Outcomes for the Future How Do We Improve






























- Slides: 52

Outcomes for the Future: How Do We Improve on the Status Quo? Ronald J. Sokol, MD Pediatric Liver Center & Digestive Health Institute Children’s Hospital Colorado University of Colorado School of Medicine Aurora, Colorado

Disclosures • Childhood Liver Disease Research Network (Chi. LDRe. N) funded by NIDDK/NIH, CF Foundation and Alpha-1 Foundation • Shire/Mirum, Retrophin, Alexion, Albireo • Investigational Drugs: ASBT inhibitors (Albireo and Shire/Mirum), chaparones, others

What is the “Status Quo”? • The state of affairs that exists at a particular time, especially in contrast to a different possible state of affairs • The current situation: the way things are now

What is the “Status Quo” for Chronic Pediatric Liver Disease? • Most diseases are Rare (orphan diseases) – exceptions NAFLD/NASH, gallstones, few others • Etiology of many diseases is unknown • Diagnostic odyssey for patients may be lengthy • Few diseases are treatable • Fewer diseases are curable • We rely on liver transplantation to rescue and save lives

“Status Quo” – Treatment and Cures • Curable: Hepatitis C

“Status Quo” – Treatment and Cures • Curable: Hepatitis C • Treatable: Hepatitis B, Tyrosinemia, Galactosemia, AIH, Bile acid synthesis defects, Wilson Disease, Hemochromatosis, Hypopituitarism, Hepatoblastoma

“Status Quo” – Treatment and Cures • Curable: • Treatable: Hepatitis C

Symptomatic - Palliation • Cholestatic Disorders: Biliary atresia, PFICs, Alagille syndrome, Alpha-1 AT deficiency, mitochondriopathies, ARC, CHARGE, etc. • • • NAFLD – NASH, ASH Genetic: ARPKD/CHF, CFLD Immune: PSC, HLH, GVHD Tumors Cirrhosis : HCC, CCA

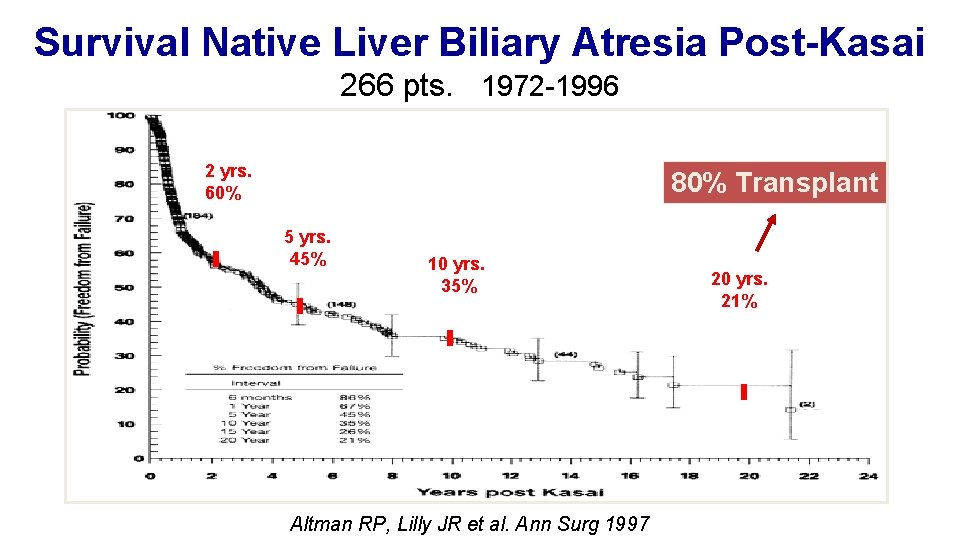
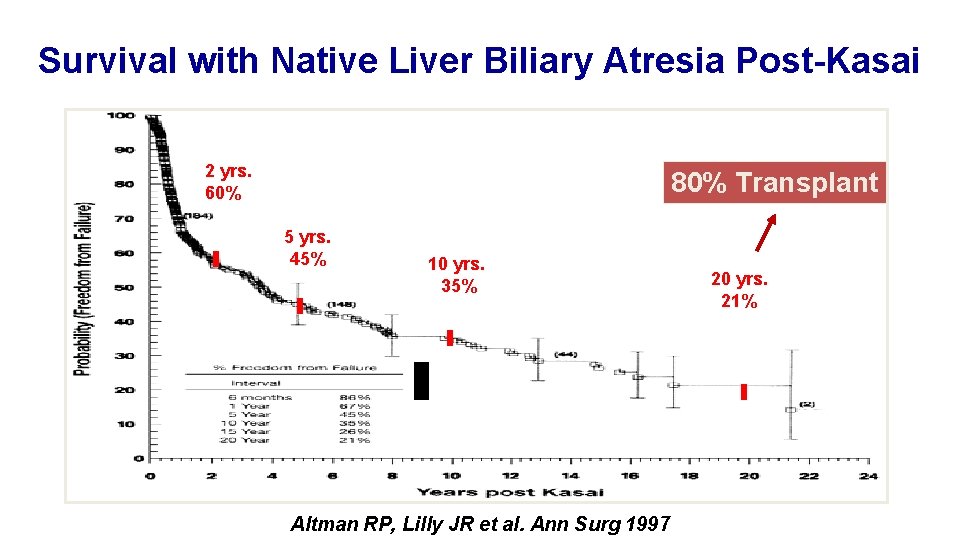
Survival Native Liver Biliary Atresia Post-Kasai 266 pts. 1972 -1996 2 yrs. 60% 80% Transplant 5 yrs. 45% 10 yrs. 35% Altman RP, Lilly JR et al. Ann Surg 1997 20 yrs. 21%

