Ovarian Cancer Tumour Markers Brig Dilshad Ahmed Khan

Ovarian Cancer Tumour Markers Brig Dilshad Ahmed Khan MBBS, MCPS, FRC Path , Ph. D Head of Chem Pathology & Endocrinology dept AFIP, Rawalpindi

Outlines o Introduction o Risk factors for development of ovarian cancer o Diagnosis of ovarian cancer o Clinical application of CA 125 o Clinical application of HE 4 o Algorithms for the estimation of the risk of ovarian cancer in women with pelvic mass (ROMA)
Introduction Ovarian cancer are developed from three categories of cells q Epithelial Cells (65 -70%) Ø Serous Ø Mucinous Ø Endometrioid Ø Transitional cell o Stromal cell– 15 -20% o Germ cell – 5 -10%:

Ovarian Cancer Epidemiology o Incidence is 2 to 15 cases per 100, 000 women o The 2 nd most common gynecologic malignancy o 4 th leading cause of cancer death in U. S. (after lung, breast and colon) American Cancer Society, 20013

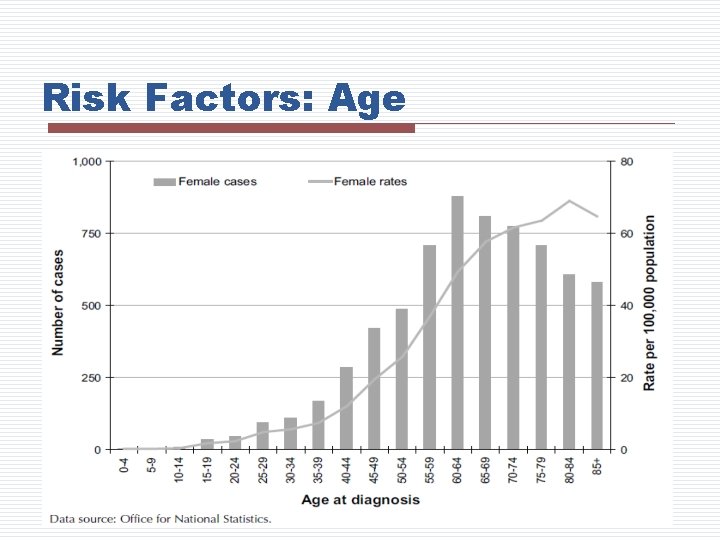
Risk Factors Age n Women over age 55 account for ~80% of all cases Reproductive history n Early menarche or age >30 years at first childbearing, and late menopause Hormone replacement therapy > 10 years n May be associated with 30% increased risk American Cancer Society, 2013
Risk Factors: Age

Risk factors: Heredity o Up to 10% of epithelial ovarian cancer are familial o Familial breast-ovarian cancer and site-specific ovarian cancer syndromes are associated with mutations of the BRCA 1 suppressor gene; account for 90% of familial ovarian cancers Rollins, G. Ann Int Med 2000; 133: 1021 -1024.
Diagnosis of Ovarian Cancer o Early detection is not an easy task o Pelvic examination o Ultrasound o CT Scan & MRI o Laparoscopic biopsy o Histological examination o Ovarian cancer tumor markers

History : Symptoms of ovarian cancer o Asymptomatic o Lower abdominal pain/pressure o Pelvic mass o Abdominal enlargement o Vaginal bleeding o Urinary/bowel symptoms

Images: U/S MRI, CT Ultrasound Ø Relatively inexpensive Ø Delineates cystic vs solid structures CT Scan Ø Assesses other organs Ø Excellent for retroperitoneum (1 -5 mm) MRI Ø Allows for ID of soft tissue lesions Ø Can differentiate normal from malignancy
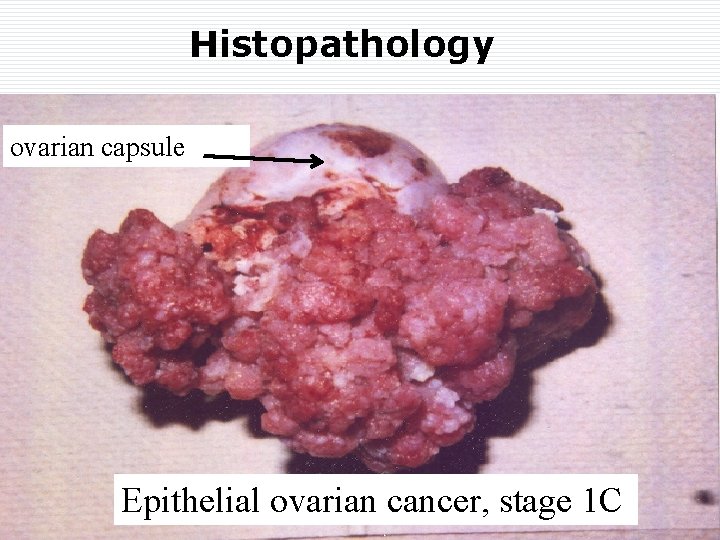
Histopathology ovarian capsule Epithelial ovarian cancer, stage 1 C

