Outpatient Antibiogram January 1 December 31 2019 Urinary
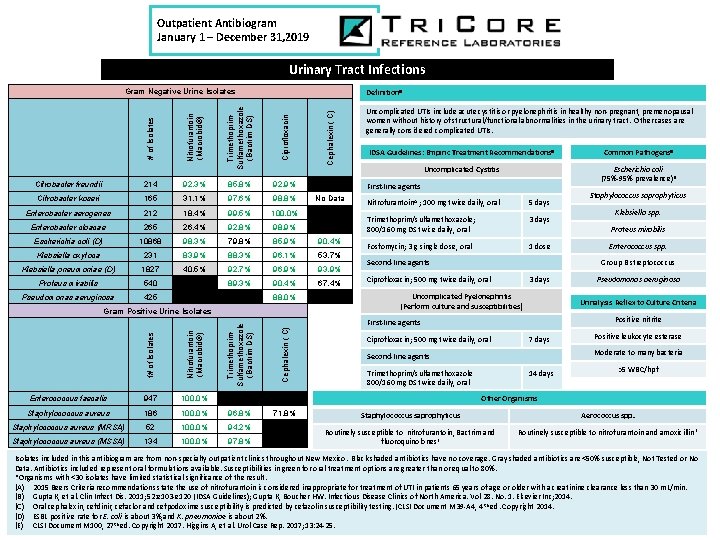
Outpatient Antibiogram January 1 – December 31, 2019 Urinary Tract Infections Gram Negative Urine Isolates 92. 3% 85. 8% 92. 9% 0. 0% Citrobacter koseri 165 31. 1% 97. 6% 98. 8% No Data Enterobacter aerogenes 212 18. 4% 99. 5% 100. 0% Enterobacter cloacae 265 26. 4% 92. 8% 98. 9% 0. 0% Escherichia coli (D) 10868 98. 3% 79. 8% 85. 9% 90. 4% Klebsiella oxytoca 231 83. 9% 88. 3% 96. 1% 53. 7% Klebsiella pneumoniae (D) 1827 40. 5% 92. 7% 96. 9% 93. 9% Proteus mirabilis 540 0. 0% 89. 3% 90. 4% 67. 4% Pseudomonas aeruginosa 425 0. 0% 88. 0% 0. 0% Nitrofurantoin (Macrobid®) Cephalexin ( C) f# of Isolates Trimethoprim. Sulfamethoxazole (Bactrim DS) Gram Positive Urine Isolates Enterococcus faecalis 947 100. 0% Staphylococcus aureus 186 100. 0% 96. 8% 71. 8% Staphylococcus aureus (MRSA) 52 100. 0% 94. 2% Staphylococcus aureus (MSSA) 134 100. 0% 97. 8% Cephalexin ( C) Ciprofloxacin 214 Nitrofurantoin (Macrobid®) Citrobacter freundii # of Isolates Trimethoprim. Sulfamethoxazole (Bactrim DS) Definition. B Uncomplicated UTIs include acute cystitis or pyelonephritis in healthy non-pregnant, premenopausal women without history of structural/functional abnormalities in the urinary tract. Other cases are generally considered complicated UTIs. IDSA Guidelines: Empiric Treatment Recommendations. B Common Pathogens. B Uncomplicated Cystitis Escherichia coli (75%-95% prevalence)B First-line agents Nitrofurantoin. A ; 100 mg twice daily, oral 5 days Trimethoprim/sulfamethoxazole; 800/160 mg DS twice daily, oral 3 days Fosfomycin; 3 g single dose, oral 1 dose Second-line agents Staphylococcus saprophyticus Klebsiella spp. Proteus mirabilis Enterococcus spp. Group B streptococcus Ciprofloxacin; 500 mg twice daily, oral 3 days Uncomplicated Pyelonephritis (Perform culture and susceptibilities) Pseudomonas aeruginosa Urinalysis Reflex to Culture Criteria Positive nitrite First-line agents Ciprofloxacin; 500 mg twice daily, oral 7 days Positive leukocyte esterase Moderate to many bacteria Second-line agents Trimethoprim/sulfamethoxazole 800/160 mg DS twice daily, oral 14 days >5 WBC/hpf Other Organisms Staphylococcus saprophyticus Aerococcus spp. Routinely susceptible to nitrofurantoin, Bactrim and fluoroquinolones E Routinely susceptible to nitrofurantoin and amoxicillin E Isolates included in this antibiogram are from non-specialty outpatient clinics throughout New Mexico. Black shaded antibiotics have no coverage. Gray shaded antibiotics are <50% susceptible, Not Tested or No Data. Antibiotics included represent oral formulations available. Susceptibilities in green for oral treatment options are greater than or equal to 80%. *Organisms with <30 isolates have limited statistical significance of the result. (A) 2015 Beers Criteria recommendations state the use of nitrofurantoin is considered inappropriate for treatment of UTI in patients 65 years of age or older with a creatinine clearance less than 30 m. L/min. (B) Gupta K, et al. Clin Infect Dis. 2011; 52: e 103 -e 120 (IDSA Guidelines); Gupta K, Boucher HW. Infectious Disease Clinics of North America. Vol 28. No. 1. Elsevier Inc; 2014. (C) Oral cephalexin, cefdinir, cefaclor and cefpodoxime susceptibility is predicted by cefazolin susceptibility testing. (CLSI Document M 39 -A 4, 4 th ed. Copyright 2014. (D) ESBL positive rate for E. coli is about 3%, and K. pneumoniae is about 2%. (E) CLSI Document M 100, 27 th ed. Copyright 2017. Higgins A, et al. Urol Case Rep. 2017; 13: 24 -25.
- Slides: 1