The Challenges for Improving Outcomes for These Patients Are Great The Opportunity is There for the Taking

How Do We Improve on the Status Quo? • Will use Neonatal Cholestatic Disorders as example • Etiologies and pathogenesis need definition • Treatments are inadequate • Living with these diseases is stressful for entire family • Many fatal if not transplanted • We need to “Move the Outcomes Needle”

6 Steps for “Moving the Outcomes Needle” (from the Status Quo) 1. Define genetics 2. Understand disease mechanisms - targets 3. Develop animal models, i. PSC, and organoids 4. Define natural history and therapeutic endpoints 5. High throughput screening vs. repurposing - Pipeline 6. Collaborative research networks and specimen biobanking

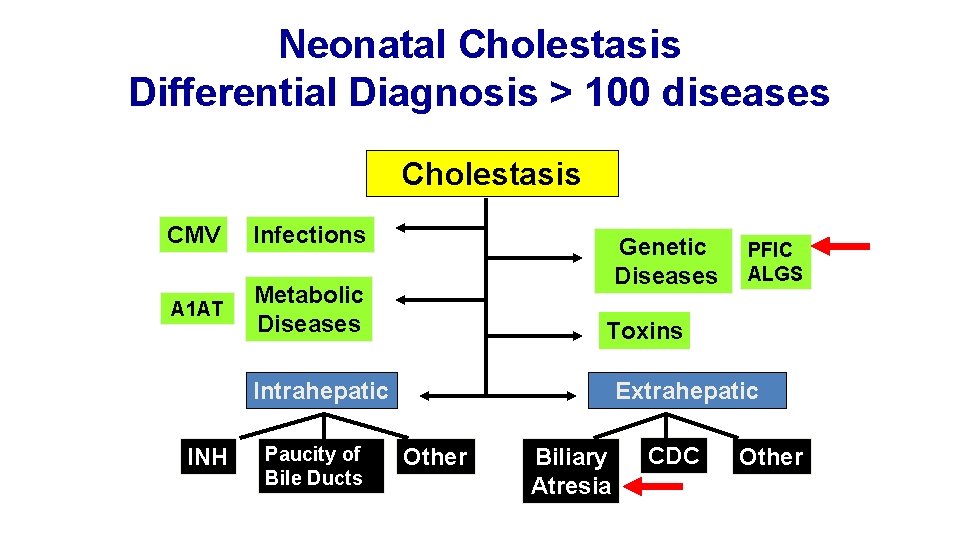
Cholestatic Liver Diseases • Hepatic or biliary condition characterized by poor or obstructed bile flow • Serum direct bilirubin > 20% of total and > 1. 0 mg/dl – Elevated serum bile acid concentrations – Evidence of fat malabsorption • 1 in 2, 500 live births affected • Over 100 conditions in infancy cause neonatal cholestasis

Neonatal Cholestasis Differential Diagnosis > 100 diseases Cholestasis CMV Infections A 1 AT Metabolic Diseases Genetic Diseases Toxins Intrahepatic INH Paucity of Bile Ducts PFIC ALGS Extrahepatic Other Biliary Atresia CDC Other

6 Steps for Moving the Outcomes Needle 1. Define genetics 2. Understand disease mechanisms - targets 3. Develop animal models, i. PSC, and organoids 4. Define natural history and therapeutic endpoints 5. High throughput screening vs. repurposing 6. Collaborative research networks and specimen biobanking

Progressive Familial Intrahepatic Cholestasis (PFIC) • 1 in 50, 000 to 100, 000 • By 6 months of life - direct hyperbilirubinemia or Jaundice, • • • malabsorption and poor growth, pruritus, FSV deficiencies ↑ serum bile acids, normal cholesterol Medical: UDCA, anti-pruritics - usually ineffective Nutritional: special infant formulae, FSV supplements Surgical: partial biliary diversion, ileal exclusion, liver transplant Unmet Needs!!

Genetics of PFIC • Autosomal recessive disorders • Canalicular transporters and regulators, tight junction proteins • • • PFIC 1 – FIC 1, ATP 8 B 1, Byler disease PFIC 2 – BSEP, ABCB 11 PFIC 3 – MDR 3, ABCB 4 PFIC 4 – TJP 2 PFIC 5 – FXR PFIC 6 – MYO 5 B PFIC 7 – OSTα/β PFIC 8 – UNC 45 PFIC 9 – PLEC PFIC 10 – MRP 9, ABCC 12

Newest Potential PFIC Genes • USP 53 and LSR - novel syndromic forms of cholestatic liver disease TGP, WES, WGS • KIF 12 - high GGT neonatal cholestasis/sclerosing cholangitis. • WDR 83 OS - intractable itching, hypercholanemia, dysmorphism, and intellectual disability • PPM 1 F - sclerosing cholangitis, short stature, hypothyroidism, and abnormal tongue pigmentation Wu SH et al. , Hepatology 2019 Jul 3 Maddirevula S, et al. Genetics in Medicine; 2019; 21: 1164


