Organic Nomenclature Organic Compounds Organic Compounds any covalently


















































































- Slides: 181

Organic Nomenclature

Organic Compounds • Organic Compounds - any covalently bonded compound containing carbon (except carbonates and oxides)

Organic Compounds Contain carbon Have covalent bonds Have low melting points Have low boiling points Burn in air (oxygen) Are soluble in nonpolar solvents Form large molecules

General Characteristics of Organic Molecules • Organic molecules exhibit three different types of hybridization at the carbon center: – sp 3 hybridized carbons for tetrahedral geometries; – sp 2 hybridized carbons for trigonal planar geometries; and – sp hybridized carbons for linear geometries.

Stabilities of Organic Molecules • Carbon forms very strong bonds between H, O, N, and halogens. • Carbon also forms strong bonds with itself. • Therefore, C can form stable long chain or ring structures. • Bond strength increases from single to double to triple bond. • Bond length decreases in the same direction.

Hydrocarbons • Hydrocarbons - Organic compounds that contain only carbon & hydrogen • Alkanes - contain only single covalent bonds • Alkenes - contain one or more carbon - carbon double bond • Alkynes - contain one or more carbon-carbon triple bond

Saturated & Unsaturated Hydrocarbons • Saturated hydrocarbons – contain only single carbon-carbon bonds (alkanes) • Unsaturated hydrocarbons – contain double carbon-carbon bonds (alkenes) or triple carbon-carbon (alkynes) bonds

Formulas • Alkanes = Cn. H 2 n+2 • Alkenes = Cn. H 2 n • Alkynes = Cn. H 2 n-2

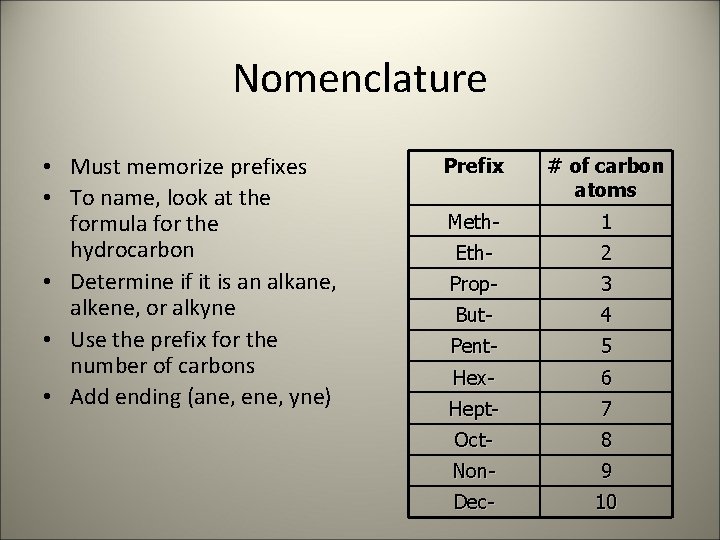
Nomenclature • Must memorize prefixes • To name, look at the formula for the hydrocarbon • Determine if it is an alkane, alkene, or alkyne • Use the prefix for the number of carbons • Add ending (ane, ene, yne) Prefix # of carbon atoms Meth- 1 Eth- 2 Prop- 3 But- 4 Pent- 5 Hex- 6 Hept- 7 Oct- 8 Non- 9 Dec- 10

Example • • • Name C 3 H 8 This falls under the equation Cn. H 2 n+2 Therefore it is an alkane Since n=3 we will use the prefix prop Since it is an alkane we will use the ending ane propane

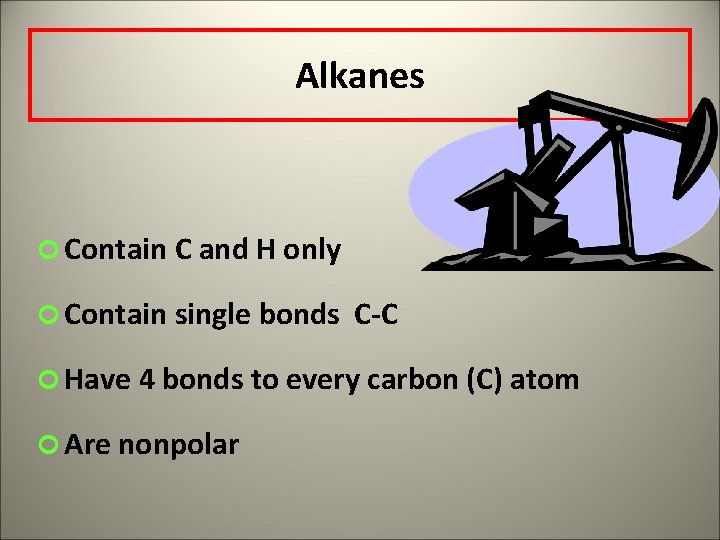
Alkanes ¢ Contain C and H only ¢ Contain single bonds C-C ¢ Have 4 bonds to every carbon (C) atom ¢ Are nonpolar

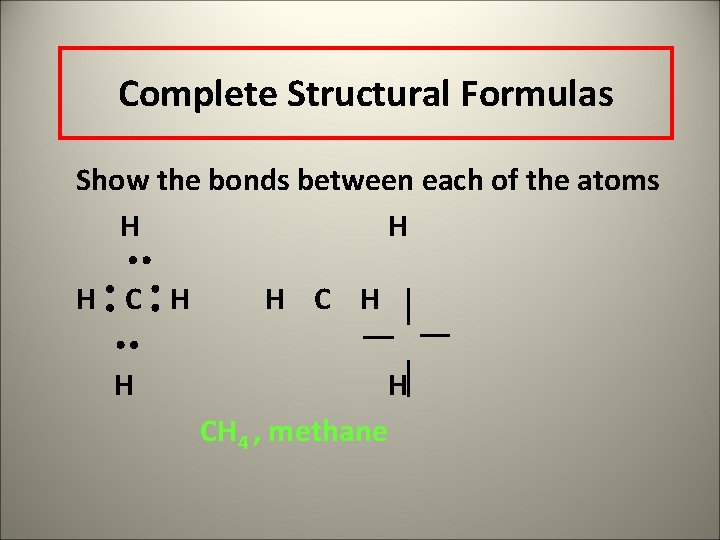
Complete Structural Formulas Show the bonds between each of the atoms H H H CH 4 , methane

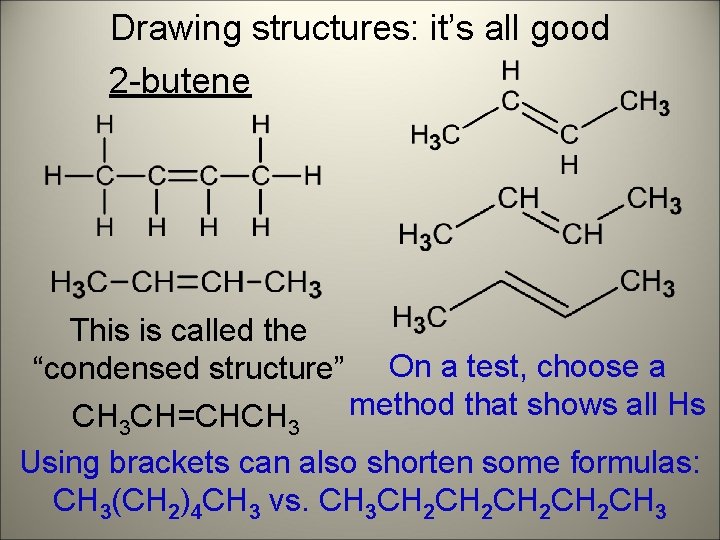
Drawing structures: it’s all good 2 -butene This is called the “condensed structure” On a test, choose a method that shows all Hs CH 3 CH=CHCH 3 Using brackets can also shorten some formulas: CH 3(CH 2)4 CH 3 vs. CH 3 CH 2 CH 2 CH 3

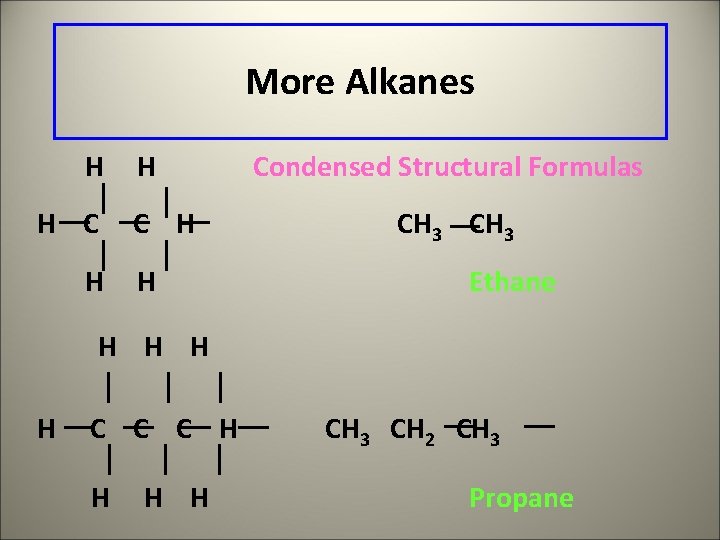
More Alkanes H H Condensed Structural Formulas CH 3 Ethane H H C C C H H CH 3 CH 2 CH 3 Propane

IUPAC Names Name # carbons Structural Formula Methane 1 CH 4 Ethane 2 CH 3 Propane 3 CH 3 CH 2 CH 3 Butane 4 CH 3 CH 2 CH 3 Pentane 5 CH 3 CH 2 CH 2 CH 3

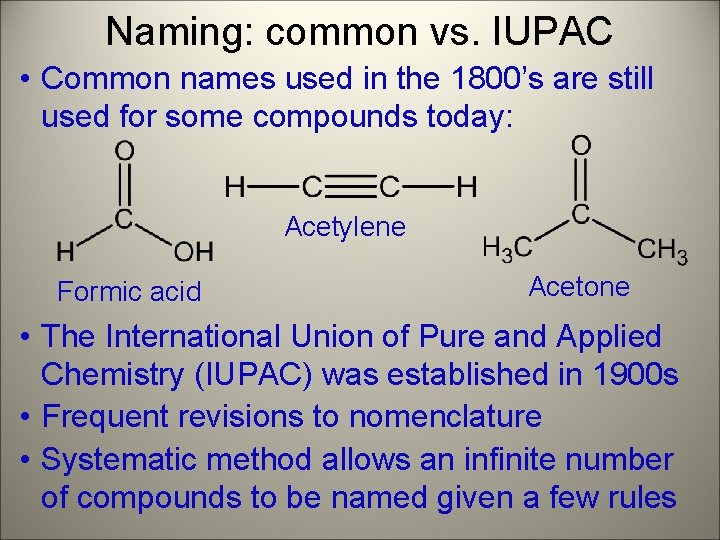
Naming: common vs. IUPAC • Common names used in the 1800’s are still used for some compounds today: Acetylene Formic acid Acetone • The International Union of Pure and Applied Chemistry (IUPAC) was established in 1900 s • Frequent revisions to nomenclature • Systematic method allows an infinite number of compounds to be named given a few rules

