Outline REACTA Forum 2014 Collaboration in industry Non


Outline • REACTA Forum 2014 • Collaboration in industry (Non. Trans. Celerate) • 제약회사 (Daiichi Sankyo) • 임상시험 센터 (삼성서울병원 CTC) • CRO (INC Research Korea)

REACTA (REgional Asian Clinical Trial Association) • CTC (주로 Asia 지역)의 모임 – Korea, Japan, Taiwan, HK, US, and Malaysia • REACTA Forum 2014 부산 동아대에서 개최 2014. 11. 6 -7 • Guiding purpose – 상호 협력 – 교육 – Regulatory harmonization





1. Yoon-Duk Han, Trans. Celerate OPTIMIZING THE CLINICAL TRIAL PROCESS THROUGH COLLABORATION

Trans. Celerate • A non-profit organization focused on advancing innovation in – R&D – Improving patient safety – Delivering more high quality medicines • Discusses issues facing the industry and solution for addressing common challenges • Support of the original 10 founding biopharmaceutical company members
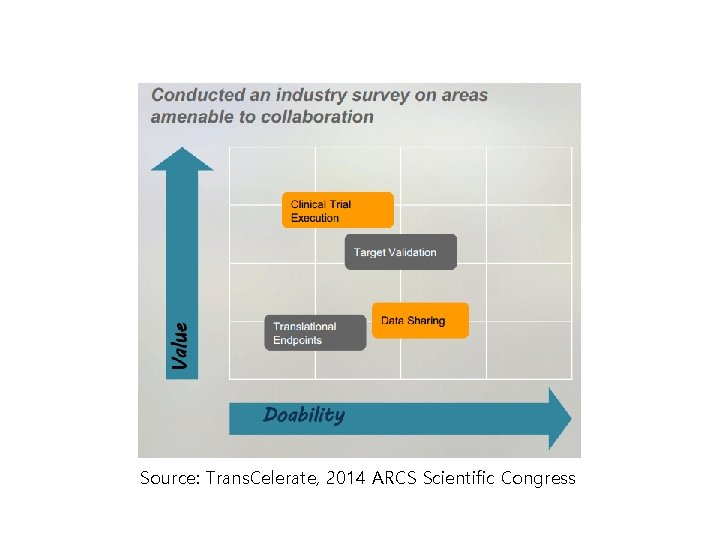
Source: Trans. Celerate, 2014 ARCS Scientific Congress

Source: Trans. Celerate, 2014 ARCS Scientific Congress

Source: Trans. Celerate, 2014 ARCS Scientific Congress
Source: Trans. Celerate, 2014 ARCS Scientific Congress
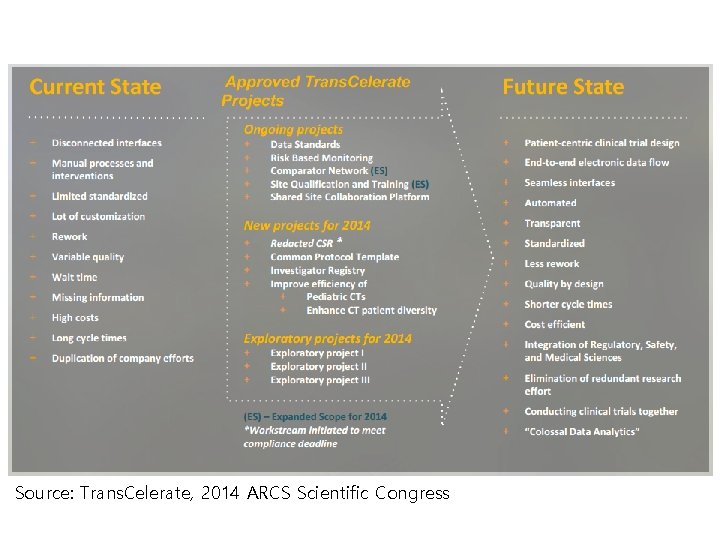
Source: Trans. Celerate, 2014 ARCS Scientific Congress