Ovarian Tumors: Classification 1. Surface epithelial – 6570%: o o o Serous (tubal) Mucinous (endocx & intestinal) Endometrioid Transitional cell - Brenners. Clear cell 2. Stromal – 15 -20%: Granulosa-cell tumor Thecoma Fibroma Sertoli-Leydig cell tumors 3. Germ cell tumors – 5 -10%: o Teratoma – n Benign cystic (dermoid cysts) n Solid immature n Monodermal – struma ovarii, carcinoid o Dysgerminoma o Yolk sac tumor Choricarcinoma o Mixed germ cell tumor 4. Metastatic tumors – 5%
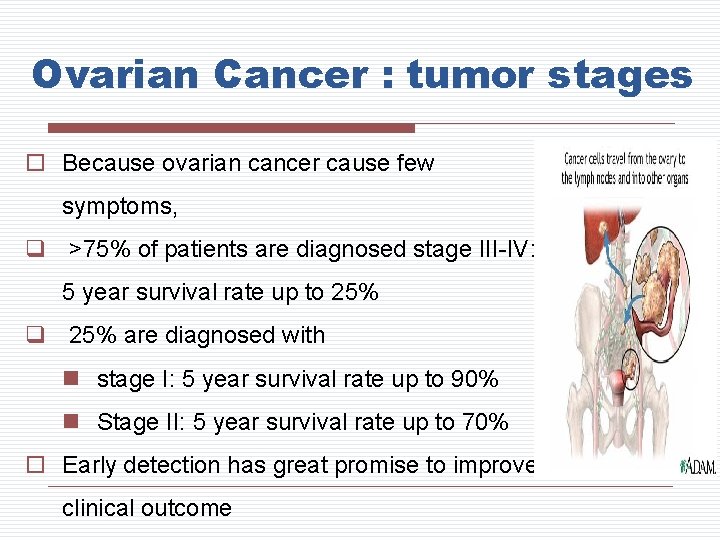
Ovarian Cancer : tumor stages o Because ovarian cancer cause few symptoms, q >75% of patients are diagnosed stage III-IV: 5 year survival rate up to 25% q 25% are diagnosed with n stage I: 5 year survival rate up to 90% n Stage II: 5 year survival rate up to 70% o Early detection has great promise to improve clinical outcome

Ovarian Cancer Biomarkers o CA 125 o Transthyretin o HE 4 o IGFBP-2 o CA 15 -3 o SMRP (Mesomark™) o CA 72 -4 o HK 6 o B 7 -H 4 (Ov-110) o Cytokeratin 19

CA-125 – Tumor Markers n CA 125 is the first tumor marker of ovarian cancer n Discovered with a mouse monoclonal antibody (OC 125) produced by immunizing a mouse with a serous ovarian cancer cell line n Glycoprotein with a molecular weight (>200 k. D) n Reference range : Serum <35 U/m. L
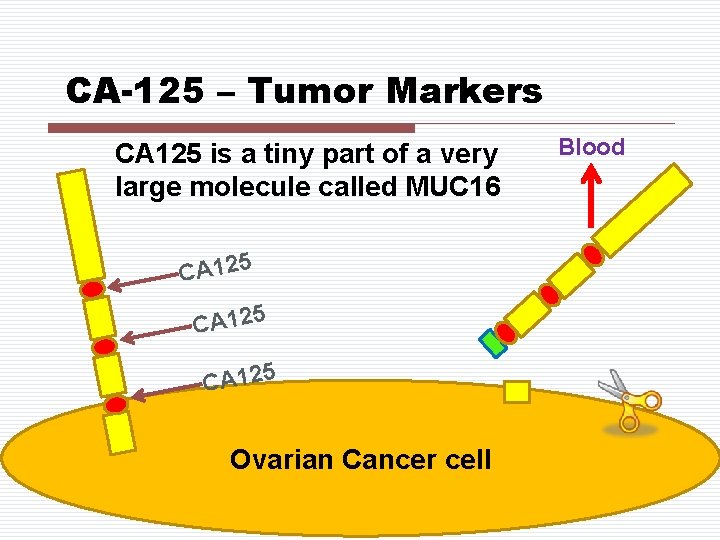
CA-125 – Tumor Markers CA 125 is a tiny part of a very large molecule called MUC 16 CA 125 Ovarian Cancer cell Blood

