Orthosilicates n n Isolated tetrahedron Common examples Olivine
























- Slides: 41

Orthosilicates n n Isolated tetrahedron Common examples Olivine, garnet, and zircon n Al 2 Si. O 5 polymorphs, staurolite, topaz, titanite n Oxygen coordinate with other anions n

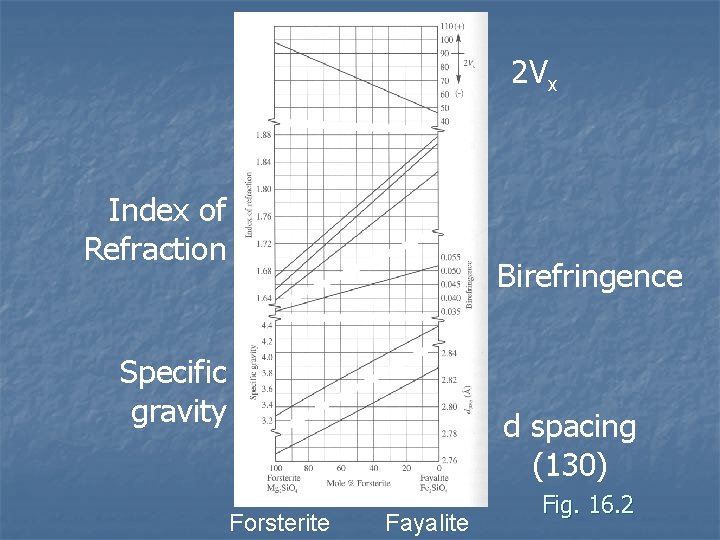
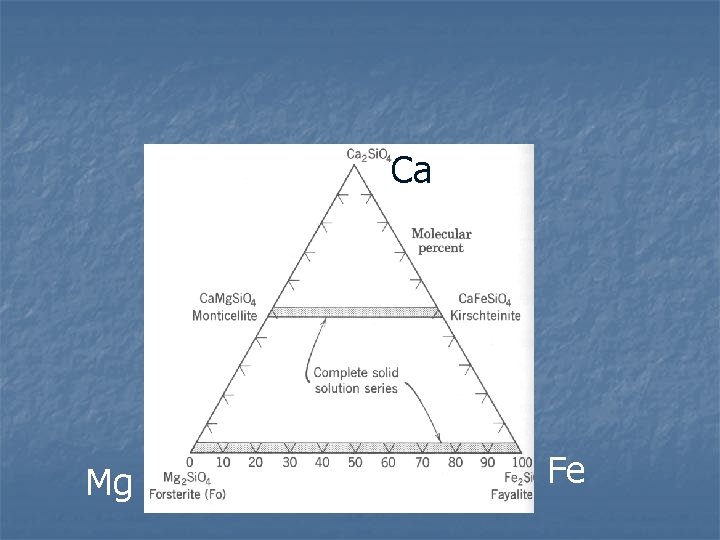
Olivine Composition n Complete solid solution between forsterite (Mg) and fayalite (Fe) Mn end members as well – rare n Ca can be around 50% of cations, still has Fe. Mg solid solution n n Fe and Mg contents cause variations in physical properties Can be used to identify composition Zoning can be common

