Organic Polymer Chemistry 29 1 Some Definitions u


























- Slides: 64

Organic Polymer Chemistry 29 -1

Some Definitions u Polymer: From the Greek, poly + meros, meros many parts. • Any long-chain molecule synthesized by bonding together single parts called monomers. u Monomer: Monomer From the Greek, mono + meros, meros single part. • The simplest nonredundant unit from which a polymer is synthesized. u Plastic: A polymer that can be molded when hot and retains its shape when cooled. 29 -2

Continued u Thermoplastic: A polymer that can be melted and molded into a shape that is retained when it is cooled. u Thermoset plastic: A polymer that can be molded when it is first prepared but, once it is cooled, hardens irreversibly and cannot be remelted. 29 -3

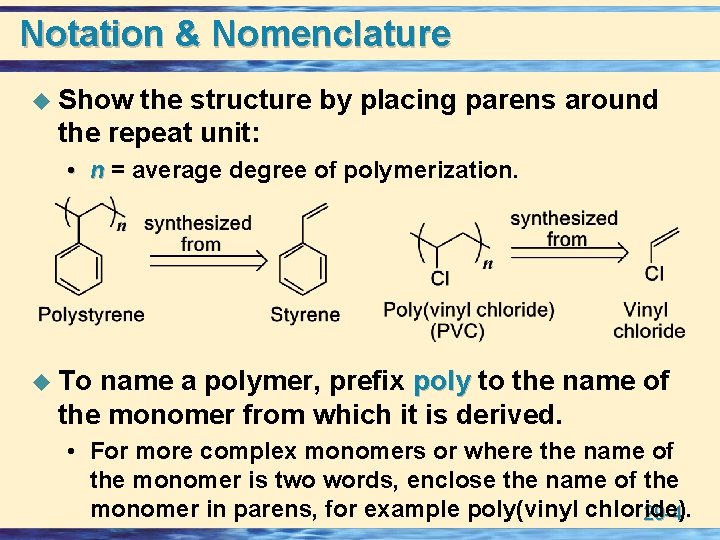
Notation & Nomenclature u Show the structure by placing parens around the repeat unit: • n = average degree of polymerization. u To name a polymer, prefix poly to the name of the monomer from which it is derived. • For more complex monomers or where the name of the monomer is two words, enclose the name of the monomer in parens, for example poly(vinyl chloride). 29 -4

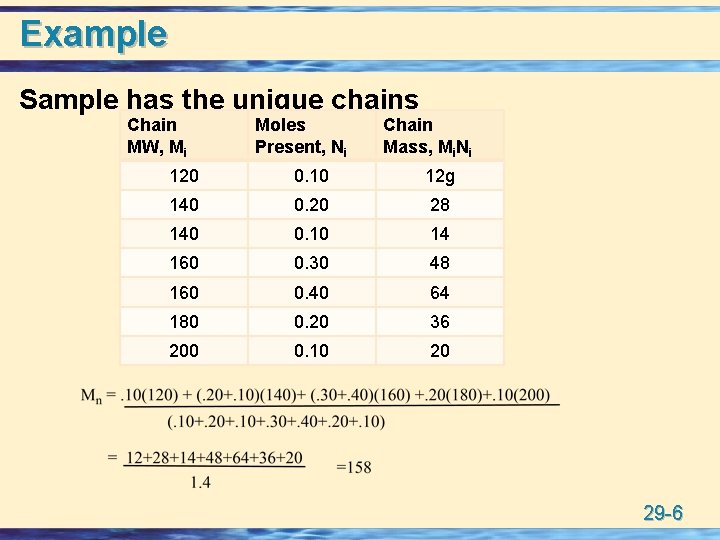
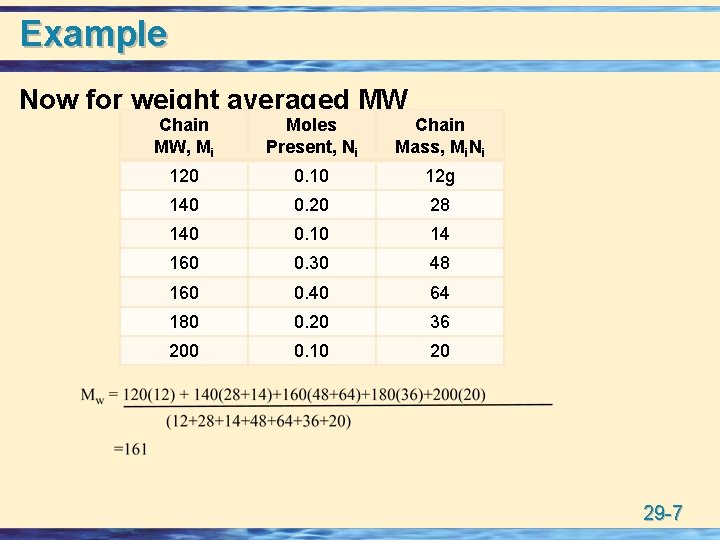
Molecular Weight u All polymers are mixtures of individual polymer molecules of variable MWs. • number average MW: Count the number of chains of a particular MW (Ni moles), multiply each number by the MW (Mi), sum these values, and divide by the total number of polymer chains. • weight average MW: 29 -5

Example Sample has the unique chains Chain MW, Mi Moles Present, Ni Chain Mass, Mi. Ni 120 0. 10 12 g 140 0. 20 28 140 0. 10 14 160 0. 30 48 160 0. 40 64 180 0. 20 36 200 0. 10 20 29 -6

Example Now for weight averaged MW Chain MW, Mi Moles Present, Ni Chain Mass, Mi. Ni 120 0. 10 12 g 140 0. 20 28 140 0. 10 14 160 0. 30 160 0. 40 180 0. 20 36 200 0. 10 20 140 48 64 29 -7

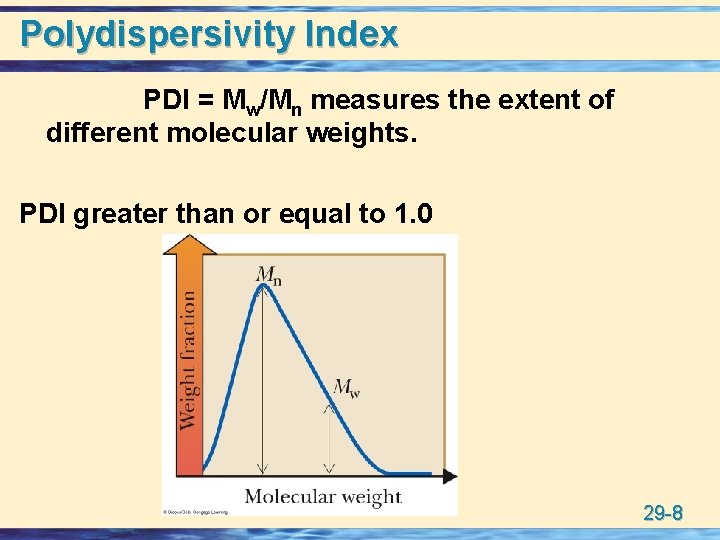
Polydispersivity Index PDI = Mw/Mn measures the extent of different molecular weights. PDI greater than or equal to 1. 0 29 -8

Morphology u Polymers tend to crystallize as they precipitate or are cooled from a melt. u Acting to inhibit crystallization are that polymers are large molecules. Complicated and irregular shapes prevent efficient packing into ordered structures. u As a result, polymers in the solid state tend to be composed of • ordered crystalline domains • disordered amorphous domains 29 -9

Morphology: Crysatline u High degrees of crystallinity are found in polymers with • regular, compact structures • strong intermolecular forces, such as hydrogen bonds and dipolar interactions. u As the degree of crystallinity increases, the polymer becomes more opaque due to scattering of light by the crystalline regions. u Melt transition temperature, Tm: The temperature at which crystalline regions melt. • As the degree of crystallinity increases, Tm increases. 29 -10

