Organic Reactions b Base hydrolysis of esters saponification

Organic Reactions (b) Base hydrolysis of esters (saponification)
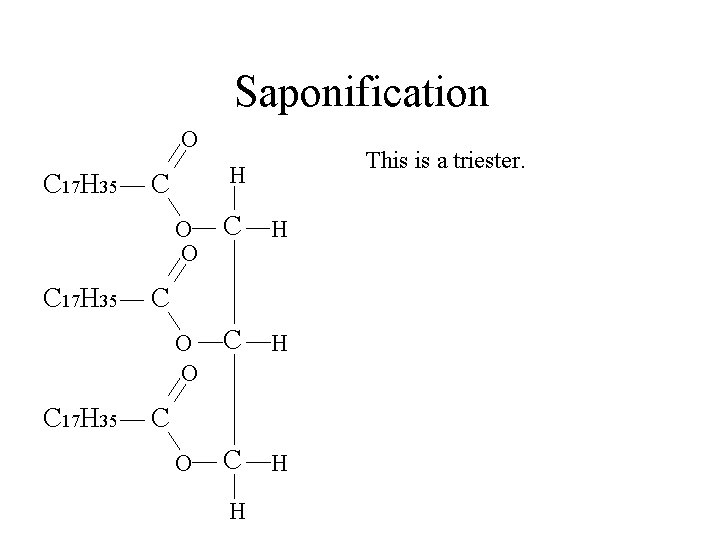
Saponification O C 17 H 35 This is a triester. H C O O C H C C H
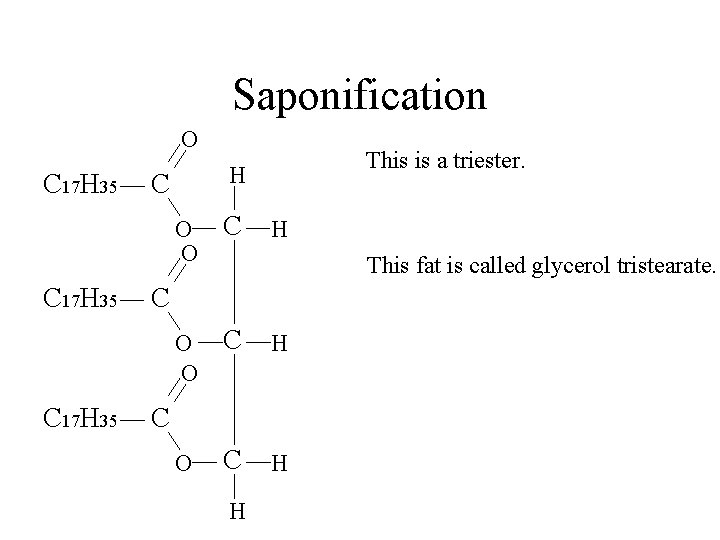
Saponification O C 17 H 35 This is a triester. H C O O C H H This fat is called glycerol tristearate. C C H
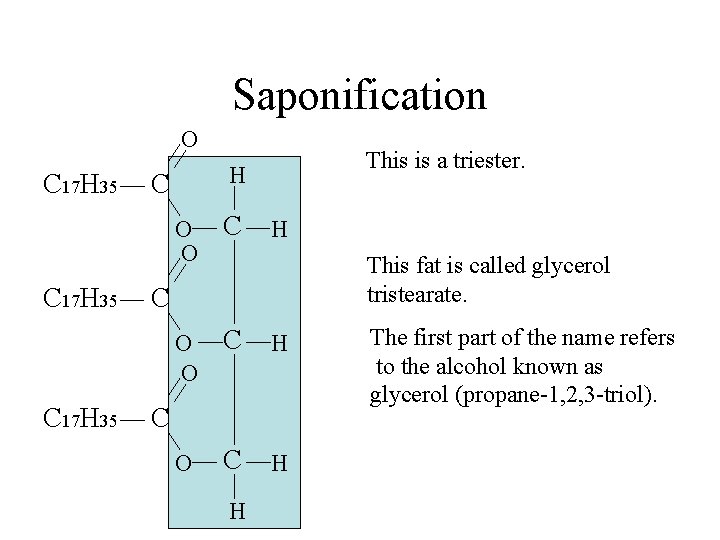
Saponification O C 17 H 35 This is a triester. H C O O C H H This fat is called glycerol tristearate. C C H The first part of the name refers to the alcohol known as glycerol (propane-1, 2, 3 -triol).