2 Vx Index of Refraction Birefringence Specific gravity d spacing (130) Forsterite Fayalite Fig. 16. 2

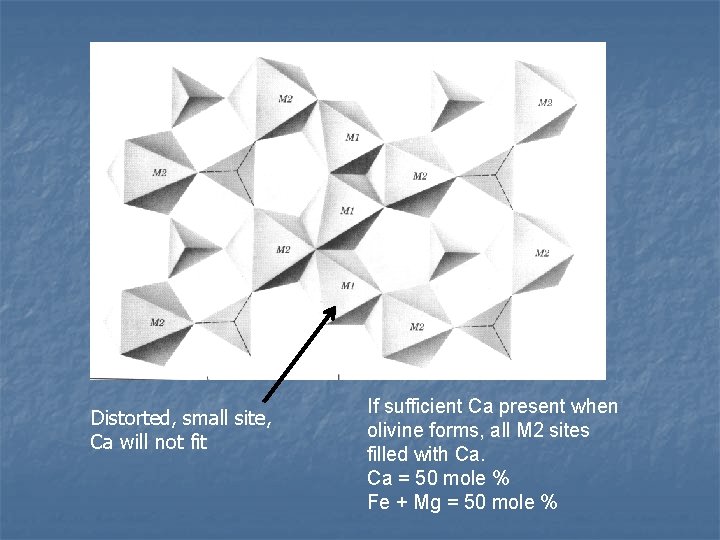
Structure and composition n Two distinct sites for cations: M 1 = distorted, so smaller than M 2 = regular octahedron n n Controls distribution of cations M 2 only site for Ca, 1. 12 Å, also may hold Fe and Mg n M 1 and M 2 n n both Fe = 0. 78 Å and Mg = 0. 72 Å

Distorted, small site, Ca will not fit If sufficient Ca present when olivine forms, all M 2 sites filled with Ca. Ca = 50 mole % Fe + Mg = 50 mole %

Ca Mg Fe

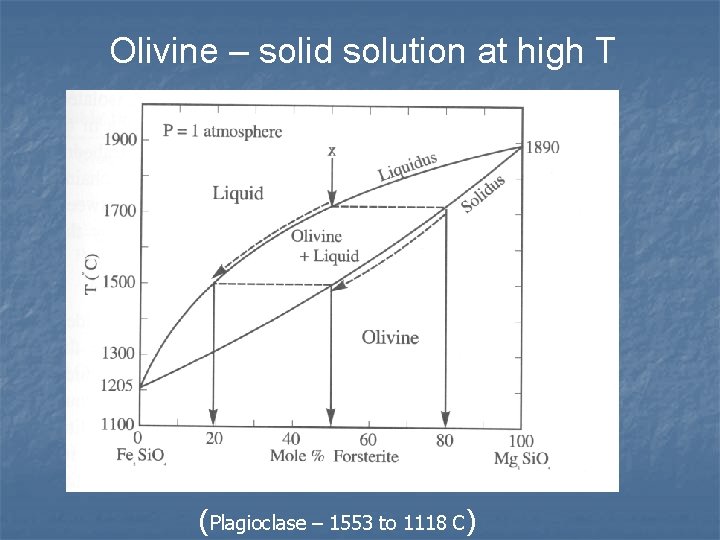
Olivine – solid solution at high T (Plagioclase – 1553 to 1118 C)

Inosilicates (chain) n n Common Fe/Mg – bearing silicates Two common groups Pyroxenes: single chains n Amphiboles: double chains n n n Pyroxenes are common in MORB Amphiboles more common on continents because of weathering

Pyroxene group n n n General formula: XYZ 2 O 6 Z/O ratio = 1/3 Z cations usually Si, occasionally Al Single chain extend along c axis Chains are stacked along a axis, alternating: Base faces base n Apex faces apex n

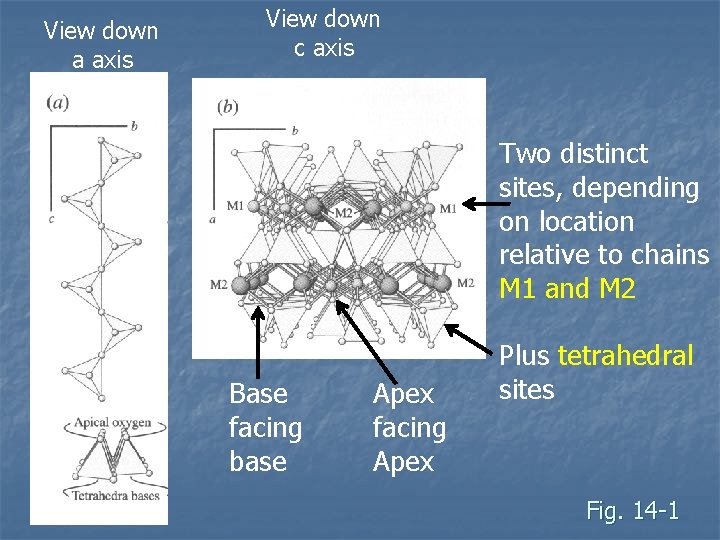
View down a axis View down c axis Two distinct sites, depending on location relative to chains M 1 and M 2 Base facing base Apex facing Apex Plus tetrahedral sites Fig. 14 -1