IUPAC NAMES Name # carbons Structural Formula Hexane 6 CH 3 CH 2 CH 2 CH 3 Heptane 7 CH 3 CH 2 CH 2 CH 2 CH 3 Octane 8 CH 3 CH 2 CH 2 CH 2 CH 3 Nonane 9 CH 3 CH 2 CH 2 CH 3 Decane 10 CH 3 CH 2 CH 2 CH 3

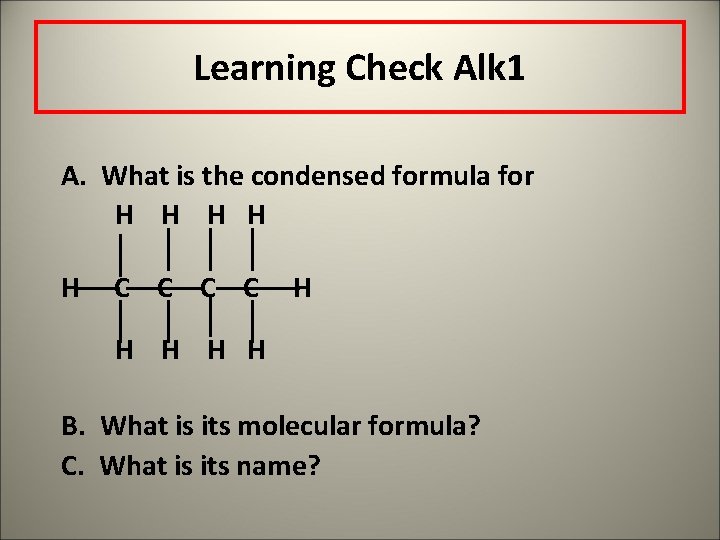
Learning Check Alk 1 A. What is the condensed formula for H H H C C H H H B. What is its molecular formula? C. What is its name?

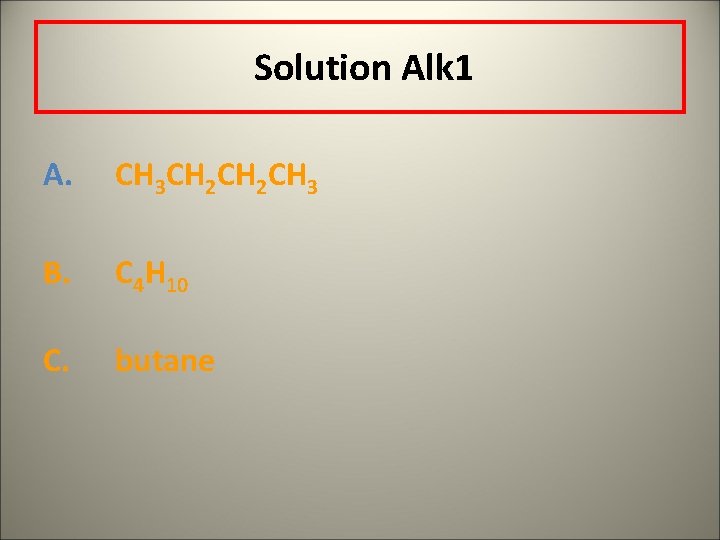
Solution Alk 1 A. CH 3 CH 2 CH 3 B. C 4 H 10 C. butane

Example • • Name C 5 H 12 5 carbons = pent Cn. H 2 n+2 = alkane (ane) pentane

• Alkenes and Alkynes – Alkenes are hydrocarbons with at least one double carbon to carbon bond. • To show the presence of the double bond, the –ane suffix from the alkane name is changed to –ene. – The alkenes are unsaturated with respect to hydrogen • This means it does not have the maximum number of hydrogen atoms as it would if it were an alkane (a saturated hydrocarbon).

– Naming is similar to naming alkanes except: • The longest continuous chain must contain the double bond. • The base name now ends in –ene. • The carbons are numbered so as to keep the number for the double bond as low as possible. • The base name is given a number which identifies the location of the double bond. – An alkyne is a hydrocarbon with at least one carbon to carbon triple bond. – Naming an alkyne is similar to the alkenes, except the base name ends in –yne.

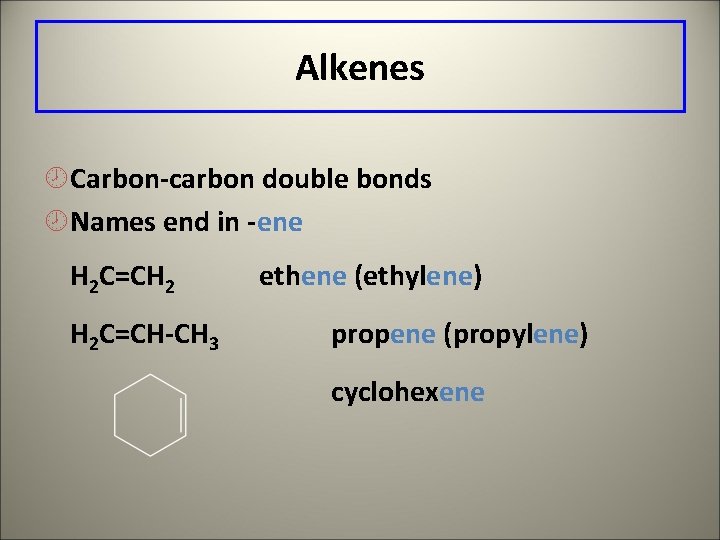
Alkenes ¾Carbon-carbon double bonds ¾Names end in -ene H 2 C=CH 2 H 2 C=CH-CH 3 ethene (ethylene) propene (propylene) cyclohexene

Alkynes ¾Carbon-carbon triple bonds ¾Names end in -yne HC CH HC C-CH 3 ethyne(acetylene) propyne

Naming Alkenes and Alkynes When the carbon chain has 4 or more C atoms, number the chain to give the lowest number to the double or triple bond. 1 2 3 4 CH 2=CHCH 2 CH 3 CH=CHCH 3 CH CHCH 3 1 -butene 2 -butyne

Numbering carbons Q- draw pentene A- Where’s the bond? We number C atoms 1 -pentene • Thus, naming compounds with multiple bonds is more complex than previously indicated • Only if 2+ possibilities exist, are #s needed • Always give double bond the lowest number • Q - Name these C 2 H 4 2 -butene Ethene 3 -nonyne

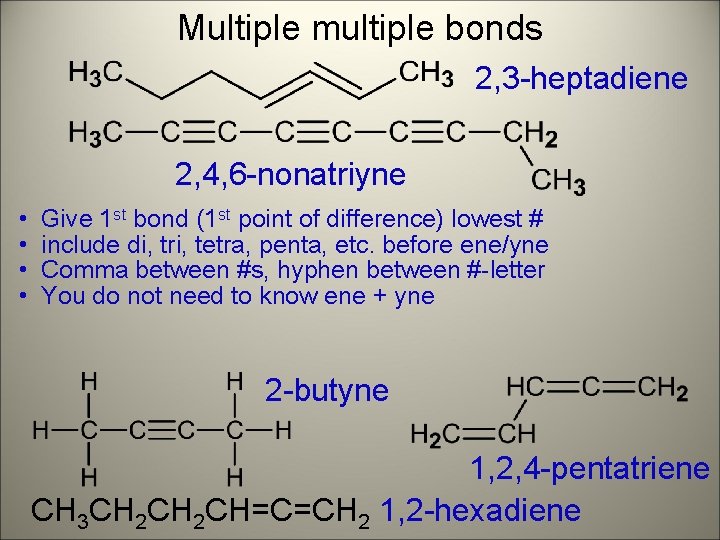
Multiple multiple bonds 2, 3 -heptadiene 2, 4, 6 -nonatriyne • • Give 1 st bond (1 st point of difference) lowest # include di, tri, tetra, penta, etc. before ene/yne Comma between #s, hyphen between #-letter You do not need to know ene + yne 2 -butyne 1, 2, 4 -pentatriene CH 3 CH 2 CH=C=CH 2 1, 2 -hexadiene

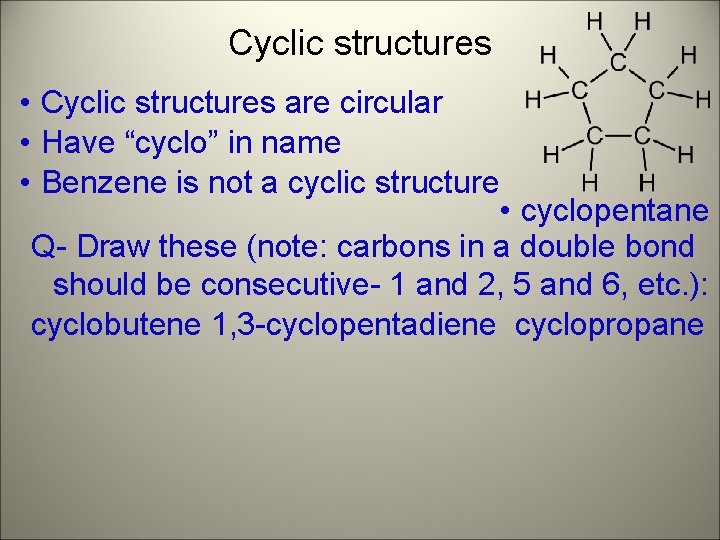
Cyclic structures • Cyclic structures are circular • Have “cyclo” in name • Benzene is not a cyclic structure • cyclopentane Q- Draw these (note: carbons in a double bond should be consecutive- 1 and 2, 5 and 6, etc. ): cyclobutene 1, 3 -cyclopentadiene cyclopropane

