Orthopedic Soft Tissues Loadbearing Connective Tissues Properties of

Orthopedic Soft Tissues Load-bearing Connective Tissues Properties of Soft Tissue Components Tendon Mechanical Properties Ligament and Tendon Notes Cartilage Types Mechanical Properties - Articular Cartilage Mechanical Properties - Meniscus Biomateriali - Prof. O. Sbaizero

TESSUTO Il tessuto è un insieme di cellule simili per forma, funzione e derivazione. I diversi tipi di tessuto del corpo umano sono: ü tessuto connettivo, specializzato nel sostenere e nel tenere insieme gli altri tessuti ü tessuto epiteliale, specializzato nella protezione, nell'assorbimento e nella secrezione ü tessuto muscolare, specializzato nella contrattilità e nella conduttibilita del protoplasma ü tessuto nervoso, specializzato nella irritabilità e nella conduttività. I diversi tipi di tessuto sono composti da tre identici componenti: le cellule, la sostanza intercellulare e il liquido. In certi casi, i tessuti epiteliali e i tessuti connettivi assumono rapporti fra loro, dando origine a strutture speciali, le mucose e le sierose. Biomateriali - Prof. O. Sbaizero

Il tessuto connettivo è un particolare tipo di tessuto delle forme viventi superiori. Esso provvede al collegamento, sostegno e nutrimento dei tessuti dei vari organi. È costituito da cellule connettivali di vario tipo (fibrociti o fibroblasti, istociti, plasmacellule, mastcellule, cellule adipose, cellule endoteliali, ecc. ) e di una sostanza fondamentale, formata da una parte omogenea detta anista e da una parte differenziata in fibre. Le fibre sono distinte in: • fibre collagene • fibre reticolari • fibre elastiche Le fibre collagene e le fibre elastiche, sono formate da un insieme di filamenti, detti protofibrille (fibrille elementari). Lo spessore delle protofibrille è di circa 0. 1 m. Biomateriali - Prof. O. Sbaizero

Vi sono diversi tipi di tessuto connettivo: ü tessuto connettivo fibrillare (es. tessuto connettivo fibrillare degli organi interni, e anche tendini) ü tessuto connettivo elastico, con prevalenza di fibre elastiche (es. pareti delle arterie di grosso calibro, come l’aorta. ) ü tessuto connettivo reticolare, con prevalenza di fibre reticolari (es. il tessuto di connessione delle cellule specifiche di molti organi, ad esempio le linfoghiandole, il fegato, la milza e le ghiandole in genere. Forma anche le guaine che avvolgono le fibre muscolari o i capillari sanguigni) ü tessuto adiposo (presente in molte parti del corpo e, in particolare, sotto alla pelle, venendo a costituire il pannicolo adiposo) Biomateriali - Prof. O. Sbaizero

ütessuto endoteliale (Gli endoteli sono tessuti che rivestono la superficie di cavita’ non comunicanti con l'esterno, ad esempio la superficie interna del cuore e dei vasi sanguigni, sotto forma di una sottile lamina) ü tessuto cartilagineo (costituito da fibre connettivali immerse in una sostanza anista molto consistente e di cellule contenute in cavita’ particolari) ü tessuto osseo (fibre collagene unite a una sostanza amorfa calcificata) ü sangue, linfa Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero

