Other Equilibrium Relationships Relative Volatility Lecture 6 1

Other Equilibrium Relationships – Relative Volatility Lecture 6 1

Calculation of Bubble-Point and Dew-Point Temperatures o o The bubble-point temperature is the temperature at which a liquid mixture begins to boil. The dew-point temperature is the temperature at which a vapor mixture first begins to condense. Lecture 6 2
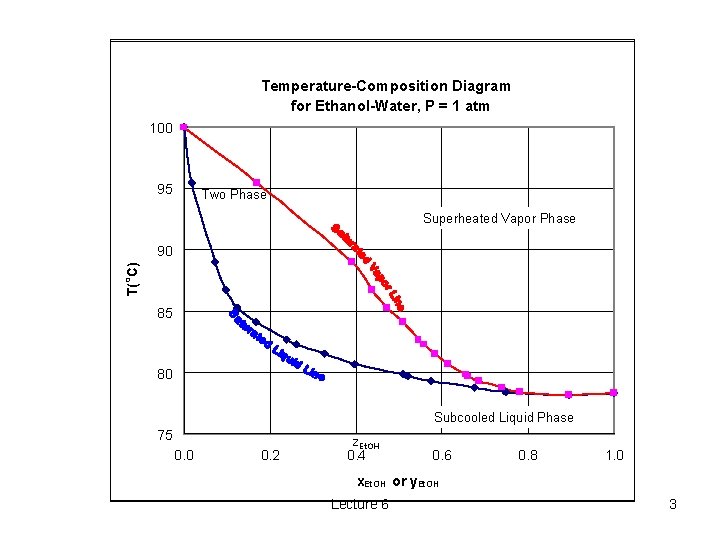
Temperature-Composition Diagram for Ethanol-Water, P = 1 atm 100 95 Two Phase Superheated Vapor Phase o T( C) 90 85 80 Subcooled Liquid Phase 75 0. 0 0. 2 z. Et. OH 0. 4 0. 6 0. 8 1. 0 x. Et. OH or y. Et. OH Lecture 6 3

Calculation of Bubble-Point Temperatures o o o If one is given a liquid mixture, one often needs to determine the bubble-point temperature of the mixture. We have done this to date using equilibrium data for binary systems – for example, from the saturated liquid line on a T vs. x, y plot for a given feed composition, zi. How do we handle multi-component systems? Lecture 6 4

Calculation of Bubble-Point Temperatures o o If the feed is in the liquid phase, the pressure p and the composition, xi’s, of the liquid phase will be given. One then needs to determine the bubble-point temperature of the mixture. Lecture 6 5

Calculation of Bubble-Point Temperatures – Where do we start? o Well, we are given the liquid-phase mole fractions, and we know that the mole fraction relationships for both the liquid and vapor phase are given by Eq. (2 -13): Lecture 6 6

Calculation of Bubble-Point Temperatures – Equilibrium Relationship o We are given the liquid-phase mole fractions, xi’s, and we need to “link” the vapor-phase mole fractions, yi’s, to the liquid-phase mole fractions – to do this, one can use the definition for the equilibrium distribution coefficient K, Eq. (2 -10) and solve for yi: or Lecture 6 7

Calculation of Bubble-Point Temperatures – Equilibrium Distribution Coefficient Relationship o o Substituting Eq. (2 -10) into Eq. (2 -13) yields: K is a function of both temperature and pressure, Eq. (2 -11): Lecture 6 8

