Osmosis Notes A solution is made of two

- Slides: 2

Osmosis Notes A solution is made of two parts: the solute and the solvent. The solute is the stuff (usually a powder) that is dissolved in the liquid. The solvent is the liquid. For example, if you are making Kool-Aid: the Kool-Aid powder is the solute and the water is the solvent. Osmosis is the movement of water across a selectively permeable membrane - a membrane that allows some things to pass through and others to be blocked from entering. The cell membrane is selectively permeable. Water always moves toward the higher concentration of solute. Osmosis does not require energy and so it is a form of passive transport. WATER IS GREEDY! Look at how the solutions below are labeled – this is your guide to how water moves in and out of cells. 5% solute 95% water 10% solute 90% water Hypotonic The cell will swell. Isotonic The cell will stay the same size. 30% solute 70% water 10% solute 90% water Hypertonic The cell will shrink.

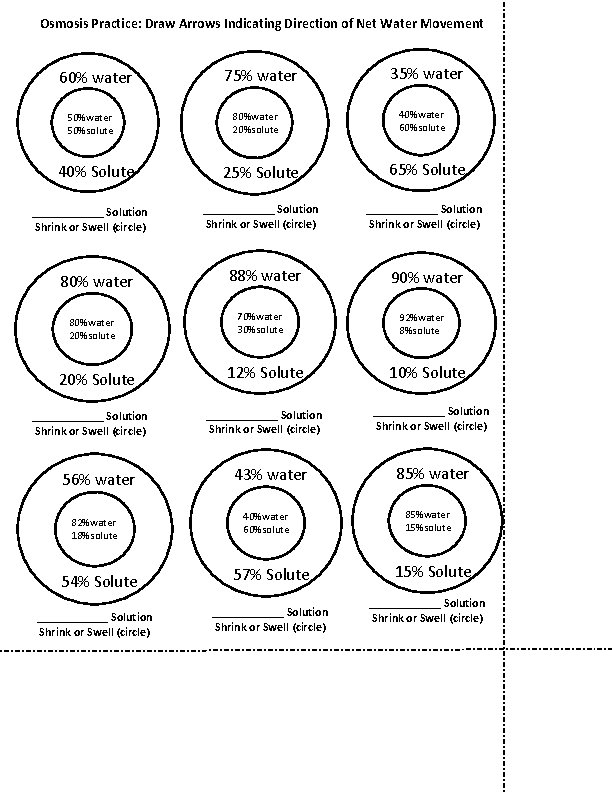
Osmosis Practice: Draw Arrows Indicating Direction of Net Water Movement 60% water 50%solute 40% Solute ______ Solution Shrink or Swell (circle) 80% water 80%water 20%solute 20% Solute ______ Solution Shrink or Swell (circle) 56% water 82%water 18%solute 54% Solute ______ Solution Shrink or Swell (circle) 75% water 80%water 20%solute 35% water 40%water 60%solute 25% Solute 65% Solute ____________ Solution Shrink or Swell (circle) 88% water 70%water 30%solute 12% Solute ______ Solution Shrink or Swell (circle) 43% water 40%water 60%solute 57% Solute ______ Solution Shrink or Swell (circle) 90% water 92%water 8%solute 10% Solute ______ Solution Shrink or Swell (circle) 85% water 85%water 15%solute 15% Solute ______ Solution Shrink or Swell (circle)