Organozinc Reagent Iodomethylzinc iodide formed by reaction of

Organozinc Reagent

Iodomethylzinc iodide formed by reaction of diiodomethane with zinc that has been coated with copper (called zinc-copper couple) CH 2 I 2 + Zn Cu ICH 2 Zn. I reacts with alkenes to form cyclopropanes reaction with alkenes is called the Simmons-Smith reaction
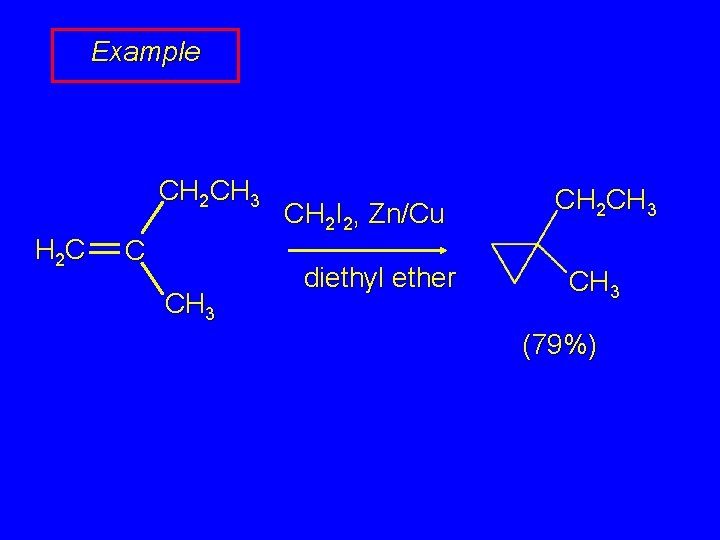
Example CH 2 CH 3 H 2 C C CH 3 CH 2 I 2, Zn/Cu diethyl ether CH 2 CH 3 (79%)

Stereospecific syn-addition CH 3 CH 2 CH 3 C H CH 2 I 2, Zn/Cu CH 3 CH 2 H C H diethyl ether CH 2 CH 3 H
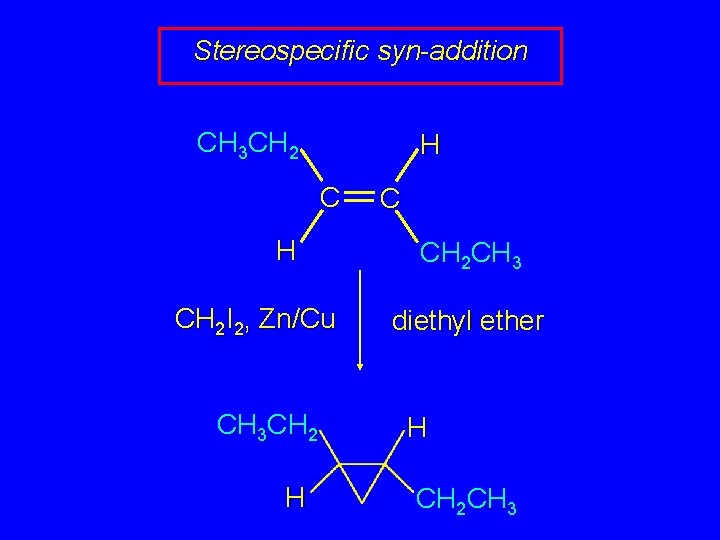
Stereospecific syn-addition CH 3 CH 2 H CH 2 I 2, Zn/Cu CH 3 CH 2 H C CH 2 CH 3 diethyl ether H CH 2 CH 3

Ziegler-Natta Catalysis of Alkene Polymerization The catalysts used in coordination polymerization are transition-metal organic compounds.