Morphology: Amorphous u Highly amorphous polymers are sometimes referred to as glassy polymers. • Lacking crystalline domains that scatter light, amorphous polymers are transparent. • They are weaker polymers, both in terms of their greater flexibility and smaller mechanical strength. • On heating, amorphous polymers are transformed from a hard glassy state to a soft, flexible, rubbery state. u Glass transition temperature, Tg: The temperature at which a polymer undergoes a transition from a hard glass to a rubbery solid. Put polystyrene cup in boiling water. 29 -11

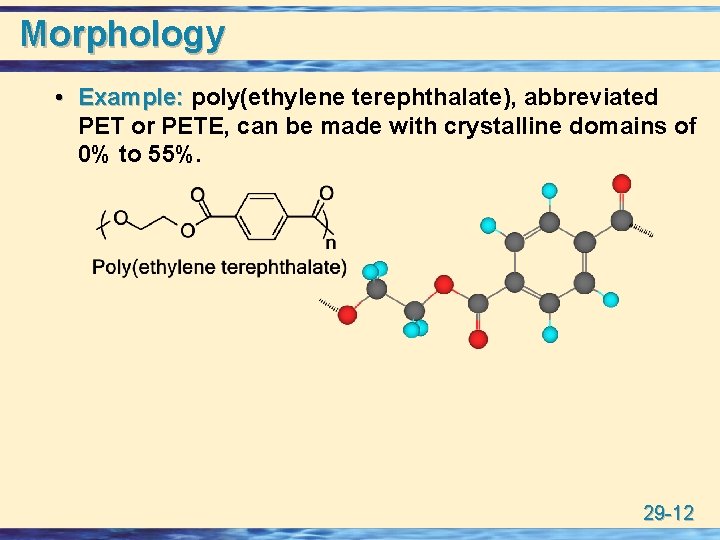
Morphology • Example: poly(ethylene terephthalate), abbreviated PET or PETE, can be made with crystalline domains of 0% to 55%. 29 -12

Morphology u Completely amorphous PET is formed by quickly cooling the melt. • PET with a low degree of crystallinity is used for plastic beverage bottles. u More crystallline formed by slow cooling, more molecular diffusion occurs and crystalline domains form as the chains become more ordered. • PET with a high degree of crystallinity can be drawn into textile fibers and tire cords. 29 -13

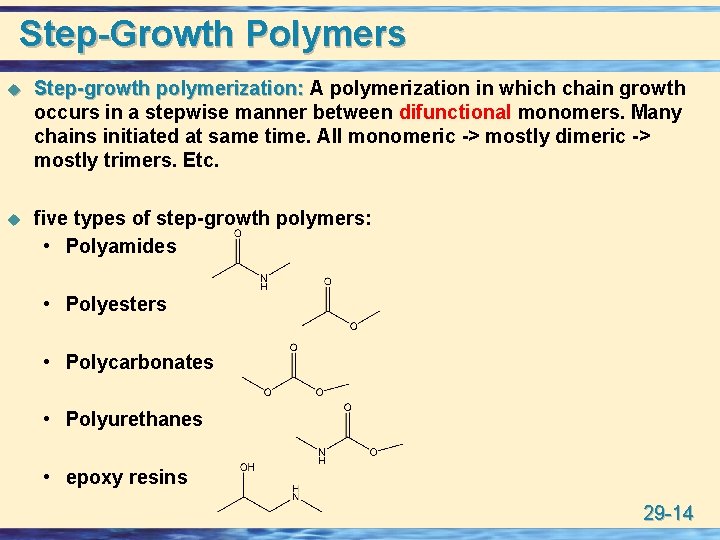
Step-Growth Polymers u Step-growth polymerization: A polymerization in which chain growth occurs in a stepwise manner between difunctional monomers. Many chains initiated at same time. All monomeric -> mostly dimeric -> mostly trimers. Etc. u five types of step-growth polymers: • Polyamides • Polyesters • Polycarbonates • Polyurethanes • epoxy resins 29 -14

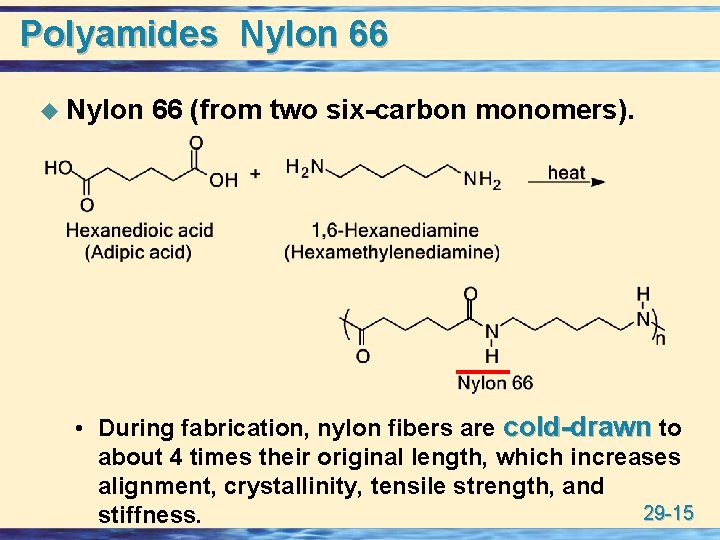
Polyamides Nylon 66 u Nylon 66 (from two six-carbon monomers). • During fabrication, nylon fibers are cold-drawn to about 4 times their original length, which increases alignment, crystallinity, tensile strength, and 29 -15 stiffness.

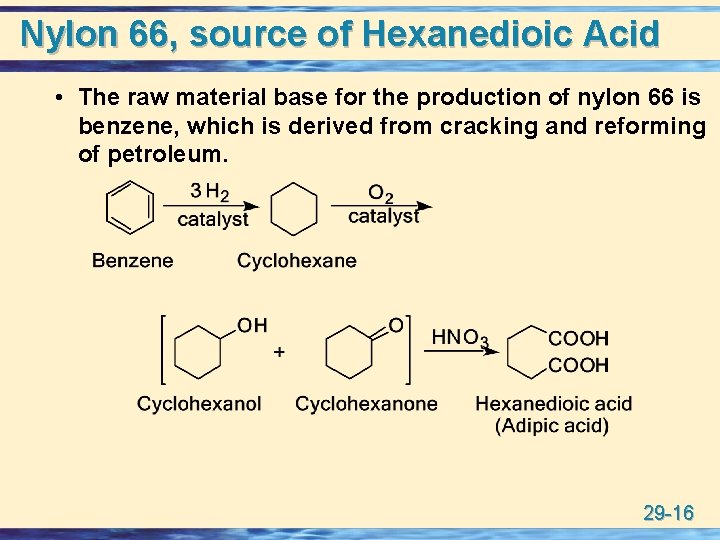
Nylon 66, source of Hexanedioic Acid • The raw material base for the production of nylon 66 is benzene, which is derived from cracking and reforming of petroleum. 29 -16