XYZ 2 O 6 n n Z/O ratio 1/3 X cations in M 2 sites Between bases of tetrahedrons n Distorted 6 - and 8 - fold coordination n Depends on stacking and the size of the cations n n Y cations in M 1 sites n 6 -fold coordination between apical oxygen

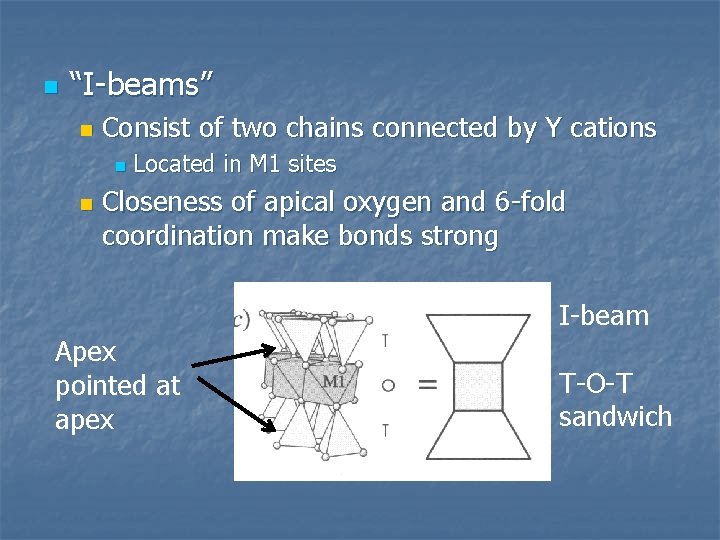
n “I-beams” n Consist of two chains connected by Y cations n n Located in M 1 sites Closeness of apical oxygen and 6 -fold coordination make bonds strong I-beam Apex pointed at apex T-O-T sandwich

n I-beams held together by X cations in M 2 site Coordination number depends on how chains line up n 6 -fold coordination gives orthorhombic symmetry – Orthopyroxenes or OPX n 8 -fold coordination gives monoclinic symmetry – Clinopyroxenes or CPX n

n Crystal shapes Blocky prisms, nearly square n Elongate along c axis n n Cleavage controlled by I-beams Cleavage typically between 87º and 93º n Only when viewed down the c axis n Mineral grain must be cut parallel to (001) n

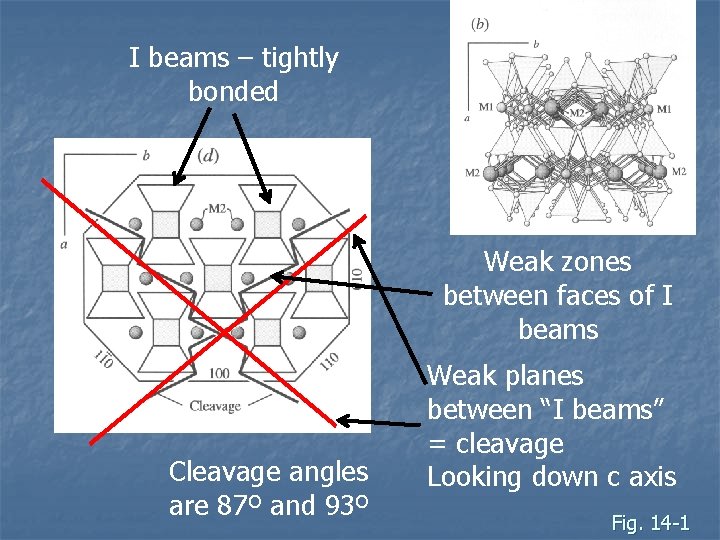
I beams – tightly bonded Weak zones between faces of I beams Cleavage angles are 87º and 93º Weak planes between “I beams” = cleavage Looking down c axis Fig. 14 -1

