OSP Quarterly Brown OSP Brown Bag September 28

OSP Quarterly Brown OSP Brown Bag September 28, 2017 Office of Sponsored Projects 1

OSP Brown Update Bag Katie Fleck Associate Director, Pre-Award Services Office of Sponsored Projects 2

OSP Brown Update Bag • OSP is moving! • OVPR’s scheduled move date is November 10, 2017 • We will be moving as part of Phase II of the overall SSL transition. Phase I includes Finance and UHR, and EHS • While we don’t anticipate any downtime during this move, this date is very close to many important sponsor deadlines. So if you’re able to route proposals early, please do so Office of Sponsored Projects 3

Pre OSP Award Brown Update Bag • New Advance Account Procedure • Codifies OSP existing process around creating Advance Accounts using UPAS o Reminder that all standard account set-up requirements, such as FCOI Training, IRB/IACUC Approvals, etc. must be complete before an Advance Account can be created Office of Sponsored Projects 4

Pre OSP Award Brown Update Bag • New Advance Account Procedure (contd. ) • Clarifies when additional information or back-up may be necessary o If Advance is Subcontract, we may ask for confirmation from PTE o If Advance is for Federal Award, we will ask that any sponsor communication be attached Office of Sponsored Projects 5

Pre OSP Award Brown Update Bag • New Advance Account Procedure (contd. ) • Expands the types of awards under which Advance Accounts are allowable • Going forward, OSP will now accept UPAS for Advance Accounts under Contracts, in addition to Grants • If the Award/Contract is currently under negotiation in OSP or Office for Industry Engagement and Commercial Venturing (formerly TVO) additional steps are required Office of Sponsored Projects 6

Pre OSP Award Brown Update Bag • New Advance Account Procedure (contd. ) • For Awards under negotiation, OSP will send a summary of the negotiation points to the departmental staff • Before the Advance Account is created, OSP must receive confirmation that the Dean, Department Chair, or Center Director is aware of these risks and would still like to proceed with the Advance Account Office of Sponsored Projects 7

Pre OSP Award Brown Update Bag • New Advance Account Procedure (contd. ) • There may be some scenarios in which an Advance Account cannot be allowed • If the University at whole would be put at risk by any award terms (such as those that may jeopardize our Fundamental Research Exemption), an Advance Account will not be created. Office of Sponsored Projects 8

Pre OSPAward Brown. Update Bag • Proposal Reminders • Reminder about NSF Form Types- please ensure you select the correct Narrative Type for your documents in COEUS o Supplementary Documents – Can include Commitment Letters, Other Documents that may be requested in the FOA o NSF Single Copy Documents –These are for NSF Use Only, not transmitted to reviewers Office of Sponsored Projects 9

Pre OSPAward Brown. Update Bag • Proposal Reminders (contd. ) • If a proposal hierarchy is used for a S 2 S submission, someone must be available from each participating Department/Unit on deadline day • If a Change/Corrected proposal is needed, each participating Unit will need to copy their individual proposal and re-link to the new parent proposal Office of Sponsored Projects 10

Pre Award OSP Brown. Update Bag • NIH Update • Forms E coming soon • Will be required for all proposals due on or after January 25 th, 2018 • Updated budget form • Will consolidate human subjects, inclusion enrollment and clinical trial information into single section of proposal • More info can be found here: https: //grants. nih. gov/grants/funding/Grant _Application_Form_Update-FORMS-E. pdf Office of Sponsored Projects 11

Pre Award OSP Brown. Update Bag • NIH Update (contd. ) • Updated Certificate of Confidentiality Policy (NOT-OD-17 -109) • Goes into effect October 1, but retroactive to awards active in December 2016 that involve human subjects • Eliminates need for NIH funded PI’s to apply for Co. C, instead Co. C will be automatically issued to applicable awards Office of Sponsored Projects 12

Pre Award OSP Brown. Update Bag • NIH Update (contd. ) • Updated Definition of Clinical Trial • Asks 4 questions; if answer to all is “yes”, the project should be identified as a Clinical Trial: o Does the study involve human participants? o Are the participants prospectively assigned to an intervention? o Is the study designed to evaluate the effect of the intervention on the participants? o Is the effect that will be evaluated a health-related biomedical or behavioral outcome? • Recently-released FAQ and Case Studies have raised additional questions, some organizations are pushing back against this new definition Office of Sponsored Projects 13

