Organization of The Periodic Table Nucleus Center of
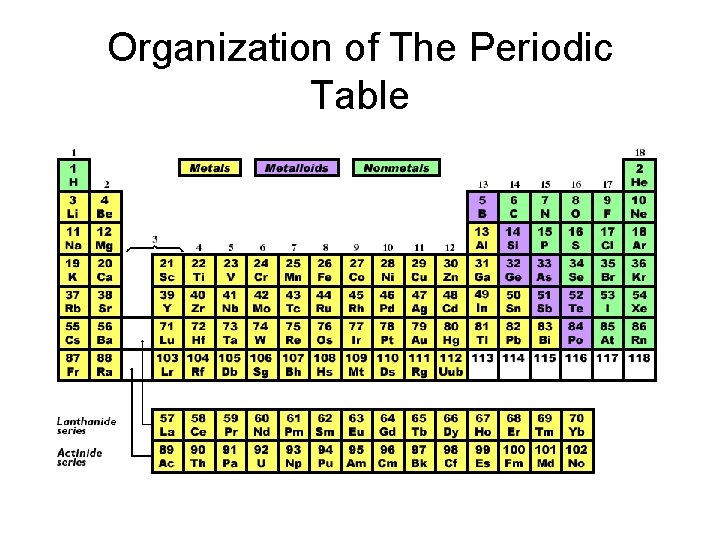
Organization of The Periodic Table
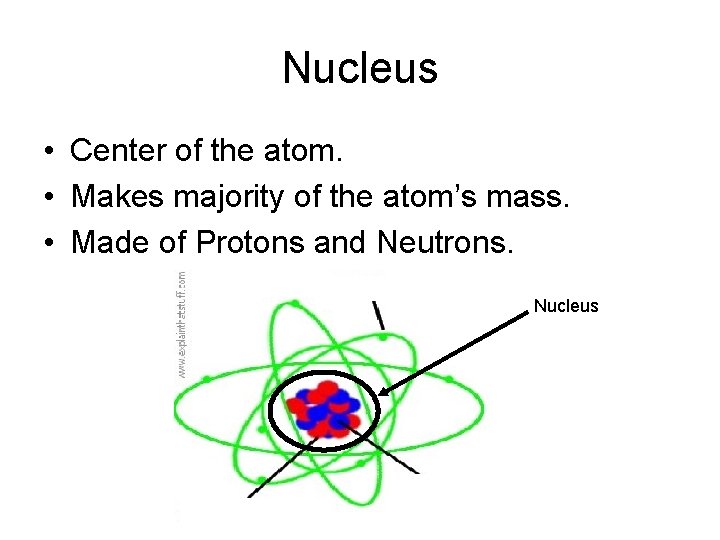
Nucleus • Center of the atom. • Makes majority of the atom’s mass. • Made of Protons and Neutrons. Nucleus
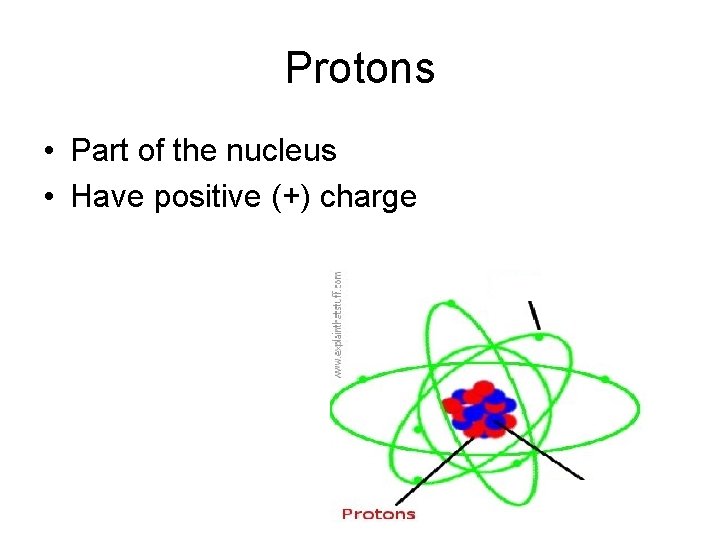
Protons • Part of the nucleus • Have positive (+) charge