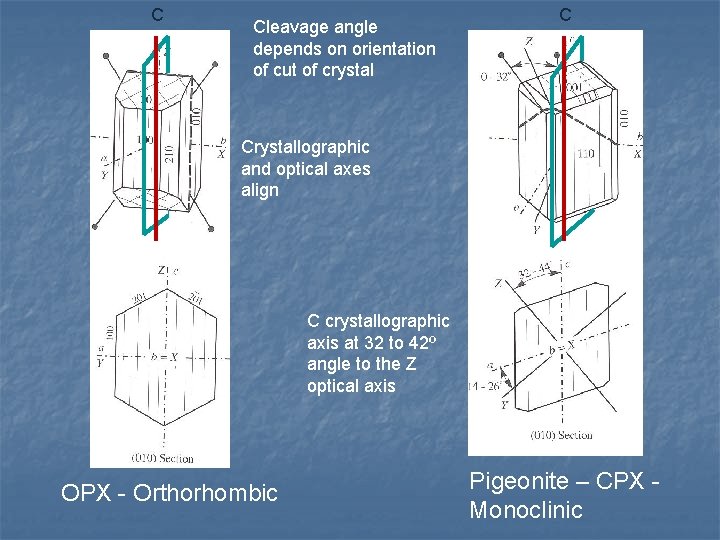
C Cleavage angle depends on orientation of cut of crystal C Crystallographic and optical axes align C crystallographic axis at 32 to 42º angle to the Z optical axis OPX - Orthorhombic Pigeonite – CPX Monoclinic

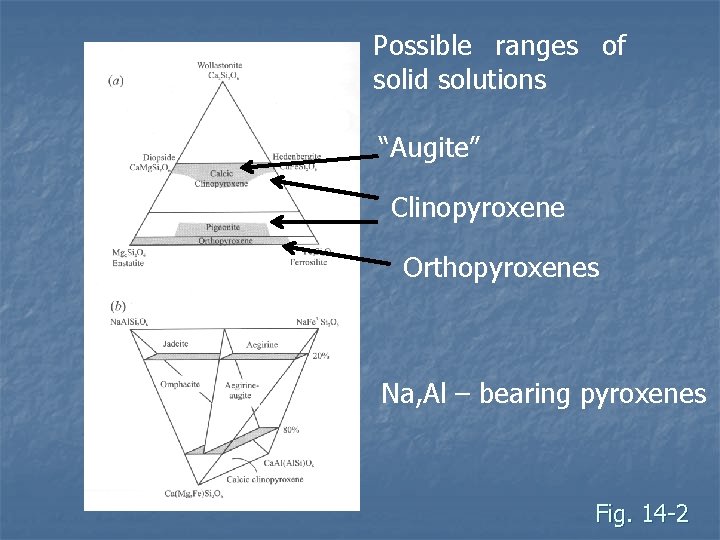
Classification n Based on two linked things Composition: which cations occurs in M 2 sites (facing bases of tetrahedron) n Symmetry: determined by composition n n Most plot on ternary diagram with apices: Wollastonite, Wo (Ca 2+) n Enstatite, En (Mg 2+) n Ferrosilite, Fe (Fe 2+) n

n Three major groups Orthopyroxenes (opx) – orthorhombic n Ca-poor clinopyroxenes (cpx) – monoclinic n Ca-rich clinopyroxenes (cpx) – monoclinic n n The amount of Ca in the mineral controls the crystal system, symmetry, and extinction angle