Ziegler-Natta Catalysts A typical Ziegler-Natta catalyst is a combination of Ti. Cl 4 and (CH 3 CH 2)2 Al. Cl, or Ti. Cl 3 and (CH 3 CH 2)3 Al. Many Ziegler-Natta catalyst combinations include a metallocene.
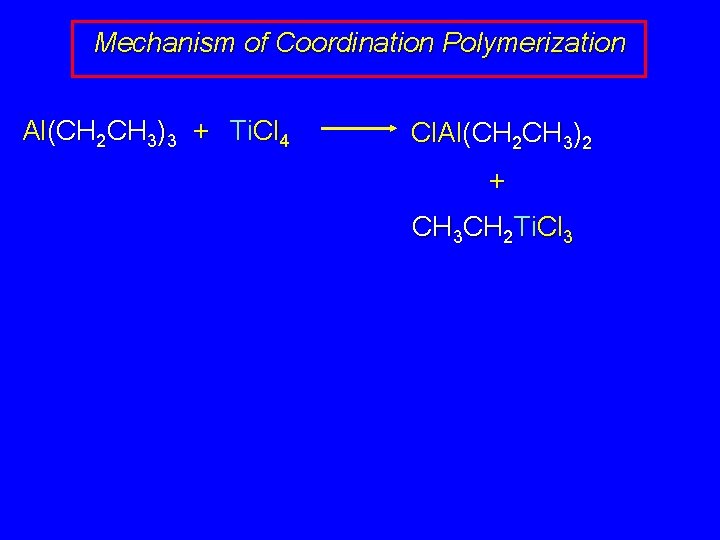
Mechanism of Coordination Polymerization Al(CH 2 CH 3)3 + Ti. Cl 4 Cl. Al(CH 2 CH 3)2 + CH 3 CH 2 Ti. Cl 3
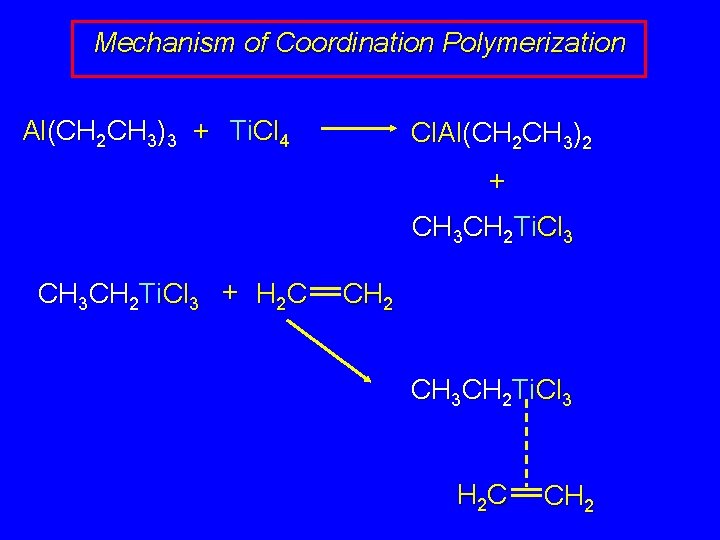
Mechanism of Coordination Polymerization Al(CH 2 CH 3)3 + Ti. Cl 4 Cl. Al(CH 2 CH 3)2 + CH 3 CH 2 Ti. Cl 3 + H 2 C CH 2 CH 3 CH 2 Ti. Cl 3 H 2 C CH 2
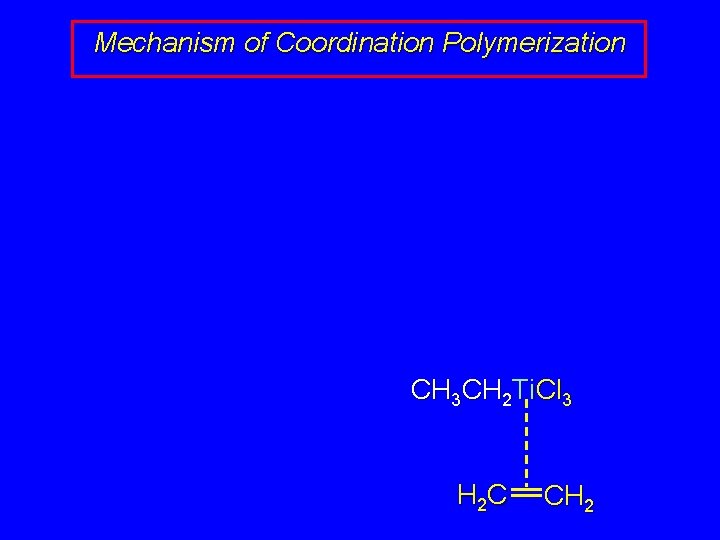