Neutrons • Part of the nucleus • Have neutral charge
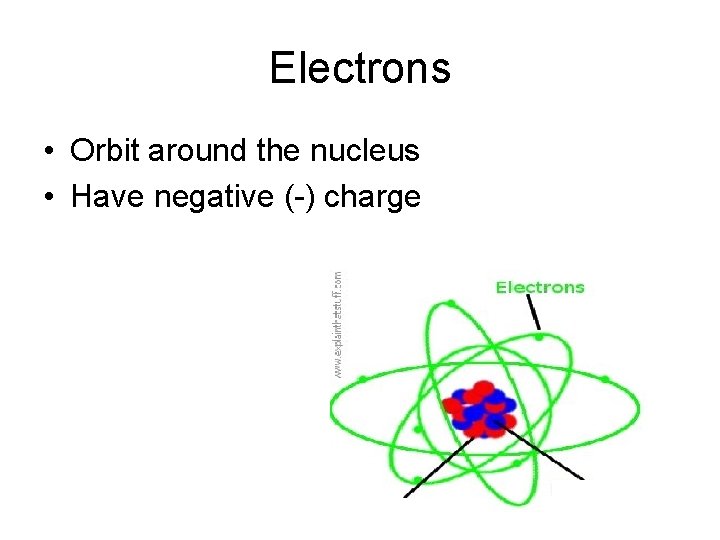
Electrons • Orbit around the nucleus • Have negative (-) charge

Atomic Number • Definition: the number of protons in the nucleus • Tells us the identity of the element. Every element is defined by the number of protons in its nucleus

Isotopes • Atoms with the same number of protons but different numbers on neutrons • Example: Carbon So you can change the number of neutrons and the element still maintains its identity

Questions • 1) What particles make up an atom? • 2) What are the charges on these particles? • 3) What particles make up the nucleus?

Questions • 1) What is Oxygen’s atomic number? • 2) How many protons does Oxygen have? • 3) If I have 4 protons what element am I? • 4) If I have 4 neutrons what element am I?
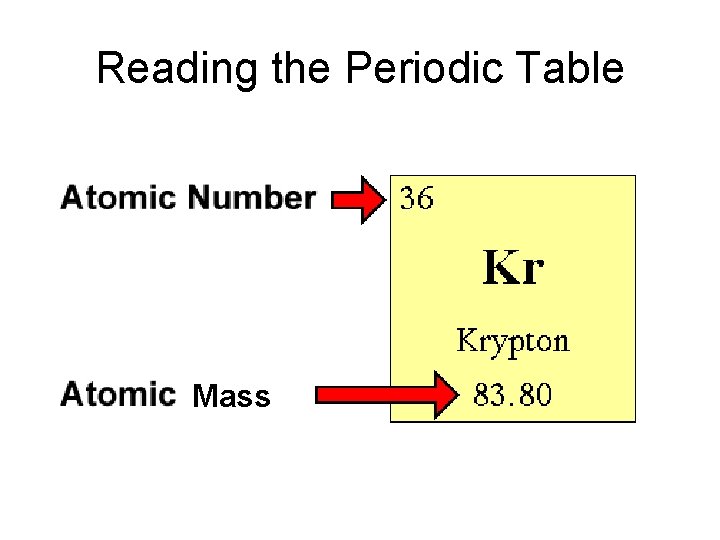
Reading the Periodic Table Mass
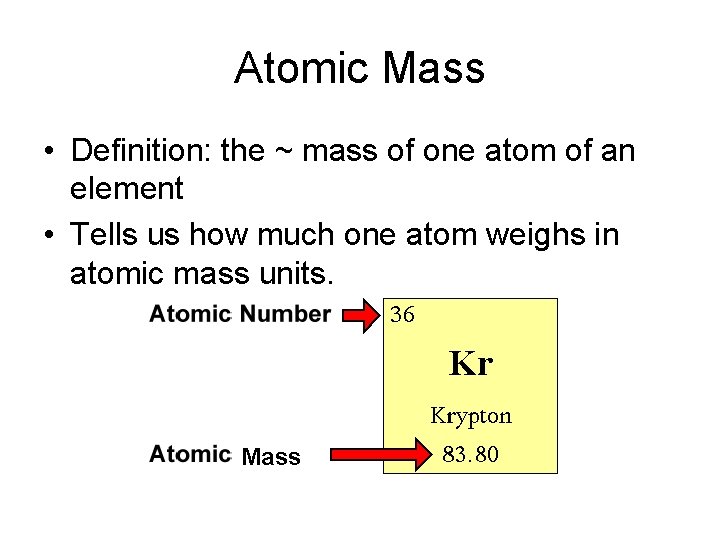
Atomic Mass • Definition: the ~ mass of one atom of an element • Tells us how much one atom weighs in atomic mass units. Mass
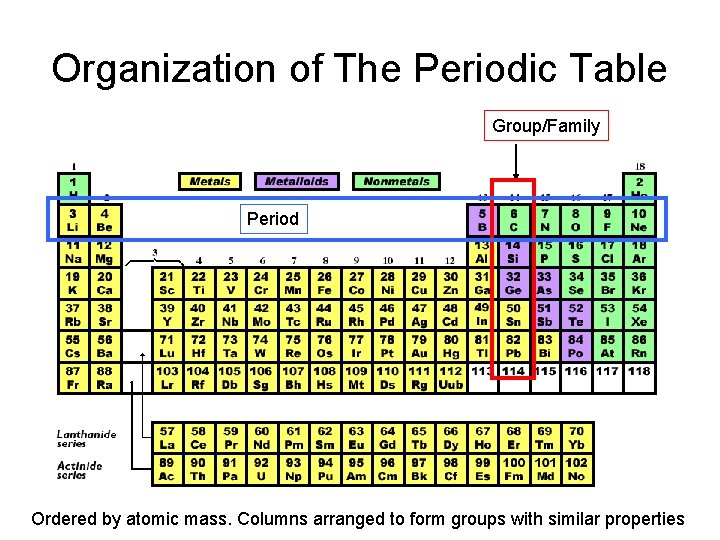
Organization of The Periodic Table Group/Family Period Ordered by atomic mass. Columns arranged to form groups with similar properties