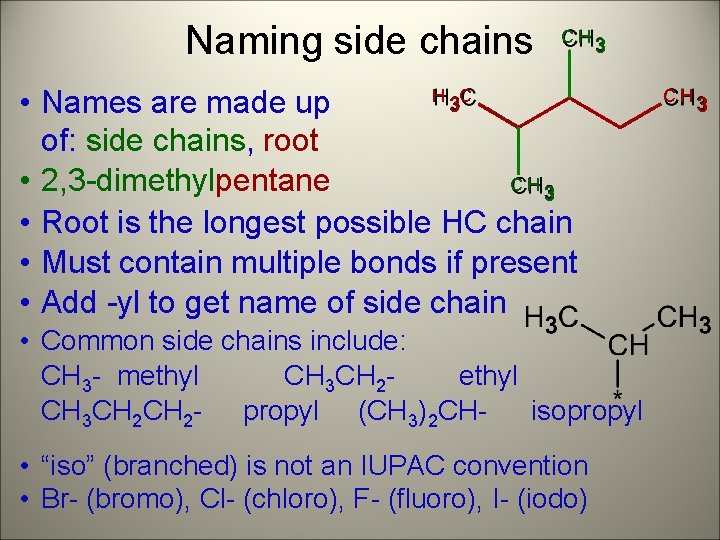
Naming side chains CH 3 H C 3 • Names are made up of: side chains, root • 2, 3 -dimethylpentane CH 3 • Root is the longest possible HC chain • Must contain multiple bonds if present • Add -yl to get name of side chain • Common side chains include: CH 3 - methyl CH 3 CH 2 CH 2 - propyl (CH 3)2 CHisopropyl • “iso” (branched) is not an IUPAC convention • Br- (bromo), Cl- (chloro), F- (fluoro), I- (iodo) CH 3

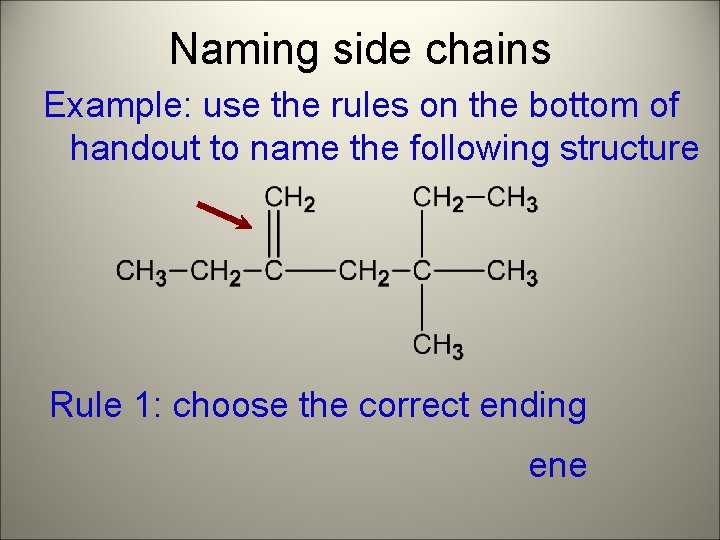
Naming side chains Example: use the rules on the bottom of handout to name the following structure Rule 1: choose the correct ending ene

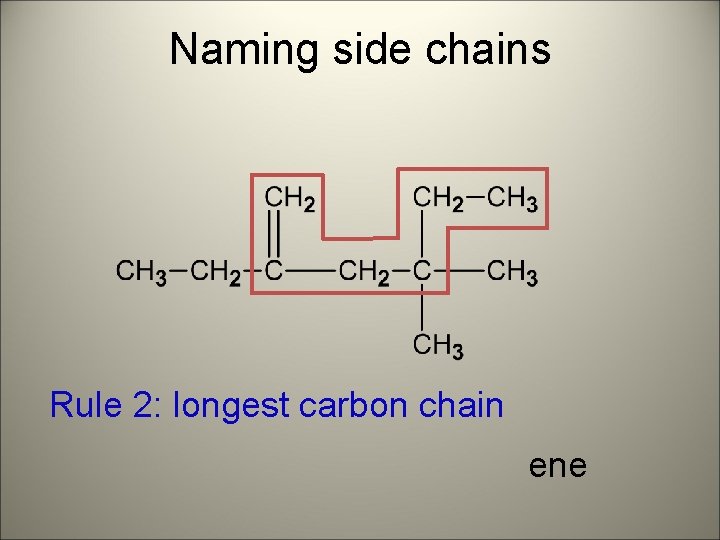
Naming side chains Rule 2: longest carbon chain ene

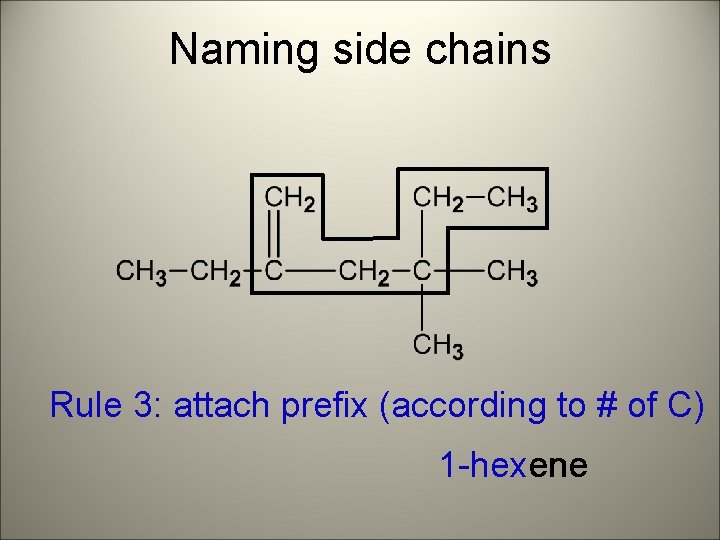
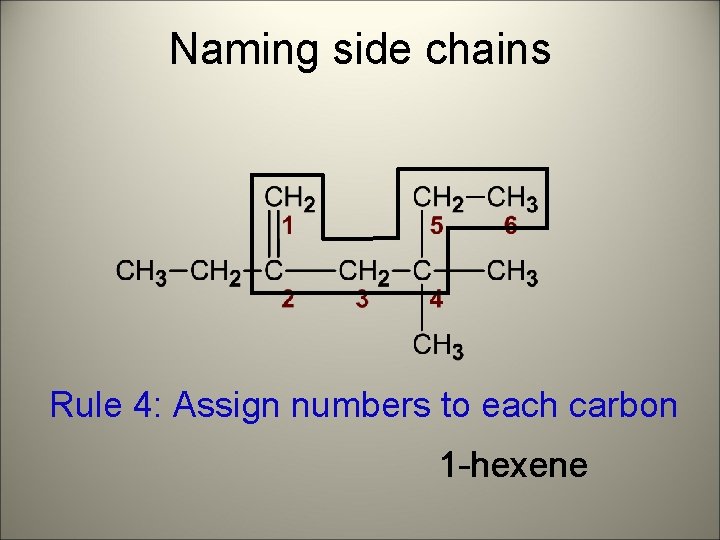
Naming side chains Rule 3: attach prefix (according to # of C) 1 -hexene

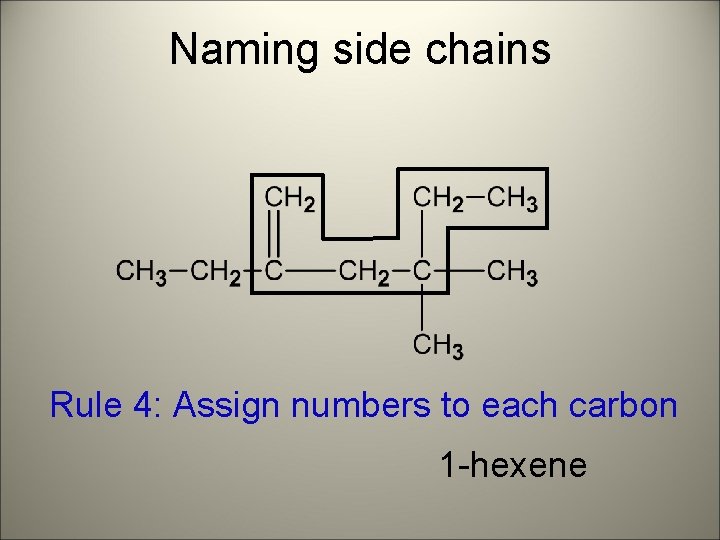
Naming side chains Rule 4: Assign numbers to each carbon 1 -hexene

Naming side chains Rule 4: Assign numbers to each carbon 1 -hexene

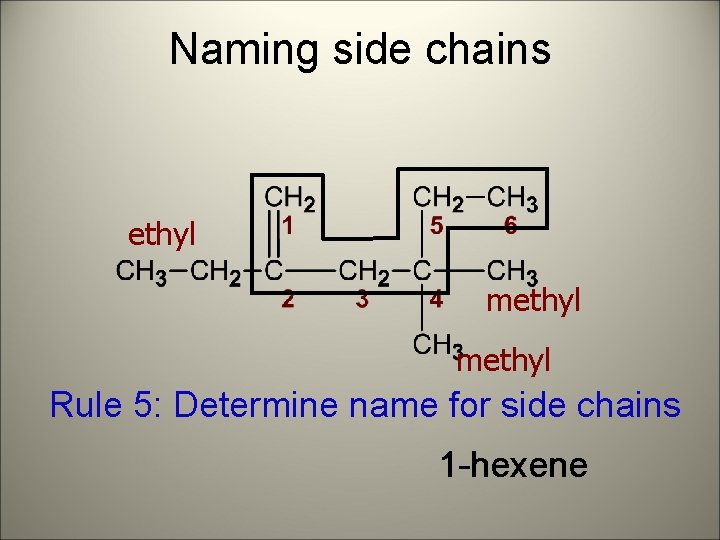
Naming side chains ethyl methyl Rule 5: Determine name for side chains 1 -hexene

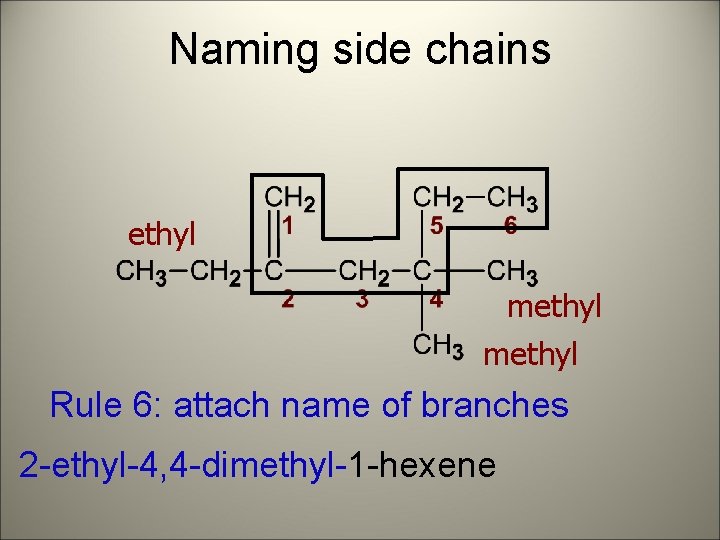
Naming side chains ethyl methyl Rule 6: attach name of branches 2 -ethyl-4, 4 -dimethyl-1 -hexene

