ORL JeanPierre DELORD Oncologue mdical 2 Presented By

ORL Jean-Pierre DELORD Oncologue médical

2 Presented By Kevin Harrington at TBD

3
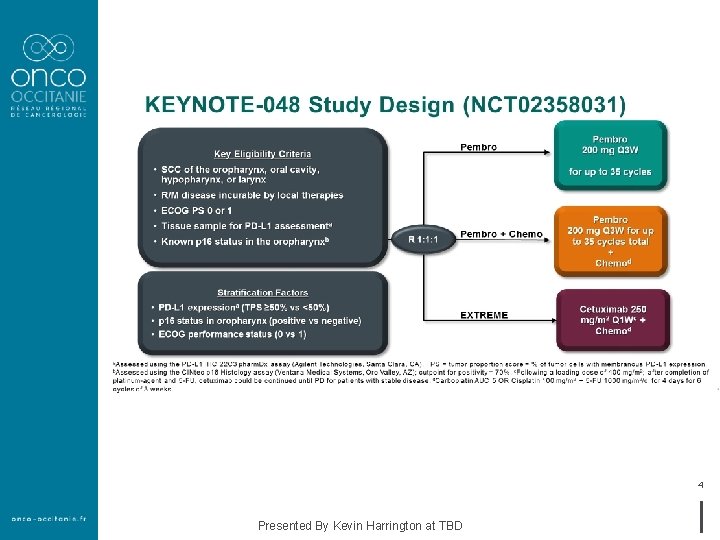
4 Presented By Kevin Harrington at TBD
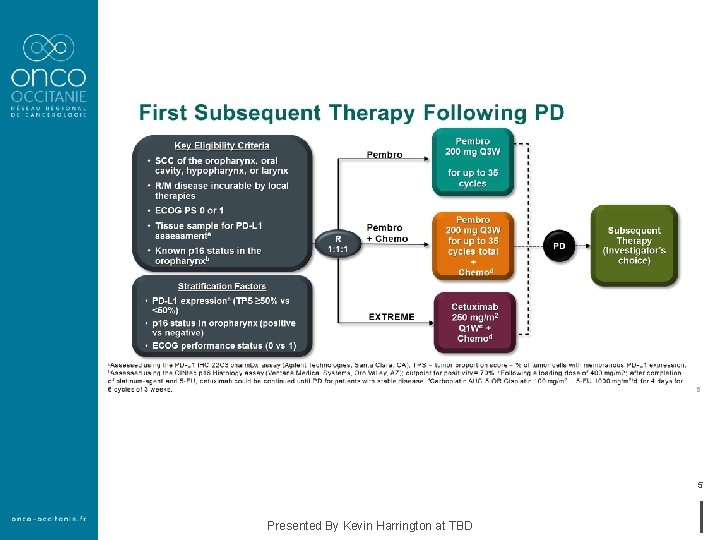
5 Presented By Kevin Harrington at TBD
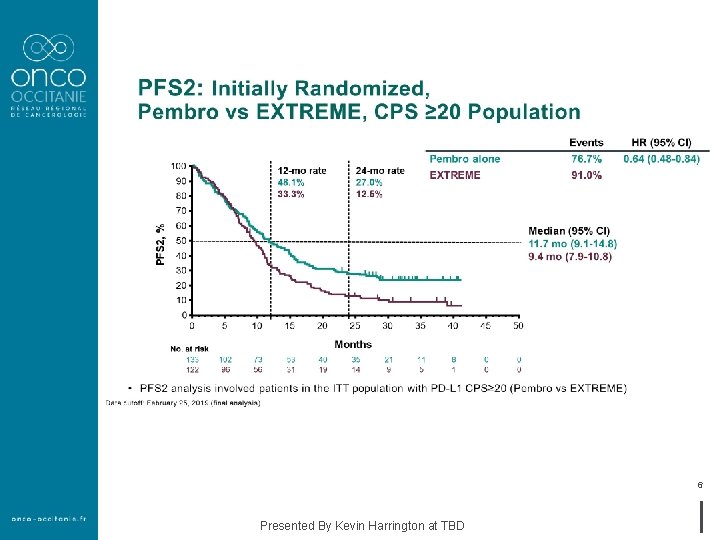
6 Presented By Kevin Harrington at TBD
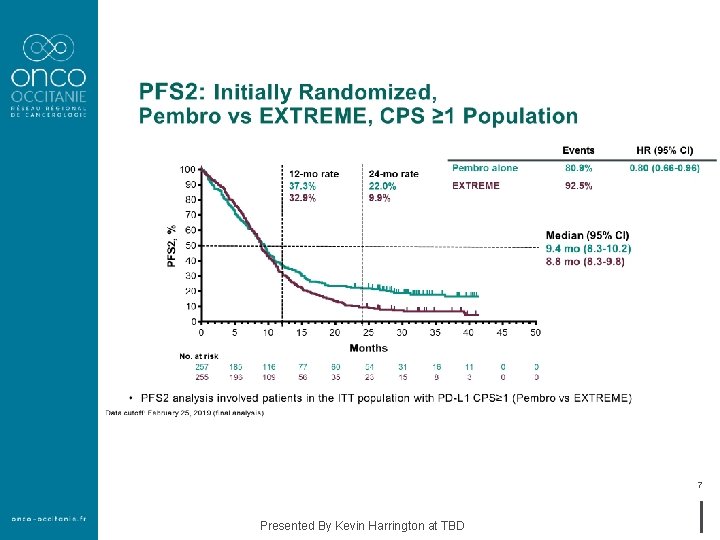
7 Presented By Kevin Harrington at TBD
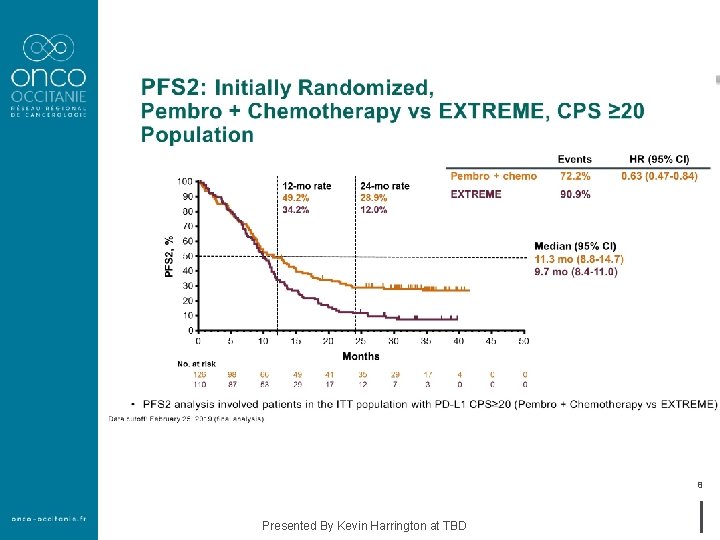
8 Presented By Kevin Harrington at TBD
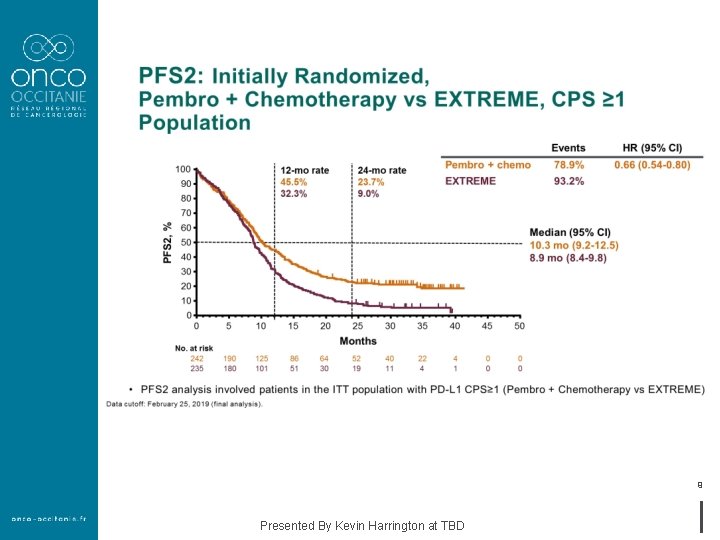
9 Presented By Kevin Harrington at TBD
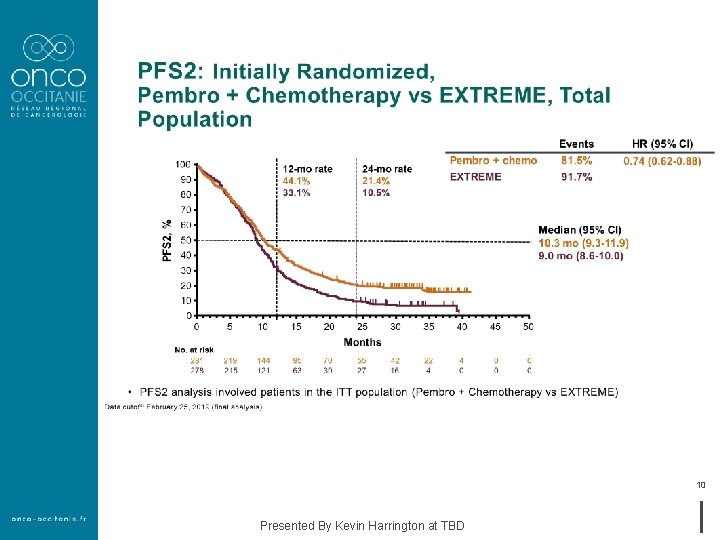
10 Presented By Kevin Harrington at TBD

11 Presented By Kevin Harrington at TBD

Phase II/III Trial of Post-operative Chemoradiotherapy Comparing 3 -Weekly Cisplatin with Weekly Cisplatin in High-risk Patients with Squamous Cell Carcinoma of the Head and Neck(JCOG 1008) Presented By Naomi Kiyota at TBD

Background (1) Presented By Naomi Kiyota at TBD
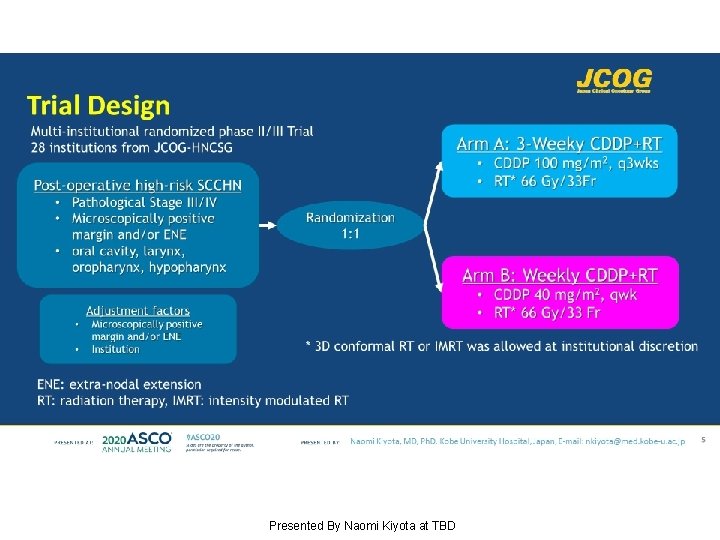
Trial Design Presented By Naomi Kiyota at TBD

Objectives and Endpoints Presented By Naomi Kiyota at TBD

Key Eligibility Criteria Presented By Naomi Kiyota at TBD

Statistical Considerations Presented By Naomi Kiyota at TBD
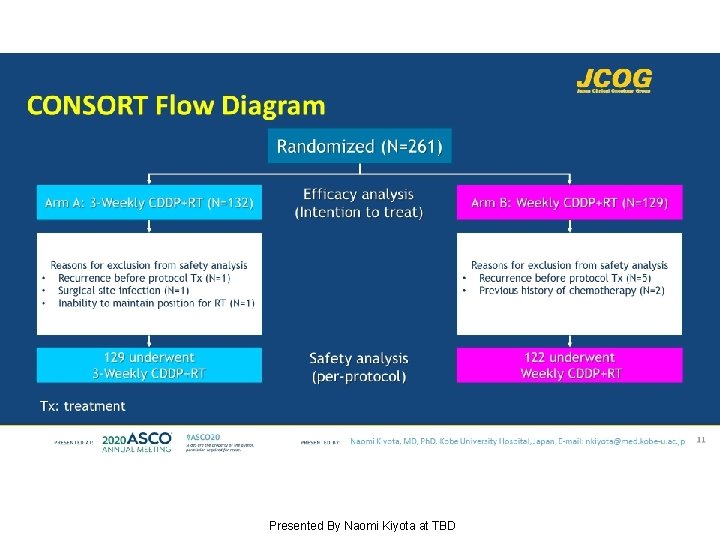
CONSORT Flow Diagram Presented By Naomi Kiyota at TBD
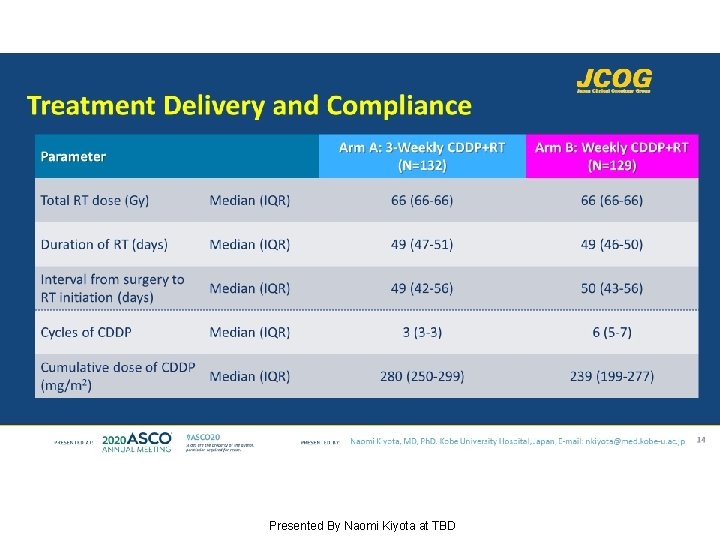
Treatment Delivery and Compliance Presented By Naomi Kiyota at TBD
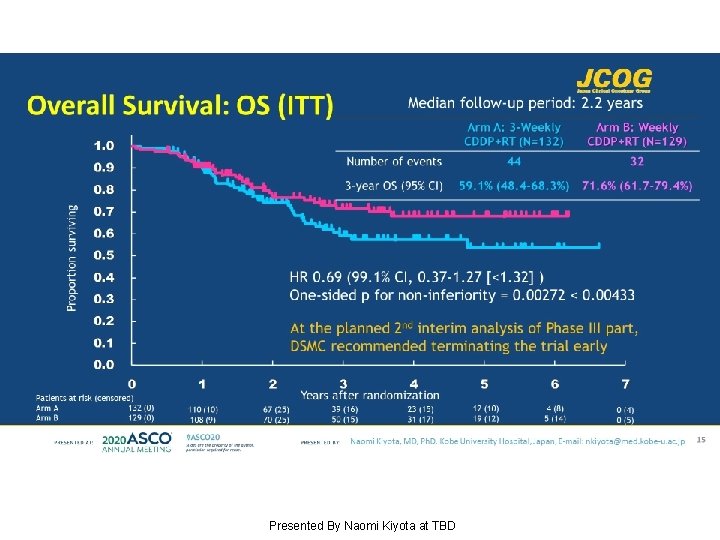