Source: Trans. Celerate, 2014 ARCS Scientific Congress

2. Koichi Miyazaki, Daichi Sankyo Co. , Ltd EFFICIENT MULTINATIONAL CLINICAL TRIALS FOR PATIENTS IN ASIA
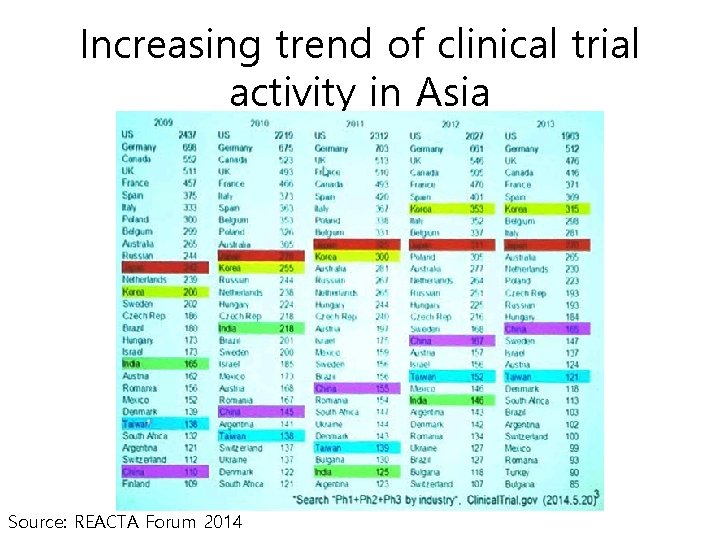
Increasing trend of clinical trial activity in Asia Source: REACTA Forum 2014
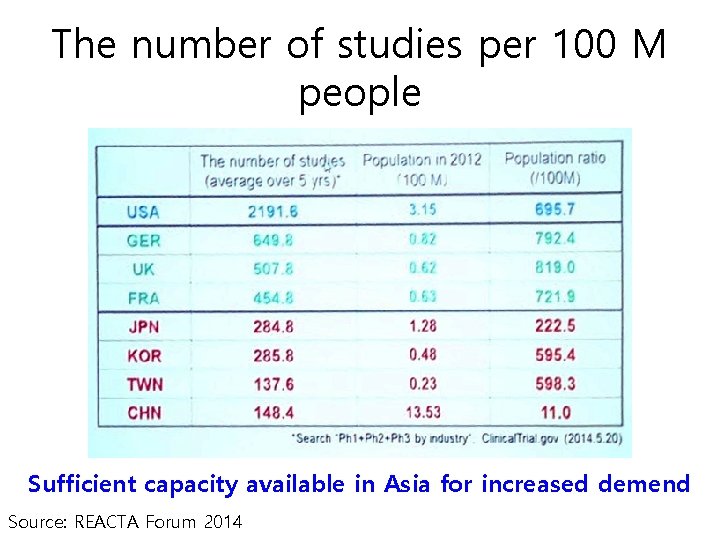
The number of studies per 100 M people Sufficient capacity available in Asia for increased demend Source: REACTA Forum 2014
