Osmoregulation Ionic and osmotic balance 221 in multicellular

Osmoregulation

Ionic and osmotic balance 2/21 • in multicellular organisms the interstitial fluid is the internal environment – composition resembles that of the ancient sea: high Na+, low K+, low Ca++ and Mg++ • osmoregulatory organs maintain this environment isovolumic, isotonic, isoionic, isohydric, etc. • further task: removal of poisonous end products of metabolism (mostly NH 3 from proteins) • marine invertebrates: osmotic pressure and ionic constitution in equilibrium with seawater • marine vertebrates: ion concentration is one third of seawater except in hagfish (Eptatretus - Cyclostomata), Mg. SO 4, Cl- much lower; in sharks-rays ion concentration is lower, but osmotic pressure is maintained by urea • fresh water, terrestrial: ion concentration is one third; hyperosmotic to freshwater, hyposmotic to seawater

Osmotic exchange 3/21 • plasma membrane separates fluids with different ionic composition, but equal osmotic pressure • epithelium separates fluids that are different in both respects • animals cannot isolate themselves from the environment: exchange of gases, absorption of nutritients; exception: Artemia salina • obligate and regulated osmotic exchange • obligate exchange depends on physical factors, that animals cannot readily regulate • regulated exchange compensates for changes caused by the obligate • only some parts of the epithelium participate in osmoregulation: gill, kidney, salt gland, enteric system

Obligate osmotic exchange 4/21 • occurs through the skin, respiratory epithelium and other epithelia in contact with environment • influencing factors: – gradient: determines direction of exchange; a frog sitting in a pond takes up water through the skin; a marine fish is loosing water in the sea, but gains Na. Cl; a freshwater fish is taking up water, but loosing salt through the gill – surface: small animal – relatively larger surface, faster exchange, e. g. dehydration – permeability: • transcellular and paracellular exchange (but: tight junction); • skin of amphibians, gill of fish have high permeability • skin of reptiles, desert amphibians, birds, mammals are much less permeable (leather containers), but mammals loose water through sweating – eating, metabolism, excretes: metabolic water is very important for desert animals, but also for marine mammals (seals put on weight eating fish, but loose weight burning fat when is feeding on invertebrates) – respiration: function of nose – condense water during exhalation – dropping nose in winter

Osmotic regulation I. 5/21 • most vertebrates are strict osmoregulators exception: shark, ray and hagfish • marine invertebrates are in equilibrium, other invertebrates, similar to vertebrates, are hyperosmotic in freshwater, hyposmotic in seawater • some of the invertebrates is conformer, others are osmoregulators • freshwater animals breathing from water – they are hyperosmotic: 200 -300 m. Osm; freshwater in general below 50 m. Osm – water inflow, salt outflow – to compensate, they produce dilute urine, take up salt with food and through active transport (fish, frog), decrease the permeability of their skin – they do not drink freshwater

Osmotic regulation II. 6/21 • marine animals breathing from water – invertebrates are in equilibrium – hagfish: Ca++, Mg++, SO 42 - regulation only – shark-ray osmotic equilibrium due to urea accumulation, excess salt removed by rectal glands – fish are loosing water, drinking seawater; excess salt removed through the gill (chloride cells) • marine animals breathing air – loosing water through respiratory epithelium and other epithelia – marine reptiles and birds are drinking seawater – cannot produce strongly hyperosmotic urine (just as fish) – excess salt removed through salt glands – marine mammals do not drink seawater, water is taken in with food and produced metabolically, urine is hyperosmotic – lion seal males spend 3 months on the beach without eating or drinking; seal pups do similarly for 8 -10 weeks while mother is on the sea

Osmotic regulation III. • terrestrial animals breathing air – if freshwater is available then water lost through breathing can be replenished by drinking, salt loss (urine, faces, sweating) compensated from food – sparing is important – problem of shipwrecked sailors: kidney can remove 6 g Na+/l urine, seawater contains 12 g Na +/l – drinking of seawater leads to salt gain – desert animals have two problems: heat and lack of water – kangaroo rat: remains in cool burrow during daytime, active only during the night; gaining water metabolically, water condensation in nose – camel: cannot hide in burrow; when dehydrated do not sweat, body temperature raises changing between 35 -41° C; hyperosmotic or no urine, urea stored in tissues; faces dry 7/21
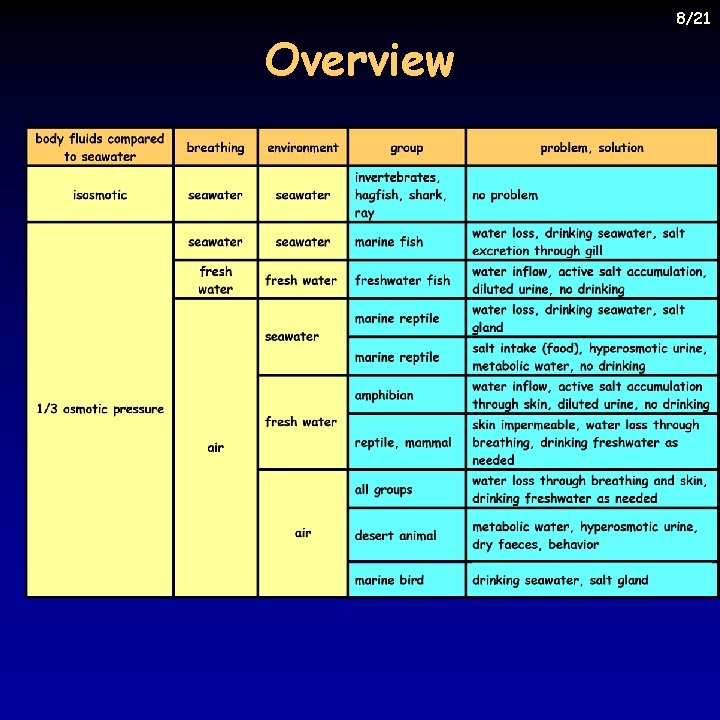
Overview 8/21