Overall Survival: OS (ITT) Presented By Naomi Kiyota at TBD
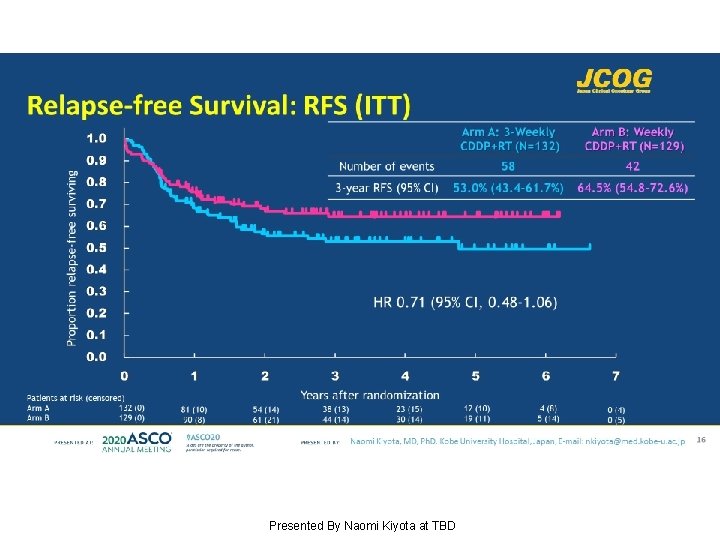
Relapse-free Survival: RFS (ITT) Presented By Naomi Kiyota at TBD
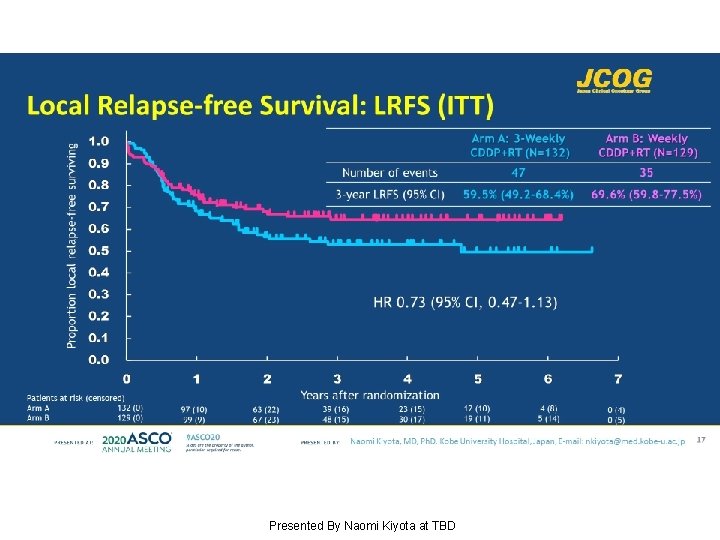
Local Relapse-free Survival: LRFS (ITT) Presented By Naomi Kiyota at TBD

Summary Presented By Naomi Kiyota at TBD
P 335 Background Efficacy and Safety of Entrectinib in Patients with NTRK Fusion-Positive (NTRK-fp) Solid Tumors: An Updated Integrated Analysis Rolfo C, 1 De Braud F, 2 Doebele RC, 3 Drilon A, 4 Siena S, 5 Patel MR, 6 Cho BC, 7 Liu SV, 8 Ahn MJ, 9 Chiu CH, 10 Farago AF, 11 Goto K, 12 Lee J, 9 Bazhenova L, 13 John T, 14 Fakih M, 15 Simmons B, 16 Pitcher B, 17 Huang X, 16 Demetri GD 18 63. 5 % kinase function that are potential oncogenic drivers across many tumor types Survival outcomes • Most patients who responded to entrectinib achieved a rapid time to first response • ORR was similar in patients with and without CNS metastases at baseline; however, median Do. R was • 41/74 (55. 4%) patients had progressive disease or died, and 24/74 patients (32. 4%) died lower in patients with baseline CNS metastases 1, 2 • Entrectinib is a potent inhibitor of TRKA/B/C and was designed to cross the blood-brain barrier common gene fusions: NTRK 1 (18/30; 60. 0%) and NTRK 3 (29/42; 69. 