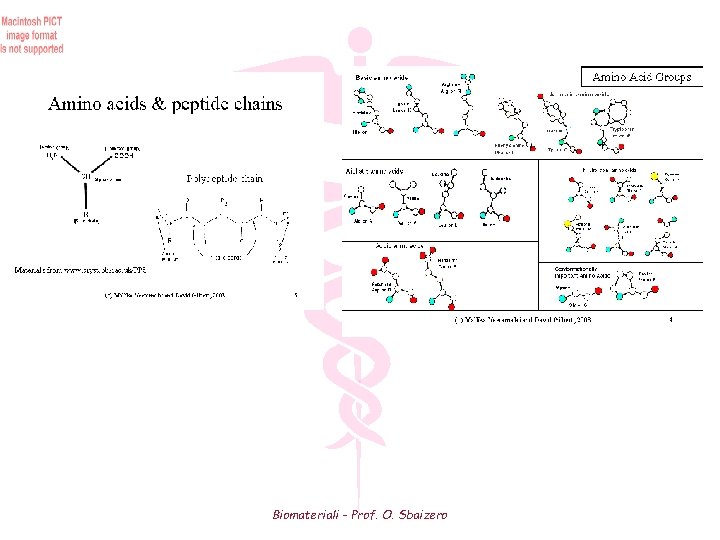
Soft Tissues Proteins are polyamides formed by reaction between amino and carboxyl groups of amino acids where R is a side group O H H (C N C) R n Depending on the side group, the molecular structure changes drastically The simplest side group is (H) which will form GLYCINE (Gly) If the side groups are larger then the resulting structure is an a helix where the hydrogen bonds occur between different parts of the same chain and hold the helix together Collagen The major structural protein is collagen, which has the amino acid sequence of Gly-Pro-Hyp-Gly-X (X can be any other amino acid) arranged in a triple a helix. Pro = Proline (Prolina) Hyp = Hydroxyproline (Idrossiprolina) Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero
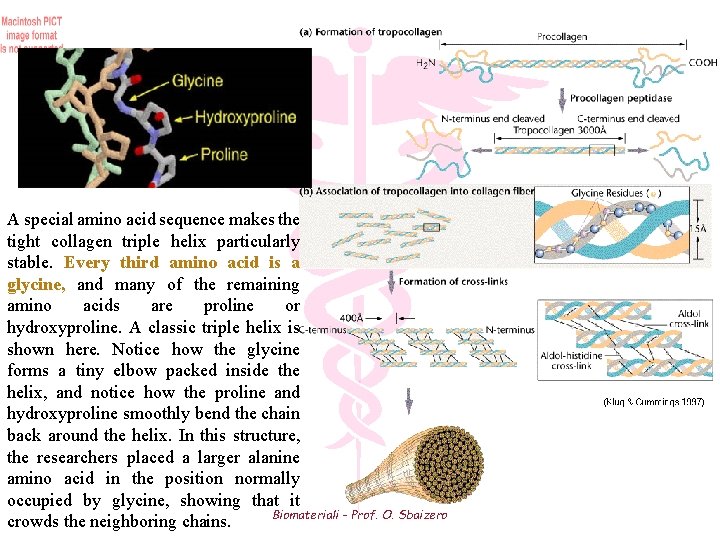
A special amino acid sequence makes the tight collagen triple helix particularly stable. Every third amino acid is a glycine, and many of the remaining amino acids are proline or hydroxyproline. A classic triple helix is shown here. Notice how the glycine forms a tiny elbow packed inside the helix, and notice how the proline and hydroxyproline smoothly bend the chain back around the helix. In this structure, the researchers placed a larger alanine amino acid in the position normally occupied by glycine, showing that it Biomateriali - Prof. O. Sbaizero crowds the neighboring chains.

Se riscaldate, le fibre di collagene si restringono (circa 2/3) e assumono una consistenza gommosa. La temperatura di transizione (denaturazione) varia a seconda del tipo di animale, ma comunque aumenta all’aumentare della quantita’ di prolina e idrossiprolina La proporzione di GLICINA e’ circa il 30% il che e’ molto elevato, altre proteine (emoglobina) ne contengono solo il 7% Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

ELASTINA L’ elastina si trova nella gran parte dei tessuti connettivi assieme al collagene. Nel caso delle fibre elastiche essa e’ il componente principale e in questo caso si possono avere deformazioni anche severe con recupero totale. Grandi quantita’ di elastina si trovano in alcuni vasi sanguigni, nei polmoni e nella pelle E’ almeno 5 volte piu’ elastica di un classico elastico Fibre di collagene e di elastina sono spesso intercalate per limitare la deformazione e prevenire la lacerazione dei tessuti Biomateriali - Prof. O. Sbaizero

In contrast to collagen, only one gene is known for elastin Elastin consists of about 800 amino acids. The protein is unusually rich in proline and glycine but, unlike collagen, is not glycosylated and contains little hydroxyproline, proline and no hydroxylysine. It does not contain the Gly-X-Y repeating sequence motif found in collagens. . Tropoelastin consists of lengths of helix separated by short nonhelical regions rich in lysine residues. The helical regions stretch upon applying tension, but revert to their original length when tension is released due to crosslinking. Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

Load-bearing connective tissues (1) Three primary tissues Tendon Ligament Cartilage • Composed of three basic structures plus few blood vessels or nerves - Collagen # One of the basic structural proteins of the human body # Fibrous protein with triple helical structure # Fibrils form fiber bundles with a diameter of 0. 2 -1. 2 micrometers (Fig 1) # Collagen is enormously strong due to a combination of biochemical bonds known as molecular cross-link # Collagen fibers vary in size due to tissue and age # Tensile strength of individual collagen fibers is about 70 MPa comparable with nylon or pure Al # Like rope, collagen offers little resistance to compression or shear Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