Assessment • Which element has the greatest atomic mass? • Lithium • Sodium • Potassium • Rubidium

Reactivity • The ease and speed with which an element combines, or reacts, with other elements or compounds Pure sodium reacts explosively with air
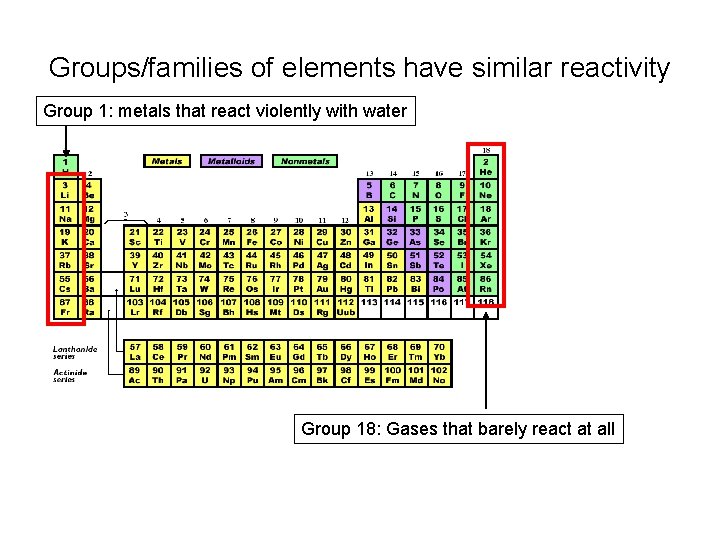
Groups/families of elements have similar reactivity Group 1: metals that react violently with water Group 18: Gases that barely react at all

Atoms have neutral charge • # of Protons (+) = # of Electrons (-) = 0 charge • Example: Hydrogen • How many electrons does C have?

Take home message • The properties of an element can be predicted from its location on the Periodic Table
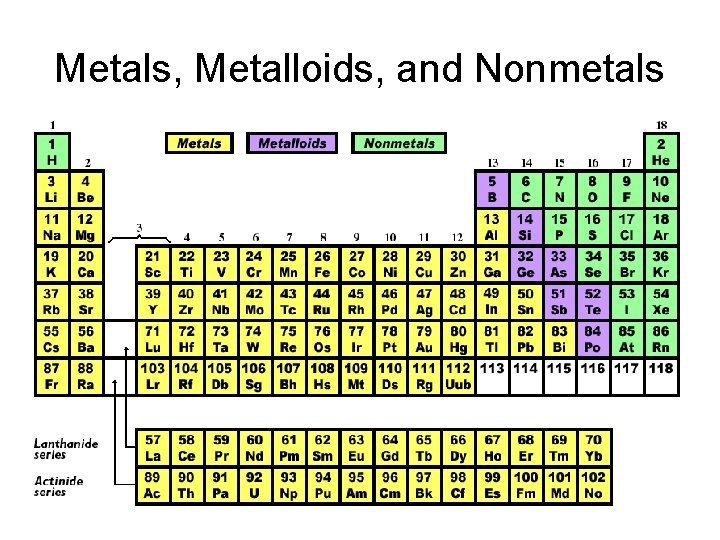
Metals, Metalloids, and Nonmetals