Clinical application of CA 125 • Increased in most ovarian cancers especially 80% of epithelial ovarian cancers 1 • Elevated in 50% of Stage I disease • Longitudinal assessment improve sensitivity • Marker to test the recurrence of cancer 1 NIH Consensus Development Conference Statement. Gynecol Oncol. 1994; 55: S 4 -S 14. 2 ACOG Practice Bulletin. Obstet Gynecol. 2007; 110: 201 -213.
CA-125 – Serum CA 125 Assay CA 125
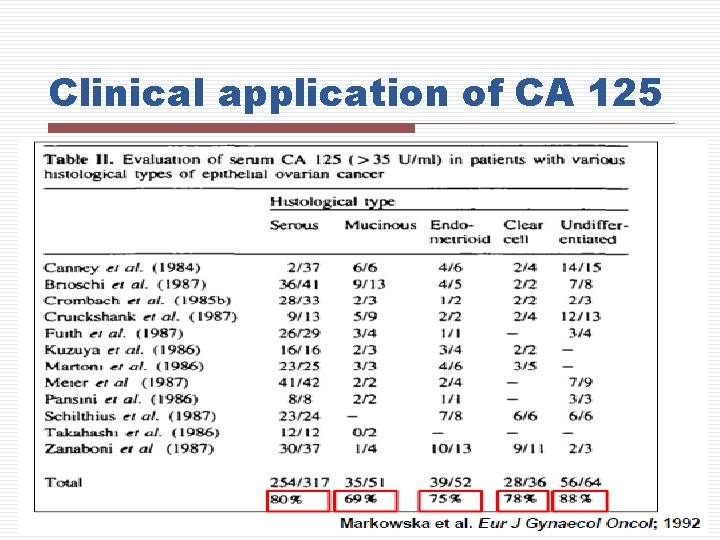
Clinical application of CA 125
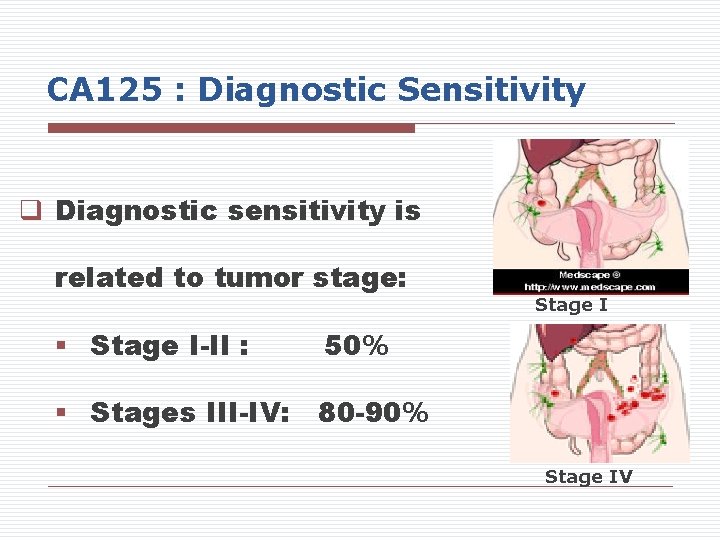
CA 125 : Diagnostic Sensitivity q Diagnostic sensitivity is related to tumor stage: § Stage I-II : Stage I 50% § Stages III-IV: 80 -90% Stage IV

CA 125 : Diagnostic Specificity Limitations Poor specificity (elevated in many gynecologic & non-gynecologic malignancies as well as benign conditions – increased in 0. 2‐ 5. 9% healthy women – increased in 2. 2‐ 27. 8% of benign 1, 2 disease NIH Consensus Development Conference Statement. Gynecol Oncol. 1994; 55: S 4 -S 14. 1 2 ACOG Practice Bulletin. Obstet Gynecol. 2007; 110: 201 -213.

