Outlines Introduction Body acidity has to be kept


Outlines

Introduction • Body acidity has to be kept at a fairly constant level. • Normal p. H range within body fluids 7. 35 7. 45. • Normal p. H is constantly being attacked by metabolic processes within the body that produces acidic metabolites.

Acid-Base Balance • An acid is a proton donor and a base is a proton acceptor. • Physiologically, there are two groups of important acids: • Carbonic acid (H 2 CO 2) • Non carbonic acid

Carbonic acid (H 2 CO 2) • Carbonic acid comes from CHO and fat metabolism and results in 15, 000 mmol of CO 2/day. Carbonic acid metabolism is mostly handled by respiration. Recall: • CO 2 + H 20 H 2 CO 3

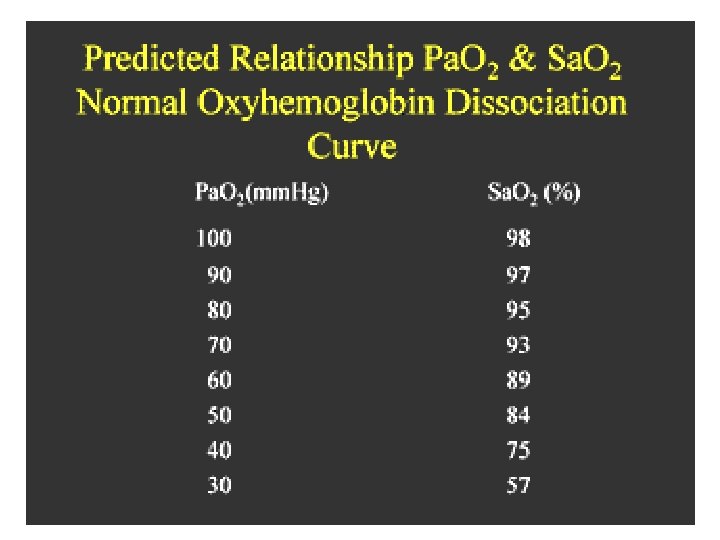
Factors involved in gas exchange • Many facotrs affects gas exchange, this process occurs between the alveoli & pulmonary capillaries & between capillaries & tissue. Its involvied : • Partial pressure • Diffusion • Ventilation-perfusion matching • Oxyhemoglobin dissociation

1. Partial pressure • When assessing a patient's oxygenation, one of the first steps is to look at Pa. O 2 is the partial pressure of oxygen dissolved in blood. • Normal Pa. O 2 is 80 to 100 mm. Hg; however this is normal for a healthy person, less than 60 years old, breathing room air.
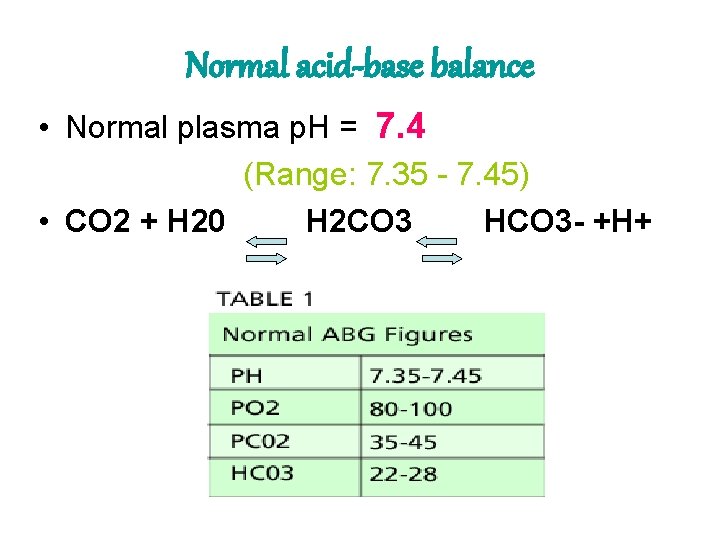
Normal acid-base balance • Normal plasma p. H = 7. 4 (Range: 7. 35 - 7. 45) • CO 2 + H 20 H 2 CO 3 HCO 3 - +H+
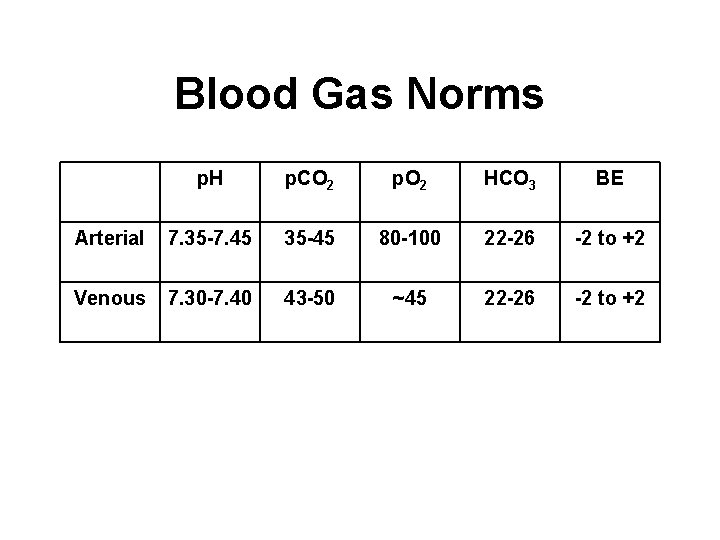
Blood Gas Norms p. H p. CO 2 p. O 2 HCO 3 BE Arterial 7. 35 -7. 45 35 -45 80 -100 22 -26 -2 to +2 Venous 7. 30 -7. 40 43 -50 ~45 22 -26 -2 to +2