Metals • Shiny • Solids (at room temp) • Malleable – can be hammered flat • Ductile – can be pulled into wire • High Conductivity – ability to transfer heat or electricity to another object
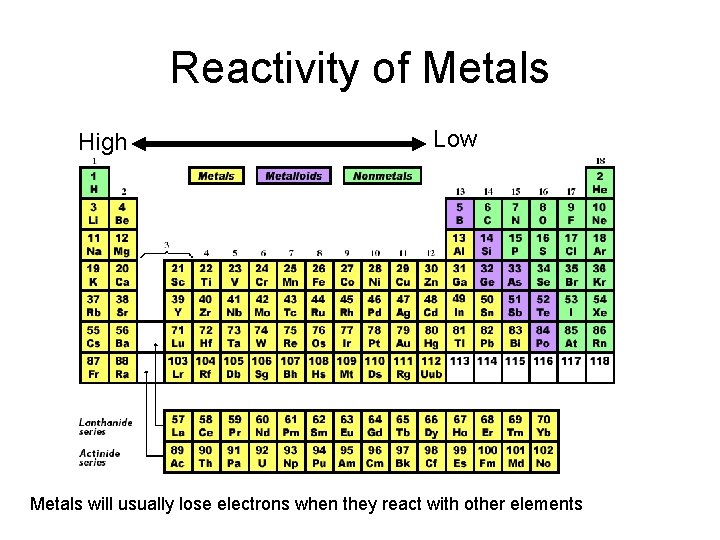
Reactivity of Metals High Low Metals will usually lose electrons when they react with other elements

Nonmetals • Opposite of properties of metals • Not shiny • Poor conductors • Mostly gases (at room temperature) • Solids are brittle Sulfur
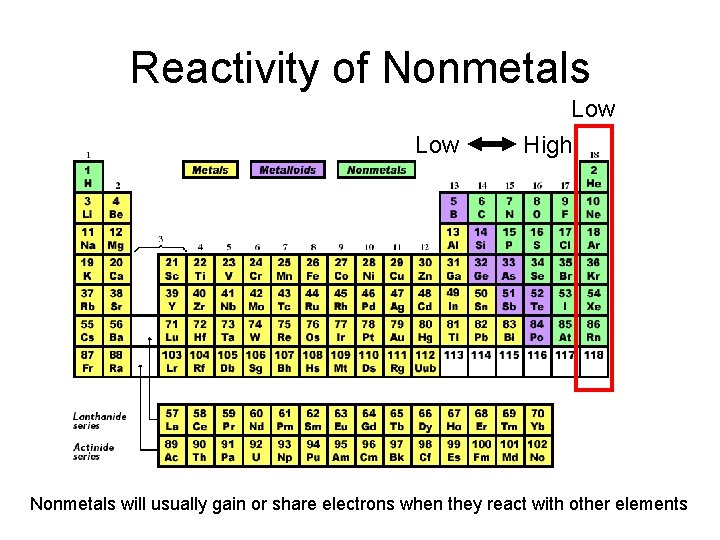
Reactivity of Nonmetals Low High Nonmetals will usually gain or share electrons when they react with other elements

Some Important Nonmetals • Carbon – important element for making up living organisms • Noble Gases – group 18 – very nonreactive. Have full outer shells.

Metalloids • Inbetween metals and nonmetals • Solids (at room temp) • brittle and hard • Semiconductors – can conduct electricity under some conditions but not others. – very important for computer chips • Most common example – Silicon – in sand glass

Assessment • The atomic number is the number of – valence electrons. – neutrons. – protons in the nucleus. – electrons in the nucleus.

Assessment • The periodic table is a chart of the elements that shows the repeating pattern of their – energies. – properties. – element symbols. – names.

Assessment • Which piece of information cannot be found in a square on the periodic table? – Atomic mass. – Chemical symbol. – Atomic number. – Number of neutrons.

Assessment • Which side of the periodic table contains most of the nonmetals? – Left side. – Right side. – Middle.

Assessment • Which is not a property of nonmetals? – Brittle. – Nonmalleable. – High conductivity. – Most are gases at room temperature
- Slides: 29