Load-bearing connective tissues (2) - Elastin • Fibrous protein with highly elastic nature • Role of elastin is to recover ligament length after stress is removed - Proteoglycans (1% in ligament, 3 -10% in cartilage) • Matrix with viscoelastic properties made up of sugar polymers • Hydrophilic properties (may be involved in regulating the amount and movement of H 2 O within tissue) • Charged structure can provide compressive strength • Relatively abundant in cartilage (10%) less in bone (1%) Biomateriali - Prof. O. Sbaizero

- Glycosaminoglycan • Part of the proteoglycan contains sugars on a protein core which have a negative elettrostatic charge and are attracted to water. • The negatively charged molecules repel each other and attempt to become as far apart as possible. • In vitro they can hold up to 50 times their weight in water, in vivo they are restrained by the collagen mesh and they can hold up only 20 times their weight in water. • The result is a stiff viscoelastic gell surrounding the collagen fibers • When the negative charges are forced together (an applied external stress) repulsive forces increases, increasing swelling pressure and frictional drag of fluid Biomateriali - Prof. O. Sbaizero

POLISACCARIDI Formati da ZUCCHERI SEMPLICI Comportamento altamente viscoso Interagiscono con le proteine (anche collagene) MUCOPOLISACCARIDI Biomateriali - Prof. O. Sbaizero
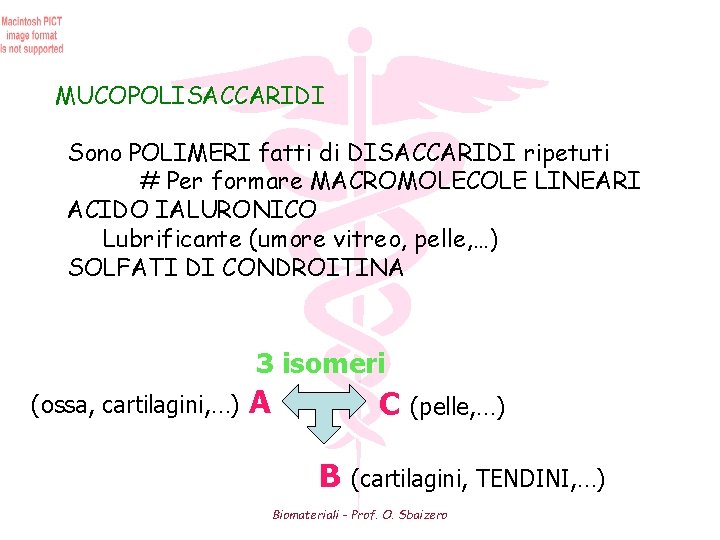
MUCOPOLISACCARIDI Sono POLIMERI fatti di DISACCARIDI ripetuti # Per formare MACROMOLECOLE LINEARI ACIDO IALURONICO Lubrificante (umore vitreo, pelle, …) SOLFATI DI CONDROITINA 3 isomeri (ossa, cartilagini, …) A C (pelle, …) B (cartilagini, TENDINI, …) Biomateriali - Prof. O. Sbaizero

Load-bearing connective tissues (3) - Water # Water makes up appr. 2/3 of the wet weight of a ligament # It can be bound to polar side chain (loosely bound) # It can be structurally bound to other matrix components # It can be freely associated with the interfibrillar gel Most water is freely bound # Its role is crucial in influencing with proteoglycans the tissue viscoelastic behaviour # Provide lubrication and facilitate inter-fascicular sliding # It carries nutrients and takes waste substances away Biomateriali - Prof. O. Sbaizero

Tendon Histology Tendon has two component: • Sparse slender cells - fibroblasts (they vary in size, shape and distribution but most are spindle-shaped and oriented parallel to the tendon longitudinal axis • The more abundant extracellular matrix - contain structural macromolecules and fluid. The fluid phase (mainly H 2 O) makes up to 58 -70% of a tendon wet weight. The structural macromolecules include the proteins collagen, elastin, proteoglycan and fibronectin. Collagen is the most abundant (30% wet weight, 80% dry). Fibronectin is a large glycoprotein that plays an important role in the interaction between fibroblasts and the extracellular matrix, particularly in cell-to-cell adhesion, fibroblast to collagen attachment and cell migration Biomateriali - Prof. O. Sbaizero

Properties of Soft Tissue Components • Elastin and collagen fibres both withstand tensile loading - Properties differ substantially • Collagen-rich tissues (tendon, ligament, cartilage) have a maximum tensile modulus which approaches that of a single collagen fibre Biomateriali - Prof. O. Sbaizero