Water compartments 9/21 • human body contains 60% water on average, differences between male-female, young-old • distributed in different compartments • intracellularly 2/3, extracellularly 1/3 • of the extracellular water: 3/4 interstitially, 1/4 in blood plasma • barriers and, transport rules are already known • measurement of volumes applying dilution principle: Evans-blue, inulin, tritiated water • homeostasis is very important. cholera, diarrhea - dehydration, working by a furnace in tropical areas – water poisoning, severe burns – dehydration due to loss of skin • the most important regulator in humans is the kidney, behavioral regulation is also important – metabolic water is limited

Human kidney 10/21 • osmoregulatory organs always contain transport epithelium (skin, gill, kidney, gut) : polarized apical (luminal, mucosal) and basal (serosal) surfaces are different • capacity of the transport epithelium is increased by its special structure: tubular organization • functioning of the mammalian kidney is well known – though it does not represent all types of vertebrate kidneys • 0, 5% of body weight, 20 -25% of cardiac output • cortex, medulla, renal pyramid, renal pelvis, ureter, urinary bladder, urethra • volume of urine is 1 l daily, slightly acidic (p. H 6), composition, volume changes with the food and the requirements of the water homeostasis beer, Amidazophen, etc.

The nephron 11/21 • functional unit of human kidney is the nephron • afferent and efferent arterioles, in between glomerulus; Bowman capsule, proximal tubule, loop of Henle, distal tubule, collecting duct • most of the nephrons (85%) are cortical, the rest juxtamedullary (15%) nephron • steps in the formation of urine: – ultrafiltration – reabsorption – secretion • the kidney is very important in p. H regulation • the kidney removes ammonia formed during the decomposition of proteins

Ultrafiltration 12/21 • in the kidney 15 -25% of water and solutes is filtrated, 180 l daily – proteins and blood cells remain • filtration depends on: – the hydrostatic pressure between the capillaries and the lumen of the Bowman capsule: 55 -15 = 40 mm. Hg – the colloid osmotic pressure of the blood: 30 mm. Hg – effective filtration pressure 40 -30 = 10 mm. Hg – the permeability of the filter: fenestrated capillaries, basal membrane (collagen + negative glycoproteins), podocytes (filtration slits between pedicels) • voluminous blood supply due to the relatively low resistance – afferent arteriole is thick and short – high pressure in the glomerulus • regulation of the blood flow: basal miogenic tone, paracrine effect of juxtaglomerular apparatus, sympathetic effect (afferent arteriole, glomerulus, podocyte)

Clearance 13/21 • clearance of a substance is the volume of plasma that is completely cleaned from the given substance in the kidney in every minute CP = VU that is VU C = -----P C - clearance, P – concentration in plasma, V – volume of urine in 1 minute, U – concentration in urine • clearance of a substance that is neither reabsorbed nor secreted (e. g. inulin) equals the glomerulus filtration rate : GFR • clearance of a substance that is not only filtrated, but completely secreted as well (e. g. PAH) equals the renal plasma flow : RPF • knowing the hematocrit, renal blood flow (RBF) can be calculated

Tubular reabsorption I. 14/21 • 180 l primary filtrate is produced every day, but only 1 l is excreted, of 1800 g filtrated Na. Cl only 10 g remains in the urine • the process of reabsorption has been successfully examined since the 1920’s using the method of micropuncture • role played by the subsequent sections of the tubules: – proximal tubule • 70% of Na+ is reabsorbed by active transport, Cl- and water follow passively, obligate reabsorption • filtrate is isosmotic, but concentration of substances that are not reabsorbed increases 4 -fold • on the apical membrane of epithelial cells microvilli • virtually all filtrated glucose and amino acids are reabsorbed using Na+ dependent symporter • tubular maximum for glucose: below 1. 8 mg/ml complete reabsorption (normal value: 1. 0 mg/ml), above 3. 0 mg/ml linear increase – sugar in urine in diabetes • Ca++, phosphate and other electrolytes are reabsorbed as needed – see later