6 Steps for Moving the Outcomes Needle 1. Define genetics 2. Understand disease mechanisms - targets 3. Develop animal models, i. PSC, and organoids 4. Define natural history and therapeutic endpoints 5. High throughput screening vs. repurposing 6. Collaborative research networks and specimen biobanking

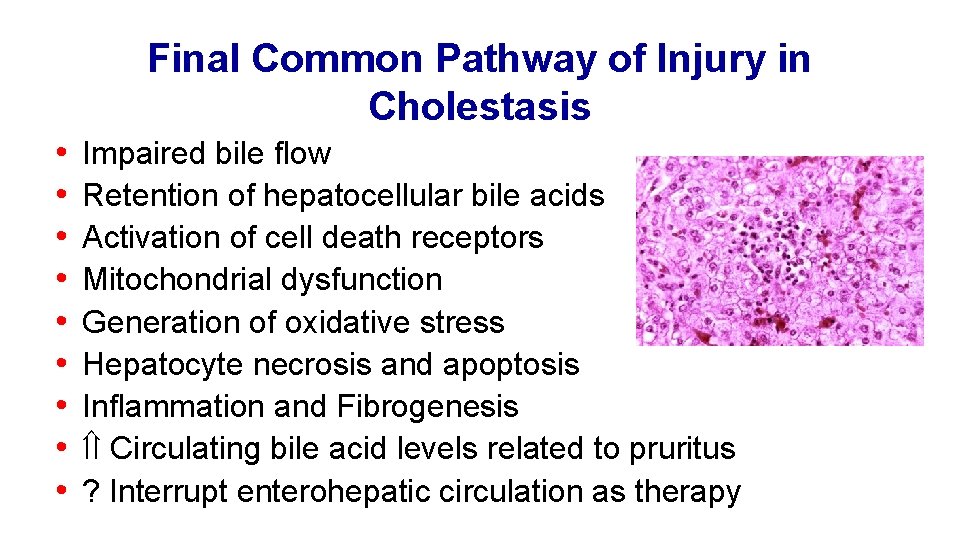
Final Common Pathway of Injury in Cholestasis • • • Impaired bile flow Retention of hepatocellular bile acids Activation of cell death receptors Mitochondrial dysfunction Generation of oxidative stress Hepatocyte necrosis and apoptosis Inflammation and Fibrogenesis Circulating bile acid levels related to pruritus ? Interrupt enterohepatic circulation as therapy

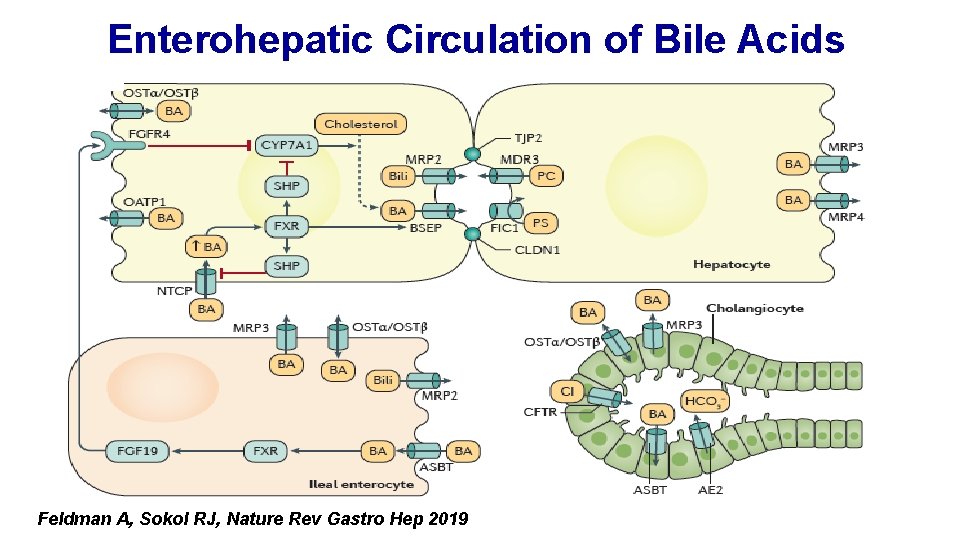
Enterohepatic Circulation of Bile Acids Feldman A, Sokol RJ, Nature Rev Gastro Hep 2019