Properties of Soft Tissue • Mechanical properties depend of composition and microstruture - Includes fibre arrangement Parallel fibres Crossed fibre arrays Felt-like fibre structure • General viscoelastic properties of orthopaedic soft tissue - Stress-strain relationship is rate dependent and exhibits hysterisis #Hysterisis due to internal energy losses - Experience stress relaxation under constant strain and creep under constant stress • Ligaments and tendons undergo preconditioning - Cycles of tension and relaxation result in progressive reduction in generated stress (for defined strain) (Fig 2) - Reaches a sustainable response after about 10 cycles - Due to fluid being extruded after a period of inactivity - Accounts for "stiff joints" after sitting around Biomateriali - Prof. O. Sbaizero

Figure 2 Effect of preconditioning soft tissue Biomateriali - Prof. O. Sbaizero

Tendons are fibrous cords Function - Connect muscle and bone - Transmit contractile forces to bone - Store elastic energy - Tendons in areas that bend sharply, such as the tendons which cause the fingers to bend (flexor digitorum) pass through a tendon sheath that directs the path of the tendon What would another function of this sheath be? Ans: Another function of the tendon sheath is to protect the tendon from repeated wear due to high friction during movement (presence of synovial fluid) • Structure (Fig 3) Densely packed collagen fibres (86% dry weight) Low elastin content Few cells Uniaxial fibre orientation Cross-sectional shape resembles attached muscle Varies between anatomical location Biomateriali - Prof. O. Sbaizero

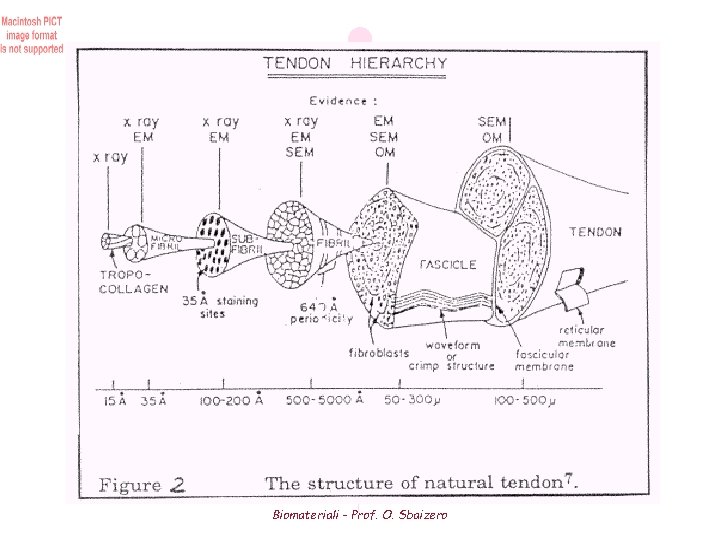
Figure 3 Hierarchical structure of a tendon. Tropocollagen is the fundamental unit of collagen. Five tropocollagen molecules group togheter to form a microfibril Microfibrils aggregate into subfibril which in turn group together into fibrils Groups of fibrils or fibres, bundle together and aggregates to form fascicles Fibroblasts are distribuited sparsely among the fibers Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero

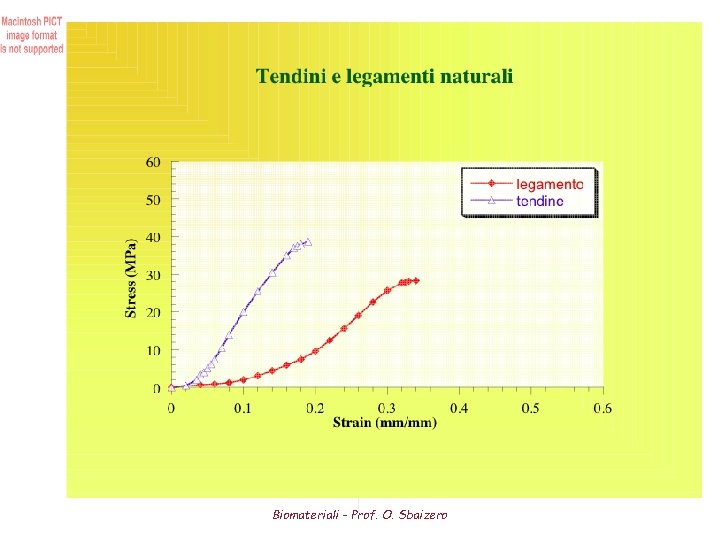
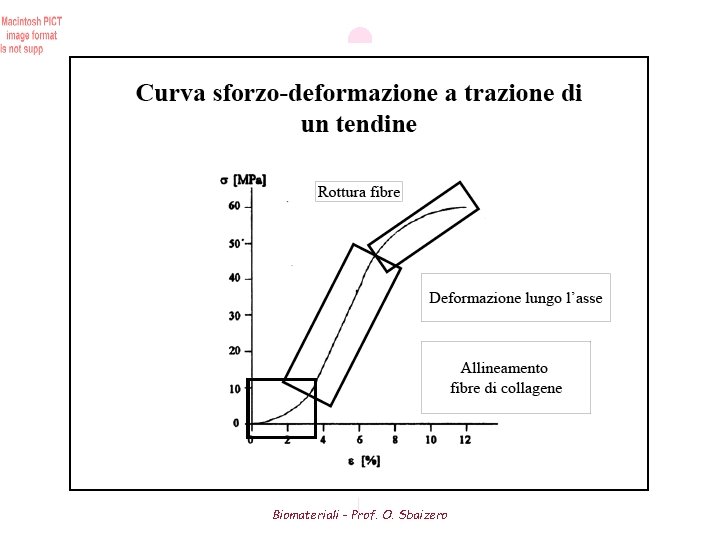
Tendon Mechanical Properties (1) The primary role of tendon is to transmit the force of its associated muscle to bone. As such tendon needs to be relatively strong and stiff in tension • High tensile strength and elastic modulus (due to its relatively high collagen content) One of highest tensile strengths of any soft tissue (50 to 150 MPa) Modulus ranges from 1. 2 to 1. 8 GPa Properties depend on the individual and the anatomic location Although tendon is relatively inextensible compared to other proteins (elastin), it is much stiffer and stronger • High collagen content results in relatively small strains Load-bearing tendons (ie. leg and spinal tendons) allow max. strain of about 10% Non-load bearing tendons allow maximum strain of about 3 -4 percent • Incompressible material • Tendons experience a small hysteresis therefore they are capable of storing and releasing considerably elastic strain energy mechanical efficiency of running Biomateriali - Prof. O. Sbaizero

Tendon Mechanical Properties (2) • During isometric contraction, the tendon will tend to lengthen due to creep Muscle is allowed to contract further while maintaining the same overall length Decreases rate of muscle fatigue • Tendon exhibits less viscoelastic (strain rate) effects than other soft tissue • Stress-strain behavior plotted as a non-linear curve (Fig 4) I. Alignment of fibers in the direction of stress II. Majority of fibres stretched along their long axes III. Individual fibres begin to break prior to catastrophic failure • Length of initial non-linear region increases with age What are disadvantages if tendon elasticity increases (reduced stiffness)? Ans: A muscle will have to exert a greater force in order to move a bone the same distance as more stretch will be induced in the tendon before movement occurs Biomateriali - Prof. O. Sbaizero
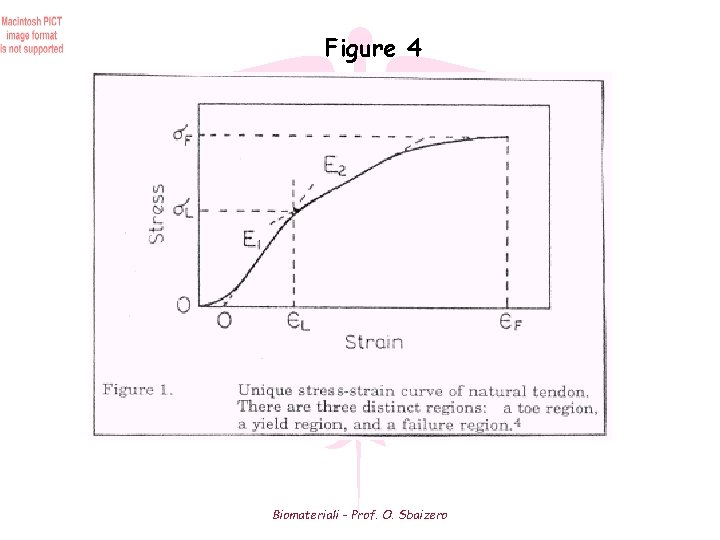
Figure 4 Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