Pre Award OSP Brown. Update Bag • NIH Update (contd. ) • Next Generation Researchers Policy (NOT-OD-17 -101) released August 31, 2017 • NIH will prioritize applications that request funding for Early Stage Investigators (ESIs) and for Early Established Investigators (EEIs) who are at risk for losing all NIH funding or who have only one active NIH award • In order to ensure that the correct status appears, it’s required that all degree information is included in PI’s e. RA Commons account Office of Sponsored Projects 14

Pre Award OSP Brown. Update Bag • NSF Update • PAPPG to be updated in Winter 2018 o o o • Implements ongoing pilot of Collaborators and Other Affiliations template. This format will be required under new PAPPG Additional explanation surrounding IRB and IACUC requirements Clarifies requirements in Prior NSF Support section regarding award dates Release of RAPID and EAGER opportunities relating to recent Hurricanes Office of Sponsored Projects 15

Pre Award OSP Brown. Update Bag • DOD/ONR Update • Revised DOD R&D General T&Cs (dated September 2017) are now available • ONR Addendum to the Do. D R&D General T&C is also available o Includes information about ONR RPPR submission using ARO system https: //www. onr. navy. mil/Contracts. Grants/submit-proposal/grantsterms-conditions Office of Sponsored Projects 16

OSP Brown Bag Tom Dillon Senior Grant/Contract Administrator Office of Sponsored Projects 17
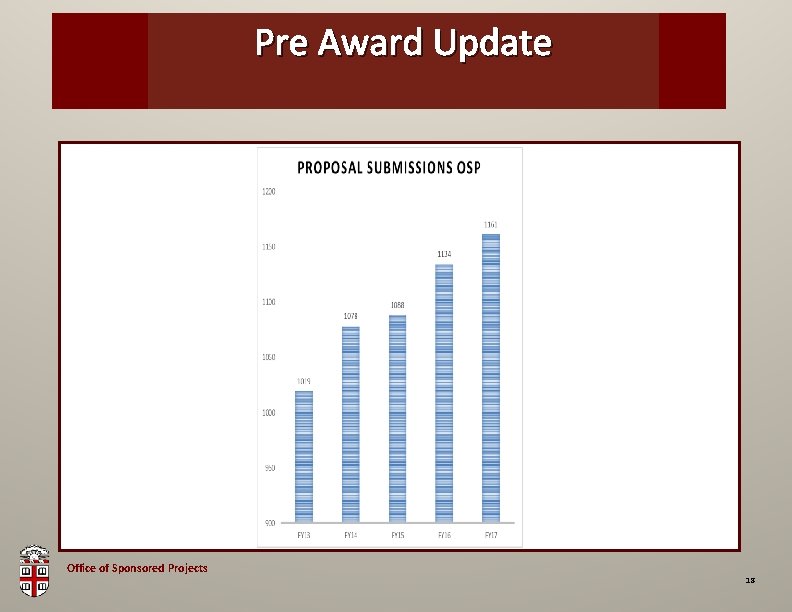
Pre Award OSP Brown. Update Bag Office of Sponsored Projects 18
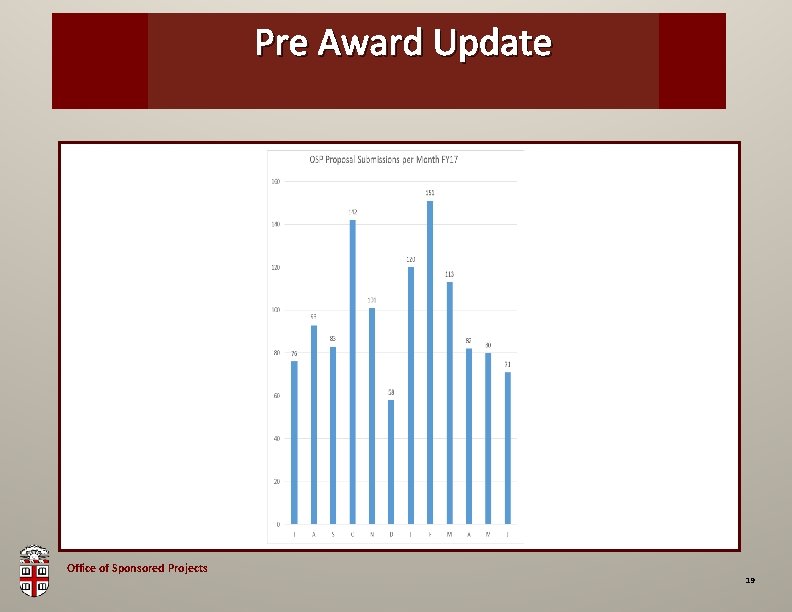
Pre Award OSP Brown. Update Bag Office of Sponsored Projects 19
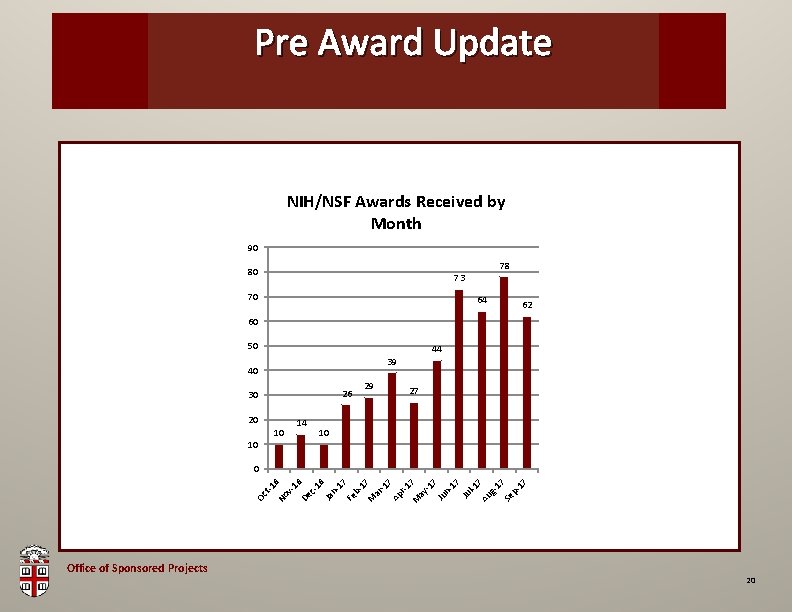
Pre Award OSP Brown. Update Bag NIH/NSF Awards Received by Month 90 78 80 73 70 64 62 60 50 44 39 40 26 30 20 10 10 14 29 27 10 Office of Sponsored Projects No Oc t-1 6 v 16 De c 16 Ja n 17 Fe b 17 M ar -1 7 Ap r-1 M 7 ay -1 7 Ju n 17 Ju l-1 7 Au g 17 Se p 17 0 20
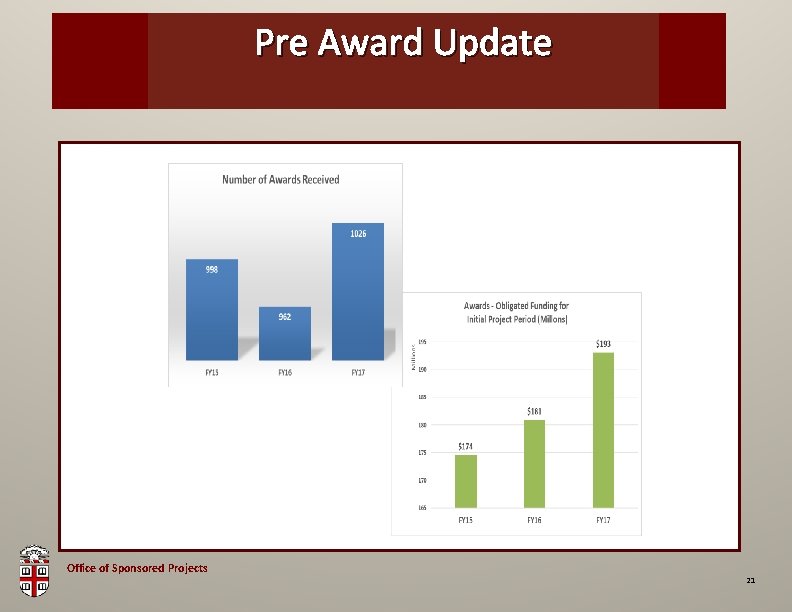
Pre Award OSP Brown. Update Bag Office of Sponsored Projects 21

OSP Post. Brown Award. Bag Update Heather Dominey Senior Grant/Contract Accountant, OSP Office of Sponsored Projects 22