n Orthopyroxenes: Fe and Mg, but little Ca Both M 1 and M 2 are octahedral n Larger Fe ion more concentrated in M 2 site n n These minerals are the enstatite – ferrosilite solid solution series

n Low-Ca clinopyroxene: more Ca, but no solid solution with Hi-Ca clinopyroxene Mineral species is Pigeonite n Ca restricted to M 2 sites, these still mostly Fe and Mg n M 1 sites all Mg and Fe n

n Ca- clinopyroxene Diopside Mg(+Ca) to Hedenbergite Fe (+Ca) n M 2 site contains mostly Ca n M 1 site contains mostly Fe and Mg n n Most common specie is augite Al can substitute in M 1 site, and for Si in tetrahedral site n Na, Fe or Mg can substitute for Ca in M 2 site n

n n Other common pyroxenes Don’t fall neatly on Ca-Fe-Mg ternary diagram: Jadeite Na. Al. Si 2 O 6 n Spodumene Li. Al. Si 2 O 6 n

Possible ranges of solid solutions “Augite” Clinopyroxene Orthopyroxenes Na, Al – bearing pyroxenes Fig. 14 -2

Amphibole Group n n n Structure, composition, and classification similar to pyroxenes Primary difference is they are double chains Z/O ratio is 4/11

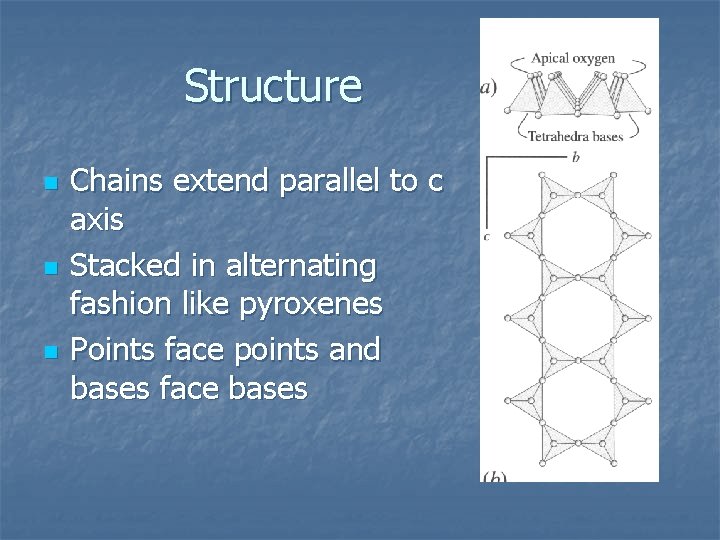
Structure n n n Chains extend parallel to c axis Stacked in alternating fashion like pyroxenes Points face points and bases face bases