CA 125 : Diagnostic Specificity Malignant conditions Benign conditions o Cervical CA o Endometriosis o Fallopian tube CA o Uterine fibroids o Endometrial CA o ovarian cysts o Pancreatic CA o PID o Colon CA o Pancreatitis o Breast CA o Liver disease o Mesothelioma o Renal failure

Monitoring Treatment Response Gynecological cancer group criterion: n Complete responder: CA 125 concentrations fall within the reference range after treatment. n At least 50% of CA 125 decrease compared with the pre treated sample
Monitoring Treatment Response Diagnosis and treatment Canc er cell Time Tumor
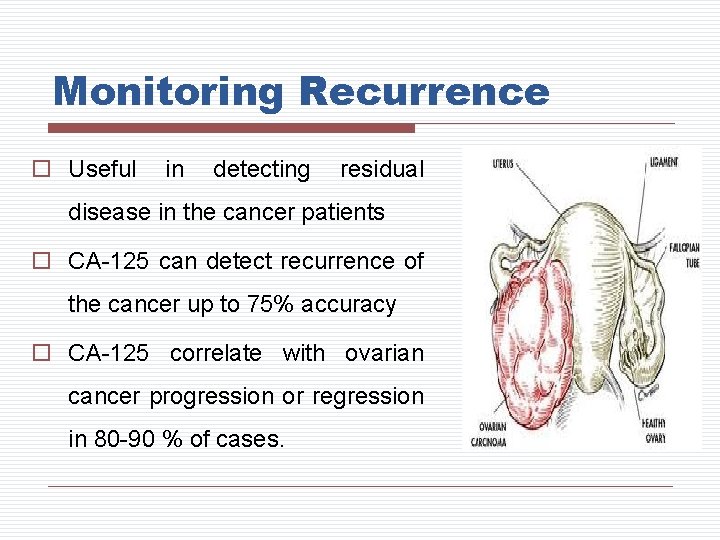
Monitoring Recurrence o Useful in detecting residual disease in the cancer patients o CA-125 can detect recurrence of the cancer up to 75% accuracy o CA-125 correlate with ovarian cancer progression or regression in 80 -90 % of cases.

Monitoring Recurrence o Patients with normalized CA 125 level: Increase in CA 125 ≥ 2 times of the upper limit of reference on two occasions after treatment. o An absolute increase of CA 125 level ≥ 5 U/m. L compared with its nadir value was a strong predictor of recurrence Gynecological Cancer Intergroup criterion (2011)

Human epididymis protein 4 (HE 4) q HE 4 is human epididymis protein 4 q A new up-regulated biomarker for ovarian cancer q Gene located in chromosome 20 q 12– 13. 1

HE 4 : Diagnostic Sensitivity q HE 4 has better diagnostic sensitivity in the early diagnosis of ovarian cancer q Overexpressed in 93% of serous, 100% of endometrioid and 50% of clear cell ovarian cancer q Not expressed in mucinous and germ‐cell ovarian cancers 1 NIH Consensus Development Conference Statement. Gynecol Oncol. 1994; 55: S 4 -S 14. Practice Bulletin. Obstet Gynecol. 2007; 110: 201 -213. 2 ACOG
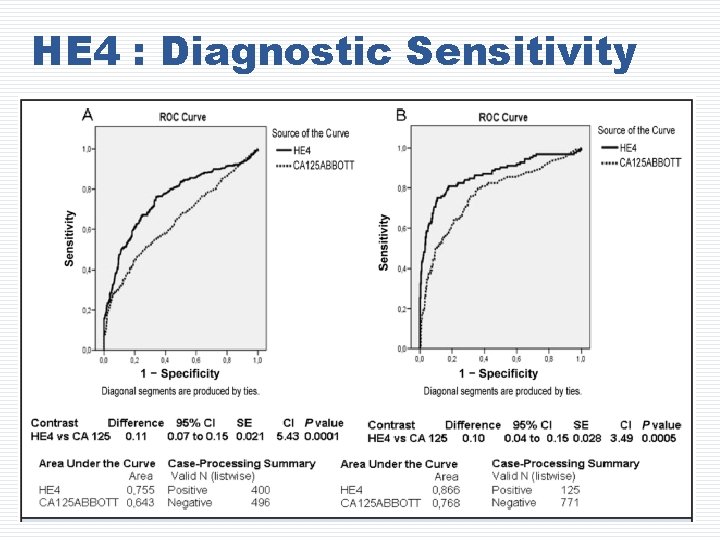
HE 4 : Diagnostic Sensitivity