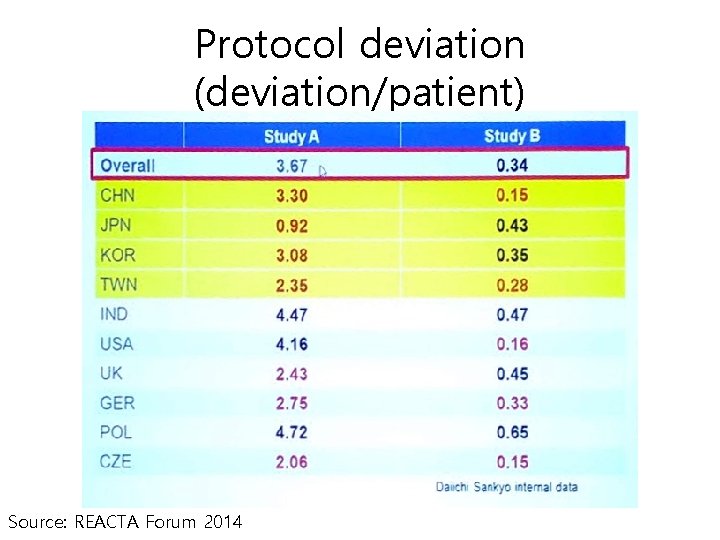
Protocol deviation (deviation/patient) Source: REACTA Forum 2014
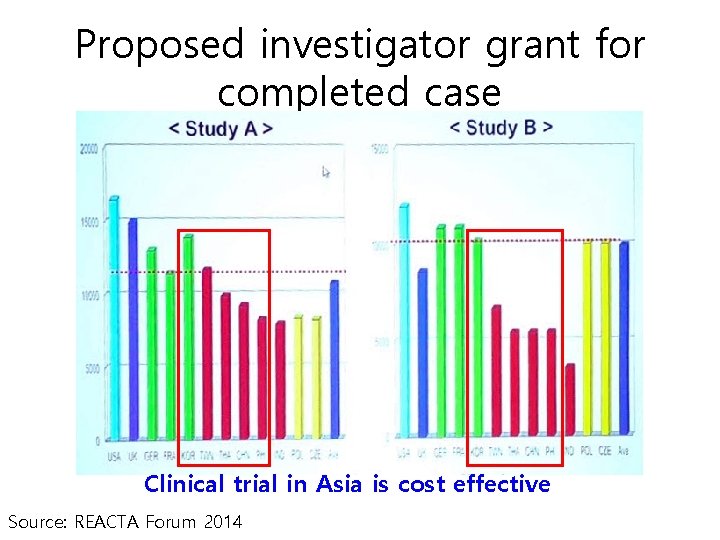
Proposed investigator grant for completed case Clinical trial in Asia is cost effective Source: REACTA Forum 2014
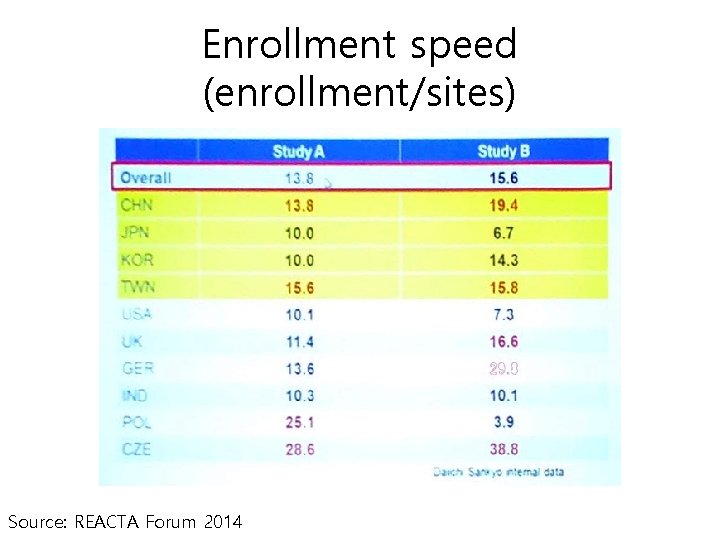
Enrollment speed (enrollment/sites) Source: REACTA Forum 2014
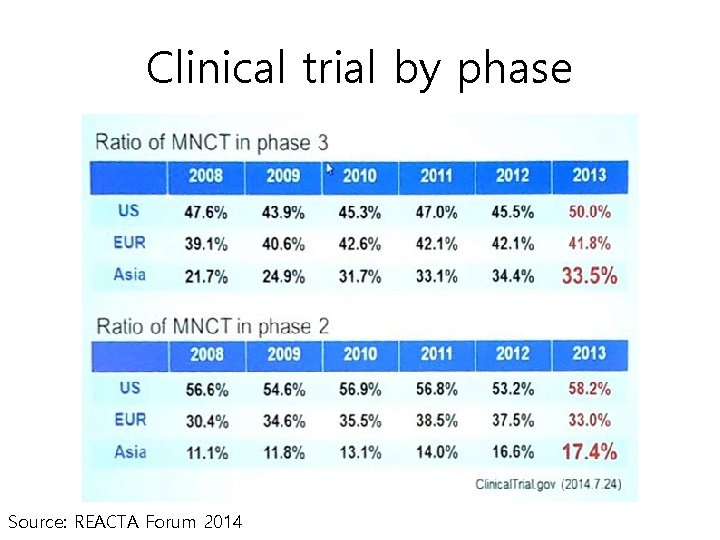
Clinical trial by phase Source: REACTA Forum 2014