Saponification O C 17 H 35 This is a triester. H C O O C H This fat is called glycerol tristearate. C H C O Commonly known as fat. C H H The first part of the name refers to the alcohol known as glycerol (propane-1, 2, 3 -triol). The second part refers to the three fatty acid molecules called stearic acid.
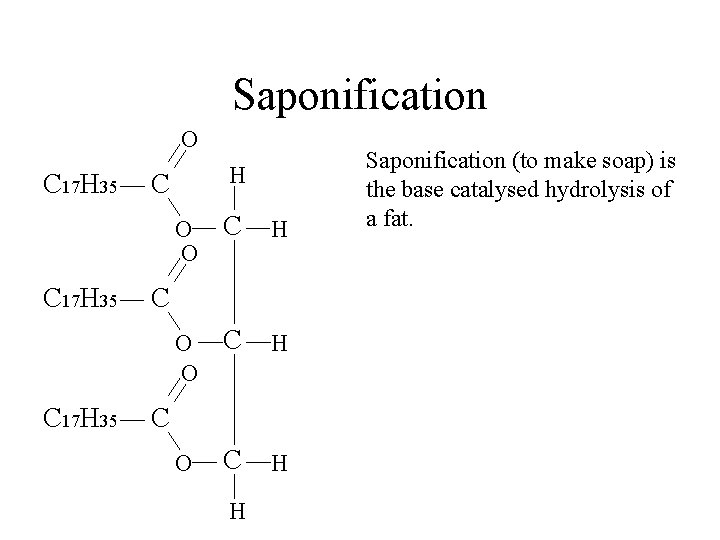
Saponification O C 17 H 35 H C O O C H C C H Saponification (to make soap) is the base catalysed hydrolysis of a fat.
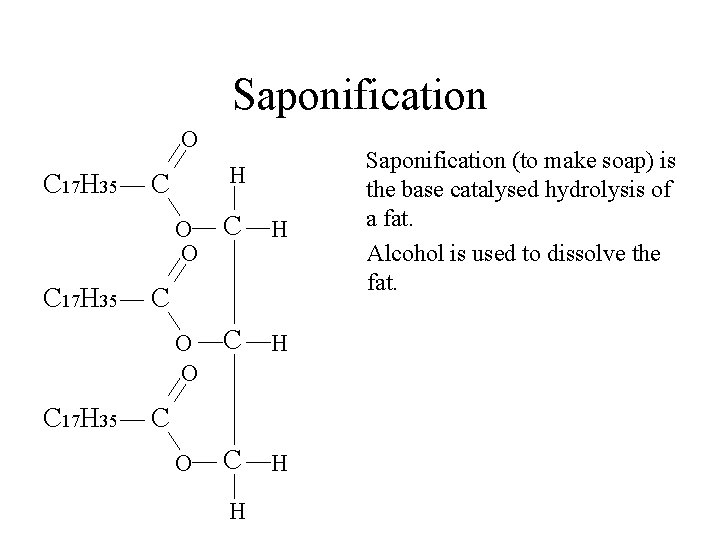
Saponification O C 17 H 35 H C O O C H C C H Saponification (to make soap) is the base catalysed hydrolysis of a fat. Alcohol is used to dissolve the fat.
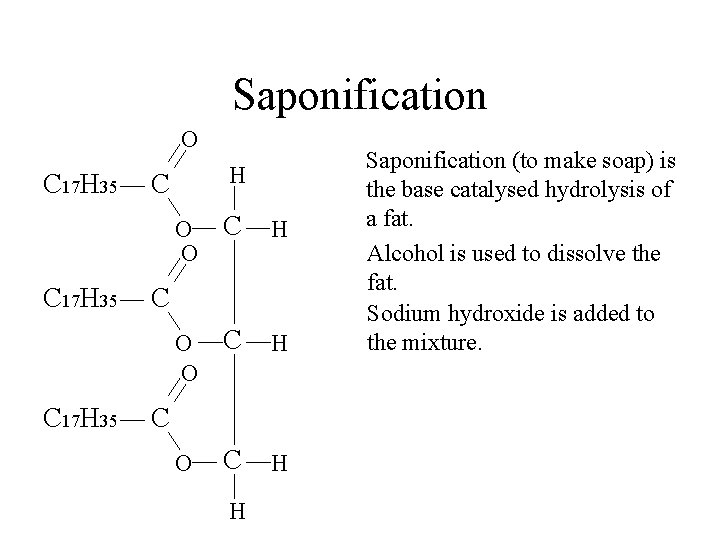
Saponification O C 17 H 35 H C O O C H C C H Saponification (to make soap) is the base catalysed hydrolysis of a fat. Alcohol is used to dissolve the fat. Sodium hydroxide is added to the mixture.