Tubular reabsorption II. 15/21 – descending part of Henle’s loop • no microvilli, few mitochondria – no active transport • low permeability for Na. Cl and urea, high for water – thin ascending part of Henle’s loop • no microvilli, few mitochondria – no active transport • low permeability for water, high for Na. Cl – thick ascending part of Henle’s loop • active reabsorption of Na+ • low water permeability – distal tubule • active reabsorption of Na+, and passive reabsorption of water • K+, H+ and NH 3 transport as needed – see later (p. H regulation) • transport is regulated by hormones – facultative reabsorption – collecting duct • active reabsorption of Na+ at the cortical part, high urea permeability in the internal medullary part • regulated water permeability (ADH)

Tubular secretion 16/21 • several substances are secreted from the plasma to the tubule in the nephron – best examined: different electrolytes (K+, H+, NH 3) organic acids and bases • active transport – recognizes substances conjugated with glucuronic acid in the liver • K+ is reabsorbed in the proximal tubule and Henle’s loop (Na/2 Cl/K transporter) • if K+ concentration is too high, secretion in the distal tubule depending on aldosterone and coupled to Na+ reabsorption - K+ acts directly on aldosterone, Na+ through renin-angiotensin • conflicting demands: insulin secretion is induced by high K+ - excess K+ is taken up by adipose tissue • secretion of H+ and NH 3 serves p. H regulation

p. H regulation I. • • 17/21 normal p. H 7. 4 – 7. 35 acidosis, 7. 45 alkalosis normal functioning is possible between 7. 0 -7. 8 regulation: buffer systems, respiration, kidney Henderson-Hasselbalch equation: [A-] p. H = p. K + log -----[HA] • for CO 2 two equations; following rearrangement: • • • [HCO 3 -] p. H = p. K’ + log -----[αCO 2] - solubility of CO 2 p. K’ = 6. 08, i. e. not good buffer at normal p. H – as CO 2 and HCO 3 - can be easily modified (respiration, kidney) plasma proteins (14 -15% Hgb, 6 -8% other) p. K is same as blood p. H – good buffers phosphate concentration low, negligible effect

p. H regulation II. 18/21 • respiratory alkalosis and acidosis: caused by hyper-, or hypoventilation • metabolic alkalosis – e. g. Cl- loss because of vomiting • metabolic acidosis – anaerobic energy production, ketosis in diabetes mellitus • in the first case: kidney compensates, in the second: breathing (short-term) and the kidney (long-term) • proximal tubule, Henle’s loop: Na+/H+ exchanger, distal tubule, collecting duct: HCO 3 - uptake through A-cells • in distal tubule and collecting duct: HCO 3 secretion through B-cells • in acidosis HCO 3 - level is low in the filtrate: NH 3 secretion – binds to H+, NH 4+ cannot go back, H+ secretion increases

Hyperosmotic urine 19/21 • birds and mammals can produce hyperosmotic urine - water reabsorption in the collecting duct due to osmotic pressure differences • common characteristic: Henle’s loop, the longer the loop, the more concentrated the urine – very long in kangaroo rat • pressure difference is achieved through the counter-current principle • Na+ transport in the ascending part of the Henle’s loop – do not enter the descending part, but attracts water leading to the same result • in addition, urea present in high concentration because of the reabsorption of water, can only leave the tubule in the internal medulla • osmotic pressure increases from the cortex to the medulla • blood supply to the tubules (vasa recta) is running in parallel to the Henle’s loop , does not decrease the osmotic gradient

Regulation of the kidney 20/21 • granular cells in the juxtaglomerular apparatus produce renin in response to a decrease in blood pressure or Na. Cl delivery to the distal tubule • renin cuts off angiotensin I (10 amino acids) from angiotensinogen (glycoprotein) • converting enzyme (mostly in the lung) cuts off 2 amino acids from angiotensin I – angiotensin II • angiotensin II enhances aldosterone secretion in the adrenal gland, increases blood pressure through vasoconstriction and increases ADH production • aldosterone increase Na+ reabsorption through 3 different ways: facilitation of the pump, ATP production, increased apical Na+ permeability • ADH producing cells detect blood pressure and osmolality and are sensitive to alcohol • atrial natriuretic peptide (ANP) – released in the atria when venous pressure increases - inhibits renin, aldosterone, ADH production