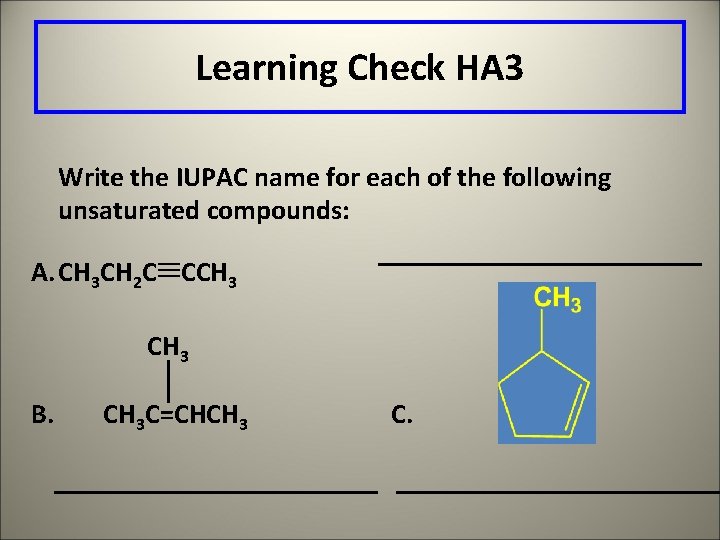
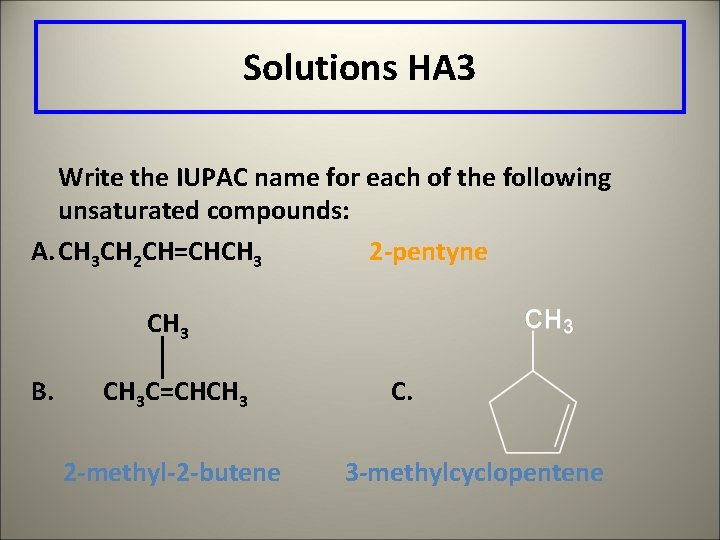
Learning Check HA 3 Write the IUPAC name for each of the following unsaturated compounds: A. CH 3 CH 2 C CCH 3 B. CH 3 C=CHCH 3 C.

Solutions HA 3 Write the IUPAC name for each of the following unsaturated compounds: A. CH 3 CH 2 CH=CHCH 3 2 -pentyne CH 3 B. CH 3 C=CHCH 3 2 -methyl-2 -butene C. 3 -methylcyclopentene

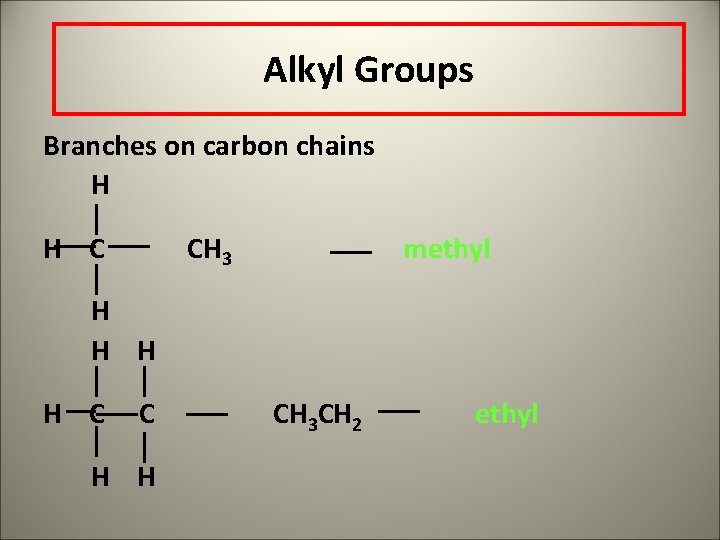
Alkyl Groups Branches on carbon chains H H C CH 3 methyl H H C C H H CH 3 CH 2 ethyl

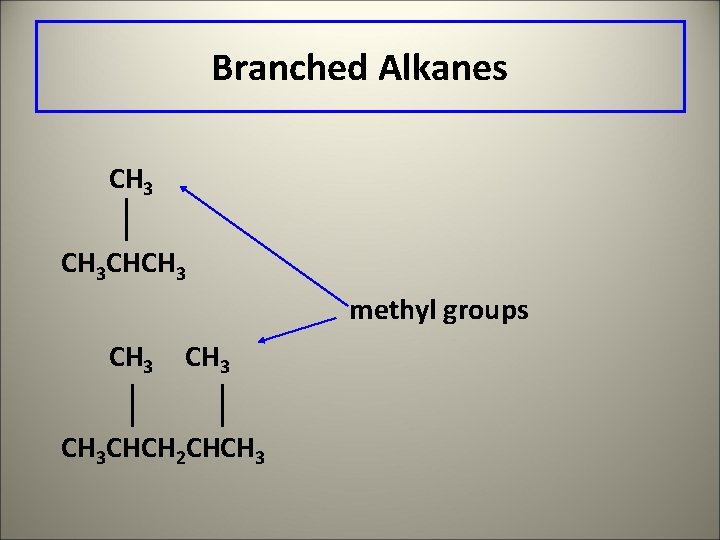
Branched Alkanes CH 3 CHCH 3 methyl groups CH 3 CHCH 2 CHCH 3

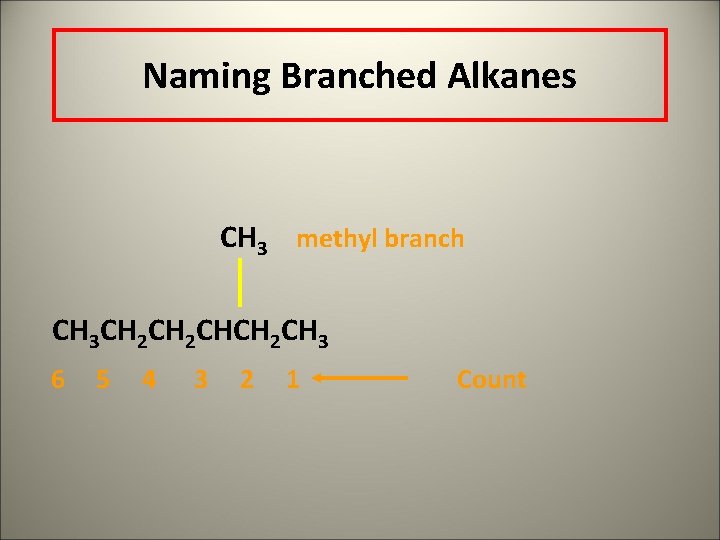
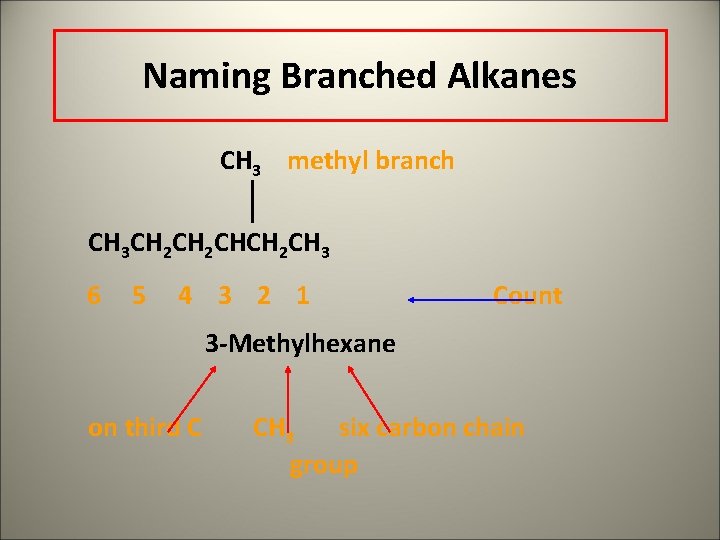
Naming Branched Alkanes CH 3 methyl branch CH 3 CH 2 CHCH 2 CH 3 6 5 4 3 2 1 Count

Naming Branched Alkanes CH 3 methyl branch CH 3 CH 2 CHCH 2 CH 3 6 5 4 3 2 1 Count 3 -Methylhexane on third C CH 3 six carbon chain group

IUPAC • Carbon chains need to be numbered in order to designate the carbon atom that the substituted group is added. • Follow these guidelines: – Find the longest continuous carbon chain – Number the chain so the substituent's have the lowest possible number. – Give priority to the multiple bonds.

Numbering priority • 1. functional groups • 2. double and triple bonds • 3. substituents

• Once all groups have been numbered, place them in alphabetical order in the final compounds name. • Numbers are separated by commas • Names and numbers are separated by dashes. • Use mathematical prefixes for multiple substituent's.