OSPOSP Post-Award Brown Bag Update Clarification on NSF Policy - Faculty Salary Compensation – “The Two Month Rule” • • • During proposal submission – any compensation in excess of two months must be disclosed in the proposal budget, justified in the budget justification, and must be approved by NSF. If no change in objective or scope, grantees are allowed to rebudget between categories including salary. No prior approval from NSF is required. Source: https: //www. nsf. gov/pubs/2017/nsf 17119. pdf? WT. mc_id=US NSF_120 Office of Sponsored Projects 23

OSPOSP Post-Award Brown Bag Update Staffing Update Office of Sponsored Projects 24

OSPOSP Post-Award Brown Bag Update Post-Award Training Schedule - Fall 2017 October 13 th Financial Closeouts October 25 th Traveling on Sponsored Projects Register via: Office of Sponsored Projects 25

Effort. OSP Reporting Brown Bag Updates Recent Changes to Effort Certification Procedures New Monthly Reporting Schedule • Previously, reporting occurred on the 1 st business day of the month for the respective reporting cycles. • Reporting now runs after the monthly close, typically on the 6 th business day of the month Office of Sponsored Projects 26

Workday OSP Brown Effort Reporting Bag • Upcoming reporting dates Certification Period Report Run Date 07/23/1709/02/17 October 10, 2017 Non-exempt staff and undergraduates 09/03/1709/30/17 November 08, 2017 Non-exempt staff and undergraduates 10/01/1710/28/17 December 08, 2017 Non-exempt staff and undergraduates Office of Sponsored Projects Employee Type 27

Effort. OSP Certification Summary Brown Bag Report • This Workday report allows you to review the status of pending effort reports for actively employed workers within any given reporting cycle. • It can be used to determine location of reports in progress to facilitate follow-up with the awaiting person. • It should be regularly reviewed to monitor progress of effort report certifications until all grant-funded workers have certified effort and the report is clear. Office of Sponsored Projects 28

Timely OSP Termination Workers in Brownof. Bag Workday After-the-fact terminating worker notifications have been on the rise… A quick reminder: Grant-funded workers should be terminated in Workday, according to their effective date, as soon as possible prior to their departure from the University to allow an opportunity for effort certification. This is especially true in cases where the worker certifies his/her own effort, which represents the majority of effort reports. Late terminations protract the off-cycle effort certification process if the worker has already left the University and an appropriate substitute must then be designated to certify on the worker’s behalf. Office of Sponsored Projects 29

Effort Reporting OSP Brown Email Bag. Reminder Centralized Email Address: Effort-Reporting@Brown. edu Departments • Contact the Effort Certification Administrator with questions • Submit manual effort report certifications OSP • Respond to inquiries • Deliver effort certification reminder emails • Send out delinquency notifications • Contact departments regarding grantfunded terminating workers Office of Sponsored Projects 30

Training OSP Brown Opportunities Bag Effort Certification Training December 13, 2017 Registration: Learning Point Location: BOB 2 nd Flr. Conf. Rm. Office of Sponsored Projects 31

Office of Research OSP Brown Integrity Bag Update Keri Godin Director, Office of Research Integrity Office of Sponsored Projects 32

IACUC Updates OSP Brown Bag • Department of Defense funded protocols: amendments • Animals under the Holding Policy & charges to federal awards • Continued commitment to admin burden reduction - Update re: pain/distress categorization Office of Sponsored Projects 33

Office of. OSP Research Brown Integrity Bag Update Susan Carton. Lopez Associate Director, ORI Office of Sponsored Projects 34

Updates OSPIRB Brown Bag • HRPP staffing changes • NIH Expanded Definition of Clinical Trials • NIH Certificates of Confidentiality • Revised Common Rule (Human Subjects Research Regulations 45 CFR 46) Office of Sponsored Projects 35

OSP IRBBrown Updates Bag Welcome Katharine Menke IRB Specialist Office of Sponsored Projects 36

OSP IRBBrown Updates Bag Revised Common Rule • Final revisions to the Federal Policy for the Protection of Human Subjects issued on January 19, 2017. • Effective date: January 19, 2018 • Freeze on new submissions from December 20, 2017 – January 19, 2018 • HRPP will be providing many opportunities to learn about the changes in policy and procedures. Office of Sponsored Projects 37

OSP IRBBrown Updates Bag NIH Definition of Clinical Trial • NIH revised definition in 2014. • In August, 2017 they released “clarification” and case studies to help investigators determine if their research is considered a clinical trial. • Many investigators may be concerned and confused about this new information. • October 19, 1 -2 pm, HRPP, RAIS and OSP offering session: NIH Clinical Trials -Interpretation and Application of the Expanded Definition Office of Sponsored Projects 38

