Osmosis and Diffusion Passive Transport Osmosis and Diffusion












- Slides: 14

Osmosis and Diffusion Passive Transport

Osmosis and Diffusion • 1. Diffusion - the process by which molecules spread from areas of high concentration, to areas of low concentration

Osmosis and Diffusion • 1. Diffusion – eventually spreading out evenly into the available space

Osmosis and Diffusion • Several factors can affect the rate the diffusion…… • One such factor is temperature • Higher temperatures tend to increase the rate of diffusion

Osmosis and Diffusion • 2. Solutes- a substance dissolved in fluid Ex: Salt is a solute, when it is concentrated inside or outside the cell, it will draw the water in its direction This is also why you get thirsty after eating something salty

Osmosis and Diffusion • 3. Solvent - is a liquid, solid, or gas that dissolves another solid, liquid, or gaseous solute – resulting in a solution

Osmosis and Diffusion 4. Osmosis A specialized type of diffusion is the movement of a solvent (frequently water) through a semi-permeable membrane Osmosis is a type of a process where a fluid passes through a semipermeable membrane and moves from an area where a solute is present in low concentrations to an area where a solute is present in a higher concentration

Osmosis • http: //www. youtube. com/watch? v=sdi. Jt. DRJQEc Osmosis is a type of a process where a fluid passes through a semi-permeable membrane, from a higher concentration of water to a lower concentration of water

Osmosis and Diffusion • Hypotonic Solutions: • contain a low concentration of solute (more water) • When a cell is placed in a hypotonic solution, the water diffuses into the cell, causing the cell to swell

Osmosis and Diffusion • Hypertonic Solutions: • contain a high concentration of solute (less water) • When a cell is placed in a hypertonic solution, the water diffuses out of the cell, causing the cell to shrivel

Osmosis and Diffusion • Isotonic Solutions: contain the same concentration of solute ( equal amounts of water) • When a cell is placed in an isotonic solution, the water diffuses into and out of the cell at the same rate • The fluid that surrounds the body cells is isotonic

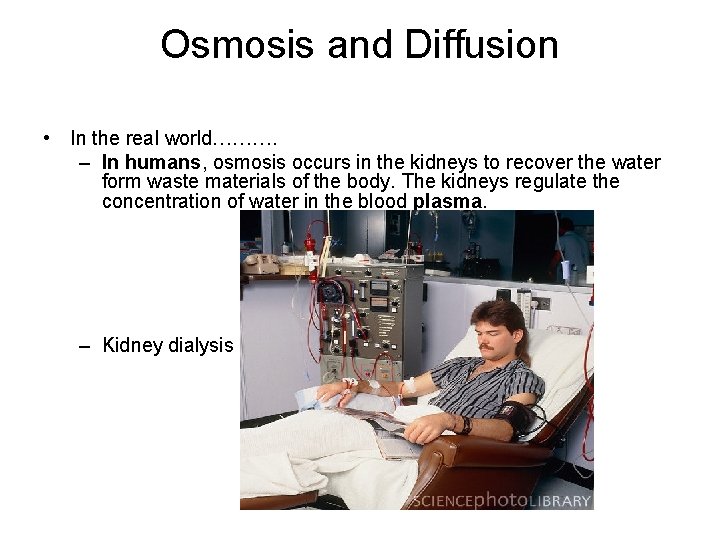
Osmosis and Diffusion • In the real world………. – In humans, osmosis occurs in the kidneys to recover the water form waste materials of the body. The kidneys regulate the concentration of water in the blood plasma. – Kidney dialysis

Osmosis and Diffusion • In the real world In plants, osmosis occurs for example at root hairs, allowing the uptake of water from the soil.

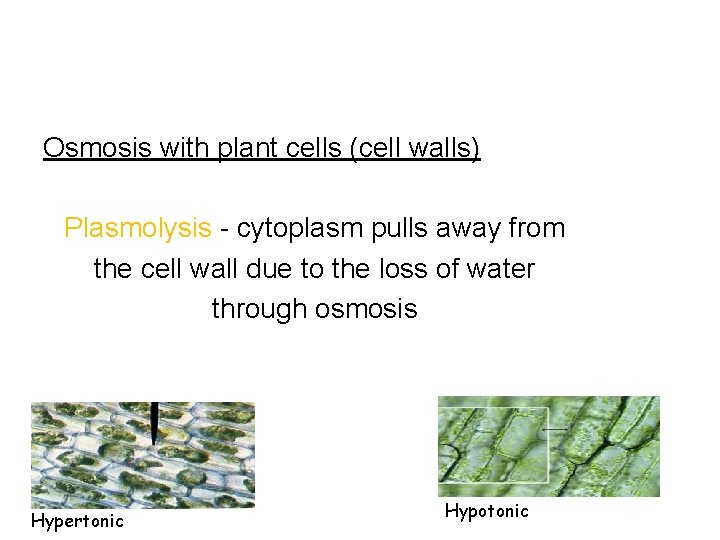
Osmosis with plant cells (cell walls) Plasmolysis - cytoplasm pulls away from the cell wall due to the loss of water through osmosis Hypertonic Hypotonic