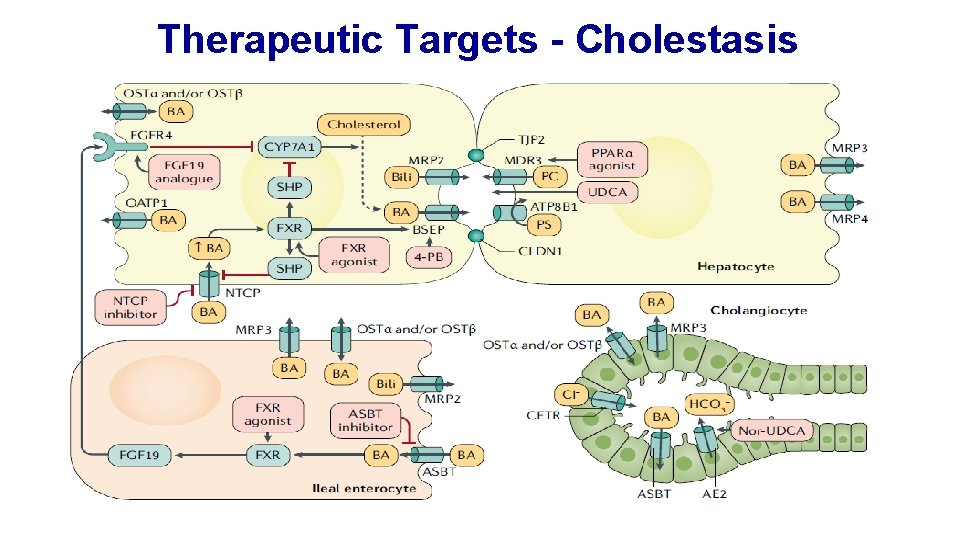
Therapeutic Targets - Cholestasis

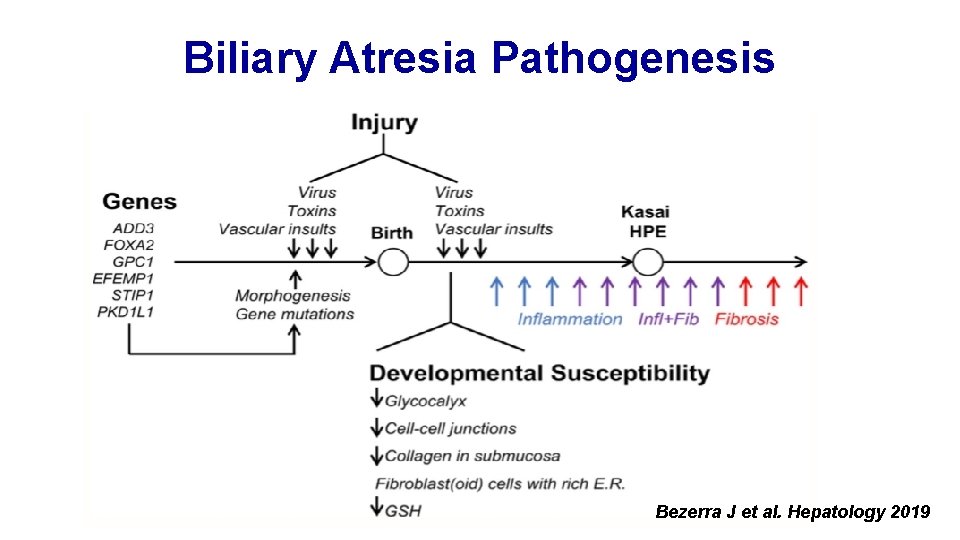
Biliary Atresia Pathogenesis Bezerra J et al. Hepatology 2019

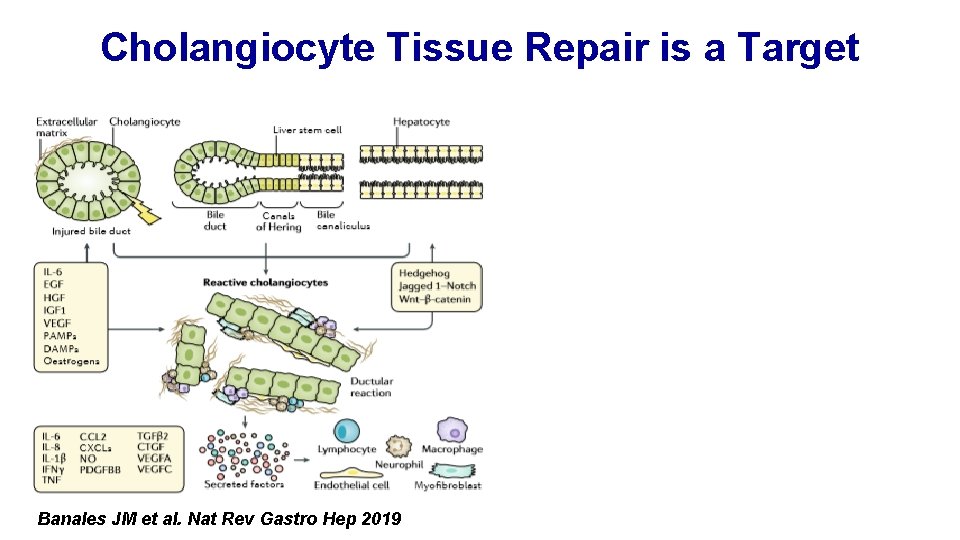
Cholangiocyte Tissue Repair is a Target Banales JM et al. Nat Rev Gastro Hep 2019

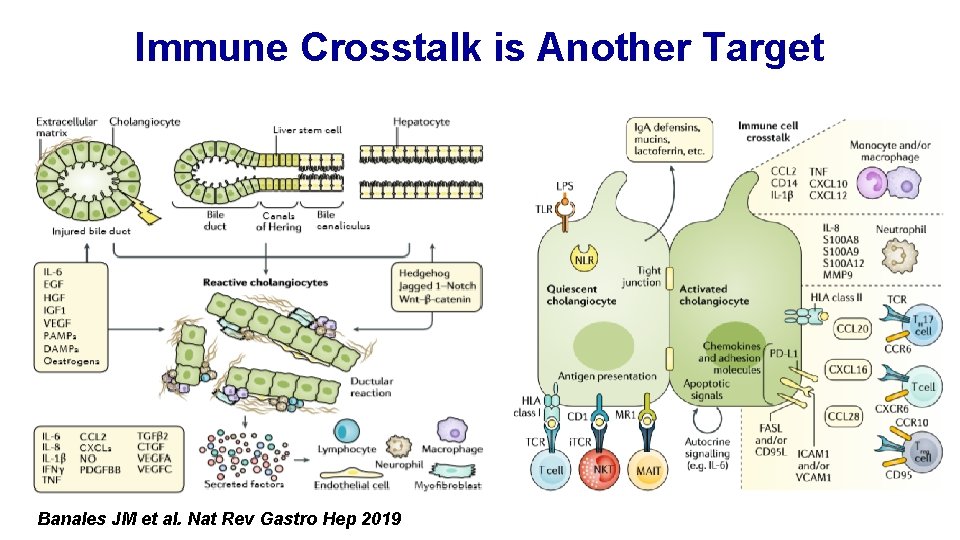
Immune Crosstalk is Another Target Banales JM et al. Nat Rev Gastro Hep 2019