Contribution from each region • … small region should contribute at least 21. 3% of the patients… • …guarantee reasonable power of … at least 20% Asian authorities may request more data from Asia Source: REACTA Forum 2014

Development strategy (past) Source: REACTA Forum 2014
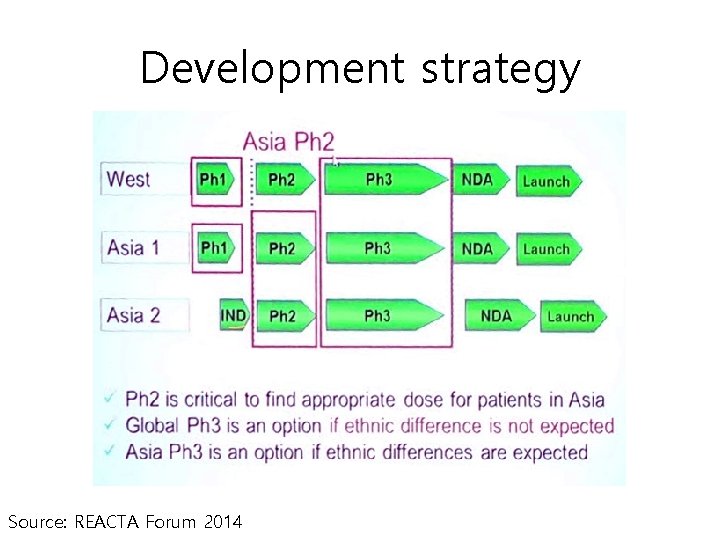
Development strategy Source: REACTA Forum 2014

Clinical study in Asia • Quality, cost and speed of Asia study are competitive • Asia should be involved from early stage trial to consider ethnic differences • Ideally, each region should contribute at lease 20% of the patients to ensure the consistency across regions Source: REACTA Forum 2014

3. Young. Suk Park, SAMSUNG Medical Center CTC SAMSUNG MEDICAL CENTER CLINICAL TRIAL CENTER

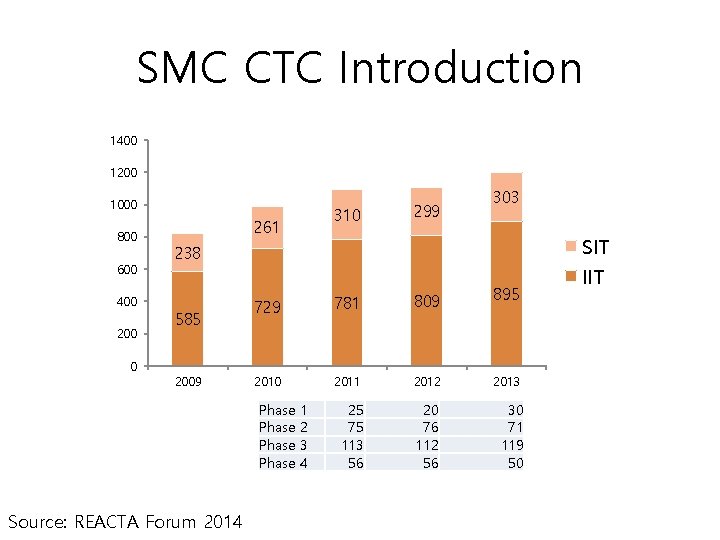
SMC CTC Introduction 1400 1200 1000 800 600 400 200 0 310 261 299 303 SIT 238 585 2009 729 781 809 2010 2011 2012 25 75 113 56 20 76 112 56 Phase Source: REACTA Forum 2014 1 2 3 4 895 2013 30 71 119 50 IIT

