OSMOSIS LAB Osmosis the movement of water molecules

OSMOSIS LAB Osmosis: the movement of water molecules from a region of lower solute concentration to a region of higher solute concentration.

SOLUTION CONCENTRATIONS: Isotonic: two solutions have the same concentration of solutes. (iso- same, ton- condition, -ic pertaining to) Two solutions of different concentration: The one with more solute is hypertonic to the solution with less solute. (hyper- more than) The solution that has less solute is hypotonic to the one with more solute. (hypo- less than)
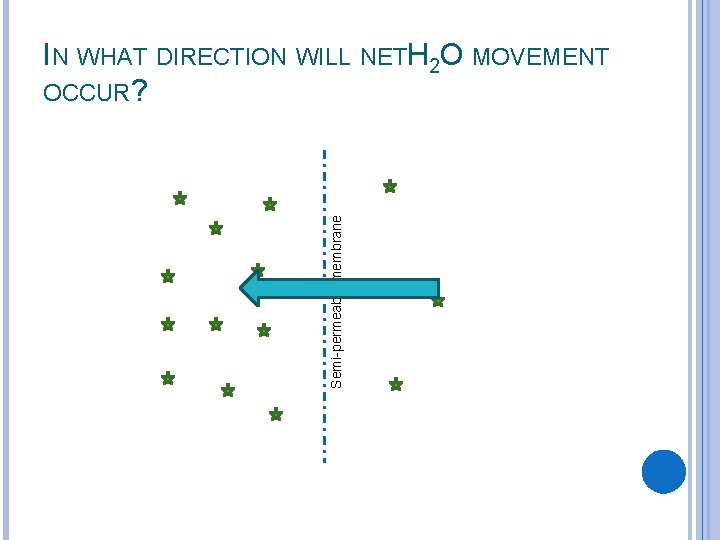
Semi-permeable membrane IN WHAT DIRECTION WILL NETH 2 O MOVEMENT OCCUR?

SET UP: Six dialysis tubing bags are filled with (15 -25 m. L) six solutions of differing sucrose concentrations and placed in beakers of distilled water. � 0 M � 0. 2 M Sucrose solution � 0. 4 M � 0. 6 M � 0. 8 M Distilled water � 1. 0 M Reminder: Molarity (M) = mol/L ex. 1 mole of sucrose per liter of water = 1. 0 M

