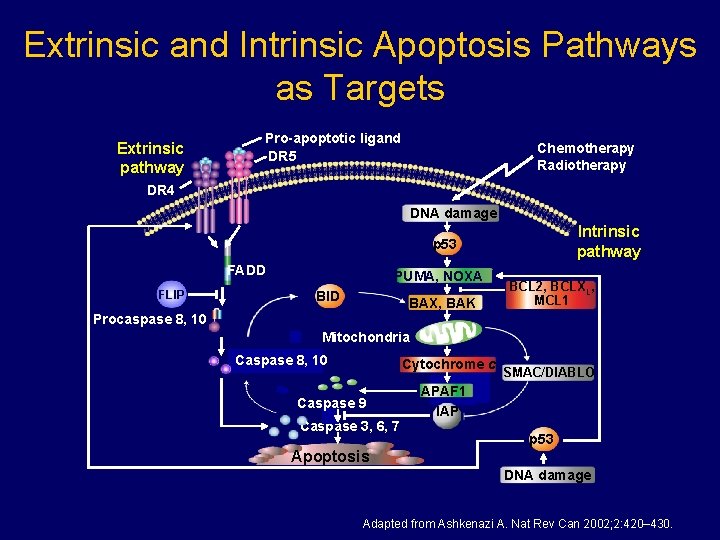
Extrinsic and Intrinsic Apoptosis Pathways as Targets Extrinsic pathway Pro-apoptotic ligand DR 5 Chemotherapy Radiotherapy DR 4 DNA damage Intrinsic pathway p 53 FADD FLIP PUMA, NOXA BID BAX, BAK BCL 2, BCLXL, MCL 1 Procaspase 8, 10 Mitochondria Caspase 8, 10 Cytochrome c Caspase 9 Caspase 3, 6, 7 Apoptosis SMAC/DIABLO APAF 1 IAP p 53 DNA damage Adapted from Ashkenazi A. Nat Rev Can 2002; 2: 420– 430.

Molecularly Targeted Agents for Sarcomas Summary • Sarcomas represent a variety of clinicopathologic subtypes for discovery and development of rationallydesigned drugs to target specific molecular pathways • Inhibition of a single pathway (KIT signaling) has proven effective for GIST • Inhibition of multiple pathways will likely provide the best results over time