0%) and remain in the central nervous system (CNS) 3, 4 • It is important to consider the value of treatments with both systemic and intracranial efficacy for patients with NTRK-fp solid tumors and CNS metastases Systemic efficacy per BICR in patients with NTRK-fp solid tumors Parameter • In an integrated analysis of phase 1/2 studies (ALKA-372 -001, STARTRK-2; Eudra. CT 2012 -000148 -88; NCT 02097810; NCT 02568267), entrectinib demonstrated systemic and intracranial efficacy in patients with NTRK-fp solid tumors. 5 We present updated data, in a larger population with longer follow-up (d ata cut-off October 31, 2018) ORR*, n (%) 95% CI Methods: study design and patients 5 Primary endpoints Secondary endpoints Do. R 12. 9 months vs previous 10. 4 Safety population (received ≥ 1 dose of entrectinib) (N=504) Adult Phase 1: ALKA-372 -001 (n=60) STARTRK-1 (n=83) Adult Phase 2: STARTRK-2 (n=332) NTRK-fp adult analysis set (N=113) ROS 1+ NSCLC adult analysis set (n=210) Other adult analysis set (n=152)* Pediatric STARTRK-NG (n=29) NTRK-fp adult safety population (N=113) No CNS metastases (per BICR / per INV: n=58 / n=55) NTRK-fp extracranial solid tumors measurable at baseline (RECIST v 1. 1) per INV efficacy population (N=74) Phase 2: STARTRK-2 (n=108) Pediatric analysis set (n=29) Baseline CNS metastases ‡ (n=16) No baseline CNS metastases ‡ (n=58) 37 (63. 8) 50. 1– 76. 0 47 (63. 5) 51. 5– 74. 4 10 (62. 5) 35. 4– 84. 8 5 (6. 8) 0 5 (8. 6) Partial response (PR), n (%) 42 (56. 8) 10 (62. 5) 32 (55. 2) Stable disease (SD), n (%) 9 (12. 2) 4 (25. 0) 5 (8. 6) Progressive disease (PD), n (%) 6 (8. 1) 1 (6. 3) 5 (8. 6) Non-CR/PD, n (%) 3 (4. 1) 0 3 (5. 2) Missing or unevaluable†, n (%) 9 (12. 