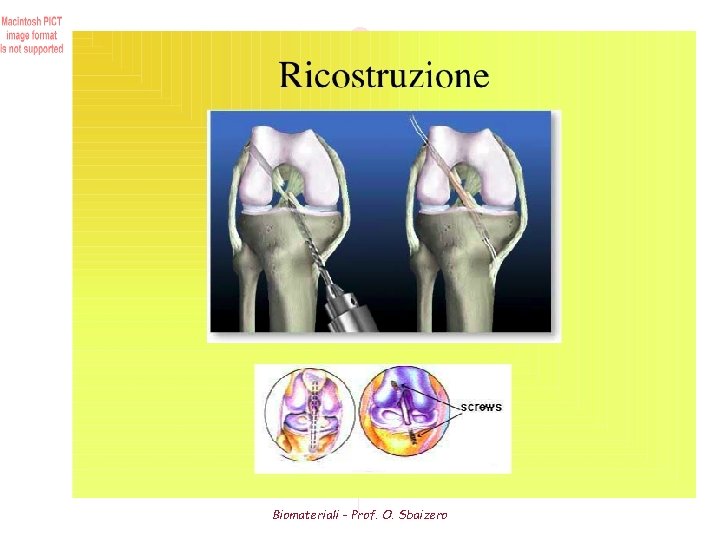
Ligament From the latin word “ligare” • Function Bone-bone connectors across a joint Stabilize joints (guide joint movement, maintain joint congruency) Possibly to act as a positional bend or strain sensor for the join • Structure Less densely packed collagen fibres (70 % dry weight) Slightly higher elastin content than tendon Flattened, elliptical cross-section • Connection to bone with one of two insertion types - Direct insertion: Ligament fibers cross "mineralization front" and progress from collagen fibrils to fibrocartilage to mineralized fibrocartilage and then bone transition zone is 1 mm deep - Indirect insertion: short collagen fibers anchor obliquely into periosteum of bone - Connection can be with either parallel fibre orientation (collateral ligaments of knee) or branching fibres (cruciate ligaments of knee) Biomateriali - Prof. O. Sbaizero
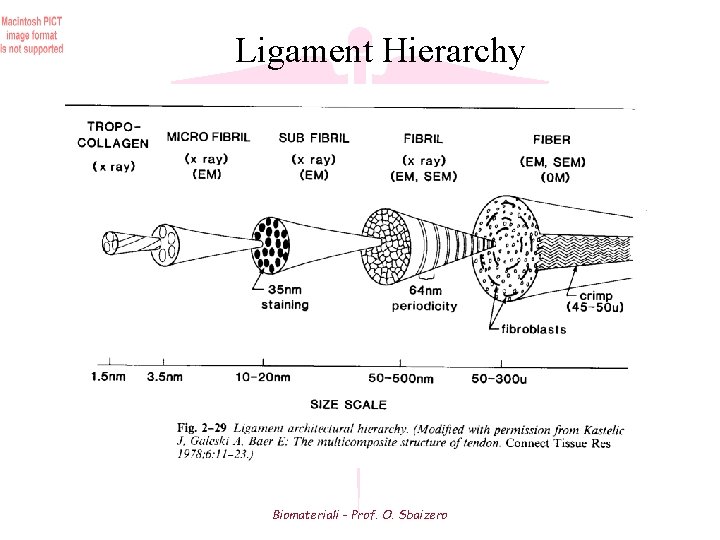
Ligament Hierarchy Biomateriali - Prof. O. Sbaizero

Ligament Fibers from SEM Biomateriali - Prof. O. Sbaizero

Ligament Mechanical Properties Less strong and less stiff than tendons - Due to lower collagen content (higher percentage of "ground substance") - More woven collagen structure compared to tendon's nearly total parallel arrangement • Same sort of stress-strain behavior attributed to fiber straightening and stretching -Ligaments exhibit force relaxation and creep. Force relaxation is a result of the viscous component of the ligaments’s response to force -Ligamets are 2/3 water. # The interfibrillar amount of water determines collagen stiffness # Water determines the distances at which fibers can interact # Because of its attraction to the highly negatively charged proteoglycan molecule water may induce pressure gradients in the tissue Biomateriali - Prof. O. Sbaizero
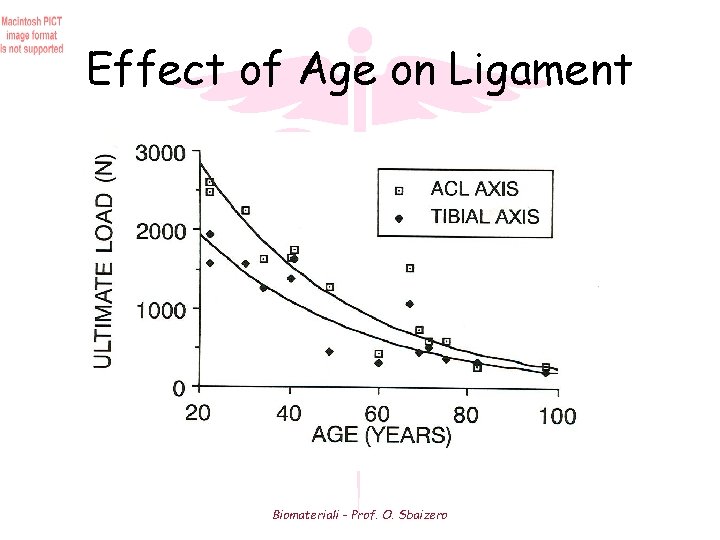
Effect of Age on Ligament Biomateriali - Prof. O. Sbaizero