Acid-Base Regulation • Three mechanisms to maintain p. H – Respiratory (CO 2) – Buffer (in the blood: carbonic acid/bicarbonate, phosphate buffers, Hgb) – Renal (HCO 3 -)

Buffers • A buffer is a substance that can give or accept protons • i. e. H+, in a manner that tends to minimise changes in the p. H of the solution. • Usually buffers are composed of a weak acid (proton donor) and a weak base (proton acceptor) as shown in the following equation.

Regulation • The process of acid-base regulation involves: 1. Chemical buffering by intracellular and extracellular buffers 2. Control of p. CO 2 by normal respiratory function 3. Control of HCO 3 - concentration and acid excretion by the kidney

The Renal function • Reclaim filtered HCO 3 - (therefore, avoid HCO 3 - loss) • Regenerate HCO 3 - in an amount equal to that used as buffer • In contrast to the respiratory system, can fully compensate without any changes in the bicarbonate pool.

Respiratory Function • the respiratory system is able to compensate for changes in the acid/base balance by increasing or decreasing ventilatory rate. • This would result in an increase or decrease of p. CO 2 in the blood. Thus changes are compensated at cost, i. e. changes in the bicarbonate pool.

Abnormal acid-base balance • Acid-base imbalances can be defined as acidosis or alkalosis. • Acidosis is a state of excess H+ • Acidemia results when the blood p. H is less than 7. 35 • Alkalosis is a state of excess HCO 3 • Alkalemia results when the blood p. H is greater than 7. 45

Acid-base disturbance Disorder type • Primary change in blood HCO 3 -→Metabolic disorder • Primary change in blood p. CO 2→Respiratory disorder

Steps for ABG interpretation • Step 1: Look at Pao 2 (hypoxemia) • Step 2: look at p. H (acid or alkaline) • Step 3: look at Paco 2 (resp. acidosis, alkalosis or normal) • Step 4: look at Hco 3 (metabolic acidosis, alkalosis, or normal) • Step 5: look back at p. H (compensated or uncompensated)

ABG interpretation ROME Use the acronym ROME (Respiratory opposite, Metabolic equal) to help you remember

Examples • • Pa. O 2: p. H: P a. CO 2: HCO 3 -: 90 mm. Hg 7. 25 50 mm. Hg 22 m. EqL (uncompensated respiratory acidosis)

Examples • • Pa. O 2: p. H: P a. CO 2: HCO 3 -: 90 mm. Hg 7. 25 40 mm. Hg 17 m. EqL (uncompensated metabolic acidosis)

Examples • • Pa. O 2: p. H: P a. CO 2: HCO 3 -: 90 mm. Hg 7. 37 60 mm. Hg 38 m. EqL (compensated respiratory acidosis with metabolic alkalosis main disordered is acidosis and alkalosis is the compensated, because the p. H is on the acid side of 7. 4))

Examples • • Pa. O 2: p. H: P a. CO 2: HCO 3 -: 90 mm. Hg 7. 42 48 mm. Hg 35 m. EqL (compensated metabolic alkalosis with respiratory acidosis main disordered is alkalosis and acidosis is the compensated, because the p. H is on the alkaline side of 7. 40)
- Slides: 23