Osmosis Objectives n Describe osmosis as passive transport

Osmosis Objectives n Describe osmosis as passive transport. n Define isotonic, hypertonic and hypotonic. n Describe what happens to an animal and plant cell in isotonic, hypertonic and hypotonic solutions. Textbook pages 185 -186
Think!!! A Google search for “water drinking deaths” returns over 18 million results! This is a common story in the news. How can someone die from drinking too much water?
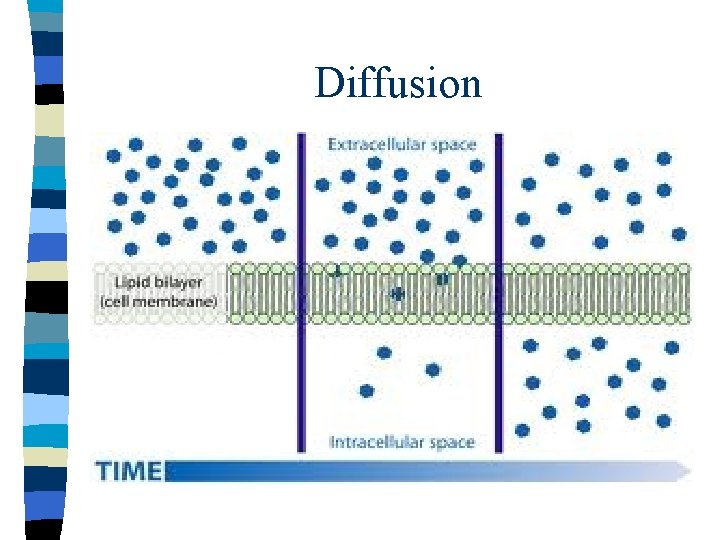
Diffusion

Osmosis n Osmosis – the diffusion of water through a selectively permeable membrane from an area of high concentration to an area of low concentration n Osmosis only happens when the membrane is NOT permeable to a solute, but is permeable to water. !!!!
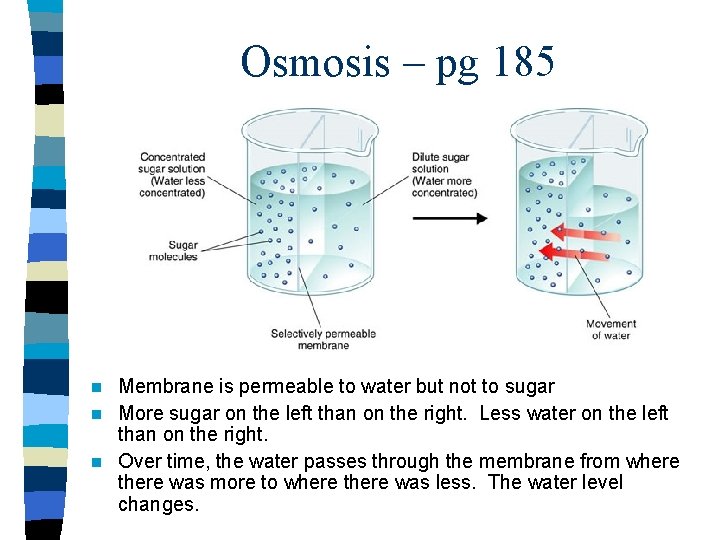
Osmosis – pg 185 Membrane is permeable to water but not to sugar n More sugar on the left than on the right. Less water on the left than on the right. n Over time, the water passes through the membrane from where there was more to where there was less. The water level changes. n
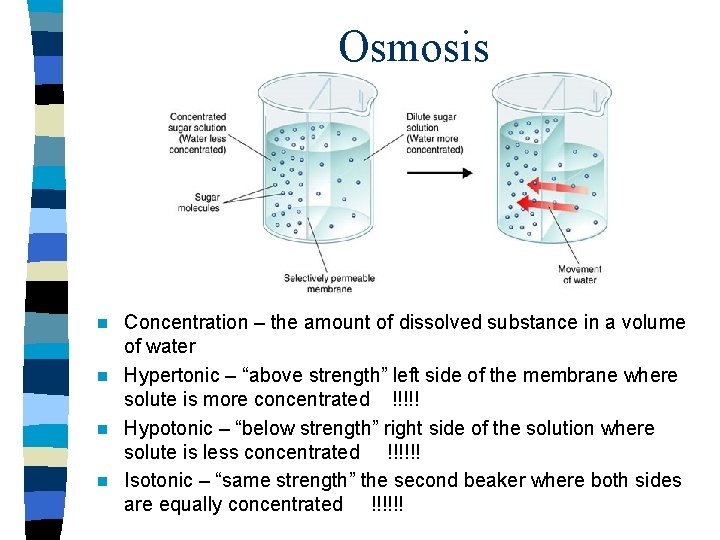
Osmosis Concentration – the amount of dissolved substance in a volume of water n Hypertonic – “above strength” left side of the membrane where solute is more concentrated !!!!! n Hypotonic – “below strength” right side of the solution where solute is less concentrated !!!!!! n Isotonic – “same strength” the second beaker where both sides are equally concentrated !!!!!! n

Osmosis n Osmosis is passive transport, like diffusion, because it occurs spontaneously without requiring energy. !!!!!

Osmosis n Animation

How Osmosis Works in Cells n http: //www. phschool. com/atschool/phbio /active_art/osmosis/index. html

How Osmosis Works on Animal Cells n Page 186 n animation n http: //www. linkpublishing. com/videotransport. htm#OSMOSIS

How Osmosis Works on Plant Cells n http: //www. linkpublishing. com/video- transport. htm#OSMOSIS

Drinking Water Deaths n When you drink too much water, are your cells being placed in a hypotonic, hypertonic or isotonic solution? n Will your cells, shrink, burst or stay the same?
- Slides: 12