6 Steps for “Moving the Outcomes Needle” (from the Status Quo) 1. Define genetics Understand pathophysiology of disease 2. Understand disease mechanisms - targets 3. Develop animal and organoids Identifymodels, and testi. PSC, therapies 4. Define natural history and therapeutic endpoints Potentially personalize therapies 5. High throughput screening vs. repurposing - Pipeline 6. Collaborative research networks and specimen biobanking

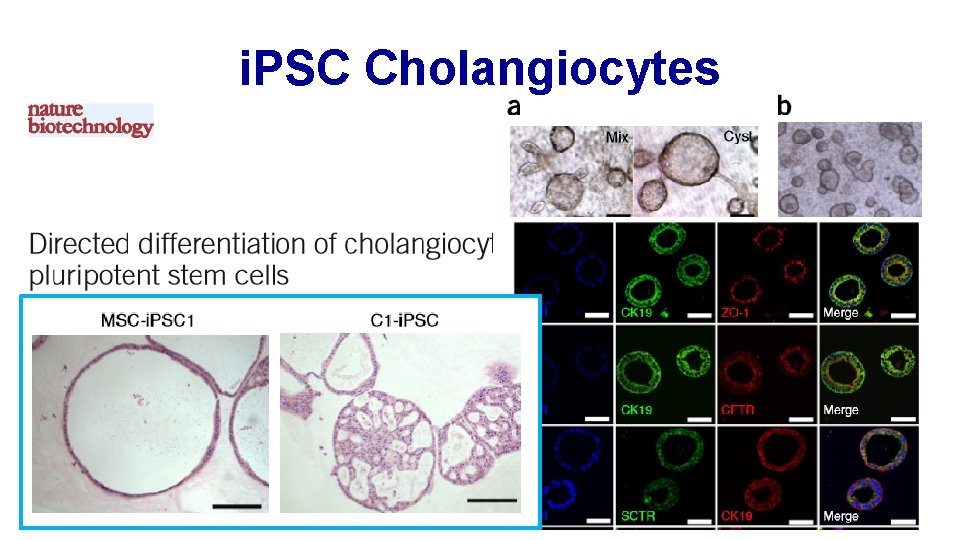
i. PSC Cholangiocytes

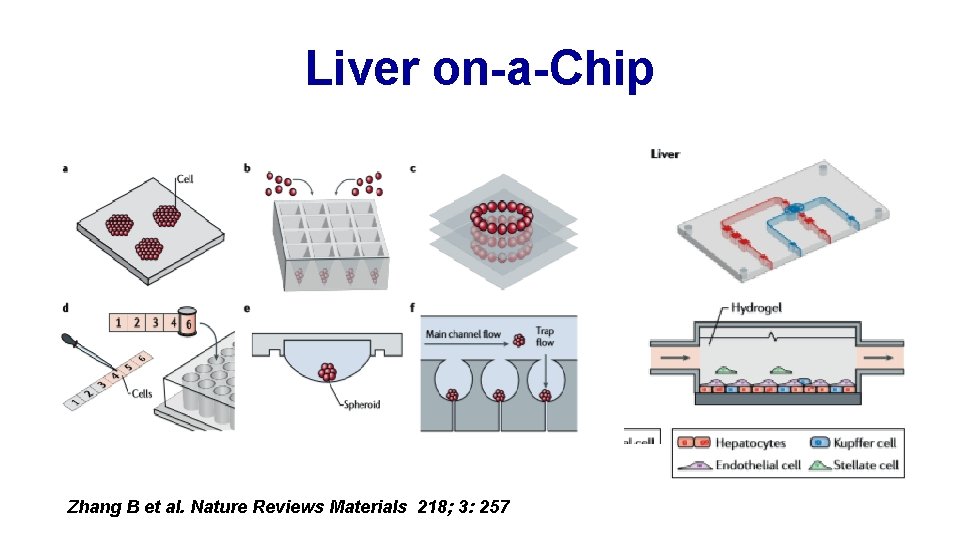
Liver on-a-Chip Zhang B et al. Nature Reviews Materials 218; 3: 257


6 Steps for “Moving the Outcomes Needle” (from the Status Quo) 1. Define genetics 2. Understand disease mechanisms - targets 3. Develop animal models, i. PSC, and organoids 4. Define natural history and therapeutic endpoints 5. High throughput screening vs. repurposing - Pipeline 6. Collaborative research networks and specimen biobanking

Natural History - Endpoints • NH generally poorly defined for most pediatric liver diseases – how can we measure effect of therapies? • Candidate Endpoints: Pruritus, FTT, varices, GI bleeding, ascites, death, liver transplant • Occur infrequently, difficult to measure (e. g. , PHT), may require invasive procedures (e. g. , endoscopy) • Need large numbers of well-phenotyped patients to establish time course of events for calculation of sample size and clinical trial design for new therapeutics