Ligament and Tendon Notes • Anisotropic tissue oriented primarily for resistance of tensile loads - Some fibers crossing between parallel fibers that contribute to the non-linear stiffness behavior as the ligament is forced • Non-linear behavior modeled by fibers of varying degrees of "crimp" - Increasing tensile deformation recruits additional fibers • Effect of creep and stress relaxation: Ans: - Effect of creep on tendons -- in an isometric contraction, the tendon will lengthen slightly and more muscle fibers will be recruited in order to maintain the position of the limb. - Effect of stress-relaxation on tendons -- in an isotonic contraction, the stress in the tendon will decrease with time - Effect of creep and stress-relaxation on ligaments -- joints will loosen with time, decreasing the possibility of injury Biomateriali - Prof. O. Sbaizero

Cartilage Articular cartilage is a thin layer of fibrous connective tissue on the articular surfaces of bone in synovial joints Consists of cells (5%) and an intercellular matrix (95%) which is substantially H 2 O (65 -80%) • Function - Maintenance of shape (Nose, Ear, Rings of trachea) - Provide a bearing surface at articulating joints # with synovial fluid, provides a very low coefficient of friction • Structure - Consists of dense proteoglycan matrix with fewer tangled collagen or elastin fibres than tendon or ligament # Proteoglycan matrix can affect mechanical properties by hindering the movements of the collagen fiber network - Cartilage forming cells (chondrocytes) found within the spaces of the matrix Biomateriali - Prof. O. Sbaizero

Cartilage Structure (1) Cartilage is structurally heterogeneous. It changes with depth from the joint surface - Superficial zone - Transitional zone - Deep (or radial) zone - Calcified zone SUPERFICIAL ZONE Is the thinnest, most superficial region. It has a surface layer and a deeper layer The surface layer is 2 m thick and it consists of random flat bundles of collagen fibrils The deeper layer consists of dense collagen fibers parallel to the plane of the joint surface The deeper layer also contains elongated chondrocytes laying parallel to the joint surface Of all the zones this one contains the highest concentration of H 2 O (80%) Water decreases almost linearly till about 65% in the deep zone The superficial zone is structured to resist the shear stress during joint motion Biomateriali - Prof. O. Sbaizero

Cartilage Structure (2) TRANSITIONAL ZONE Consists of collagen fibrils with larger diameters than those in the superficial zone They lie parallel to the plane of joint motion but they are less parallel than those on the surface Chondrocytes are spherical It contains cells able to sinthetize matrix DEEP ZONE Features large numbers of big collagen bundles running perpendicular to the plane of joint motion Proteoglycan content is highest in the deep zone Water content is relatively low Chondrocytes are round and stacked on top of each other in column perpendicular to the joint surface This zone produces proteins Biomateriali - Prof. O. Sbaizero

Cartilage Structure (3) CALCIFIED ZONE It marks the transition from soft cartilage to stiffer subchondral bone It is characterize by the presence of hydroxyapatite The collagen fibers anchor the cartilage to the bone The heterogeneous composition and structural framework result in a highly anisotropic material HISTOLOGY CELLS They account for less than 10% and are called chondrocytes Chondrocytes are similar to the osteocytes of bone in that they play an active metabolic role MATRIX Consists of structural macromolecules (20 -40%) and tissue fluid (60 -80%) Macromolecules are collagen, proteoglycan and other proteins The interactions of tisse fluid with macromolecules produce mechanical entanglement, elettrostatic bonding and excluded volume effects. Biomateriali - Prof. O. Sbaizero