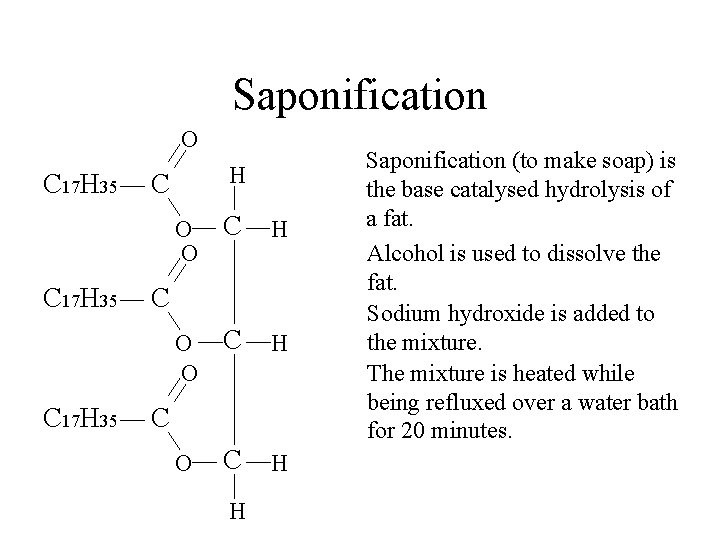
Saponification O C 17 H 35 H C O O C H C C H Saponification (to make soap) is the base catalysed hydrolysis of a fat. Alcohol is used to dissolve the fat. Sodium hydroxide is added to the mixture. The mixture is heated while being refluxed over a water bath for 20 minutes.
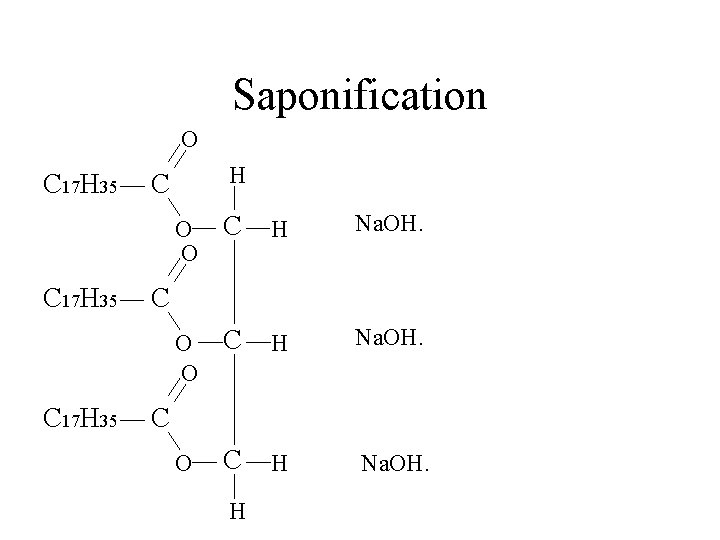
Saponification O C 17 H 35 H C O O C H Na. OH. C C H
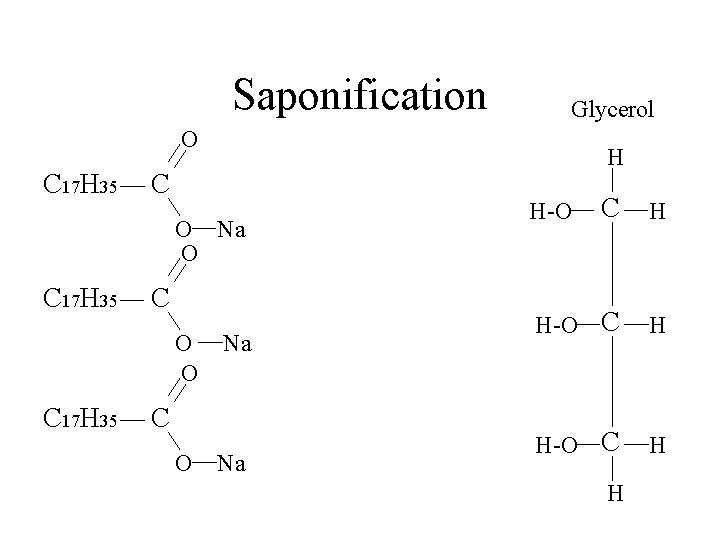
Saponification Glycerol O C 17 H 35 H C O Na O C 17 H 35 C O O C 17 H 35 Na C O Na C H H-O H