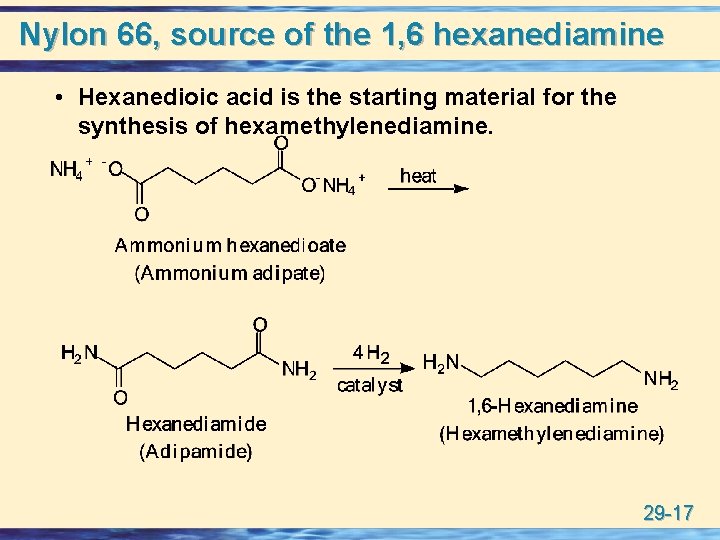
Nylon 66, source of the 1, 6 hexanediamine • Hexanedioic acid is the starting material for the synthesis of hexamethylenediamine. 29 -17

Polyamides, Nylon 6 u Nylons are a family of polymers, the two most widely used of which are nylon 66 and nylon 6. • Nylon 6 is synthesized from a six-carbon monomer. • Nylon 6 is fabricated into fibers, brush bristles, highimpact moldings, and tire cords. 29 -18

Polyamides, Kevlar u Kevlar is a polyaromatic amide (an aramid). • Cables of Kevlar are as strong as cables of steel, but only about 20% the weight. • Kevlar fabric is used for bulletproof vests, jackets, and raincoats. 29 -19

Polyesters, PET u Poly(ethylene terephthalate), abbreviated PET or PETE, is fabricated into Dacron fibers, Mylar films, and plastic beverage containers. 29 -20

PET, source of glycol and terepthalic acid • Ethylene glycol is obtained by air oxidation of ethylene followed by hydrolysis to the glycol. • Terephthalic acid is obtained by catalyzed air oxidation of petroleum-derived p-xylene. 29 -21

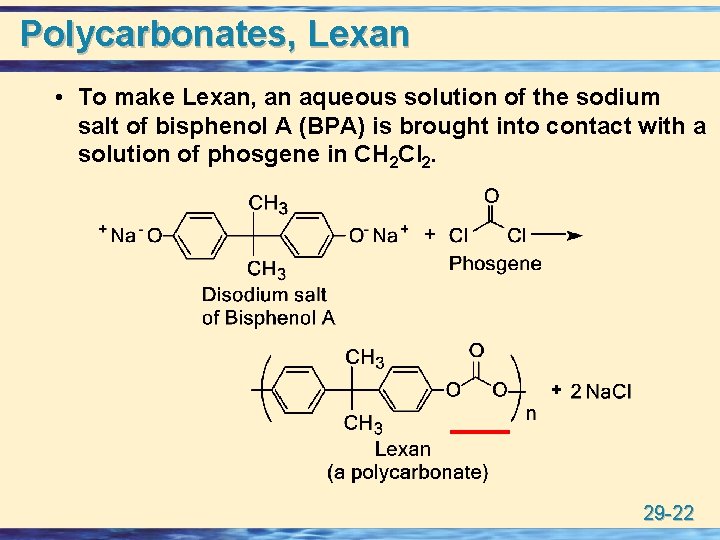
Polycarbonates, Lexan • To make Lexan, an aqueous solution of the sodium salt of bisphenol A (BPA) is brought into contact with a solution of phosgene in CH 2 Cl 2. 29 -22

Phase transfer catalysis. The sodium salt of bisphenol A is water soluble while the phosgene is not. Immiscible. No reaction. Solution: Phase transfer catalysis. NBu 4+ and a negative ion can go back and forth between the two phases. u NBu 4+ brings bisphenolate ion into organic phase u Reaction occurs with phosgene producing Cl u NBu 4+ brings chloride ion into water phase. 29 -23

Polycarbonates, Lexan u Lexan is a tough transparent polymer with high impact and tensile strengths and retains its shape over a wide temperature range. • It is used in sporting equipment, such as bicycle, football, and snowmobile helmets as well as hockey and baseball catcher’s masks. • It is also used in the manufacture of safety and unbreakable windows. 29 -24

Polyurethanes u. A urethane, urethane or carbamate, is an ester of carbamic acid, H 2 NCH 2 COOH. • They are most commonly prepared by treatment of an isocyanate with an alcohol. Addition to the N=C bond u Polyurethanes consist of flexible polyester or polyether units alternating with rigid urethane units. • The rigid urethane units are derived from a diisocyanate. 29 -25

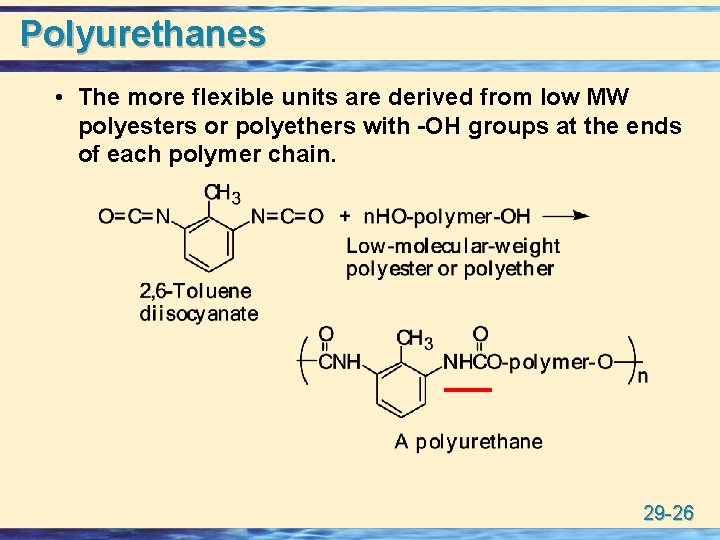
Polyurethanes • The more flexible units are derived from low MW polyesters or polyethers with -OH groups at the ends of each polymer chain. 29 -26

Epoxy Resins u Epoxy resins are materials prepared by a polymerization in which one monomer contains at least two epoxy groups. • Epoxy resins are produced in forms ranging from lowviscosity liquids to high-melting solids. 29 -27

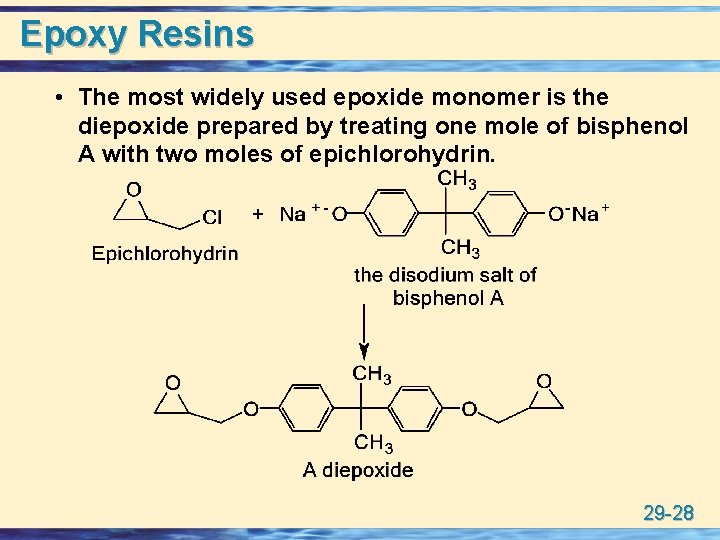
Epoxy Resins • The most widely used epoxide monomer is the diepoxide prepared by treating one mole of bisphenol A with two moles of epichlorohydrin. 29 -28