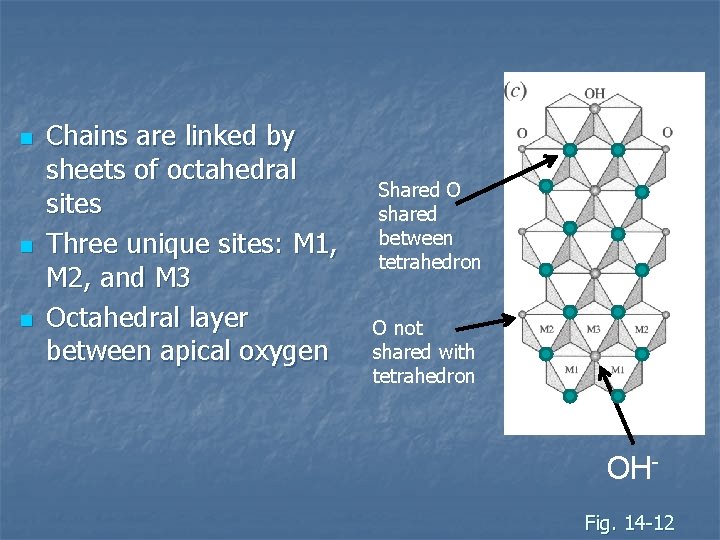
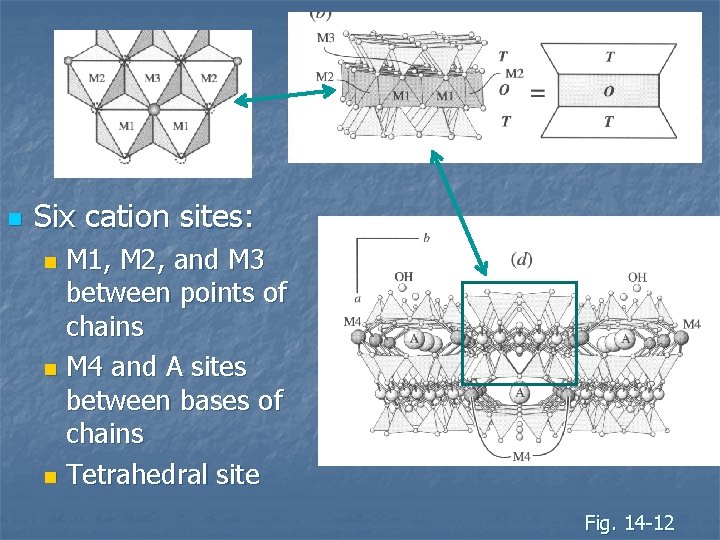
n n n Chains are linked by sheets of octahedral sites Three unique sites: M 1, M 2, and M 3 Octahedral layer between apical oxygen Shared O shared between tetrahedron O not shared with tetrahedron OHFig. 14 -12

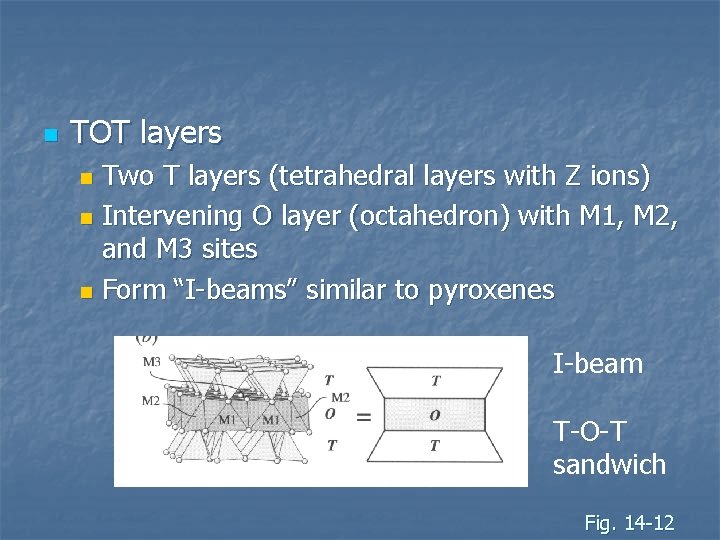
n TOT layers Two T layers (tetrahedral layers with Z ions) n Intervening O layer (octahedron) with M 1, M 2, and M 3 sites n Form “I-beams” similar to pyroxenes n I-beam T-O-T sandwich Fig. 14 -12