OSP IRBBrown Updates Bag Certificates of Confidentiality • Co. Cs help researchers protect privacy and confidentiality of human subjects enrolled in sensitive, health-related research • Allow researchers to refuse to disclose names or other identifying characteristics of research subjects in response to legal demands. • Issued by NIH and other HHS agencies • Policy change goes into effect October 1, 2017 Office of Sponsored Projects 39

OSP IRBBrown Updates Bag Certificates of Confidentiality, continued • All biomedical, behavioral, clinical, or other research that was commenced or ongoing on or after December 13, 2016, funded wholly or in part by the NIH, that collects or uses identifiable, sensitive information is deemed to be issued a Co. C and is therefore required to protect the privacy of individuals who are subjects of such research. • Will require change in consent documents. • Find out more on the Brown IRB website and NIH website: https: //humansubjects. nih. gov/coc/index Office of Sponsored Projects 40

RAIS Update OSP Brown Bag Jennifer Quinn Research Services Manager Office of Sponsored Projects 41

OSP Coeus Brown Update Bag NIH FORMS-E Implementation What’s Changing: • Consolidation of human subjects, inclusion enrollment, and clinical trial information previously collected across multiple forms into a new PHS Human Subjects and Clinical Trials Information Form. • Removal of Human Subjects Section and Human Subject related attachments on various forms: • PHS 398 Career Development Award Supplemental Form 4. 0 • PHS 398 Cover Page Supplement 4. 0 • PHS 398 Research Plan 4. 0 • PHS 398 Research Training Program Plan 4. 0 • PHS Fellowship Supplemental Form 4. 0 Office of Sponsored Projects 42

OSP Coeus Brown Update Bag NIH FORMS-E Implementation What’s Changing: • Updated R&R Other Project Information form to incorporate the addition of human subject exemption codes (Waiting on OMB Approval) • Updated OMB Expiration Dates across various form • Incorporation of recent Grants. gov changes to R&R Budget forms (V 1. 4) – These are already in Coeus! Office of Sponsored Projects 43

OSP Coeus Brown Update Bag NIH FORMS-E Implementation Timeline of FORMS-E Roll-Out: • Applicants must use FORMS-E application packages for due dates on or after January 25, 2018. (FORMS-E can not be used prior to January 25, 2018). • NIH will begin posting funding opportunities with the new FORMS-E packages beginning October 25 th. • FORMS-E packages will be added to all active nonclinical trial FOAs with due dates on/after Jan. 25 between Oct. 25 th and Nov. 25 th • FOAs that will allow clinical trials using FORMS-E on/after Jan. 25 will be reissued. Office of Sponsored Projects 44

OSP Coeus Brown Update Bag NIH FORMS-E Implementation in Coeus Timeline of FORMS-E Roll-Out in Coeus: • RAIS is in the process of testing the revised PHS Forms & R&R Other Project Information forms in Coeus Test environment. • The new PHS Human Subjects and Clinical Trials Information Form is currently underdevelopment. Office of Sponsored Projects 45

OSP Coeus Brown Update Bag NIH FORMS-E Implementation in Coeus Timeline of FORMS-E Roll-Out in Coeus: • Between October 25 th and January 24 th NIH Funding Opportunities may list 2 application packages for the same opportunity. • FORMS-D package Competition ID – FORMS-D Competition Title – change to “Use for due dates on or before January 24, 2018” Close date – January 24, 2018 • FORMS-E package Competition ID – FORMS-E Competition Title – “Use for due dates on or after January 25, 2018” Close date – original close date of FOA Office of Sponsored Projects 46