Mechanism of Coordination Polymerization CH 3 CH 2 Ti. Cl 3 H 2 C CH 2
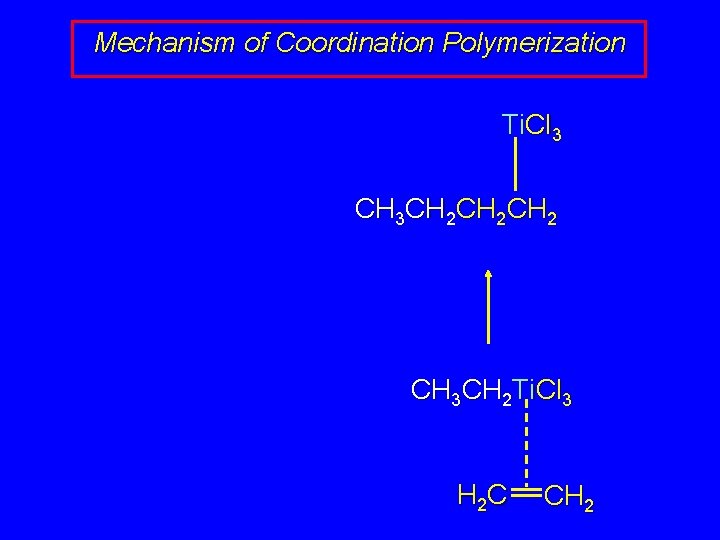
Mechanism of Coordination Polymerization Ti. Cl 3 CH 3 CH 2 CH 2 CH 3 CH 2 Ti. Cl 3 H 2 C CH 2
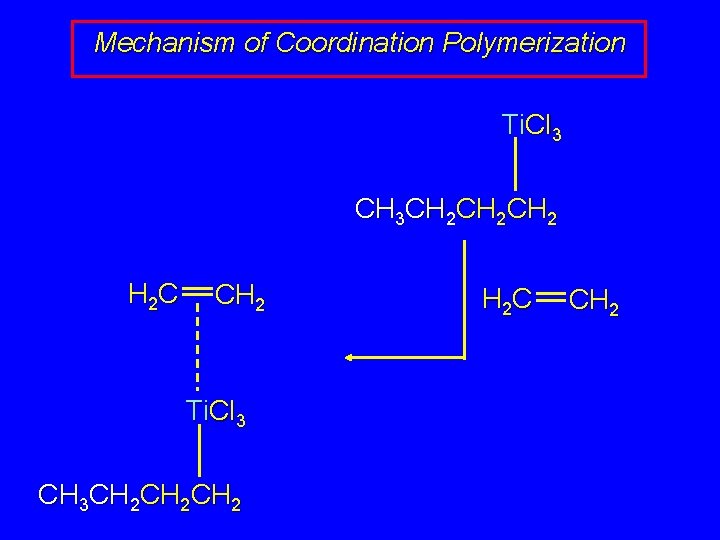
Mechanism of Coordination Polymerization Ti. Cl 3 CH 3 CH 2 CH 2 CH 2 H 2 C CH 2
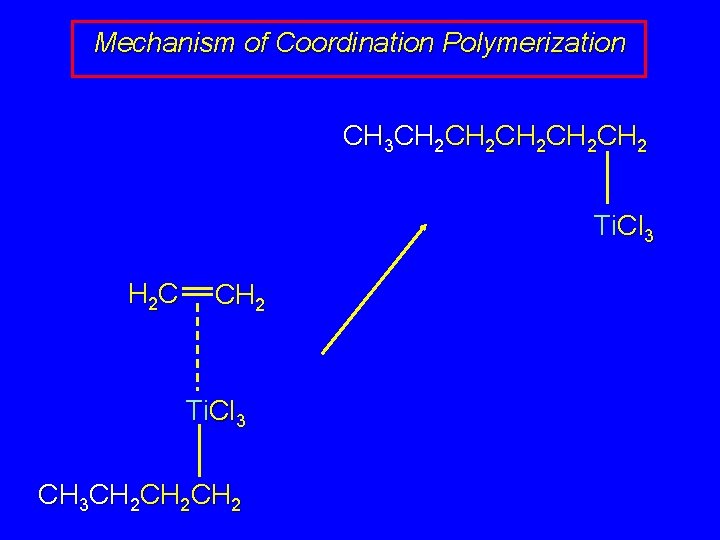
Mechanism of Coordination Polymerization CH 3 CH 2 CH 2 CH 2 Ti. Cl 3 H 2 C CH 2 Ti. Cl 3 CH 3 CH 2 CH 2
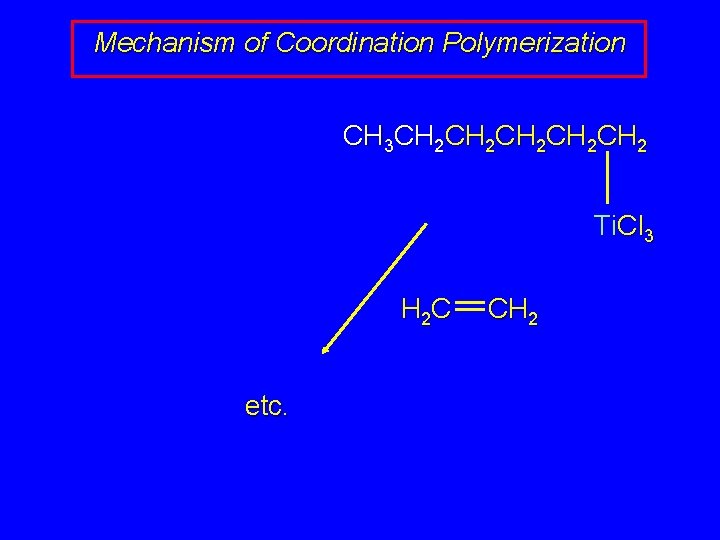
Mechanism of Coordination Polymerization CH 3 CH 2 CH 2 CH 2 Ti. Cl 3 H 2 C etc. CH 2
- Slides: 14