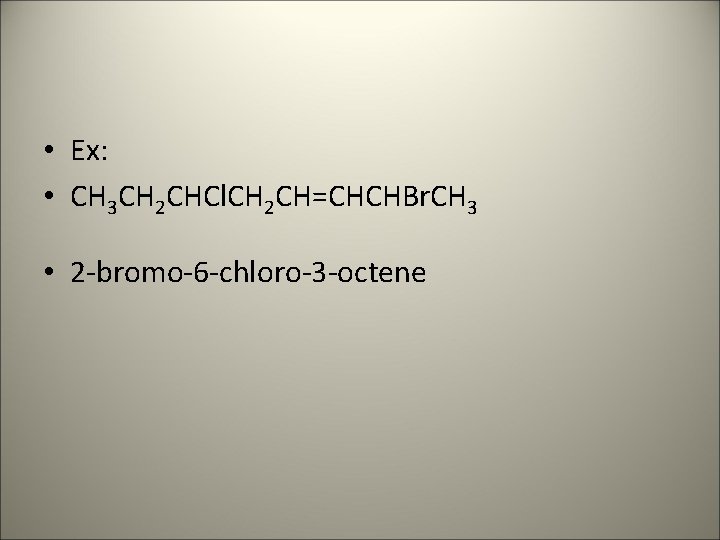
• Ex: • CH 3 CH 2 CHCl. CH 2 CH=CHCHBr. CH 3 • 2 -bromo-6 -chloro-3 -octene

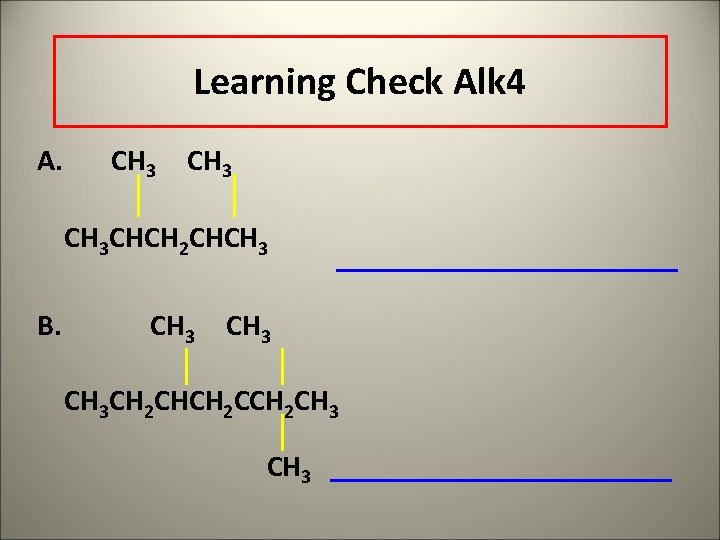
Learning Check Alk 4 A. CH 3 CHCH 2 CHCH 3 B. CH 3 CH 2 CHCH 2 CH 3

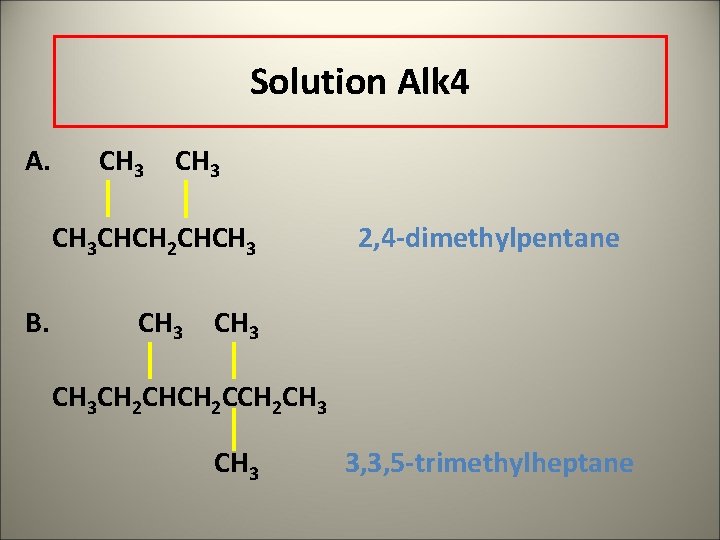
Solution Alk 4 A. CH 3 CHCH 2 CHCH 3 B. CH 3 2, 4 -dimethylpentane CH 3 CH 2 CHCH 2 CH 3 3, 3, 5 -trimethylheptane

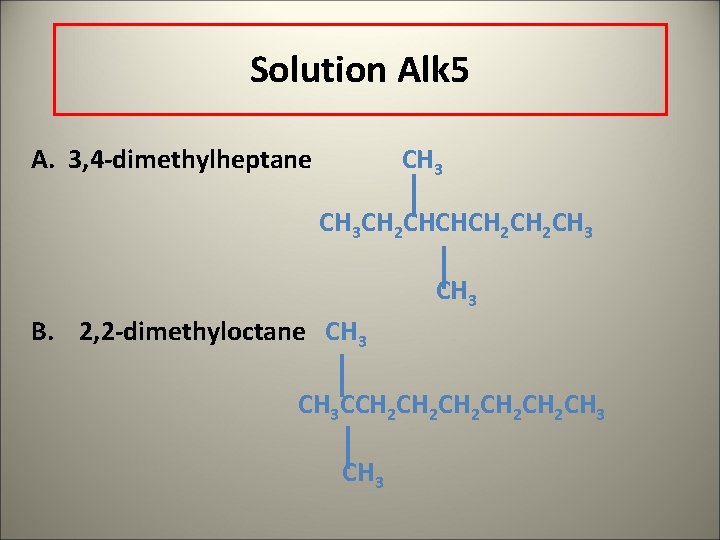
Learning Check Alk 5 Write a condensed structure for A. 3, 4 -dimethylheptane B. 2, 2 -dimethyloctane

Solution Alk 5 A. 3, 4 -dimethylheptane CH 3 CH 2 CHCHCH 2 CH 3 B. 2, 2 -dimethyloctane CH 3 CCH 2 CH 2 CH 2 CH 3

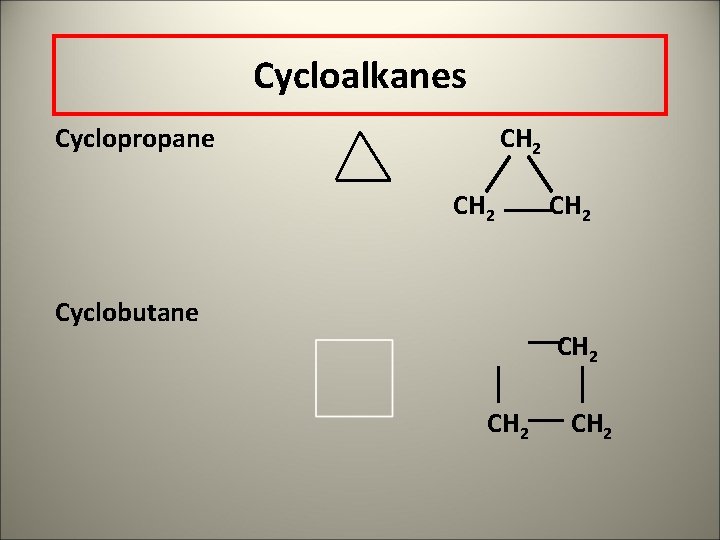
Cycloalkanes Cyclopropane CH 2 Cyclobutane CH 2

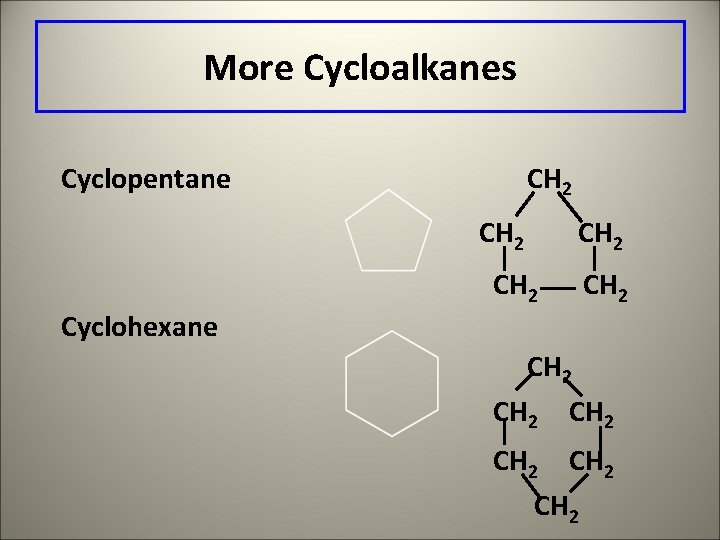
More Cycloalkanes Cyclopentane CH 2 CH 2 Cyclohexane CH 2 CH 2

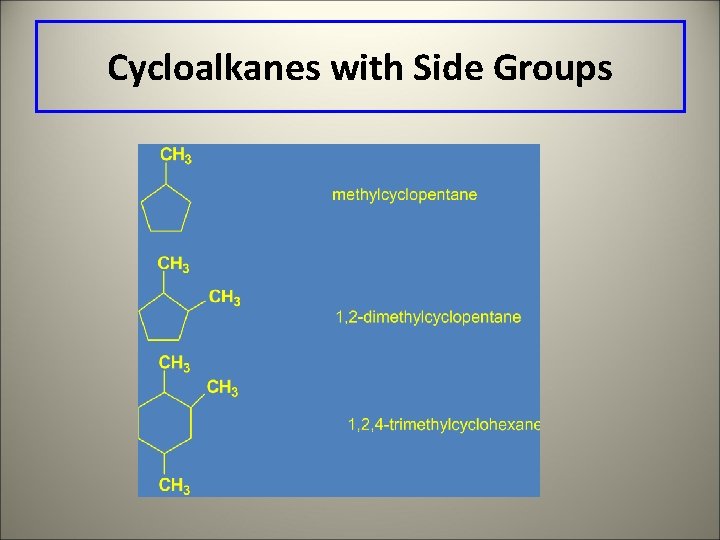
Naming Cycloalkanes with Side Groups Number of Naming side groups 1. Side group name goes in front of the cycloalkane name. 2. Number the ring in the direction that gives the lowest numbers to the side groups.