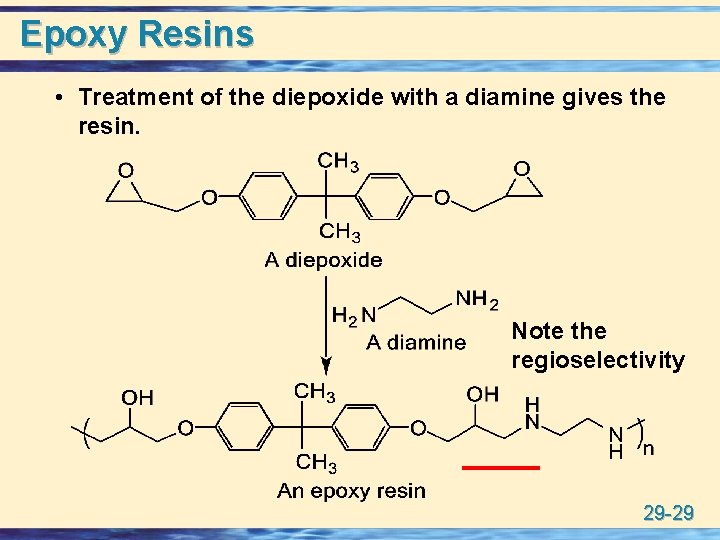
Epoxy Resins • Treatment of the diepoxide with a diamine gives the resin. Note the regioselectivity 29 -29

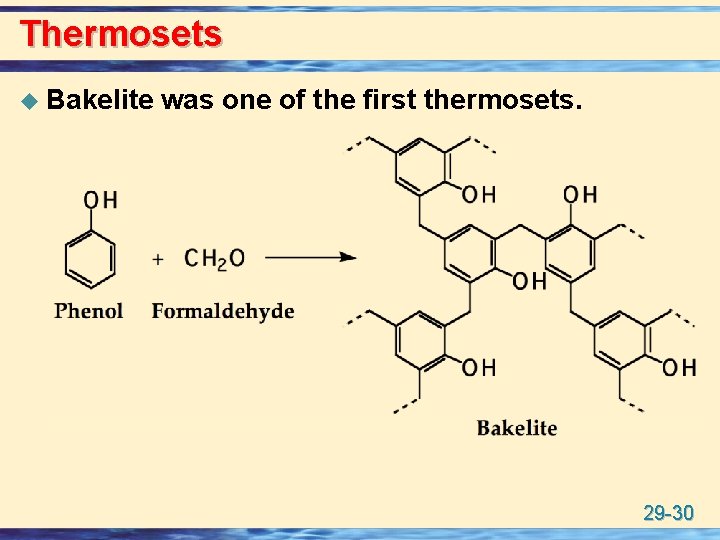
Thermosets u Bakelite was one of the first thermosets. 29 -30

Chain-Growth Polymers u Chain-growth polymerization: A polymerization that involves sequential addition reactions, either to unsaturated monomers or to monomers possessing other reactive functional groups. u Reactive intermediates in chain-growth polymerizations include radicals, carbanions, carbocations, and organometallic complexes. 29 -31

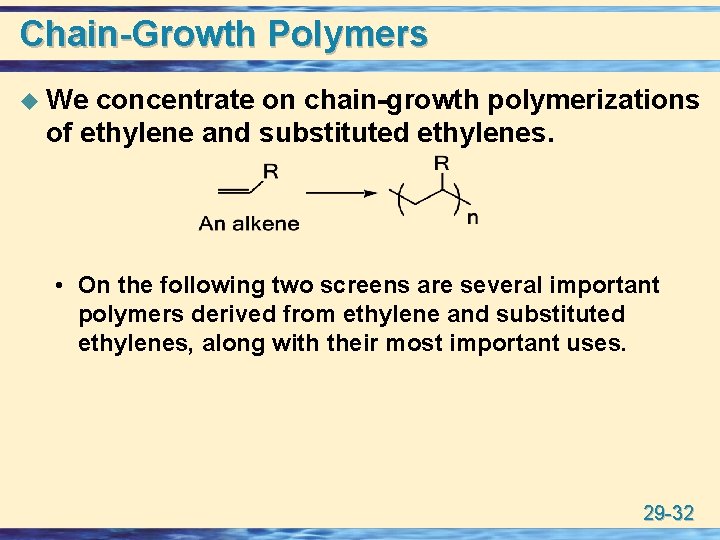
Chain-Growth Polymers u We concentrate on chain-growth polymerizations of ethylene and substituted ethylenes. • On the following two screens are several important polymers derived from ethylene and substituted ethylenes, along with their most important uses. 29 -32

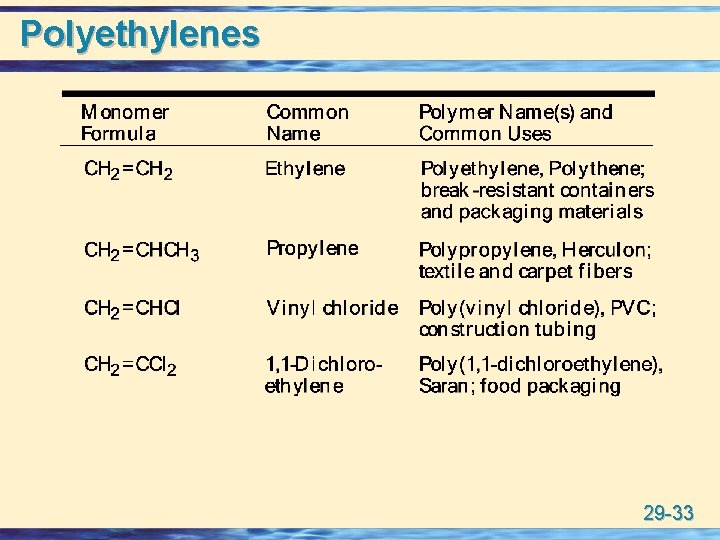
Polyethylenes 29 -33

Polyethylenes 29 -34

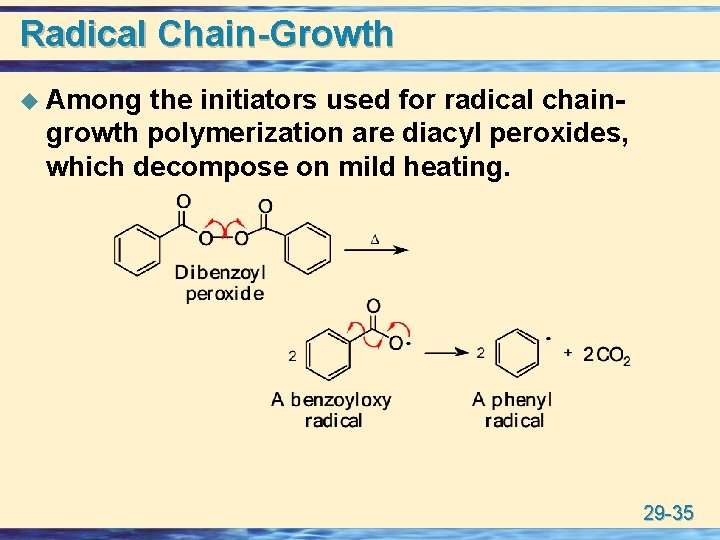
Radical Chain-Growth u Among the initiators used for radical chaingrowth polymerization are diacyl peroxides, which decompose on mild heating. 29 -35

Radical Chain-Growth u Another common class of initiators are azo compounds, which also decompose on mild heating or with absorption of UV light. 29 -36