Types of Cartilage • Hyaline cartilage /cartilagine IALINA)- - Small, evenly distributed collagen fibres (50 - 80 % dry weight) - Negligible elastin content - Glassy smooth appearance Ex. Articular cartilage Growth plate Rib/costal cartilage Support of trachea (wind pipe) Nasal septum • Fibrous cartilage - More numerous, densely packed collagen fibres than hyaline cartilage (about 90% dry weight) Examples Outer portion of intervertebral disc (annulus fibrousus) Meniscus of knee (provides padding, shock absorption) • Elastic cartilage - Similar to hyaline cartilage, but also contains elastin fibres -- therefore more flexible - Present in structures subject to repeated bending Ex: Epiglottis External auditory canal Biomateriali - Prof. O. Sbaizero Eustachian tube
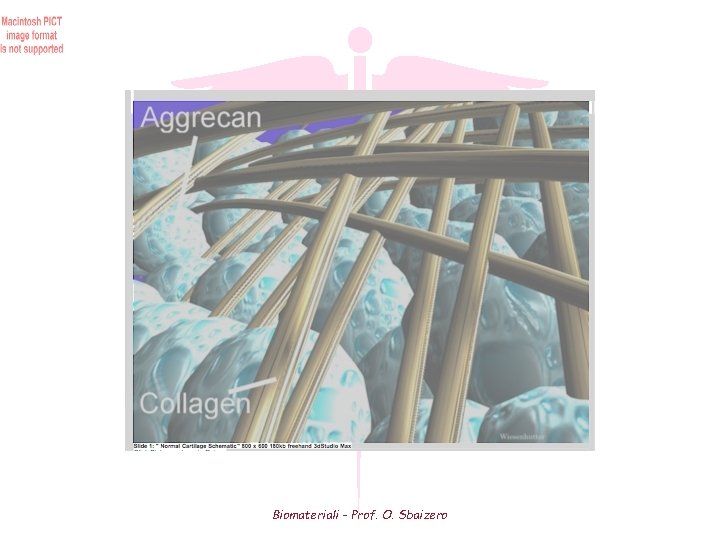
Biomateriali - Prof. O. Sbaizero

Mechanical Properties - Articular Cartilage (1) • Large variation in mechanical properties based on location and subject age • Collagen gives much of its tensile strength • Anisotropic due to variation in collagen structure • Even non-articular cartilage is load bearing • Choice of properties for a replacement material would depend on an analysis of stresses experienced at the site with normal physiological activity • Tensile behavior: Initial toe region followed by linear extension I. Straightening of fibres II. Stretching of fibres Stiffness independent of proteoglycan content Low tensile strength (compared to ligament and tendon ) Biomateriali - Prof. O. Sbaizero

Mechanical Properties - Articular Cartilage (2) • Compressive behavior: Stress-strain plot linear to failure (very little yield) Independent of collagen content -- dependent on proteoglycan concentration Proteoglycans are related to the negative charge in the matrix High weight bearing areas stiffer than low weight bearing areas Have higher proteoglycan content • Average Properties Tensile modulus: 1 - 10 MPa Compressive modulus: about 1 MPa in healthy individuals • Both Young's modulus and strength decrease with depth in the cartilage pad - Due to changes in the collagen structure and content (Fig 5) - Superficial tangential zone: fibres parallel to surface - Highest collagen content - Middle zone: random fibres, preference towards 45° - Lowest collagen content - Deep zone: perpendicular to surface, cross over and connect to calcified tissue Biomateriali - Prof. O. Sbaizero

Mechanical Properties - Articular Cartilage (3) • Articular cartilage experiences creep to an equilibrium point under compression - Equilibrium may not be reached until about 30 minutes after load application - Permeability decreases with increased compressive strain - Prevents excessive fluid loss under prolonged compression - Creep is recoverable - Water re-absorbed into the cartilage's proteoglycan matrix - Under static load, initial strain is about 25% of equilibrium strain - The viscous drag on the fluid being exruded provides the energy dissipation in articular cartilage and meniscus • Low coefficient of friction (< 0. 01) - Due to squeeze-film effect - Synovial fluid is squeezed out of the cartilage under compressive loading - Fluid resorped into cartilage under tension - Provides lubricating film Biomateriali - Prof. O. Sbaizero

Mechanical properties - Meniscus • Compressive modulus about 0. 4 MPa • Lower fluid permeability than articular cartilage • Meniscus and articular cartilage have same basic function - Provide shock absorption and a bearing surface - Difference in properties indicates that they perform these functions differently - Meniscus must withstand large tensile hoop stresses caused by radial "extrusive" forces during loading • Loss of meniscus in the knee results in possible overloading of articular cartilage Biomateriali - Prof. O. Sbaizero

SYNOVIAL FLUID Synovial fluid is a transparent alkaline, viscous fluid secreted by the synovial membrane contained in joint cavities It contains a protein polysaccharides (hyaluronic acid, lubricin) The thickness of the synovial fluid between two articulating surfaces is about 0. 5 -1 m The synovial fluid is highly non-Newtonian. At low shear rates its viscosity is 1/10 Pa but At higher raters it is only 1/1000 Pa Biomateriali - Prof. O. Sbaizero
- Slides: 54