Natural History - Endpoints • NH generally poorly defined for most pediatric liver diseases – how can we measure effect of therapies? • Candidate Endpoints: Pruritus, FTT, varices, GI bleeding, ascites, death, liver transplant • Occur infrequently, difficult to measure (e. g. , PHT), may require invasive procedures (e. g. , endoscopy) • Need large numbers of well-phenotyped patients to establish time course of events for calculation of sample size and clinical trial design for new therapeutics

Survival with Native Liver Biliary Atresia Post-Kasai 2 yrs. 60% 80% Transplant 5 yrs. 45% 10 yrs. 35% Altman RP, Lilly JR et al. Ann Surg 1997 20 yrs. 21%

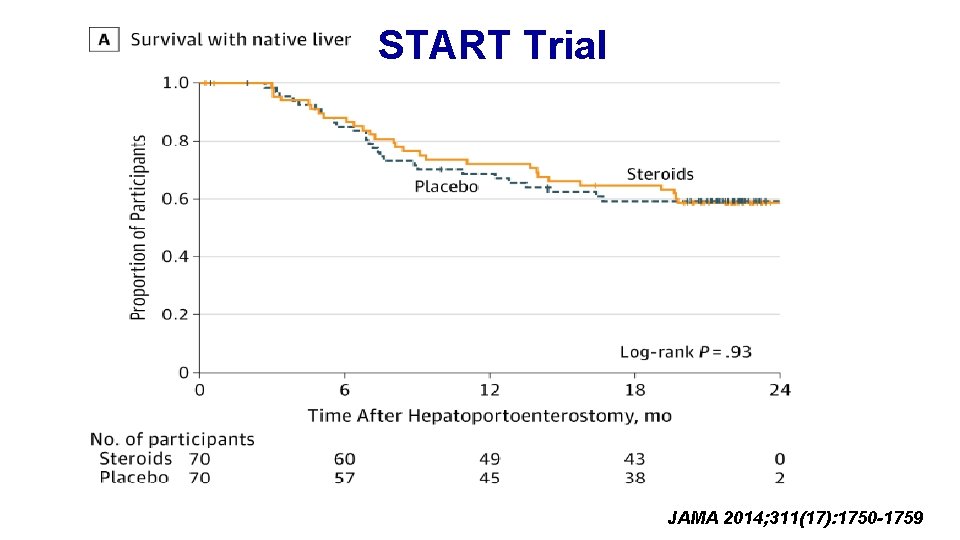
START Trial JAMA 2014; 311(17): 1750 -1759

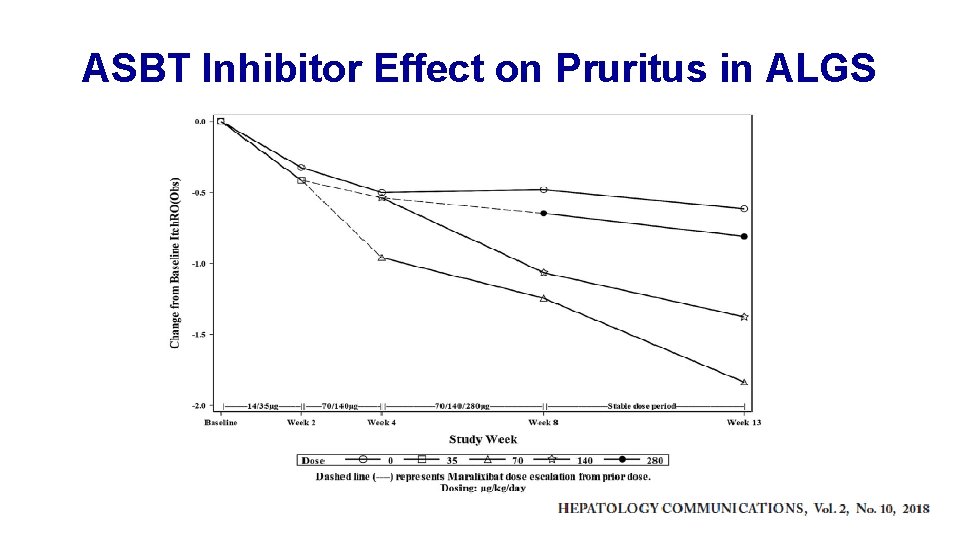
ASBT Inhibitor Effect on Pruritus in ALGS