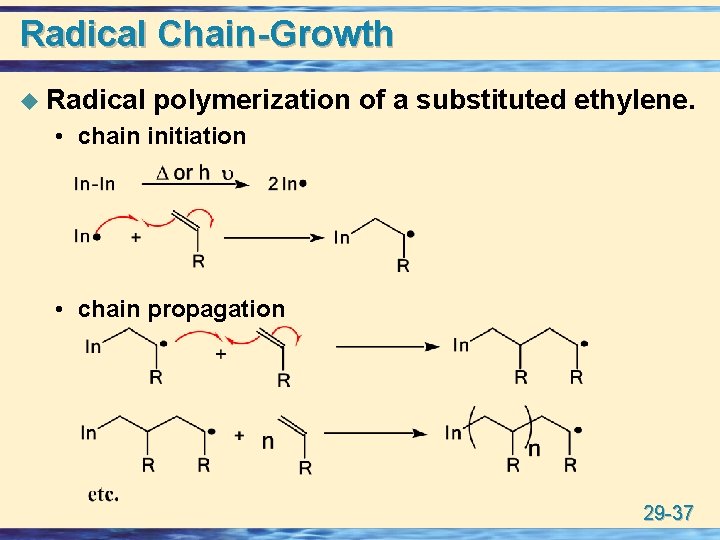
Radical Chain-Growth u Radical polymerization of a substituted ethylene. • chain initiation • chain propagation 29 -37

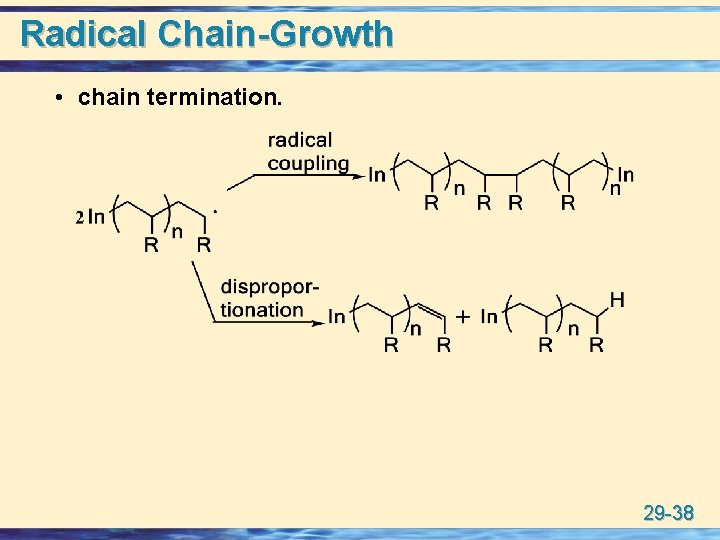
Radical Chain-Growth • chain termination. 29 -38

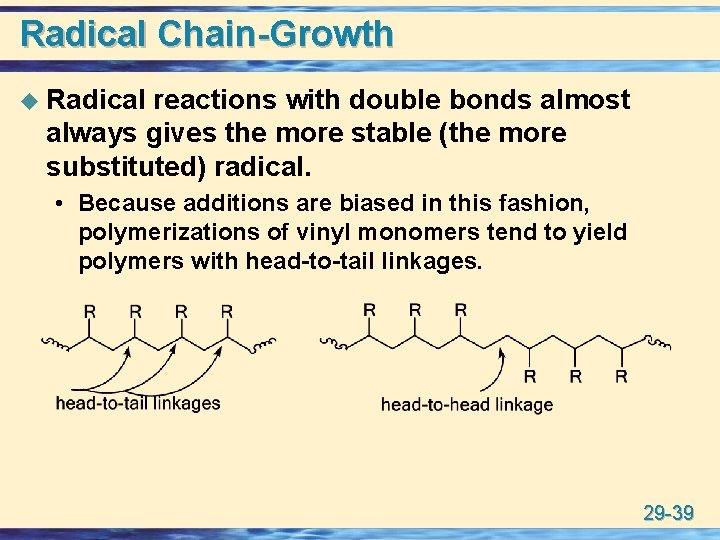
Radical Chain-Growth u Radical reactions with double bonds almost always gives the more stable (the more substituted) radical. • Because additions are biased in this fashion, polymerizations of vinyl monomers tend to yield polymers with head-to-tail linkages. 29 -39

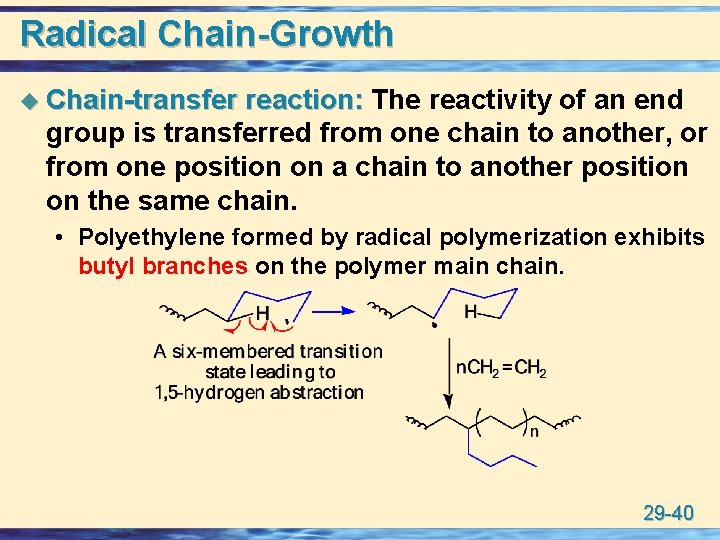
Radical Chain-Growth u Chain-transfer reaction: The reactivity of an end group is transferred from one chain to another, or from one position on a chain to another position on the same chain. • Polyethylene formed by radical polymerization exhibits butyl branches on the polymer main chain. 29 -40

Radical Chain-Growth u The first commercial polyethylenes produced by radical polymerization were soft, tough polymers known as low-density polyethylene (LDPE). • LDPE chains are highly branched due to chain-transfer reactions. • Because this branching prevents polyethylene chains from packing efficiently, LDPE is largely amorphous and transparent. • Approx. 65% is fabricated into films for consumer items such as baked goods, vegetables and other produce, and trash bags. 29 -41

Ziegler-Natta Polymers u Ziegler-Natta chain-growth polymerization is an alternative method that does not involve radicals. • Ziegler-Natta catalysts are heterogeneous materials composed of a Mg. Cl 2 support, a Group 4 B transition metal halide such as Ti. Cl 4, and an alkylaluminum compound. 29 -42

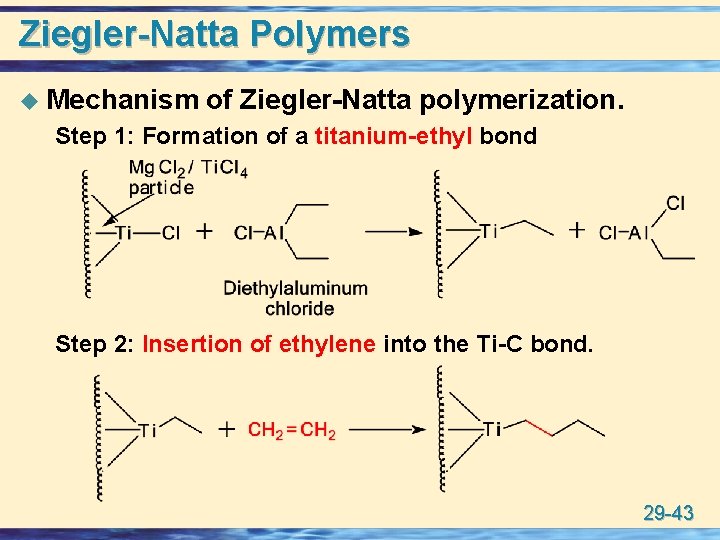
Ziegler-Natta Polymers u Mechanism of Ziegler-Natta polymerization. Step 1: Formation of a titanium-ethyl bond Step 2: Insertion of ethylene into the Ti-C bond. 29 -43