2) 1 (6. 3) 8 (13. 8) *BICR assessed, RECIST v 1. 1. †Includes patients with unevaluable on-study scans or those who discontinued prior to ‡CNS metastases status determined by BICR. NE, not estimable. obtaining adequate scans to evaluate or confirm response. Median, months 12. 9 6. 0 12. 9 (95% CI) (9. 3–NE) Best individual response per BICR, by tumor t ype (4. 2–NE) Cholangiocarcinoma (n=1) Neuroendocrine (n=4) Sarcoma (n=16) GI-other (n=1) Pancreatic (n=3) CNS metastases (per BICR / per INV: n=16 / n=19) Phase 1: ALKA-372 -001 (n=1) STARTRK-1 (n=4) Efficacy population (N=74) Complete response (CR), n (%) Do. R* Efficacy and safety populations enrolled in the integrated analysis Durable disease control Survival outcomes in patients with NTRK-fp solid tumors • Entrectinib was active in a range of tumor types, with similar responses observed in patients with the most • Overall survival • Objective response rate (ORR) • Progression-free survival (PFS) (OS) • Duration of response (Do. R) • Efficacy by CNS disease status • Safety • Median duration of follow-up from first entrectinib dose: 14. 2 months (range 0. 1– 29. 7) Summary Clinically meaningful OR responses in NTRKfp solid tumors R • NTRK gene fusions lead to transcription of chimeric TRK proteins with constitutively active Systemic efficacy (9. 3–NE) Breast (n=6) Gynecological (n=2) Salivary MASC (n=13) Thyroid (n=7) CRC (n=7) NSCLC (n=13) Efficacy population (N=74) Baseline CNS metastases ‡ (n=16) No baseline CNS metastases ‡ (n=58) Median PFS*, months (95% CI) 11. 