Calculation of Bubble-Point Temperatures – Equilibrium Distribution Coefficient T, P Relationship o o One needs an expression for K as a function of T and P. One convenient expression for K as a function of T and P for light hydrocarbons is the De. Priester equation, Eq. (2 -12): Lecture 6 9
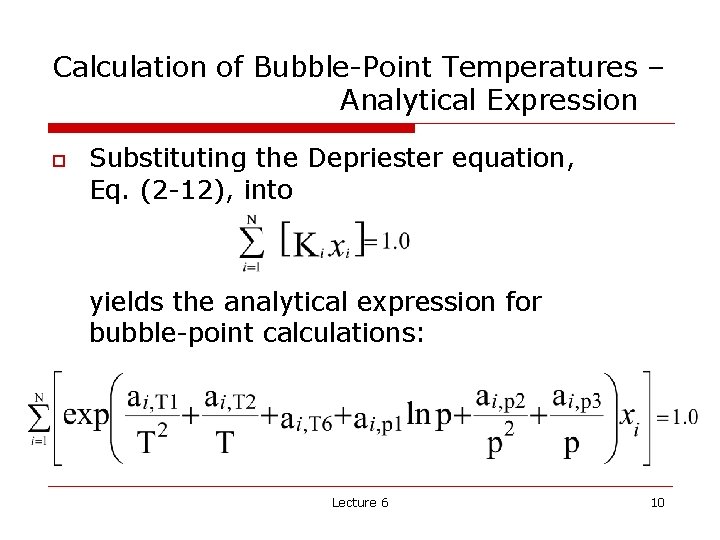
Calculation of Bubble-Point Temperatures – Analytical Expression o Substituting the Depriester equation, Eq. (2 -12), into yields the analytical expression for bubble-point calculations: Lecture 6 10

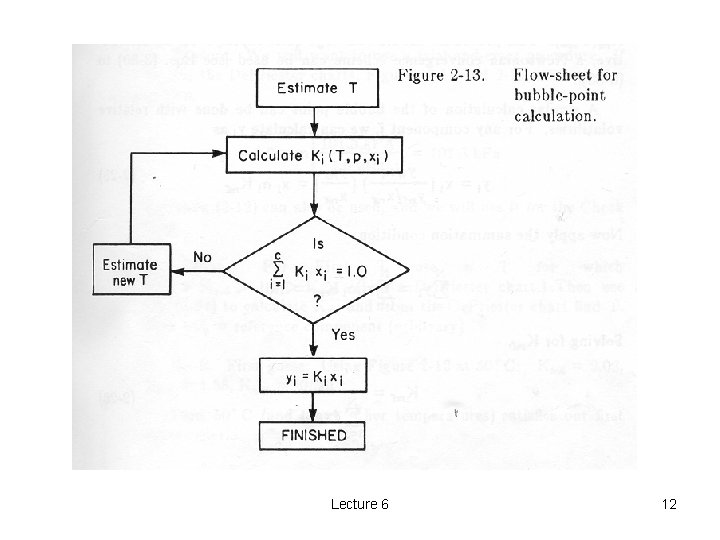
Calculation of Bubble-Point Temperatures o o One must solve the bubble-point expression for T. There are several ways to solve for T: 1. ) If the expression for K is simple enough, one may be able to algebraically solve for T – e. g. , if some of the constants in the De. Priester equation are 0. 2. ) One may use a trail and error method as outlined in Fig. 2 -13, Wankat, p. 29. 3. ) One may solve numerically. Lecture 6 11
Lecture 6 12

Calculation of Dew-Point Temperatures o o If the feed is in the vapor phase, the pressure p and the composition, yi’s, of the vapor phase will be given. One then needs to determine the dewpoint temperature of the mixture. Lecture 6 13

Calculation of Dew-Point Temperatures o Just as one may derive the bubble-point temperature relationship, one can use a similar derivation using the equilibrium coefficient equation definition, Eq. (2 -10), but this time solving for xi, since one would be given the vapor-phase mole fractions, yi’s: or Lecture 6 14

Calculation of Dew-Point Temperatures – Equilibrium Distribution Coefficient Relationship o o Substituting Eq. (2 -10) into Eq. (2 -13) yields: K is a function of both temperature and pressure, Eq. (2 -11): Lecture 6 15

Calculation of Dew-Point Temperatures– Analytical Expression o Substituting the Depriester equation, Eq. (2 -12), into yields the analytical expression for dewpoint calculations: Lecture 6 16

Calculation of Bubble-Point and Dew-Point Temperatures – Numerical Solutions o o One can conveniently use numerical methods for these types of problems, using, for example, Mathcad uses non-linear numerical methods such as the Quasi. Newtonian or Levenberg-Marquardt algorithms to solve equations. Lecture 6 17

A Final Note! o While we will solve bubble-point and dew-point temperature problems for a given pressure, there is no reason why this same methodology cannot be applied to determining bubblepoint and dew-point pressure problems for a given temperature! Lecture 6 18
- Slides: 18