Nitrogen removal 21/21 • part of the digested amino acids are reused, amino groups from the others have to be removed as NH 3 and NH 4+ are poisonous • three forms: ammonia, urea, uric acid • ammonia – poisonous – huge volume is needed to provide low concentration in the cell and high outward gradient – 0. 5 l water/1 g nitrogen – fish, aquatic invertebrates, mammals in low amount – transport in the form of glutamine from the liver to the kidney • urea – less poisonous, 0. 05 l water/1 g nitrogen – synthesis requires ATP – vertebrates, except fish, synthesize urea in the ornithine-urea cycle, fish and invertebrates from uric acid – hominoids cannot metabolize uric acid (from nucleic acids) – can accumulate and lead to gout • uric acid – low solubility: 0. 001 l water/1 g nitrogen – white precipitate - guano in birds (uric acid, guanine) – fish, reptiles, terrestrial arthropods
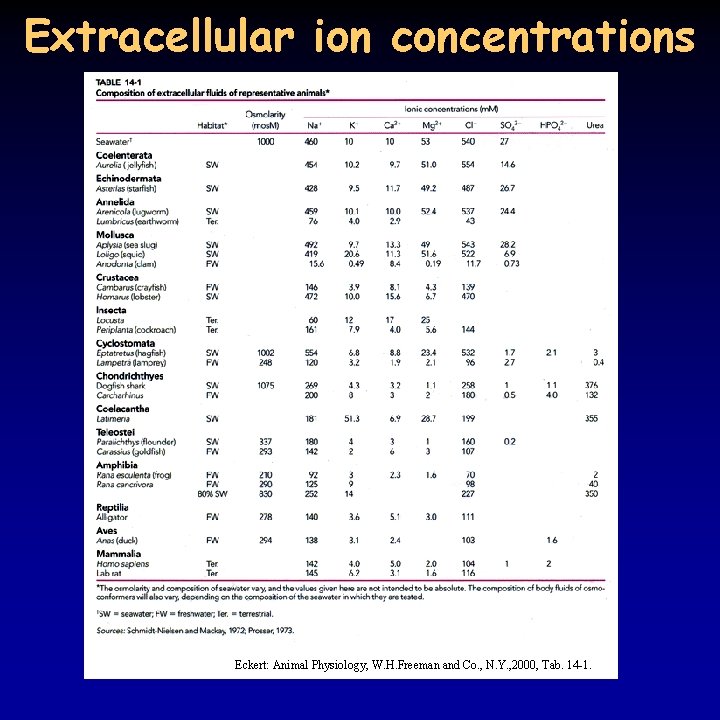
Extracellular ion concentrations Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Tab. 14 -1.