2 (8. 0– 15. 7) 6. 7 (4. 7–NE) 12. 0 (8. 7– 16. 0) Median OS, months (95% CI) 23. 9 (16. 0–NE) 14. 3 (7. 6–NE) 23. 9 (16. 8–NE) Parameter *BICR assessed, RECIST v 1. 1. ‡CNS metastases status determined by BICR. Intracranial efficacy • Intracranial ORR was 50. 0% (8/16 patients), including 4 patients with a complete response Intracranial efficacy per BICR in patients with baseline CNS metastases Parameter* Responders, n Intracranial ORR, % (95% CI) *ROS 1+ non-NSCLC, ALK fusion-positive, no gene fusion. INV, investigator; BICR, blinded independent central review. Strong intracranial efficacy in patients with CNS metastases at baseline Intracran ial ORR 50. 0% Systemic efficacy irrespective of presence or absence of CNS metastases at baseline ORR with 62. 5% withou t 63. 8% Presented at the 2020 American Society of Clinical Oncology (ASCO) Annual Meeting | 29 May– 2 June 2020 Copies of this poster obtained through Quick Response (QR) Code are for personal use only and may not be reproduced without permission from ASCO ® and the author of this poster. https: //bit. ly/2 W Da. Ej. G Visit Medically. Roche. com for more information. Affiliations 1. University of Maryland School of Medicine, Baltimore, MD, USA; 2. Department of Medical Oncology and Hematology, Fondazione IRCCS Istituto Nazionale dei Tumori, Milan, Italy; 3. University of Colorado, Aurora, CO, USA; 4. Memorial Sloan Kettering Cancer Center, and Weill Cornell Medical College, New York, NY, USA; 5. Grande