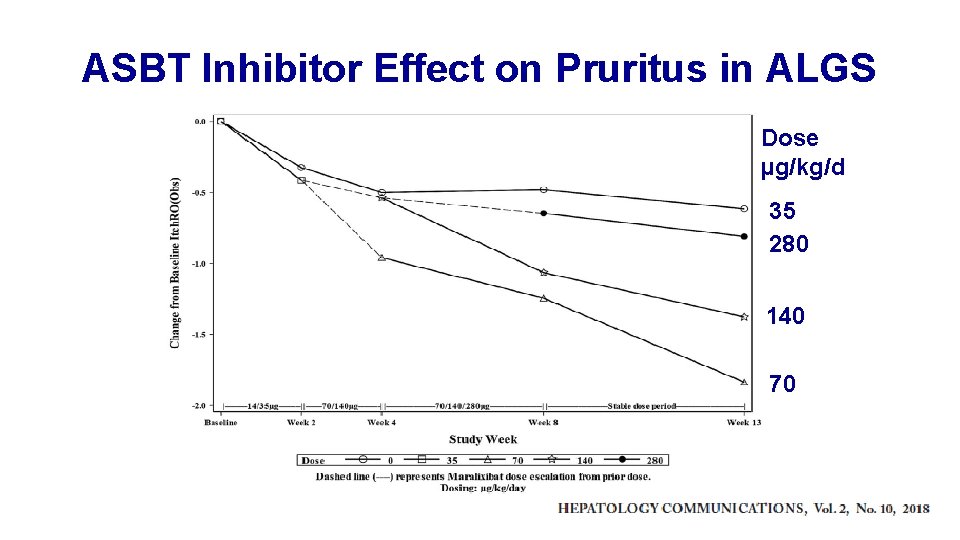
ASBT Inhibitor Effect on Pruritus in ALGS Dose µg/kg/d 35 280 140 70

Study Endpoints • Death/Transplant – slow to enroll, 2 -year endpoint, 5 -6 years to close study and analyze • Pruritus – imprecise metrics, participant vs. parent vs. clinician assessment • Need biomarkers Serum bile acids for ASBT inhibitor? Elastography for hepatic fibrosis? • Novel surrogate biomarkers needed!!!!

6 Steps for “Moving the Outcomes Needle” (from the Status Quo) 1. 2. 3. 4. Define genetics Understand disease mechanisms - targets Develop animal models, i. PSC, and organoids Define natural history and therapeutic endpoints 5. High throughput screening vs. repurposing Pipeline 6. Collaborative research networks and specimen biobanking


6 Steps for “Moving the Outcomes Needle” (from the Status Quo) 1. Define genetics 2. Understand disease mechanisms - targets 3. Develop animal models, i. PSC, organoids Solution to and # 15 4. Define natural history and therapeutic endpoints 5. High throughput screening vs. repurposing - Pipeline 6. Collaborative research networks and specimen biobanking

Pediatric Liver Disease Research Networks • • • SPLIT – liver transplant PALF – acute liver failure BARC - biliary atresia, INH NASH CRN Peds. C – hepatitis C CLi. C – ALGS, A 1 AT, BASD, PFIC, MITOHEPs Chi. LDRe. N = BARC + CLi. C + PSC CFLD – cystic fibrosis liver disease NAPPED – Europe – PFIC Special Interest Groups – NASPGHAN, AASLD, others

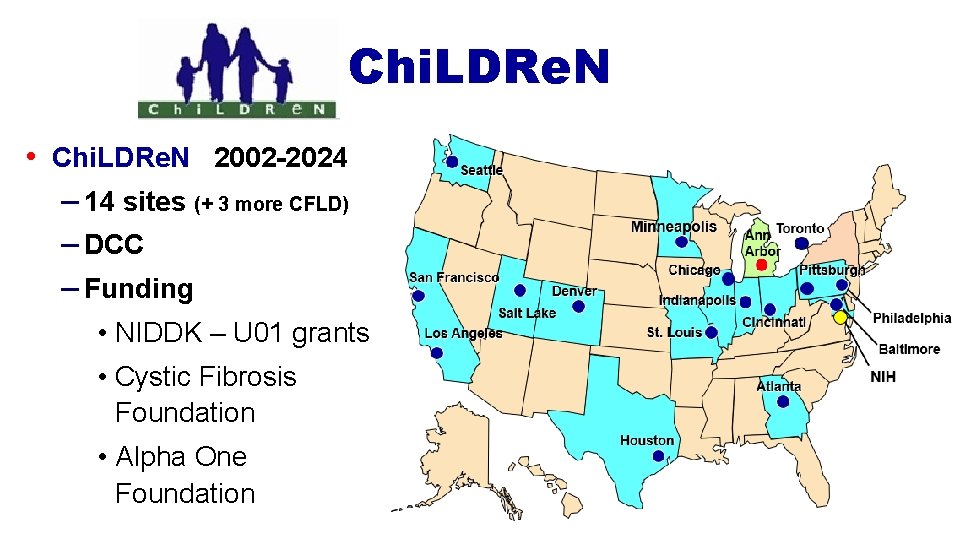
Chi. LDRe. N • Chi. LDRe. N 2002 -2024 – 14 sites (+ 3 more CFLD) – DCC – Funding • NIDDK – U 01 grants • Cystic Fibrosis Foundation • Alpha One Foundation

Chi. LDRe. N Diseases Studied 1. 2. Biliary atresia Idiopathic neonatal hepatitis (transient neonatal cholestasis) 3. 4. 5. Alpha-one antitrypsin deficiency 6. 7. Bile acid synthesis defects Alagille syndrome Progressive familial intrahepatic cholestasis (PFIC) and BRIC Mitochondrial hepatopathies – respiratory chain and fatty acid oxidation defects 8. Cystic fibrosis liver disease 9. Primary Sclerosing Cholangitis (PSC)

Chi. LDRe. N Participant Population Cohorts • Large cohorts with these 9 rare diseases studied for 20 • • • years longitudinally – Big Data Thoroughly phenotyped Standardized ontologies, data collection points, data elements Biospecimens linked to visits - Biomarkers Highly motivated families and subjects Could not be accomplished in single site Natural history and endpoints defined; Clinical Trials