Osmoregulation in animals Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -8.
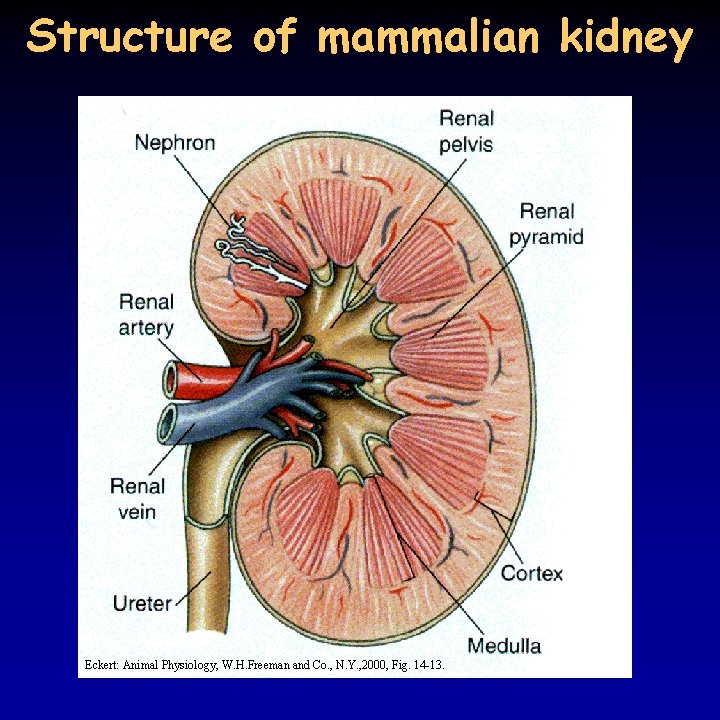
Structure of mammalian kidney Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -13.
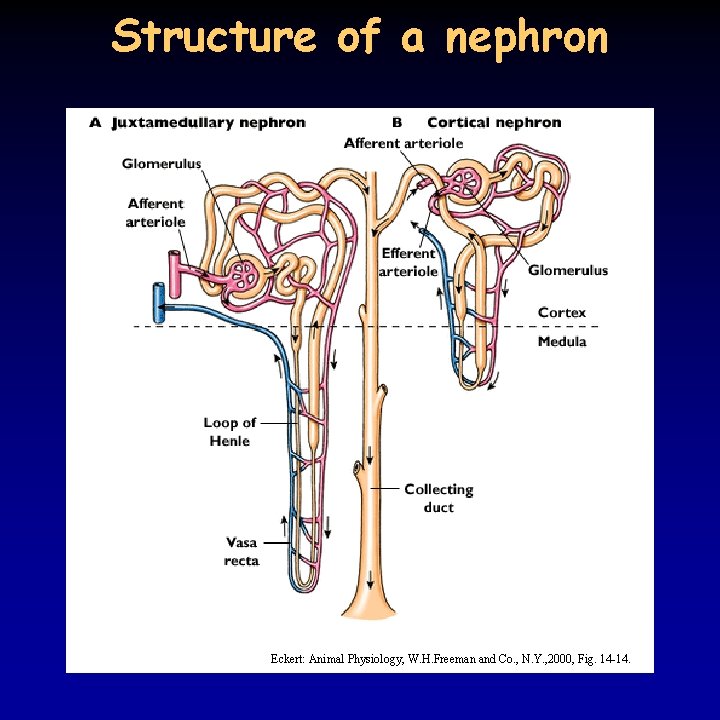
Structure of a nephron Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -14.
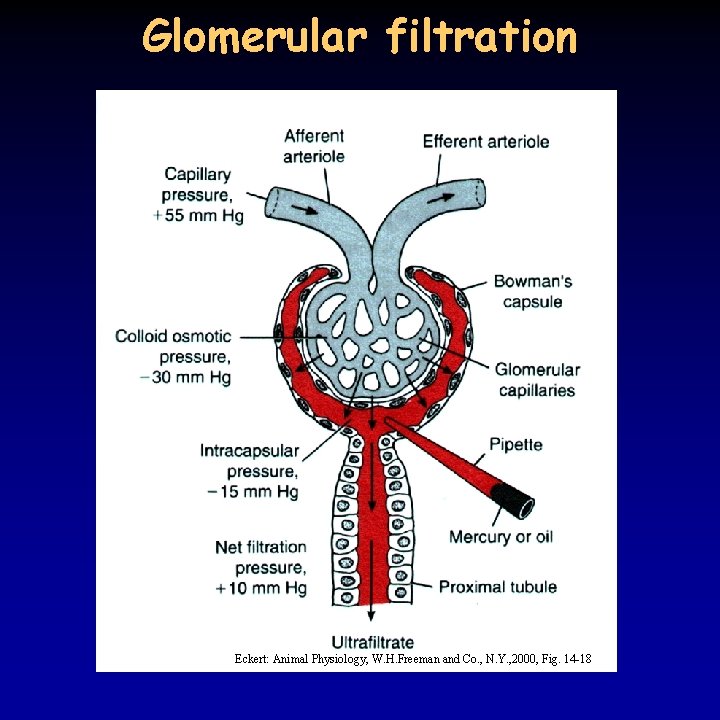
Glomerular filtration Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -18
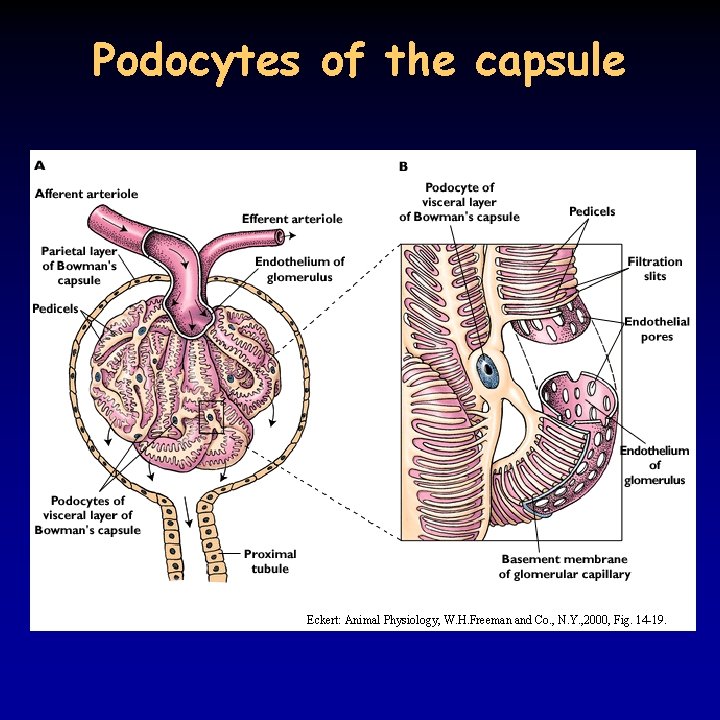
Podocytes of the capsule Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -19.

Podocytes of the capsule - EM Berne and Levy, Mosby Year Book Inc, 1993, Fig. 41 -7
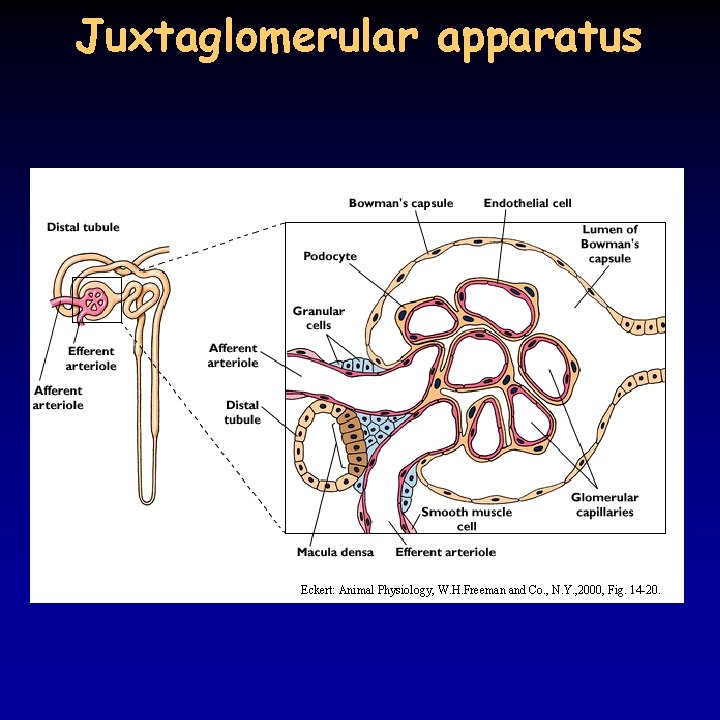
Juxtaglomerular apparatus Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -20.