Ziegler-Natta Polymers u Polyethylene from Ziegler-Natta systems is termed high-density polyethylene (HDPE). • It has a considerably lower degree of chain branching than LDPE and a result has a higher degree of crystallinity, a higher density, a higher melting point, and is several times stronger than LDPE. • Appox. 45% of all HDPE is molded into containers. • With special fabrication techniques, HDPE chains can be made to adopt an extended zig-zag conformation. HDPE processed in this manner is stiffer than steel and has 4 x the tensile strength! 29 -44

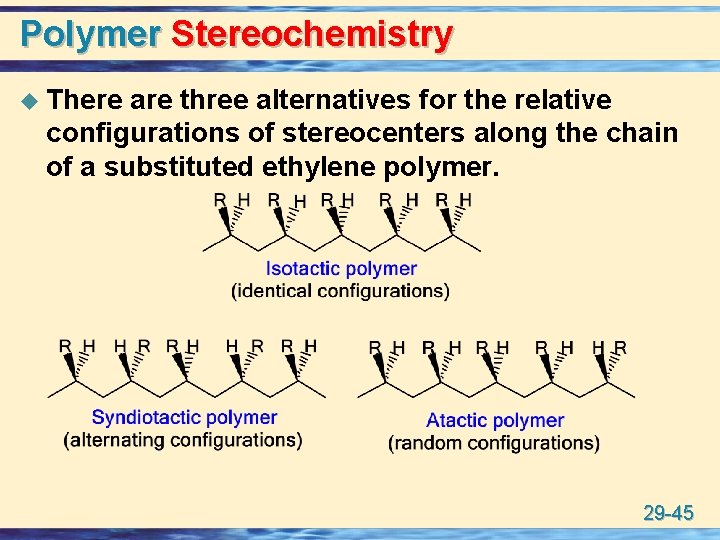
Polymer Stereochemistry u There are three alternatives for the relative configurations of stereocenters along the chain of a substituted ethylene polymer. 29 -45

Polymer Stereochemistry u In general, the more stereoregular the stereocenters are (the more highly isotactic or syndiotactic the polymer is), the more crystalline it is. • Atactic polypropylene, for example, do not pack well and the polymer is an amorphous glass. • Isotactic polypropylene is a crystalline, fiber-forming polymer with a high melt transition. 29 -46

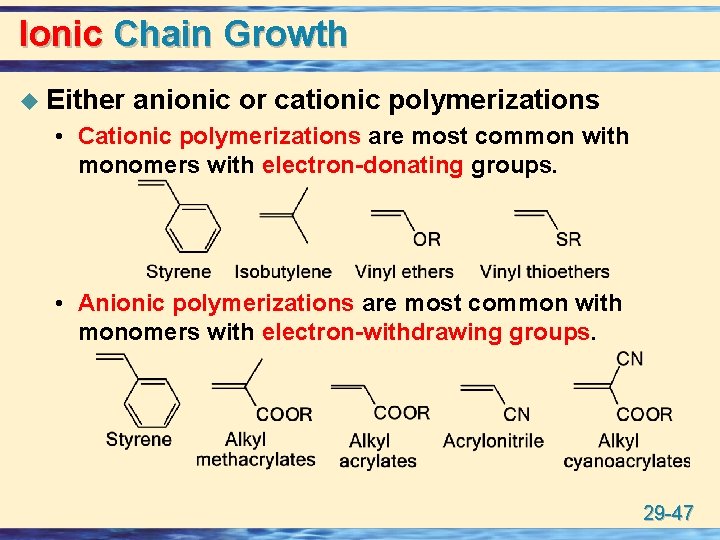
Ionic Chain Growth u Either anionic or cationic polymerizations • Cationic polymerizations are most common with monomers with electron-donating groups. • Anionic polymerizations are most common with monomers with electron-withdrawing groups. 29 -47

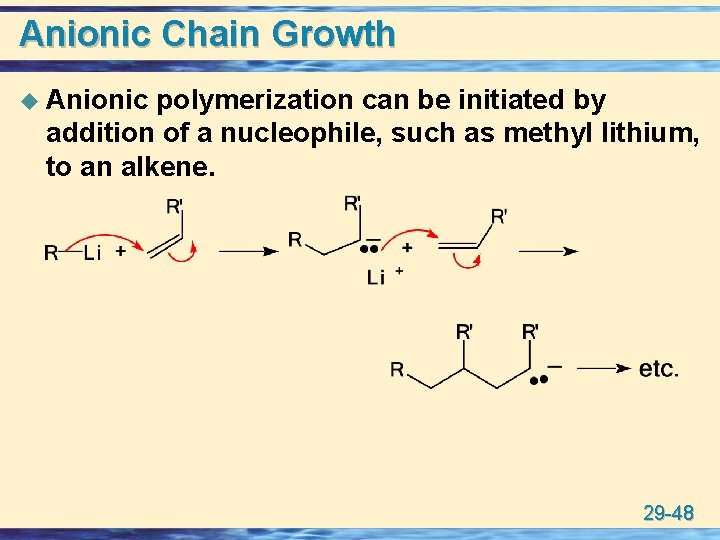
Anionic Chain Growth u Anionic polymerization can be initiated by addition of a nucleophile, such as methyl lithium, to an alkene. 29 -48

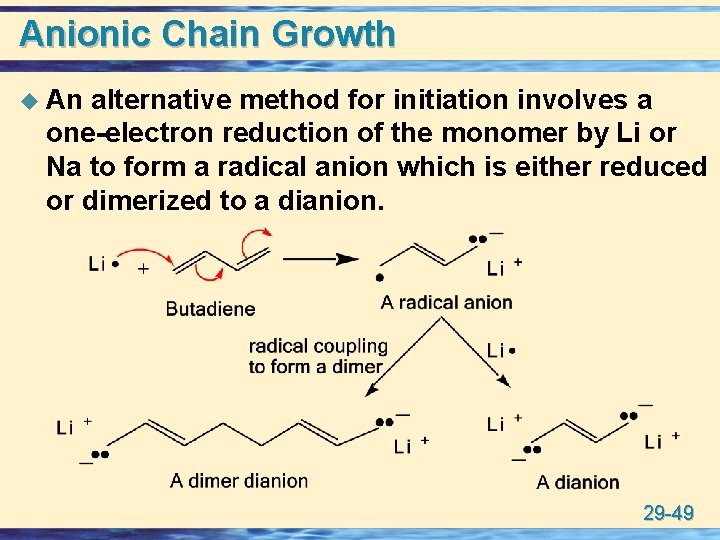
Anionic Chain Growth u An alternative method for initiation involves a one-electron reduction of the monomer by Li or Na to form a radical anion which is either reduced or dimerized to a dianion. 29 -49