Cycloalkanes with Side Groups

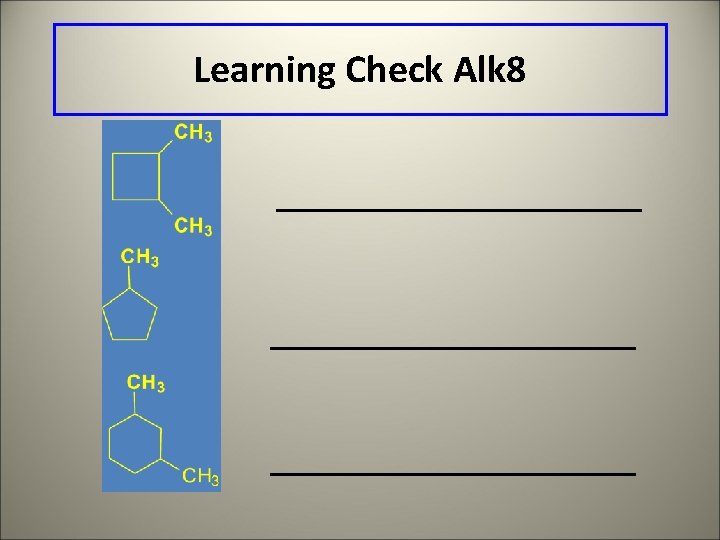
Learning Check Alk 8

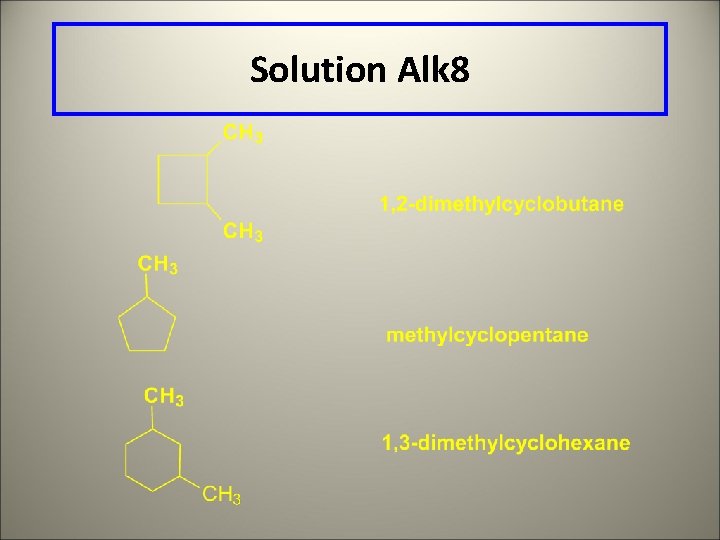
Solution Alk 8

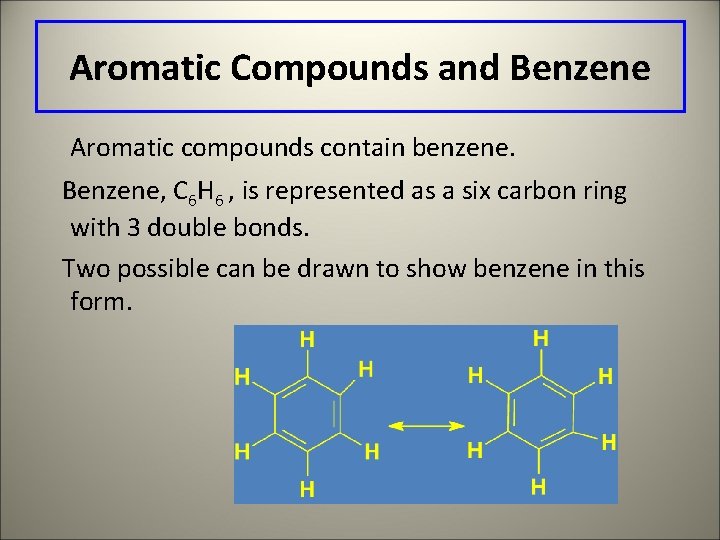
Aromatic Compounds and Benzene Aromatic compounds contain benzene. Benzene, C 6 H 6 , is represented as a six carbon ring with 3 double bonds. Two possible can be drawn to show benzene in this form.

Benzene Structure The structures for benzene can also be written as a single structure where the alternating double bonds are written as a circle within the ring. Benzene structure

Aromatic Compounds in Nature and Health Many aromatic compounds are common in nature and in medicine.

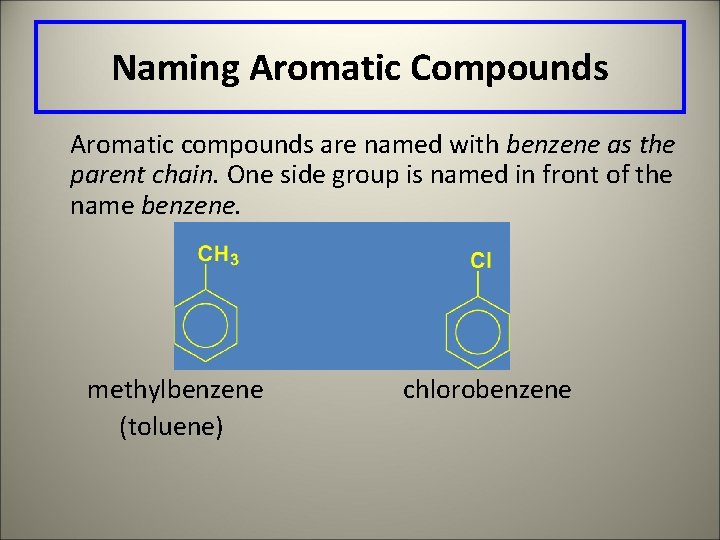
Naming Aromatic Compounds Aromatic compounds are named with benzene as the parent chain. One side group is named in front of the name benzene. methylbenzene (toluene) chlorobenzene

Naming Aromatic Compounds When two groups are attached to benzene, the ring is numbered to give the lower numbers to the side groups. The prefixes ortho (1, 2), meta (1, 3 -) and para (1, 4 -) are also used.

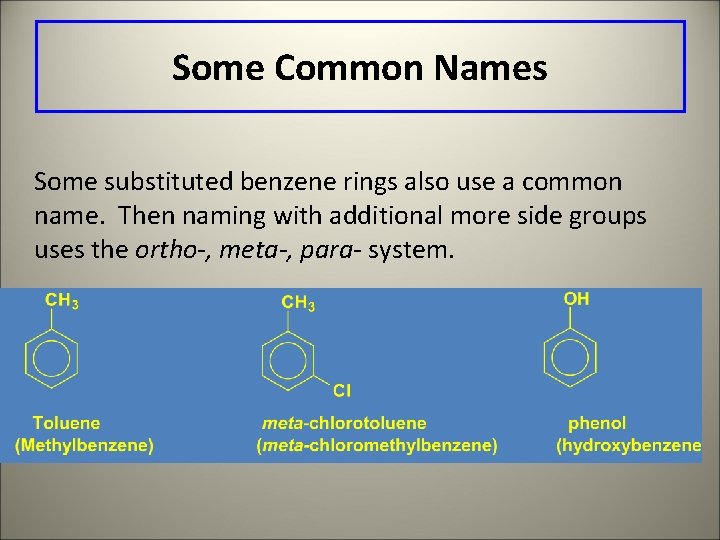
Some Common Names Some substituted benzene rings also use a common name. Then naming with additional more side groups uses the ortho-, meta-, para- system.

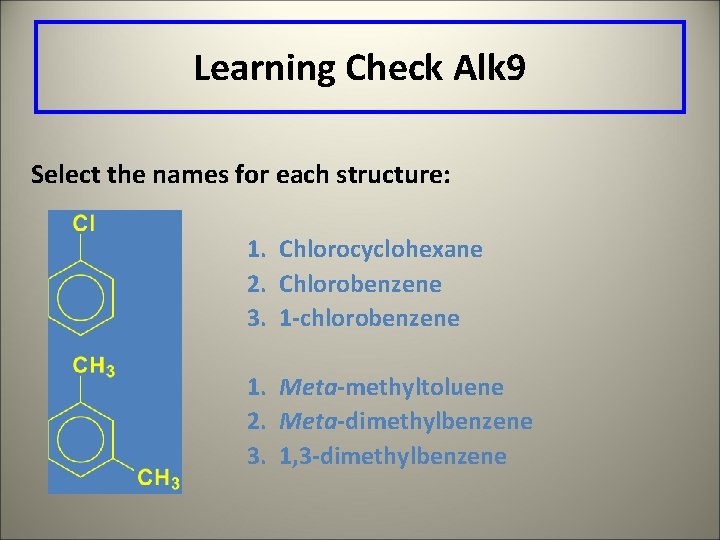
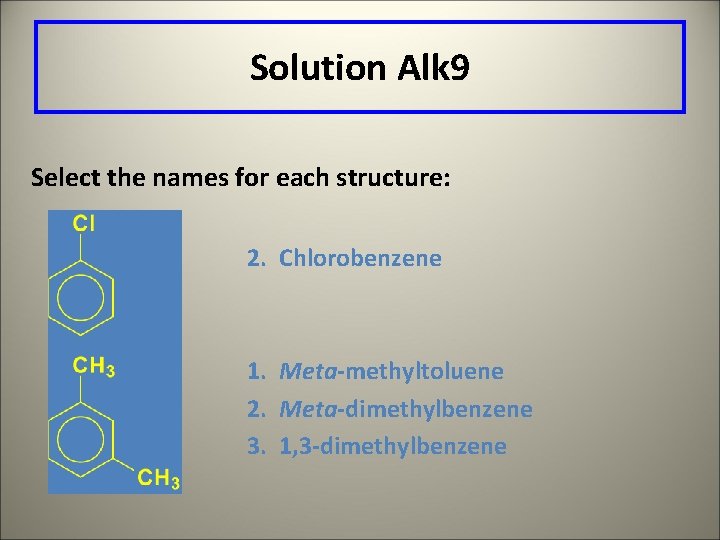
Learning Check Alk 9 Select the names for each structure: 1. Chlorocyclohexane 2. Chlorobenzene 3. 1 -chlorobenzene 1. Meta-methyltoluene 2. Meta-dimethylbenzene 3. 1, 3 -dimethylbenzene

Solution Alk 9 Select the names for each structure: 2. Chlorobenzene 1. Meta-methyltoluene 2. Meta-dimethylbenzene 3. 1, 3 -dimethylbenzene

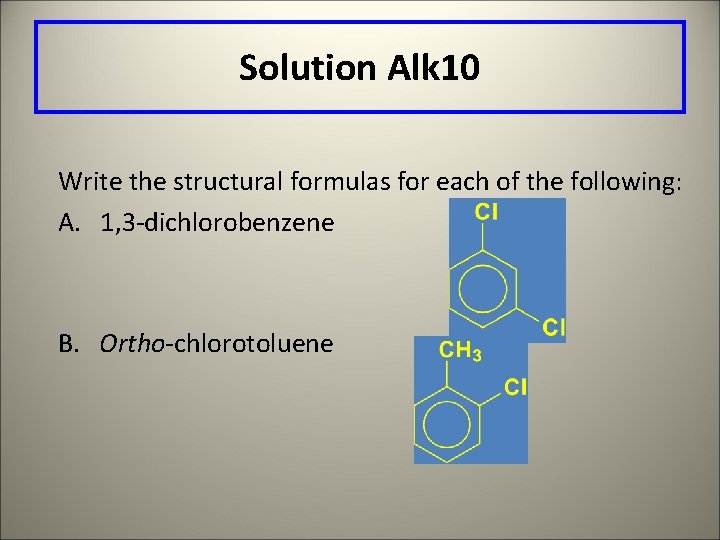
Learning Check Alk 10 Write the structural formulas for each of the following: A. 1, 3 -dichlorobenzene B. Ortho-chlorotoluene

Solution Alk 10 Write the structural formulas for each of the following: A. 1, 3 -dichlorobenzene B. Ortho-chlorotoluene

Functional Groups § Part of an organic molecule where chemical § § § reactions take place Composed of an atom or group of atoms Replace a H in the corresponding alkane Provide a way to classify organic compounds