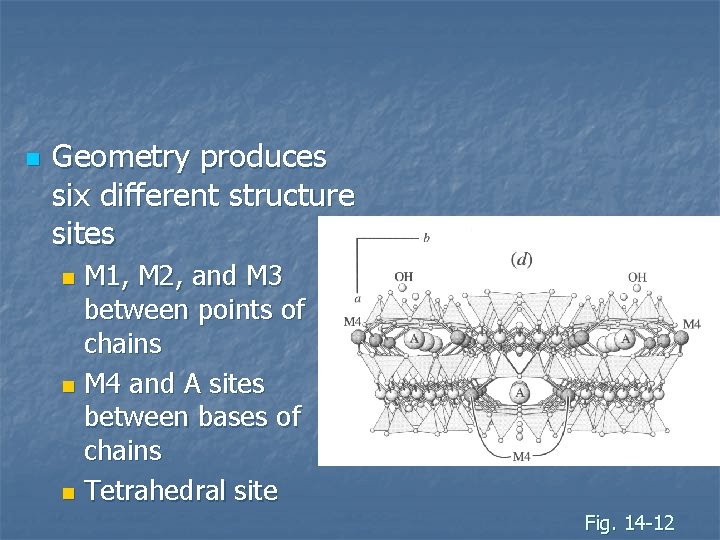
n Geometry produces six different structure sites M 1, M 2, and M 3 between points of chains n M 4 and A sites between bases of chains n Tetrahedral site n Fig. 14 -12

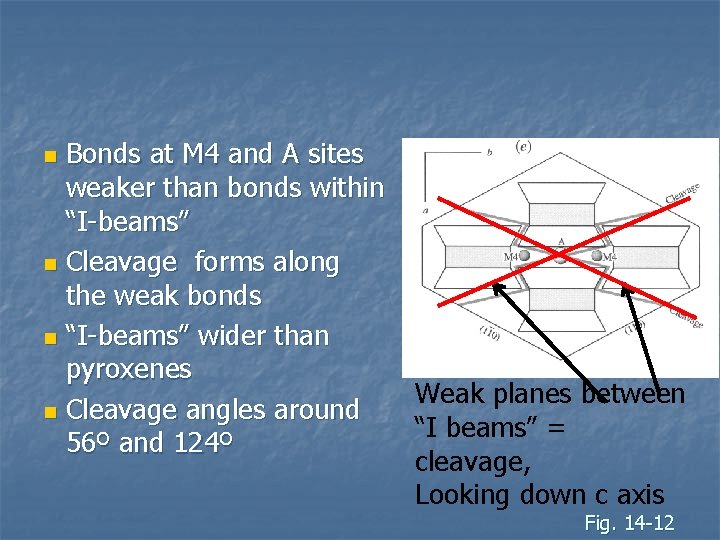
Bonds at M 4 and A sites weaker than bonds within “I-beams” n Cleavage forms along the weak bonds n “I-beams” wider than pyroxenes n Cleavage angles around 56º and 124º n Weak planes between “I beams” = cleavage, Looking down c axis Fig. 14 -12

n Six cation sites: M 1, M 2, and M 3 between points of chains n M 4 and A sites between bases of chains n Tetrahedral site n Fig. 14 -12

Composition W 0 -1 X 2 Y 5 Z 8 O 22(OH)2 n n n Note: Z/O ratio 4/11 Each cation fits a particular site W cation Occurs in A site n Has ~10 fold coordination n Generally large, usually Na+ n

W 0 -1 X 2 Y 5 Z 8 O 22(OH)2 n X cations Located in M 4 sites n Analogous to M 2 sites in pyroxenes n Have 6 or 8 fold coordination depending on arrangement of chains n If 8 -fold, X usually Ca n If 6 -fold, X usually Fe or Mg n

W 0 -1 X 2 Y 5 Z 8 O 22(OH)2 n Y cations Located in M 1, M 2, and M 3 sites; Octahedral cations in TOT strips n Similar to M 1 sites in pyroxenes n Usually Mg, Fe 2+, Fe 3+, Al n n Z cations n Usually Si and Al

W 0 -1 X 2 Y 5 Z 8 O 22(OH)2 n Water – hydrous phase Form from magma that contains water n Form from weathering of pyroxenes at surface n

n Composition Most common amphiboles shown on ternary diagram n Wide variety of substitution, simple and coupled n Divided into ortho and clino amphiboles n Depends on X cations in M 4 site (largely amount of Ca), distorts structure n Reduces symmetry from orthorhombic to monoclinic n