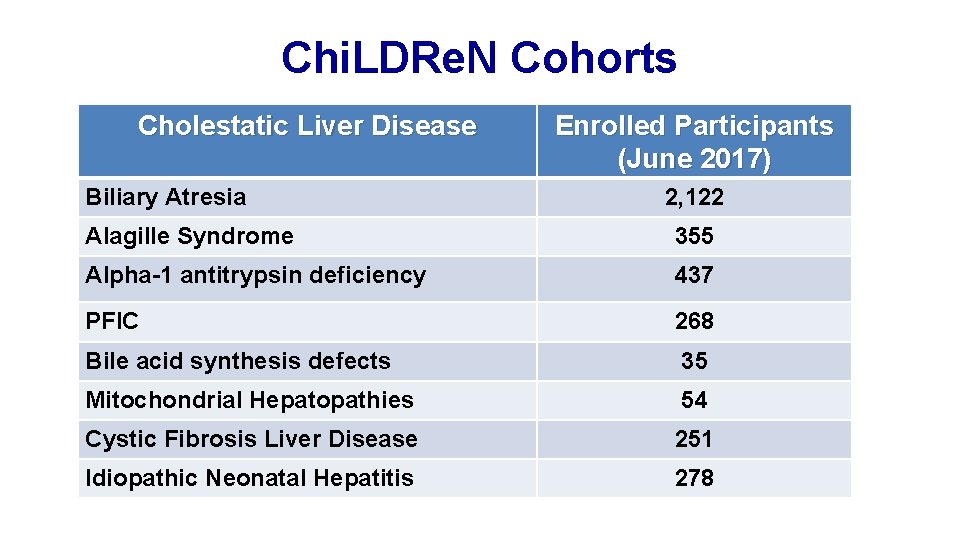
Chi. LDRe. N Cohorts Cholestatic Liver Disease Biliary Atresia Enrolled Participants (June 2017) 2, 122 Alagille Syndrome 355 Alpha-1 antitrypsin deficiency 437 PFIC 268 Bile acid synthesis defects 35 Mitochondrial Hepatopathies 54 Cystic Fibrosis Liver Disease 251 Idiopathic Neonatal Hepatitis 278

Bio-repository Specimens • > 185, 000 serum, plasma, urine, tissue specimens – linked to visits • > 6, 700 DNA (cell lines or DNA) • De-identified, aliquotted and bar coded • Standard collection methods & processing • Being used to identify susceptibility genes in BA; modifier genes in BA, ALGS and A 1 AT; exome sequencing for etiology of BASM, MITOHEP and PFIC, biomarker development • Clinical Trials

6 Steps for “Moving the Outcomes Needle” (from the Status Quo) 1. Define genetics 2. Understand disease mechanisms - targets 3. Develop animal models, i. PSC, and organoids 4. Define natural history and therapeutic endpoints 5. High throughput screening vs. repurposing - Pipeline 6. Collaborative research networks and specimen biobanking

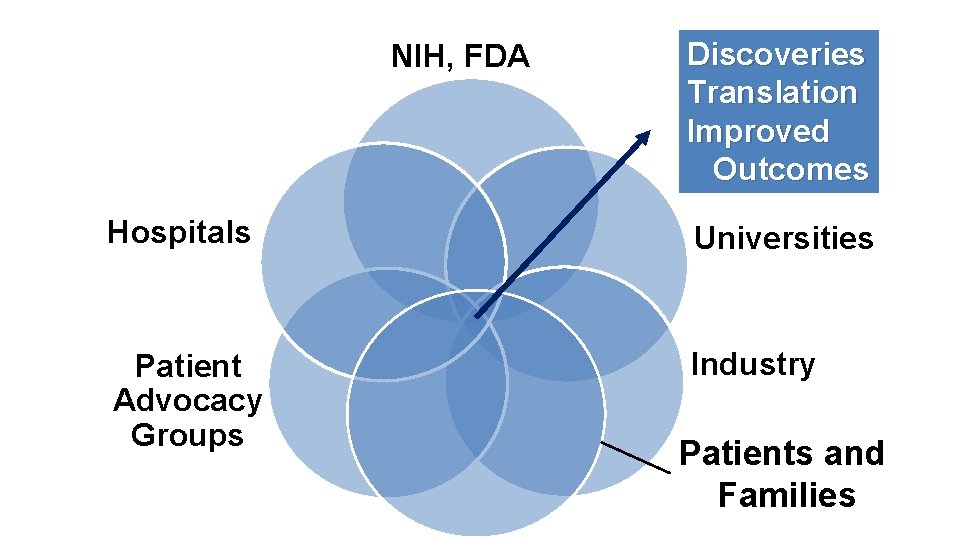
7 Steps for “Moving the Outcomes Needle” (from the Status Quo) 1. 2. 3. 4. 5. 6. Define genetics Understand disease mechanisms - targets Develop animal models, i. PSC, and organoids Define natural history and therapeutic endpoints High throughput screening vs. repurposing - Pipeline Collaborative research networks and specimen biobanking 7. Engage patients, families, PAGs, Industry, FDA, as partners

NIH, FDA Discoveries Translation Improved Outcomes Hospitals Universities Patient Advocacy Groups Industry Patients and Families

The Future • “Golden Age” of cholestasis therapeutics is around the corner • At least 9 drugs in the pipeline, in trials in adults, and some in children • New technology enables discovery of mechanisms, targets and development of therapeutics for all liver diseases • Encourage trainees and young investigators to become involved – get educated, trained and mentored

Thank you