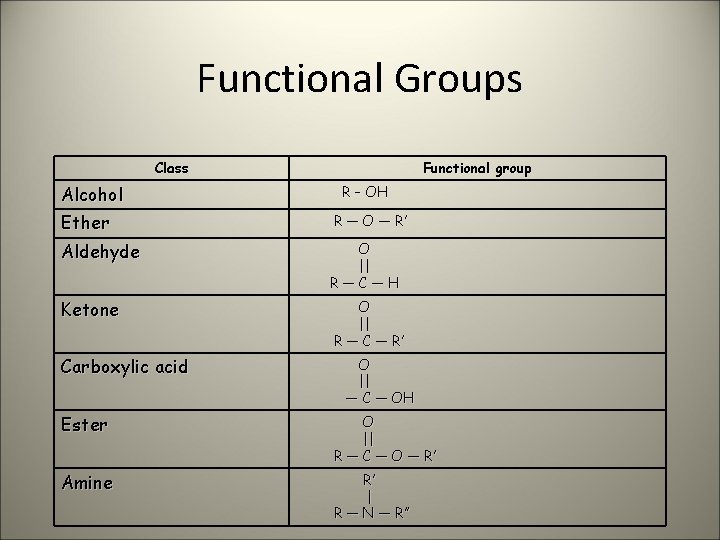
Functional Groups Class Alcohol Functional group R – OH Ether R — O — R’ Aldehyde O || R—C—H Ketone O || R — C — R’ Carboxylic acid O || — C — OH Ester O || R — C — O — R’ Amine R’ | R — N — R’’

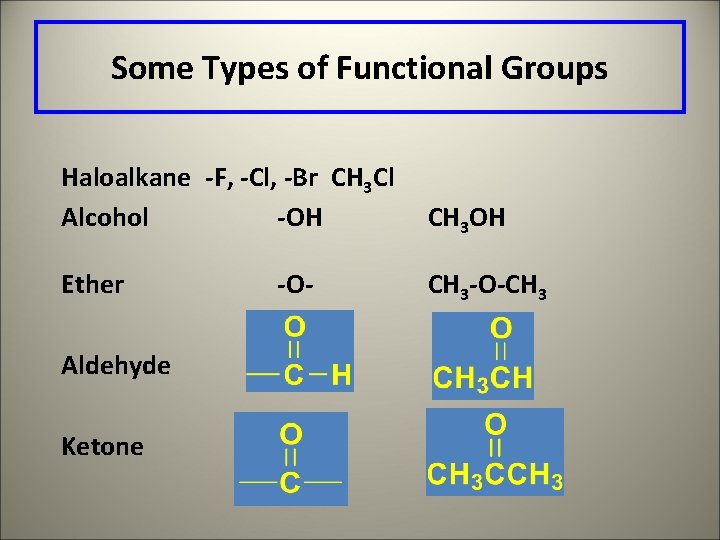
Some Types of Functional Groups Haloalkane -F, -Cl, -Br CH 3 Cl Alcohol -OH CH 3 OH Ether CH 3 -O-CH 3 Aldehyde Ketone -O-

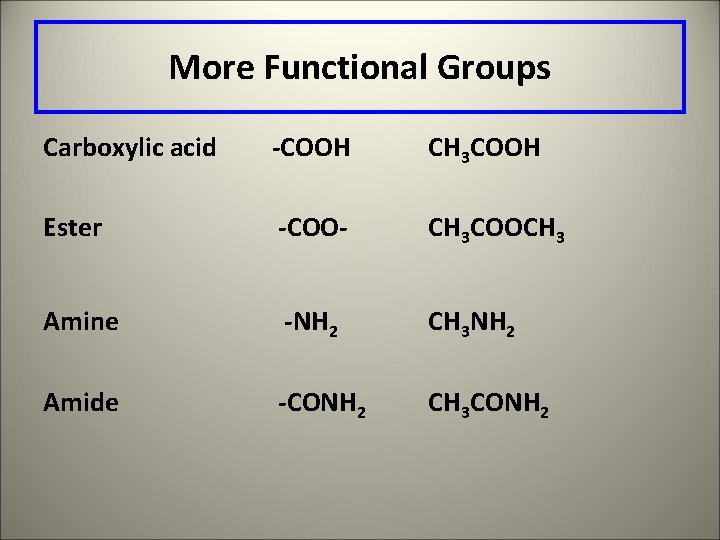
More Functional Groups Carboxylic acid -COOH CH 3 COOH Ester -COO- CH 3 COOCH 3 Amine -NH 2 CH 3 NH 2 Amide -CONH 2 CH 3 CONH 2

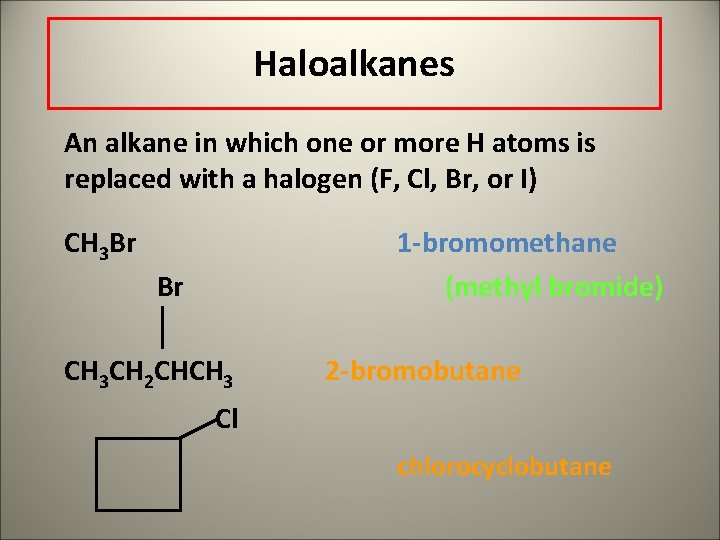
Haloalkanes An alkane in which one or more H atoms is replaced with a halogen (F, Cl, Br, or I) CH 3 Br 1 -bromomethane (methyl bromide) Br CH 3 CH 2 CHCH 3 2 -bromobutane Cl chlorocyclobutane

Topic 27 Hydrocarbons: Basic Concepts Organic Compounds Containing Halogens • Any organic compound that contains a halogen substituent is called a halocarbon. • If you replace any of the hydrogen atoms in an alkane with a halogen atom, you form an alkyl halide.

Topic 27 Hydrocarbons: Basic Concepts Organic Compounds Containing Halogens • An alkyl halide is an organic compound containing a halogen atom covalently bonded to an aliphatic carbon atom. • The first four halogens— fluorine, chlorine, bromine, and iodine—are found in many organic compounds.

Topic 27 Hydrocarbons: Basic Concepts Organic Compounds Containing Halogens • For example, chloromethane is the alkyl halide formed when a chlorine atom replaces one of methane’s four hydrogen atoms.

Topic 27 Hydrocarbons: Basic Concepts Organic Compounds Containing Halogens • An aryl halide is an organic compound containing a halogen atom bonded to a benzene ring or other aromatic group. • The structural formula for an aryl halide is created by first drawing the aromatic structure and then replacing its hydrogen atoms with the halogen atoms specified.

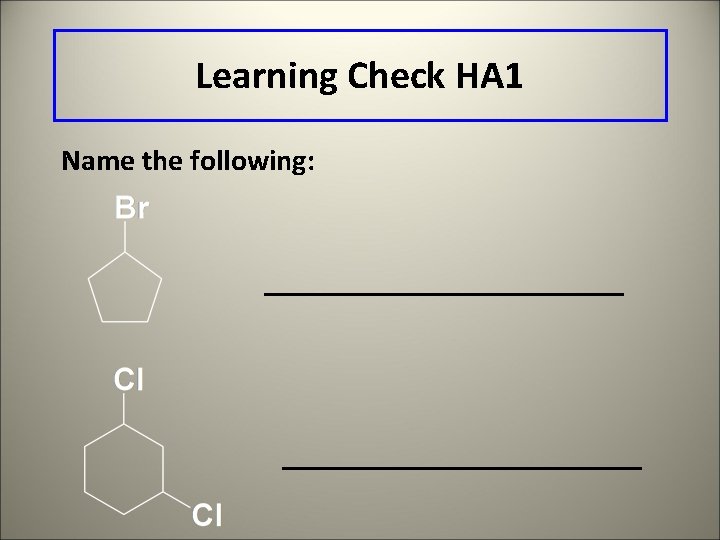
Learning Check HA 1 Name the following:

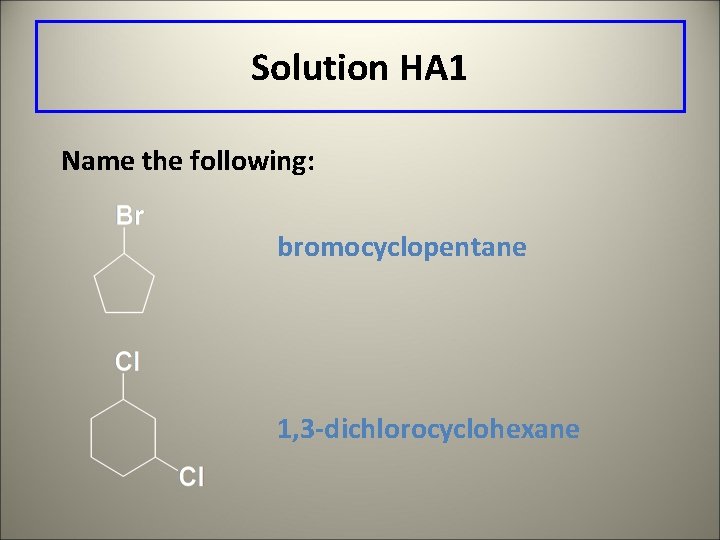
Solution HA 1 Name the following: bromocyclopentane 1, 3 -dichlorocyclohexane

Substituents List other attached atoms or group in alphabetical order Br = bromo, Cl = chloro Cl Br CH 3 CHCH 2 CH 2 CH 3 4 -bromo-2 -chloroheptane

Learning Check HA 2 The name of this compound is: Cl CH 3 CH 2 CHCH 3 1) 2, 4 -dimethylhexane 2) 3 -chloro-5 -methylhexane 3) 4 -chloro-2 -methylhexane

Solution HA 2 The name of this compound is: Cl CH 3 CH 2 CHCH 3 3) 4 -chloro-2 -methylhexane

Haloalkanes as Anesthetics Halothane (Fluothane) F Cl F C C Br F H Fluothane is a haloalkane that is widely used as an anesthetic, which is a compound that decreases the ability of the nerve cells to conduct pain.

Ozone Layer Ozone layer Stratosphere (10 -30 miles Above Earth)

Chlorofuorocarbons (CFCs and the Ozone Layer =ozone O 3 layer absorbs most of the sun’s harmful radiation. =CFCs - chlorofluorocarbons - are depleting that ozone layer. =CFCs are used as Freons in refrigeration, air conditioning, and foam insulation. =Their use in spray cans is no longer allowed.