Ospedale Metropolitano Niguarda, and Università degli Studi di Milano, Milan, Italy; 6. University of Minnesota, Department of Medicine, Minneapolis, MN, USA; 7. Yonsei Cancer Center, Seoul, Republic of Korea; 8. Georgetown University, Washington, DC, USA; 9. Samsung Medical Center, Seoul, Republic of Korea; 10. Department of Chest Medicine, Taipei Veterans General Hospital, Taipei, INTERACTIVE Taiwan; 11. Massachusetts General Hospital, Boston, MA, USA; 12. National Cancer Center Hospital East, Kashiwa, Japan; 13. University of California San Diego, CA, USA; 14. Olivia Newton-John Cancer Research Institute, Austin Health, Heidelberg, Australia; 15. City of Hope Comprehensive Cancer Center, Duarte, CA, USA; 16. Genentech Inc, South San Francisco, CA, USA; 17. Hoffmann-La Roche Ltd Mississauga, Canada; 18. Dana-Farber Cancer Institute, Boston, MA, USA Patients with missing SLD percent change are excluded from the plot. SLD, sum of longest diameters. GI, gastrointestinal. CRC, colorectal cancer. NSCLC, non-small-cell lung cancer. MASC, mammary analogue secretory carcinoma. Results: patient demographics and baseline characteristics Characteristic Age, years • Sex, n (%) 39 (52. 7) 35 (47. 3) White Asian Black Not reported 52 (70. 3) 13 (17. 6) 2 (2. 7) 7 (9. 5) ECOG performance status, n (%) 0 1 2 30 (40. 5) 34 (45. 9) 10 (13. 5) Prior lines of systemic therapy, n (%) 0 1 2 ≥ 3 20 (27. 0) 21 (28. 4) 20 (27. 0) 13 (17. 6) Any previous therapy, n (%) Chemotherapy Targeted therapy Hormonal therapy Immunotherapy 60 (81. 