HE 4 : Diagnostic Specificity q HE 4 has an increased diagnostic specificity compared with CA 125, in the ovarian malignancies q Overexpressed in pulmonary, endometrial, and breast cancers and mesotheliomas q Renal failure and pleural effusions are the most important sources of false positive HE 4.
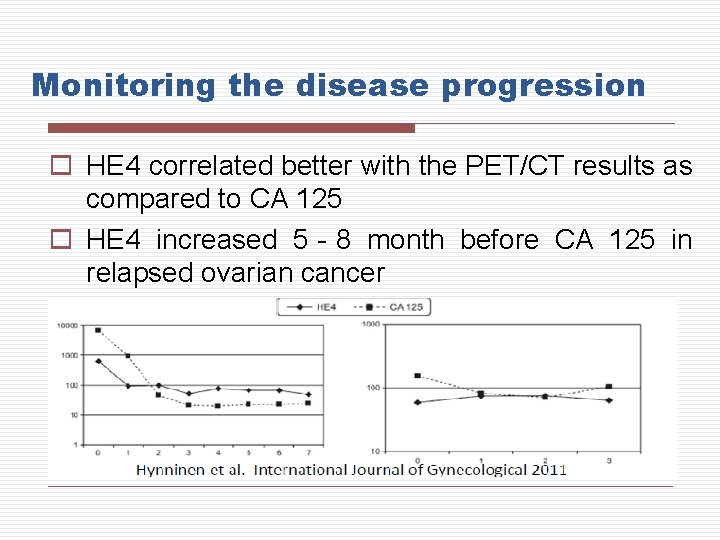
Monitoring the disease progression o HE 4 correlated better with the PET/CT results as compared to CA 125 o HE 4 increased 5‐ 8 month before CA 125 in relapsed ovarian cancer

Combination of HE 4 and CA 125 o Tumor markers CA 125 and HE 4 are approved by FDA for monitoring the disease progression o The combination of HE 4 and CA 125 are more sensitive than either marker alone o Both tumor markers relate to stage and histology of ovarian cancer
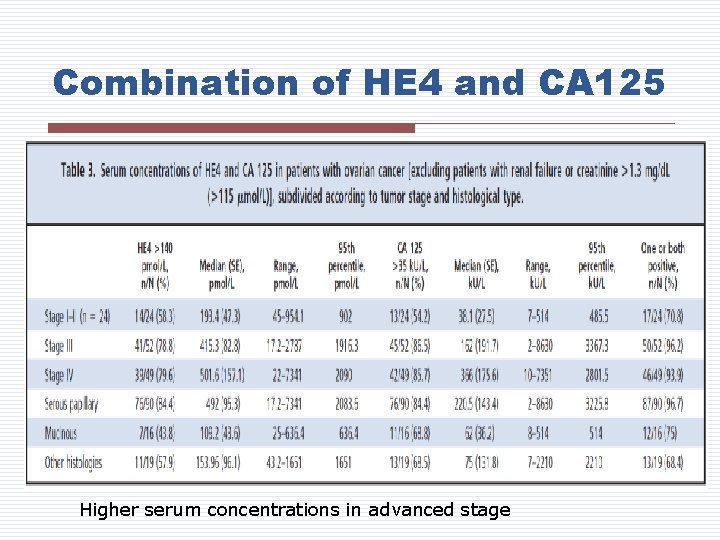
Combination of HE 4 and CA 125 Higher serum concentrations in advanced stage

Algorithms for the Estimation of risk of ovarian cancer in women with pelvic mass (ROMA) o To assess whether a woman who presents with an ovarian adnexal mass is at high or low likelihood of having malignancy o A quantitative test that combines serum HE 4, CA 125 & menopausal status into a numerical score
![ROMA (Risk of Ovarian Malignancy Algorithm : Calculation o ROMA = exp(PI) [1+exp(PI)]*10 o ROMA (Risk of Ovarian Malignancy Algorithm : Calculation o ROMA = exp(PI) [1+exp(PI)]*10 o](http://slidetodoc.com/presentation_image_h/75f963f14d1eeed7ecdaff6a8a1ef2db/image-35.jpg)
ROMA (Risk of Ovarian Malignancy Algorithm : Calculation o ROMA = exp(PI) [1+exp(PI)]*10 o Premenopausal: Predicative Index (PI) = cut off of ≥ 1. 31 o Postmenopausal: Predicative Index (PI) = cut off of ≥ 2. 77 o Provide a specificity level of 75%.

Conclusions q CA 125 is the tumor marker of choice for monitoring ovarian cancer q HE 4 diagnostic sensitivity is better than CA 125 in early stages of ovarian cancer q Combination of both tumor markers improved the detection of ovarian cancer & specificity than either alone q Both correlated well with the tumour stage, histology and prognosis q ROMA estimate the risk of ovarian cancer in women with pelvic mass
- Slides: 36