RQ: HOW DOES THE MOLARITY OF A SOLUTION EFFECT THE RATE OF OSMOSIS THROUGH A SEMI PERMEABLE MEMBRANE? Independent Variable: Dependent Variable:
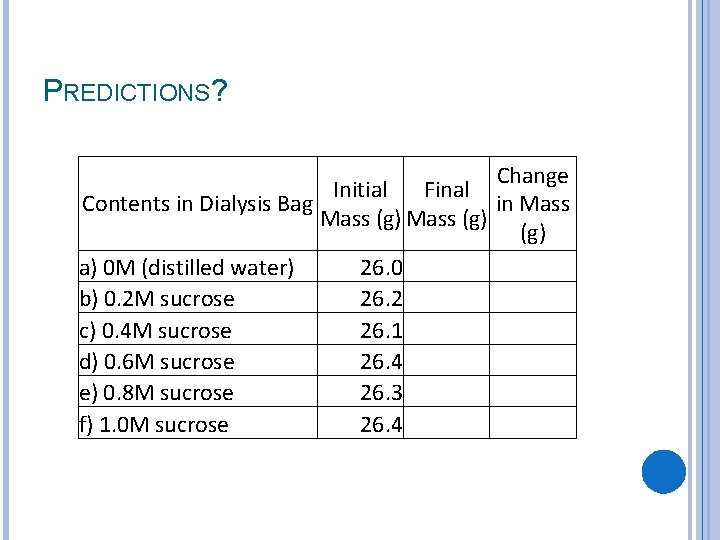
PREDICTIONS? Change Initial Final Contents in Dialysis Bag in Mass (g) a) 0 M (distilled water) 26. 0 b) 0. 2 M sucrose 26. 2 c) 0. 4 M sucrose 26. 1 d) 0. 6 M sucrose 26. 4 e) 0. 8 M sucrose 26. 3 f) 1. 0 M sucrose 26. 4
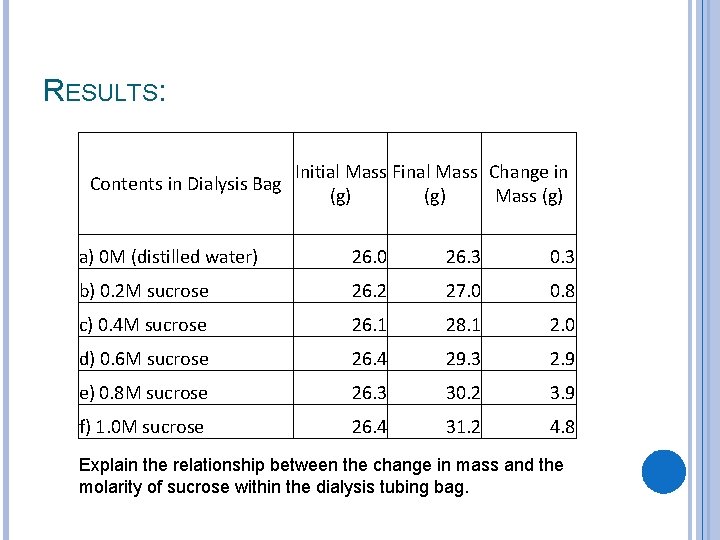
RESULTS: Contents in Dialysis Bag Initial Mass Final Mass Change in (g) Mass (g) a) 0 M (distilled water) 26. 0 26. 3 0. 3 b) 0. 2 M sucrose 26. 2 27. 0 0. 8 c) 0. 4 M sucrose 26. 1 28. 1 2. 0 d) 0. 6 M sucrose 26. 4 29. 3 2. 9 e) 0. 8 M sucrose 26. 3 30. 2 3. 9 f) 1. 0 M sucrose 26. 4 31. 2 4. 8 Explain the relationship between the change in mass and the molarity of sucrose within the dialysis tubing bag.
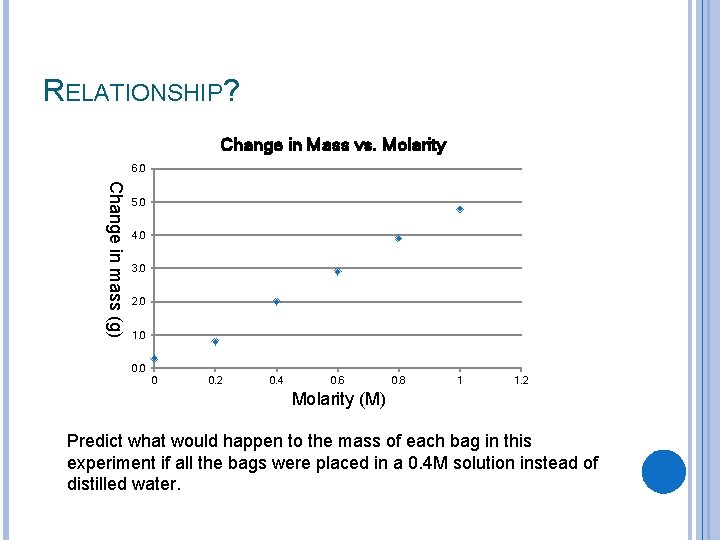
RELATIONSHIP? Change in Mass vs. Molarity 6. 0 Change in mass (g) 5. 0 4. 0 3. 0 2. 0 1. 0 0 0. 2 0. 4 0. 6 0. 8 1 1. 2 Molarity (M) Predict what would happen to the mass of each bag in this experiment if all the bags were placed in a 0. 4 M solution instead of distilled water.
- Slides: 8