1) 18 (24. 3) 9 (12. 2) Race, n (%) 4 (25. 0) Discontinua tion day On treatment (PR) On treatment (CR) First CR/PR 8. 0 (6. 7–NE) Intracranial PFS 57. 0 (21– 83) Female Male 4 (25. 0) Partial response, n (%) Intracranial Do. R Time on treatment: responders and non-responders 8 50. 0 (24. 7– 75. 4) Complete response, n (%) Median, months (95% CI) NTRK-fp (N=74) Median (range) Baseline CNS metastases ‡ (n=16) Intracranial ORR NTRK-fp nonmeasurable disease / <6 months follow-up from start of treatment (n=39) Median, months (95% CI) Intracranial events at data cut-off, n (%) 8. 9 (5. 9– 14. 3) 10 (62. 5) Intracranial ORR derived using RECIST v 1. 1 criteria applied only to CNS lesions. *BICR assessed, RECIST v 1. 1. ‡CNS metastases status determined by BICR. Still on treatment First PD Safety Death • Overall, the safety profile for entrectinib was similar to previous findings, and aligned with those of other TRK-targeted agents 5, 6 Conclusions Discontinua tion day On treatment (PR) On treatment (CR) On treatment (missing) CNS metastases at baseline*, n (%) Yes No 16 (21. 6) / 19 (25. 7) 58 (78. 4) / 55 (74. 3) Prior radiotherapy (RT) of the brain †, n (%) Yes No 11 (68. 8) 5 (31. 2) Time from end of brain RT to first dose ‡, n (%) <2 months >2 months–<6 months ≥ 6 months 5 (45. 5) 4 (36. 4) 2 (18. 