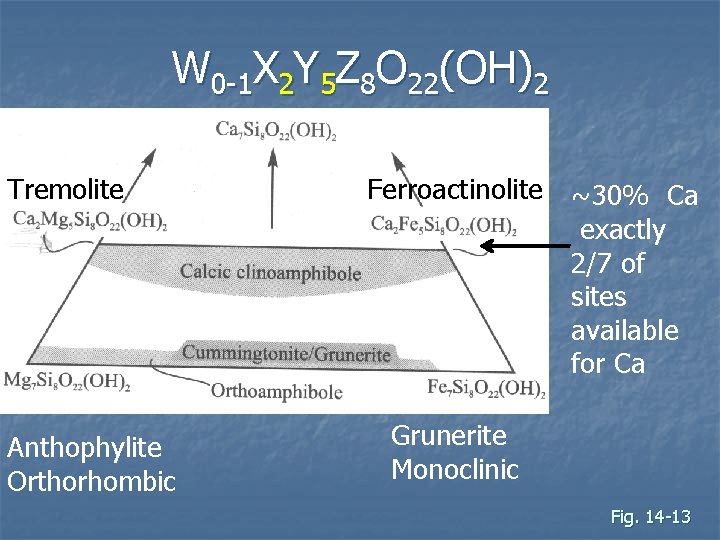
W 0 -1 X 2 Y 5 Z 8 O 22(OH)2 Tremolite Anthophylite Orthorhombic Ferroactinolite ~30% Ca exactly 2/7 of sites available for Ca Grunerite Monoclinic Fig. 14 -13

Pyroxenes and Amphiboles

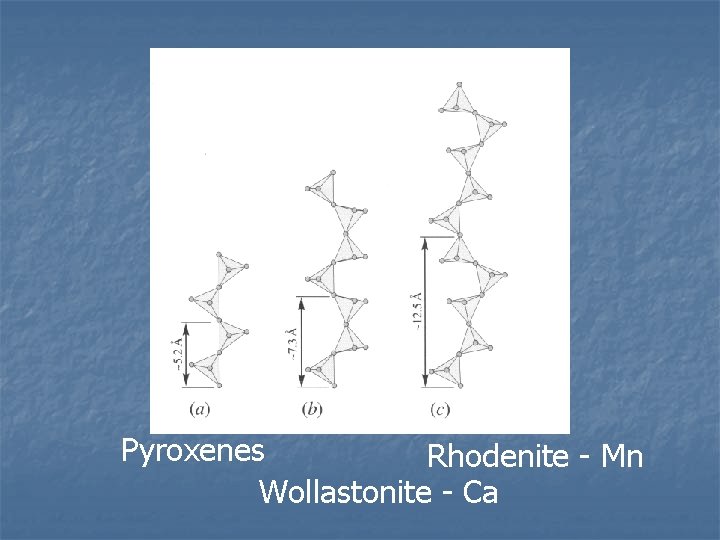
Pyroxenoid Group n Similar to pyroxenes Single chains n Z/O ratio 1/3 n n Differ in repeat distance along c axis Pyroxene – 2 tetrahedron repeat (5. 2 Å) n Pyroxenoid – 3 or more repeat (more than 7. 3 Å) n Difference is the pyroxenes are straight pyroxenoids are kinked n n Cased by larger linking cations

Pyroxenes Rhodenite - Mn Wollastonite - Ca

n n Only a few minerals Most common have Ca, Mn, or Ca plus Na filling the M 1 and M 2 sites Wollastonite – Ca, fairly common, metamorphosed qtz and carbonate systems n Rhodonite – Mn n Pectolite – Ca and Na n

n Wollastonite Composition: Ca with some Mn and Fe substitution n Common in altered carbonate rocks, particularly with reaction with qtz n Useful industrial mineral, replacing asbestos, also used in paints and plastics n