Phase 1 Unit Team at CTC □ Technology for early phase trials □ Member Researcher Number Pharmacologist 6 Pharmacist 2 Operation 1 CRC Oncology 6 Healthy Volunteer 6 Nurse 2 PK 6 Source: REACTA Forum 2014 • Phase 1 Study – – Single ascending dose Multiple ascending dose Drug-Drug Interaction BA/BE • Proof of Concept Study with New Biomarkers • Through QT/QTc Study • Multi Omics (Genomics/Proteomics/Metabolomics) • PK/PD M&S • New Statistical Tools for Innovative Design

Personalized Tx Clinical at ICMI Source: REACTA Forum 2014
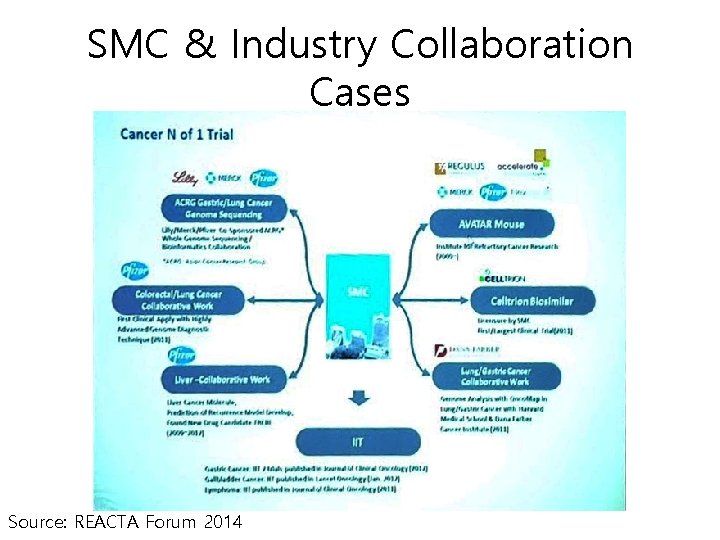
SMC & Industry Collaboration Cases Source: REACTA Forum 2014

Translational CRO Functions Source: REACTA Forum 2014

3. Hailey Chae, INC Research Korea SITE SELECTION

Site Selection Process • • Potential Investigators Evaluate Criteria Feasibility Study Pre-study Visit Source: REACTA Forum 2014

Identifying Potential Investigators • • • Literature Review Medical Society Directories Investigator Database Reference from Investigator Professional Colleagues Regulatory Lists Source: REACTA Forum 2014

Site Evaluation • Experience with similar trials • Interest Level (Prompt response for feasibility & any queries) • Patient pool • Competing trials • Resources (study coordinator/Sub-I/Rater) • EC (SOP/approval timeline/contract process) Source: REACTA Forum 2014

Site Evaluation • Staff – Study coordinator/Sub-I/Rater – General interest & attitude – Training • Facility – Storage for study supplies – Archiving practices: study documents – Device calibration: Freezer/Blood pressure Source: REACTA Forum 2014
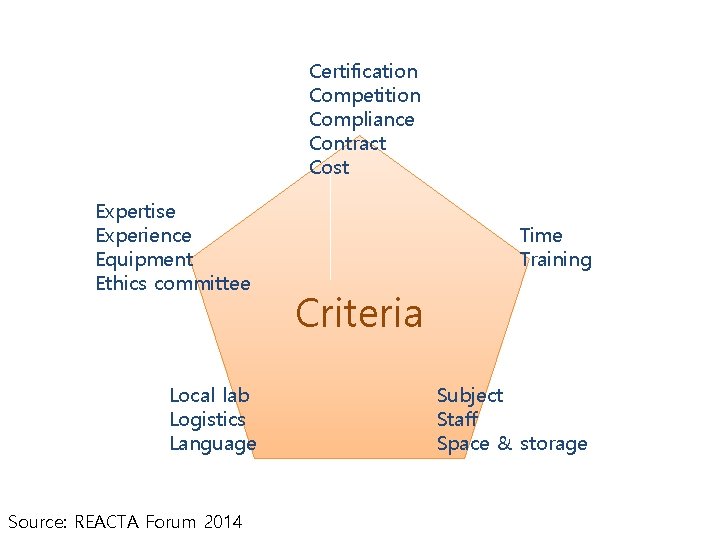
Certification Competition Compliance Contract Cost Expertise Experience Equipment Ethics committee Local lab Logistics Language Source: REACTA Forum 2014 Time Training Criteria Subject Staff Space & storage