Na+ reabsorption Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -25.
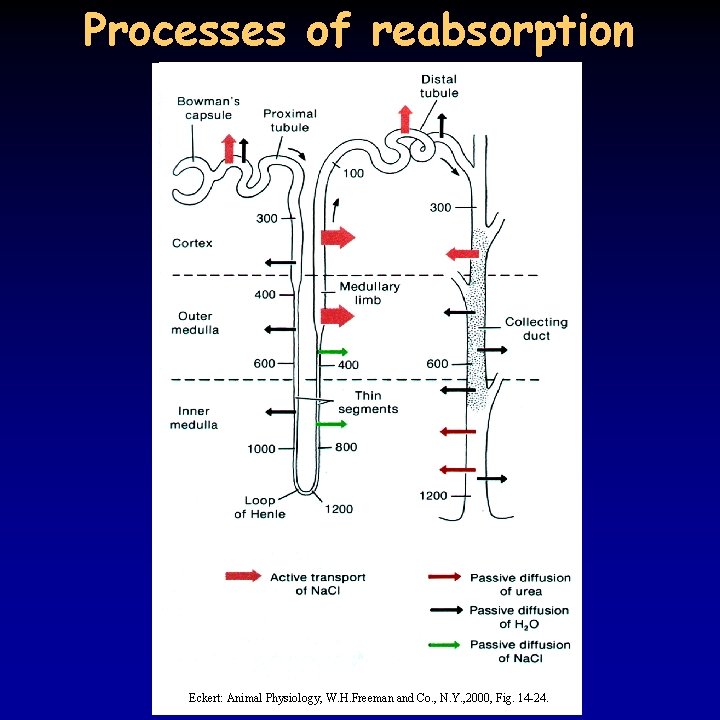
Processes of reabsorption Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -24.
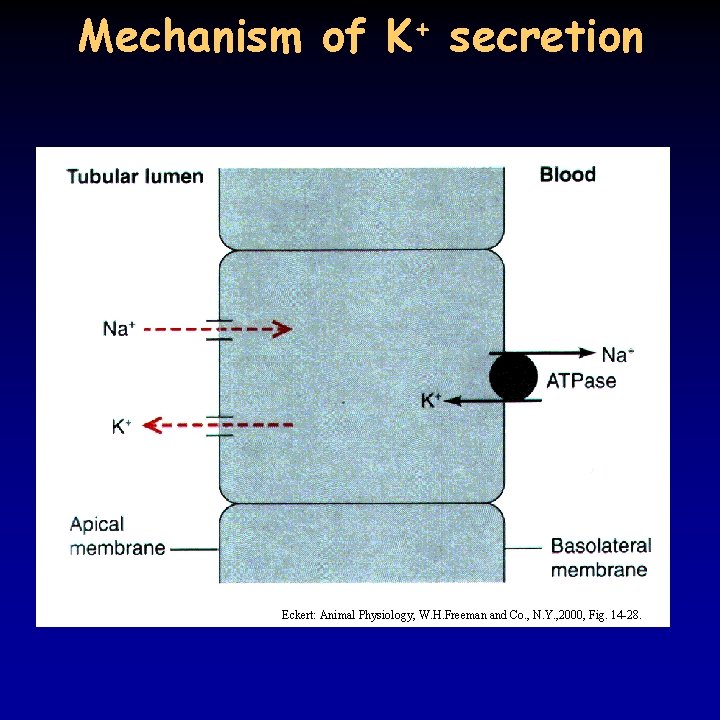
Mechanism of K+ secretion Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -28.