2) On treatment (NE) On treatment (PD) On treatment (SD) On treatment (non-CR/PD) First CR/PR Still on treatment First PD Death These data provide additional evidence confirming that entrectinib is associated with clinically meaningful and durable systemic responses in patients with NTRK-fp solid tumors Response rates were independent of the presence or absence of CNS metastases at baseline Entrectinib demonstrated durable, clinically meaningful intracranial activity in patients with CNS metastases at baseline Entrectinib is a potential option to fulfill the need for a CNS-active treatment for patients with NTRK-fp solid tumors and CNS metastases *BICR assessed / INV assessed. †Patients with baseline CNS metastases per BICR. ‡Patients with baseline CNS metastases per BICR and prior brain RT. References Acknowledgments Disclosures 1. Vaishnavi A, et al. Cancer Discov 2014; 2. Amatu A, et al. ESMO Open 2016; 3. Menichincheri M, et al. J Med Chem 2016; 4. Fischer H, et al. Neuro-Oncol 2020; 5. Doebele RC, et al. Lancet Oncol 2020; 6. Hong DS, et al. Lancet Oncol 2020. We thank the patients, their families, and participating study centers. Third-party medical writing assistance, under the direction of the authors, was provided by Alix Biancardi of Gardiner-Caldwell Communications, and was funded by F. Hoffmann-La Roche Ltd. CR is a speaker for MSD and Astra. Zeneca; a scientific advisor for Mylan, Oncompass, and Astra. Zeneca; has research collaborations with Biomarkers and Guardant Health; and has undertaken advisory board activity for Archer, Inivata and MD Serono. For co-author disclosures please refer to the abstract: https: //meetinglibrary. asco. org/
- Slides: 24