OSP Coeus Brown Update Bag NIH FORMS-E Implementation in Coeus Timeline of FORMS-E Roll-Out in Coeus: • Between October 25 th and January 24 th NIH Funding Opportunities may list 2 application packages for the same opportunity. • You Must only use FORMS-D packages. • You will NOT be able to connect to FORMS-E packages. Office of Sponsored Projects 47
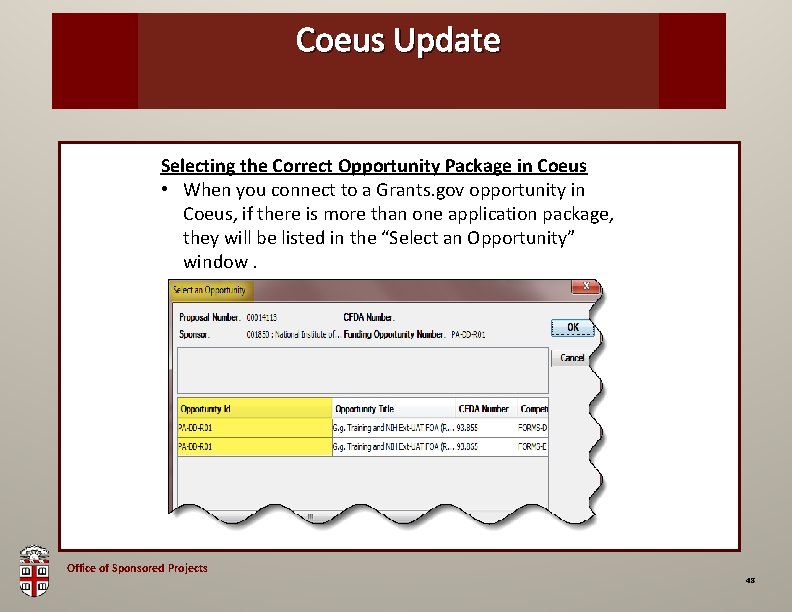
Coeus Update OSP Brown Bag Selecting the Correct Opportunity Package in Coeus • When you connect to a Grants. gov opportunity in Coeus, if there is more than one application package, they will be listed in the “Select an Opportunity” window. Office of Sponsored Projects 48

Coeus Update OSP Brown Bag Selecting the Correct Opportunity Package in Coeus • To ensure you are selecting the FORMS-D package, Look at the Competition ID field for which Form set is being used. Office of Sponsored Projects 49

Coeus Update OSP Brown Bag NIH FORMS-E Implementation Resources: • High-level Summary of Form Changes in FORMS-E Application Packages • Annotated Preview of FORMS-E Grant Application Form Changes • A Walk-through of the PHS Human Subjects and Clinical Trials Form (9 min. video) Office of Sponsored Projects 50

Grants. gov Workspace OSP Brown Bag. Update Grants. gov is Transitioning to using Grants. gov Workspace for Grant Submissions On December 31, 2017, Grants. gov will retire its legacy, downloadable PDF application packages and make a full transition to Grants. gov Workspace is a shared, online environment managed by Grants. gov where multiple users can simultaneously work on the application package on an online platform. While most of our Federal grant applications are submitted System-to-System (S 2 S) via Coeus; if you have a submission that cannot be submitted by Coeus, you may need to use Grants. gov Workspace. Office of Sponsored Projects 51

Grants. gov Workspace OSP Brown Bag. Update RAIS currently Reviewing the Grants. gov Workspace Process: • A Grants. gov Workspace is an online space where you can complete the application package via completing forms online or downloading the PDF and re-uploading into Workspace. • Multiple users can collaborate on one application package in Workspace. • Grants. gov Workspace will require people to have a Grants. gov account to be able to Create or be a “participant” in a Workspace. *In the coming months, RAIS will be providing more details about the Grants. gov Workspace process to the Research Community. Office of Sponsored Projects 52
Grants. gov Workspace OSP Brown Bag. Update Resources on Grants. gov Workspace : • Grants. gov Workspace Overview • Grants. gov Workspace Process • Grants. gov Workspace You Tube Channel Office of Sponsored Projects 53

NSF Redesigning Account OSP Brown Bag Management • NSF is working on streamlining and modernizing the registration functionality and processes around user Accounts. NSF is focused on Improving: • Login: consolidate to one centralized login to access either Research. gov or Fastlane • Account: Enforce that each individual user only has one NSF ID • Self Registration: User initiates request for NSF ID within System. • Role Request: Automate requests for user role approval • Profile Management: provide one unique profile maintained by the user. Tentative Timeline for Implementation: • Winter/Spring 2018 Office of Sponsored Projects 54

Coeus Training OSP Brown Bag Fall Training Schedule Track Coeus Class Coeus Viewing Proposals & Award Info Date 10/03/20 17 Coeus Premium Proposal Development 10/26/20 17 Coeus Lite Proposal Development 11/03/20 17 Coeus Premium Advanced Budgeting 12/05/20 17 Office of Sponsored Projects 55

Brown Bag Listserv OSP Brown Bag To be added to OSP’s Brown Bag Listserv, please email: resadmin@brown. edu Office of Sponsored Projects 56
- Slides: 56