p. H regulation Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -29.
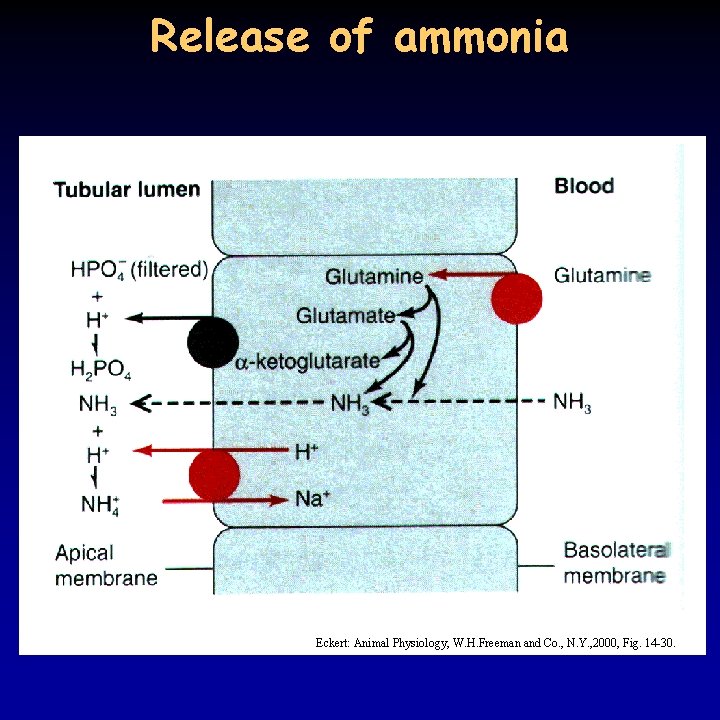
Release of ammonia Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -30.
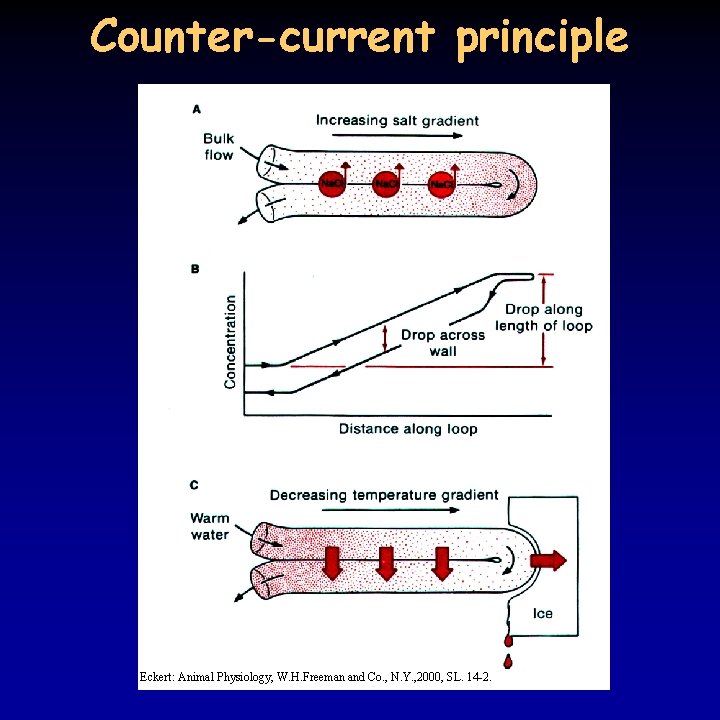
Counter-current principle Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, SL. 14 -2.
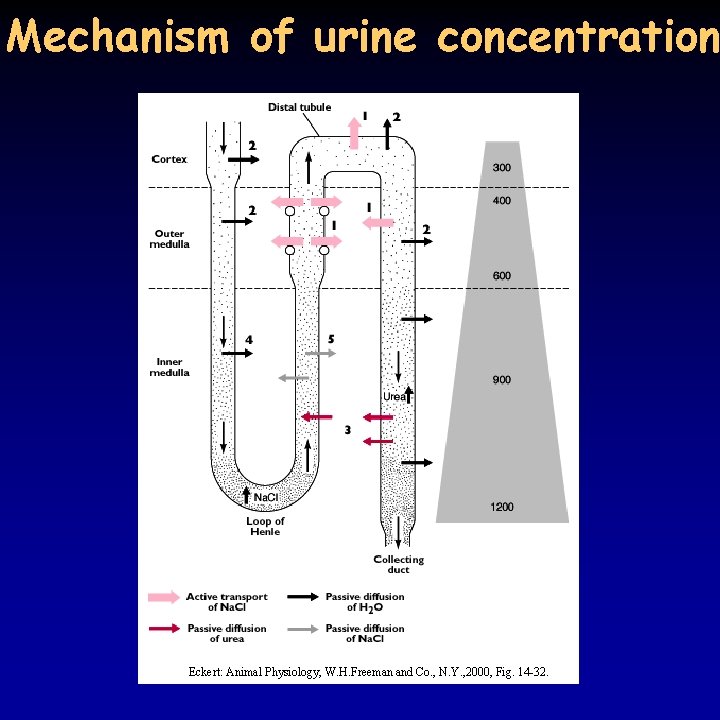
Mechanism of urine concentration Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -32.
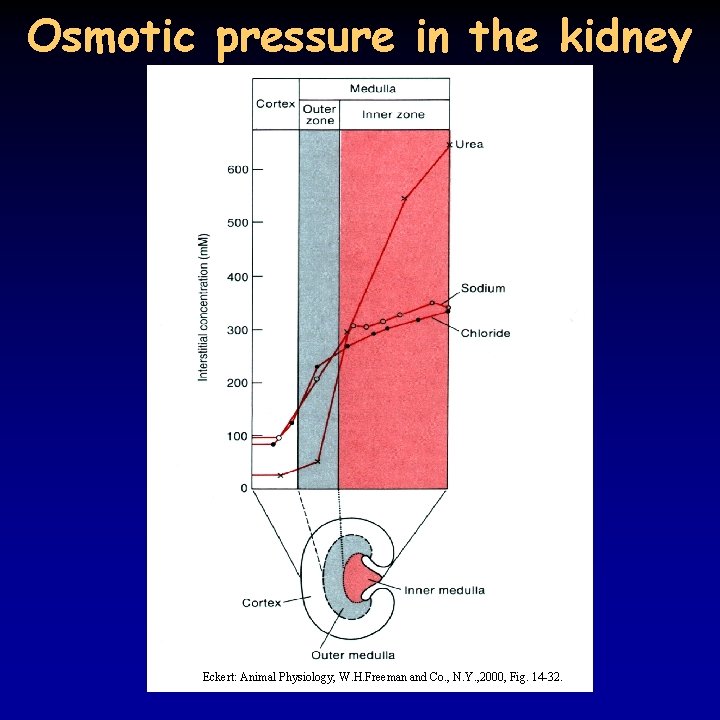
Osmotic pressure in the kidney Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -32.
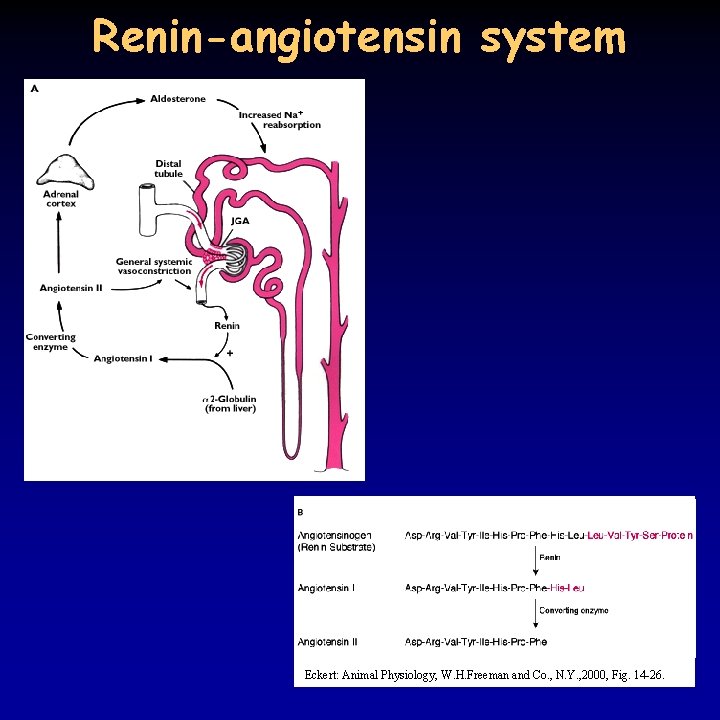
Renin-angiotensin system Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -26.