Selection of site is both the Art and Science of Investigating Investigator Source: REACTA Forum 2014

Factors influencing clinical trial site selection in Europe • Applications to run clinical trials in Europe fell 25% between 2007 and 2011 • 19 factors explored: across investigator-driven, hospital-driven and environment-driven criteria, and costs. It also surveyed perceptions of the European trial environment • Investigator-dependent, environment-dependent and hospitaldependent factors were rated highly important, costs being less important • Within environment-driven criteria, pool of eligible patients, speed of approvals and presence of disease-management networks were significantly more important than costs or government financial incentives Source: REACTA Forum 2014

Investigator-driven criteria in the selection of phase II-III trial sites Criteria Mean Investigator recruitment/retention track record 27. 3 Investigator experience in previous trials 22. 7 Investigator interest 22. 4 Investigator concurrent workload 17. 2 Investigator publication track record 10. 4 Source: REACTA Forum 2014

Environment-driven criteria in the selection of phase II-III trial sites Criteria Mean Size of marketing/eligible patients in a region 23. 8 Speed of Mo. H/Ethics committees approval 23. 4 Disease management system/networks 18. 9 Cost of running trial 15. 2 Presence of country on ‘core country list’ 11. 8 Source: REACTA Forum 2014

Hospital-driven criteria in the selection of phase II-III trial sites Criteria Mean Site personnel experience and training 22. 0 Previous experience with site 20. 0 Facilities/equipment required by trial 19. 7 Hospital approval/contracting system 17. 4 Site personnel language proficiency 10. 8 Source: REACTA Forum 2014
Source: REACTA Forum 2014

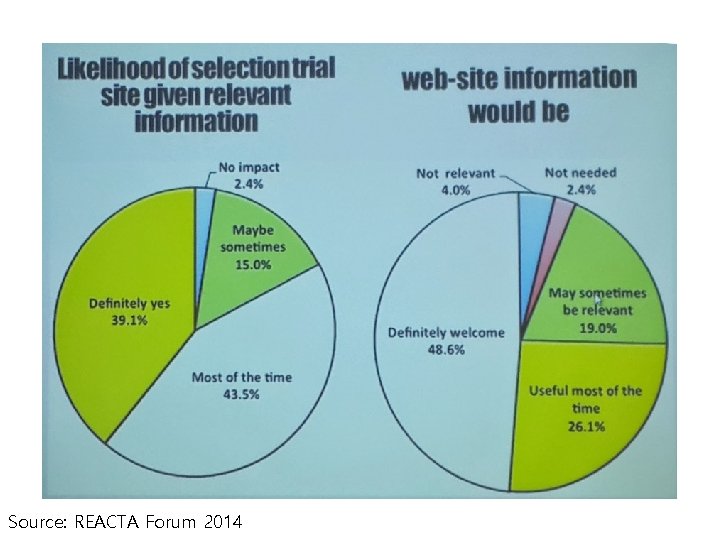
Institutional Considerations to procure more projects • Expand experience and qualified investigator pool by active involvement of sub-investigator group • Establish and maintain qualified and welltrained study coordinator resource via clinical trial center • Update regular trial experience and patient pool (Web-based/Newsletter) • Streamline and control feasibility process • Obtain regular feedback from industry Source: REACTA Forum 2014

- Slides: 47