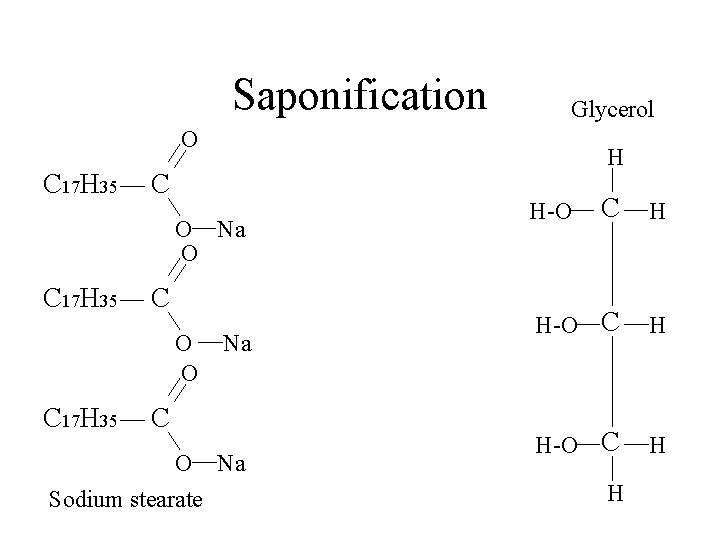
Saponification Glycerol O C 17 H 35 H C O Na O C 17 H 35 C O O C 17 H 35 Na C O Na Sodium stearate C H H-O H
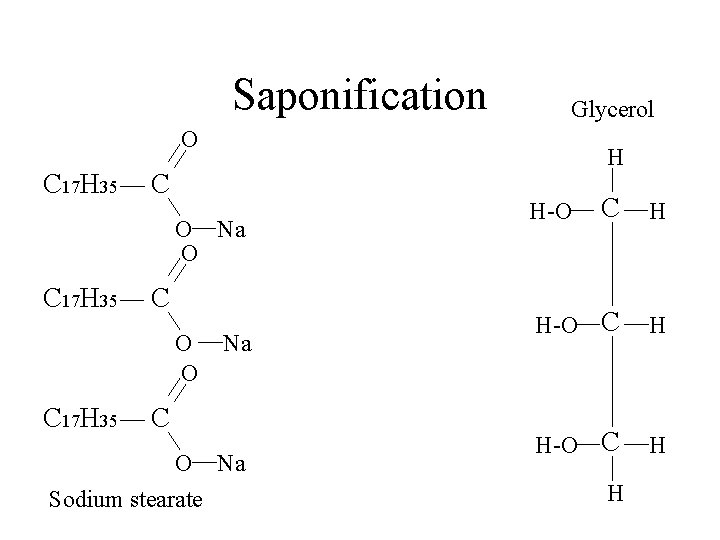
Saponification Glycerol O C 17 H 35 H C O Na O C 17 H 35 C O O C 17 H 35 Na C O Na Sodium stearate C H H-O H
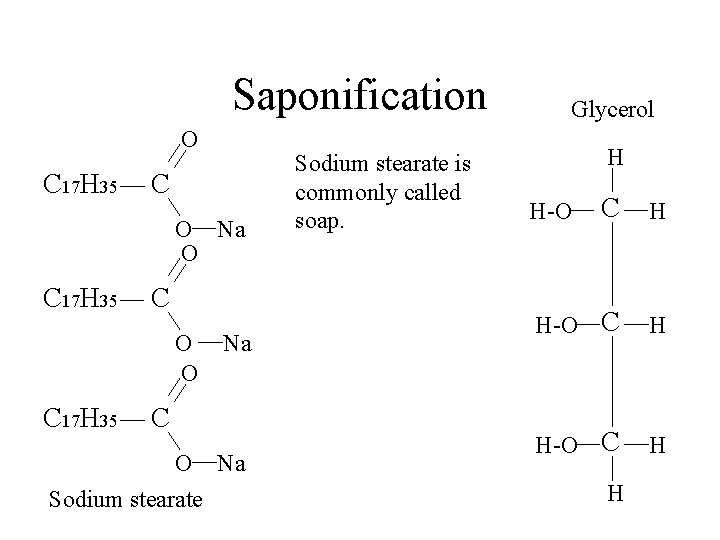
Saponification O C 17 H 35 C O Na O C 17 H 35 C O O C 17 H 35 Na C O Na Sodium stearate is commonly called soap. Glycerol H C H H-O H
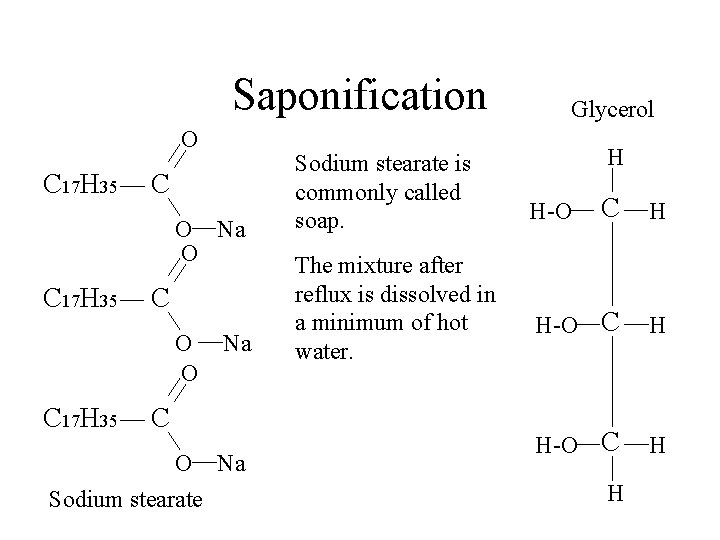