Actions of aldosterone Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -27.

Regulation of ADH secretion Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -35.
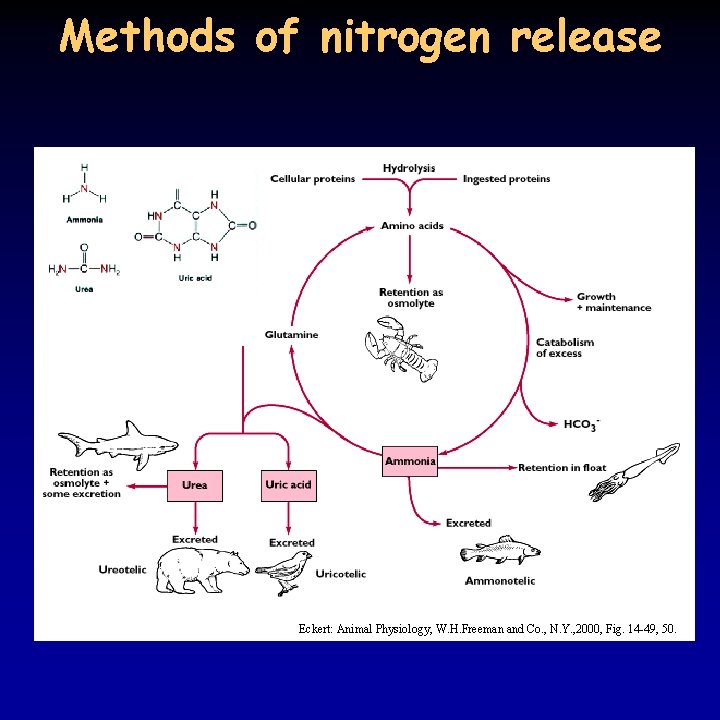
Methods of nitrogen release Eckert: Animal Physiology, W. H. Freeman and Co. , N. Y. , 2000, Fig. 14 -49, 50.
- Slides: 41