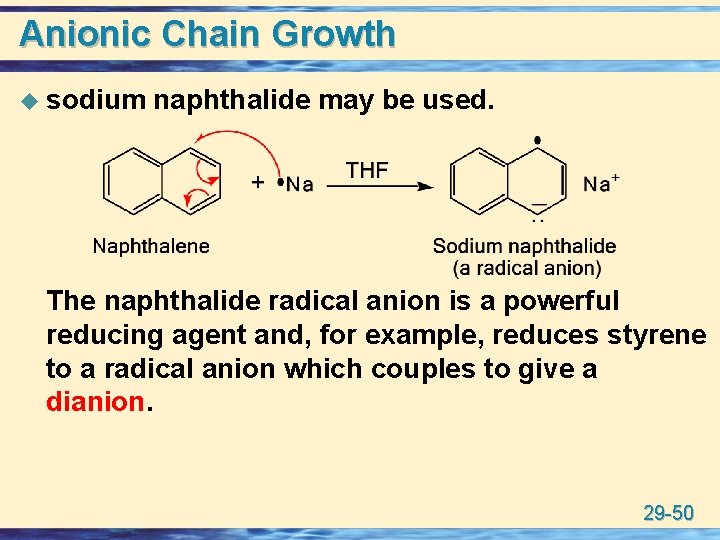
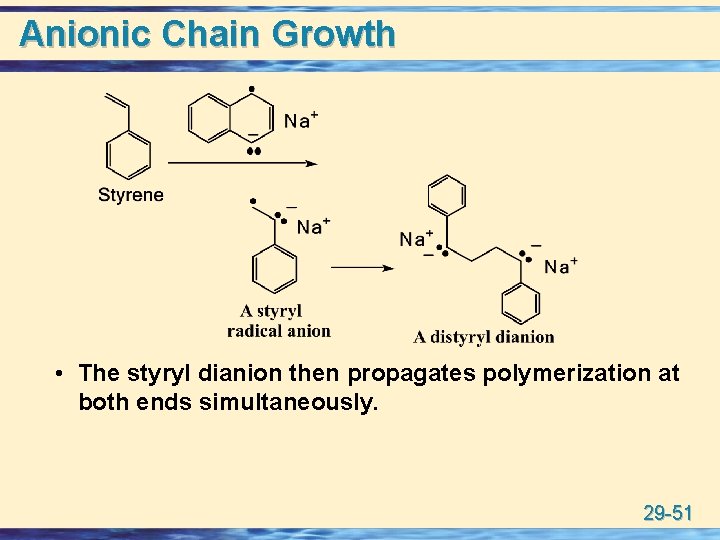
Anionic Chain Growth u sodium naphthalide may be used. The naphthalide radical anion is a powerful reducing agent and, for example, reduces styrene to a radical anion which couples to give a dianion. 29 -50

Anionic Chain Growth • The styryl dianion then propagates polymerization at both ends simultaneously. 29 -51

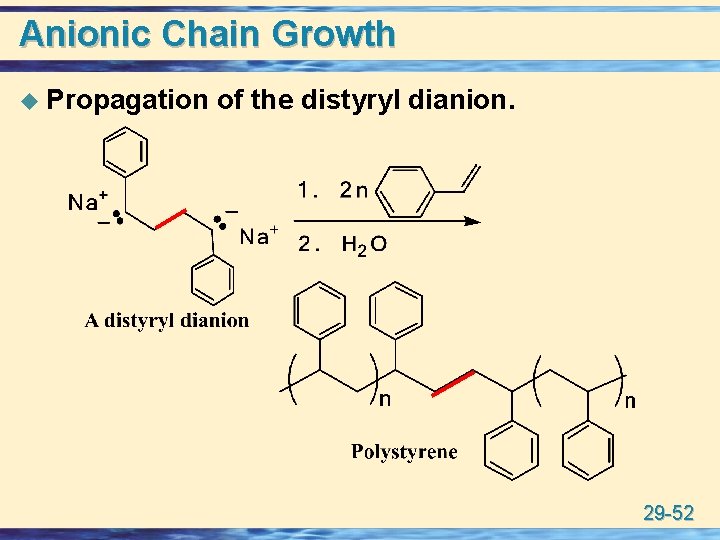
Anionic Chain Growth u Propagation of the distyryl dianion. 29 -52

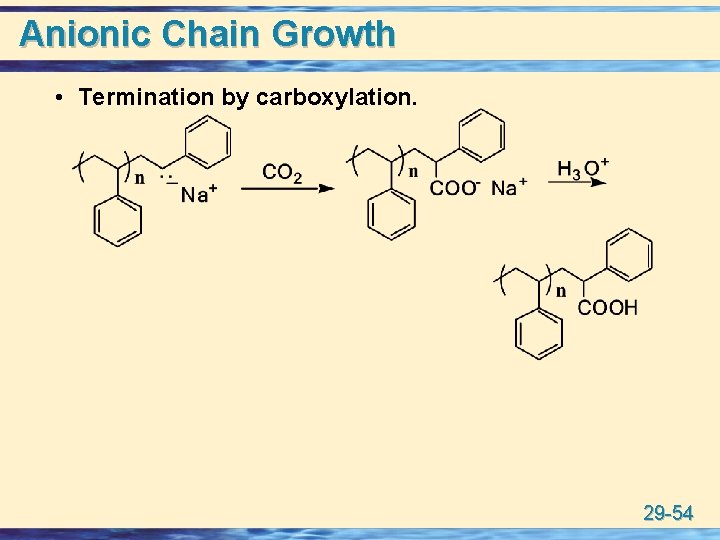
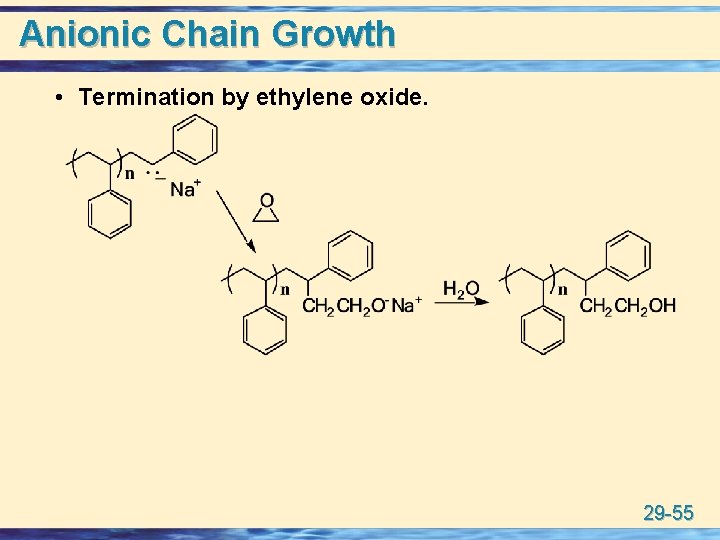
Anionic Chain Growth u Living polymer: A polymer chain that continues to grow without chain-termination steps until either all of the monomer is consumed or some external agent is added to terminate the chains. • after consumption of the monomer under living anionic conditions, electrophilic agents such as CO 2 or ethylene oxide are added to functionalize the chain ends. 29 -53

Anionic Chain Growth • Termination by carboxylation. 29 -54

Anionic Chain Growth • Termination by ethylene oxide. 29 -55

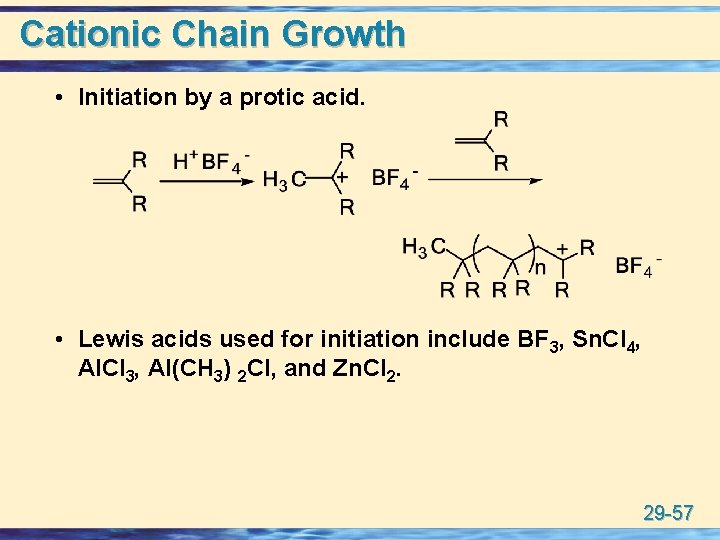
Cationic Chain Growth u The two most common methods for initiating cationic polymerization are: • Addition of H+. Reaction of a strong proton acid with the monomer. • Ionization, as in SN 1. Abstraction of a halide from the organic initiator by a Lewis acid. u Initiation by a proton acid requires a strong acid with a nonnucleophilic anion in order to avoid completion of the addition to the double bond • Suitable acids include HF/As. F 5 and HF/BF 3. 29 -56