Saponification O C 17 H 35 C O Na O C 17 H 35 C O O C 17 H 35 Na C O Na Sodium stearate is commonly called soap. The mixture after reflux is dissolved in a minimum of hot water. Glycerol H C H H-O H
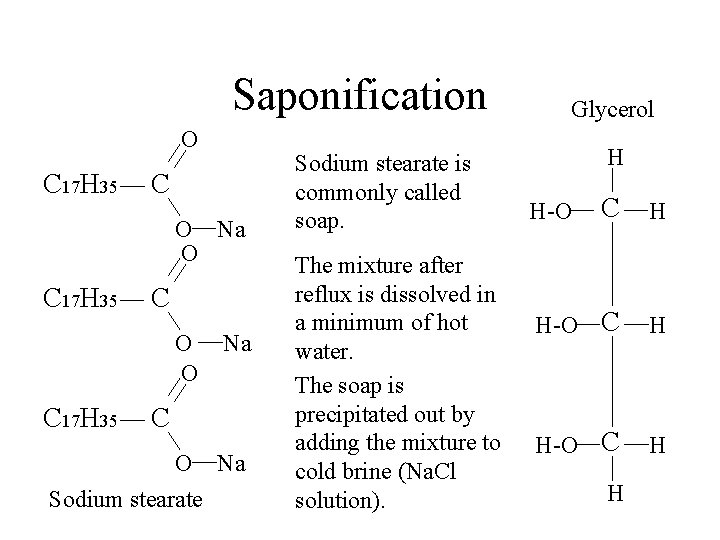
Saponification O C 17 H 35 C O Na O C 17 H 35 C O O C 17 H 35 Na C O Na Sodium stearate is commonly called soap. The mixture after reflux is dissolved in a minimum of hot water. The soap is precipitated out by adding the mixture to cold brine (Na. Cl solution). Glycerol H C H H-O H
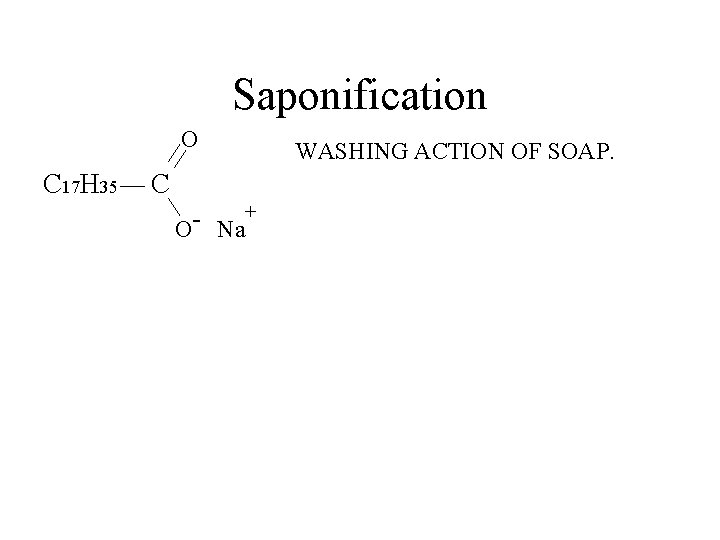
Saponification O C 17 H 35 C + O Na WASHING ACTION OF SOAP.
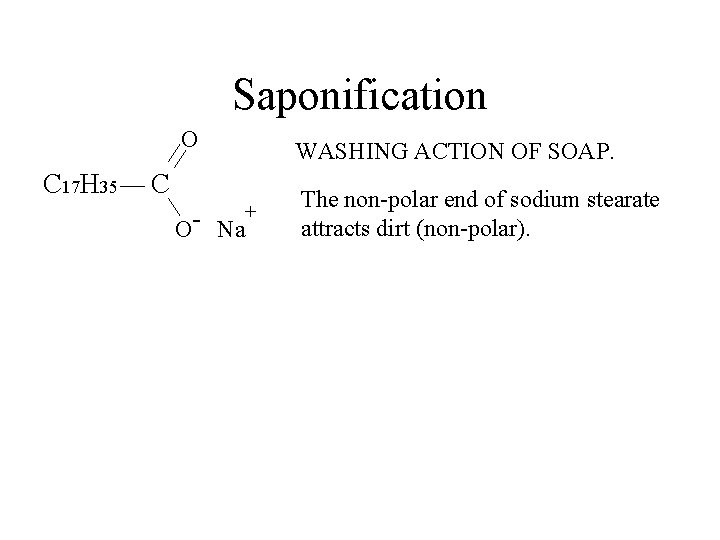
Saponification O C 17 H 35 WASHING ACTION OF SOAP. C + O- Na The non-polar end of sodium stearate attracts dirt (non-polar).
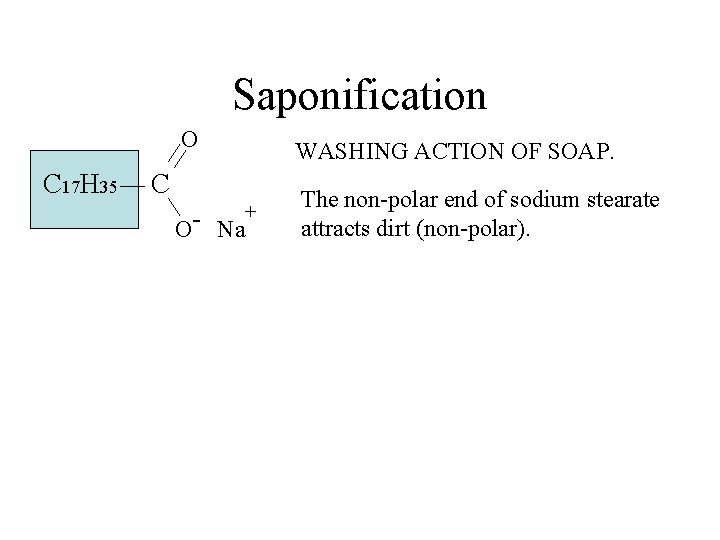
Saponification O C 17 H 35 WASHING ACTION OF SOAP. C + O- Na The non-polar end of sodium stearate attracts dirt (non-polar).

Saponification O C 17 H 35 WASHING ACTION OF SOAP. C + O- Na The non-polar end of sodium stearate attracts dirt (non-polar). The ionic end of sodium stearate attracts water (polar).

Saponification O C 17 H 35 WASHING ACTION OF SOAP. C + O- Na The non-polar end of sodium stearate attracts dirt (non-polar). The ionic end of sodium stearate attracts water (polar). The sodium stearate molecules surround the dirt in three dimensions and pull the dirt off surfaces.

Saponification O C 17 H 35 WASHING ACTION OF SOAP. C + O- Na The non-polar end of sodium stearate attracts dirt (non-polar). The ionic end of sodium stearate attracts water (polar). The sodium stearate molecules surround the dirt in three dimensions and pull the dirt off surfaces.
- Slides: 22