Cationic Chain Growth • Initiation by a protic acid. • Lewis acids used for initiation include BF 3, Sn. Cl 4, Al. Cl 3, Al(CH 3) 2 Cl, and Zn. Cl 2. 29 -57

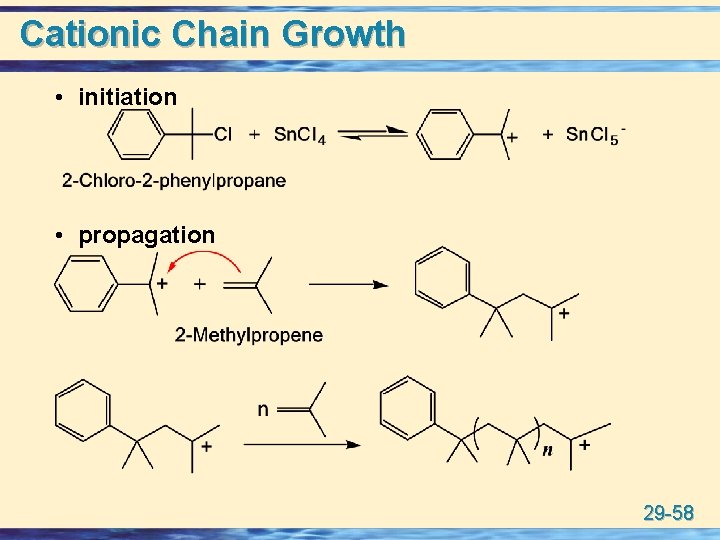
Cationic Chain Growth • initiation • propagation 29 -58

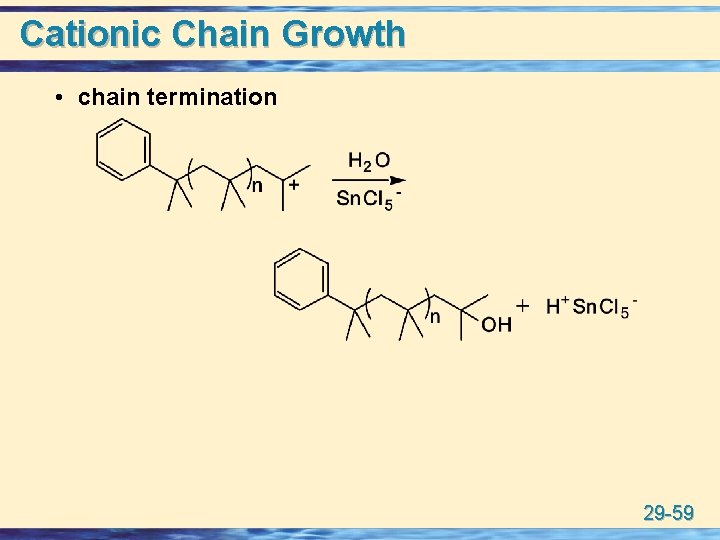
Cationic Chain Growth • chain termination 29 -59

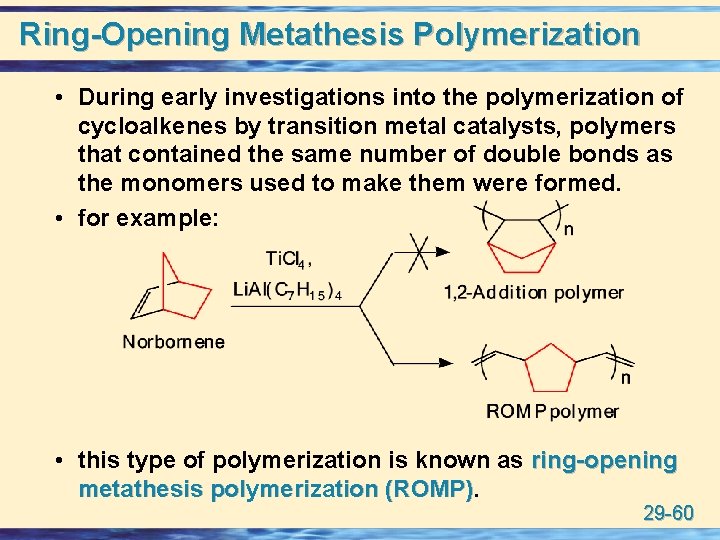
Ring-Opening Metathesis Polymerization • During early investigations into the polymerization of cycloalkenes by transition metal catalysts, polymers that contained the same number of double bonds as the monomers used to make them were formed. • for example: • this type of polymerization is known as ring-opening metathesis polymerization (ROMP) 29 -60

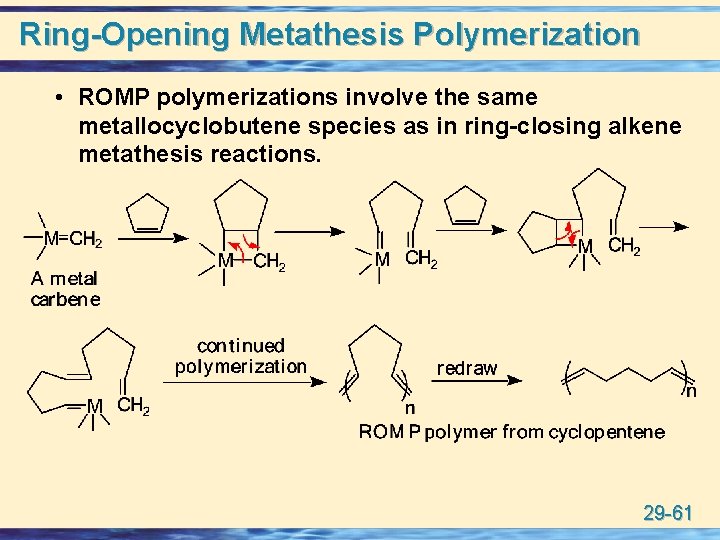
Ring-Opening Metathesis Polymerization • ROMP polymerizations involve the same metallocyclobutene species as in ring-closing alkene metathesis reactions. 29 -61

Ring-Opening Metathesis Polymerization • all steps in ROMP are reversible, and the reaction is driven in the forward direction by the release of ring strain that accompanies the opening of the ring. 29 -62

Ring-Opening Metathesis Polymerization • ROMP is unique is that all unsaturation present in the monomer is conserved in the polymer. • polyacetylene is prepared by the ROMP technique. 29 -63

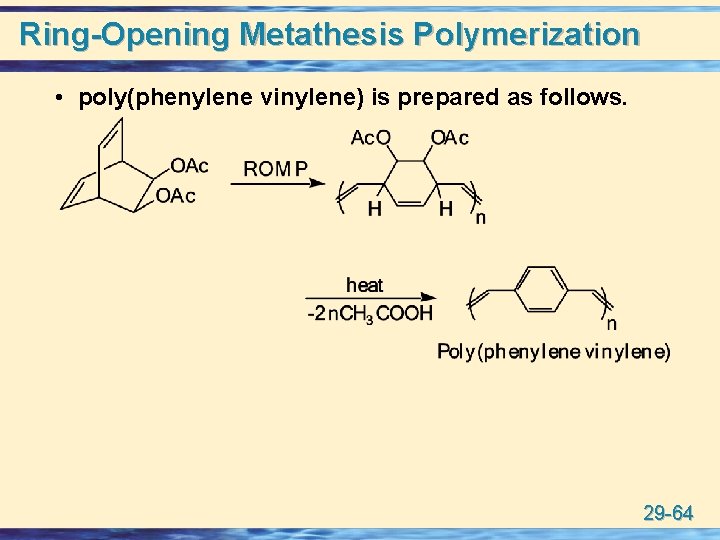
Ring-Opening Metathesis Polymerization • poly(phenylene vinylene) is prepared as follows. 29 -64