Alcohols • Alcohols have a OH group attached to a C somewhere along the chain. • The ending changes to “ol” • If there are 2 OH groups the are referred to as a “diol” • The OH group has to be identified using the IUPAC numbering system • Ex: CH 2 Cl. CHOHCHCl. CH 3 • 1, 3 -dichloro-2 -butanol

Topic 27 Hydrocarbons: Basic Concepts Alcohols • An oxygen-hydrogen group covalently bonded to a carbon atom is called a hydroxyl group ( —OH). • An organic compound in which a hydroxyl group replaces a hydrogen atom of a hydrocarbon is called an alcohol. • The general formula for an alcohol is ROH.

Naming Alcohols n A carbon compound that contain -OH (hydroxyl) group n In IUPAC name, the -e in alkane name is replaced with -ol. CH 4 methane CH 3 OH CH 3 methanol (methyl alcohol) ethane CH 3 CH 2 OH ethanol (ethyl alcohol)

Ethanol CH 3 CH 2 OH l Acts as a depressant l Kills or disables more people than any other drug l 12 -15 mg/d. L ethanol metabolized by a social drinkers in one hour l 30 mg/d. L ethanol metabolized by an alcoholic in one hour.

Alcohol in Some Products % Ethanol Product 50% 40% 15 -25% 12% 3 -9% Whiskey, rum, brandy Flavoring extracts Listerine, Nyquil, Scope Wine, Dristan, Cepacol Beer, Lavoris

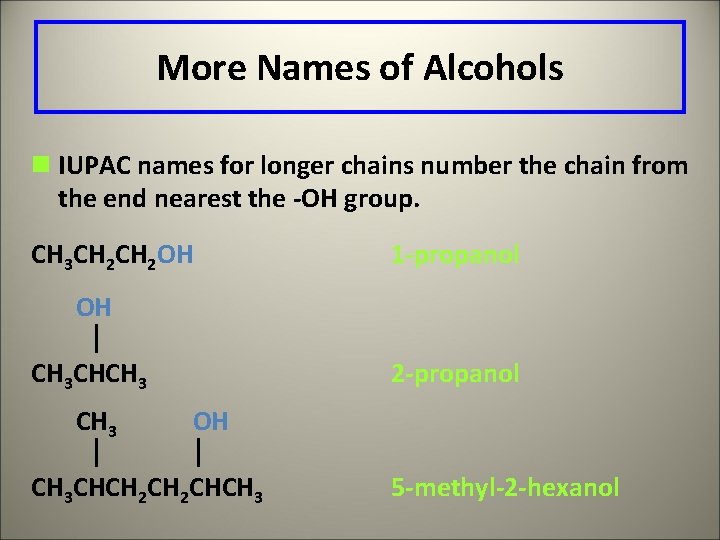
More Names of Alcohols n IUPAC names for longer chains number the chain from the end nearest the -OH group. CH 3 CH 2 OH 1 -propanol OH CH 3 CHCH 3 2 -propanol OH CH 3 CHCH 2 CHCH 3 5 -methyl-2 -hexanol

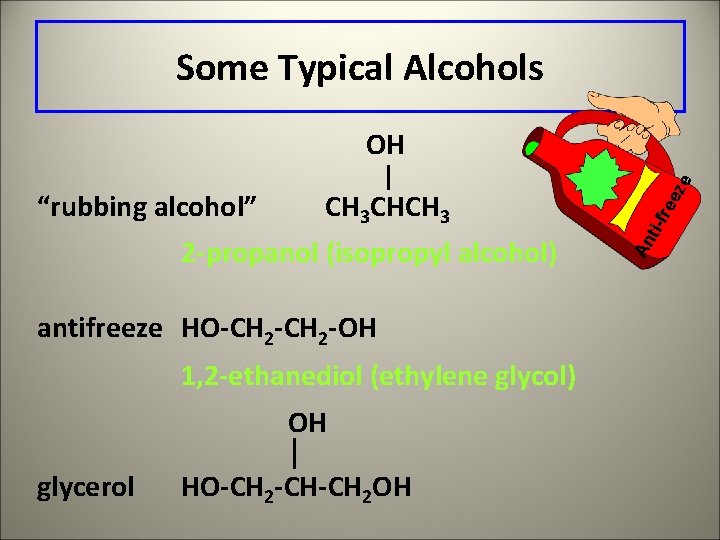
Some Typical Alcohols 2 -propanol (isopropyl alcohol) antifreeze HO-CH 2 -OH 1, 2 -ethanediol (ethylene glycol) OH glycerol HO-CH 2 -CH-CH 2 OH ree ti-f CH 3 CHCH 3 An “rubbing alcohol” ze OH

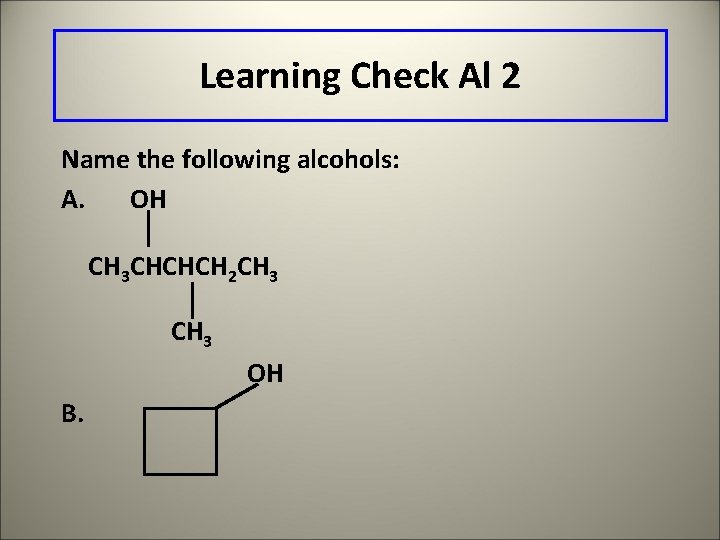
Learning Check Al 2 Name the following alcohols: A. OH CH 3 CHCHCH 2 CH 3 OH B.

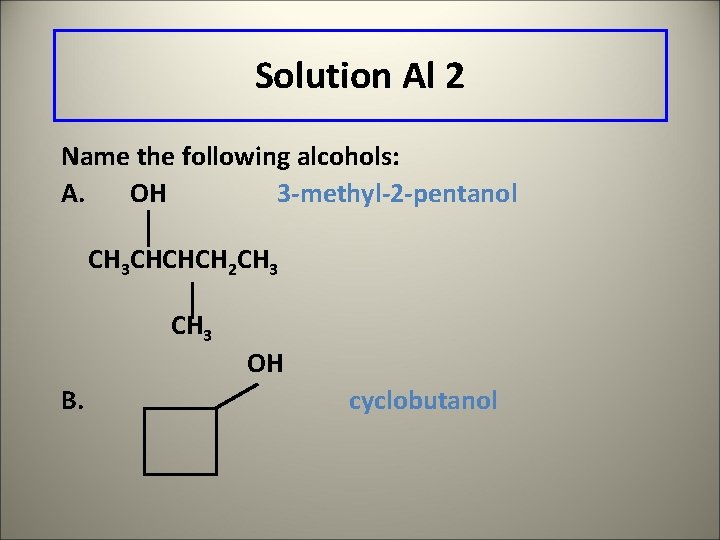
Solution Al 2 Name the following alcohols: A. OH 3 -methyl-2 -pentanol CH 3 CHCHCH 2 CH 3 B. OH cyclobutanol

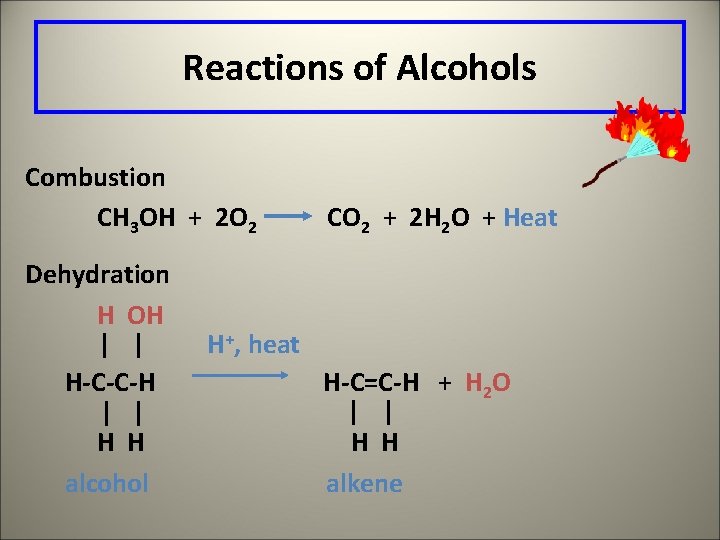
Reactions of Alcohols Combustion CH 3 OH + 2 O 2 Dehydration H OH CO 2 + 2 H 2 O + Heat H+, heat H-C-C-H H-C=C-H + H 2 O H H alcohol H H alkene

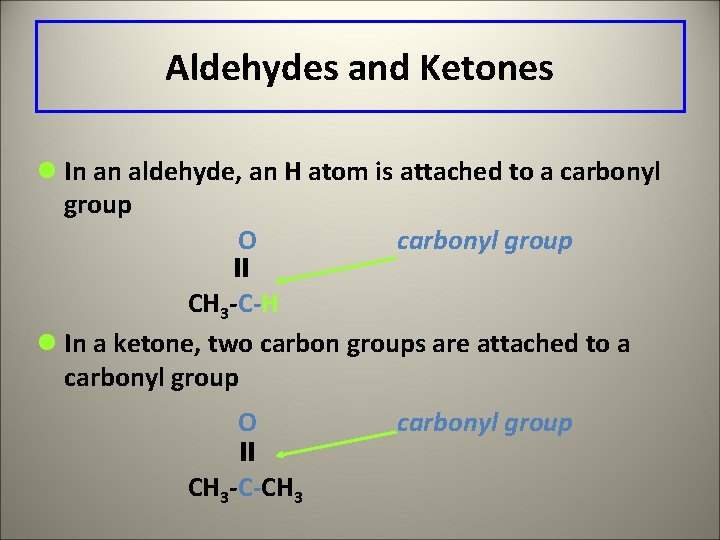
Aldehydes and Ketones l In an aldehyde, an H atom is attached to a carbonyl group O carbonyl group CH 3 -C-H l In a ketone, two carbon groups are attached to a carbonyl group O CH 3 -C-CH 3 carbonyl group

aldehydes • Aldehydes contain a CHO attached to the end of a carbon chain The C has a double bonded O and a single bonded H attached to it. Since it is at the end of a chain there is no need to number. • The ending changes to “al” • Ex: CH 3 CHICH 2 CHICHO • 2, 5 -diiodohexanal

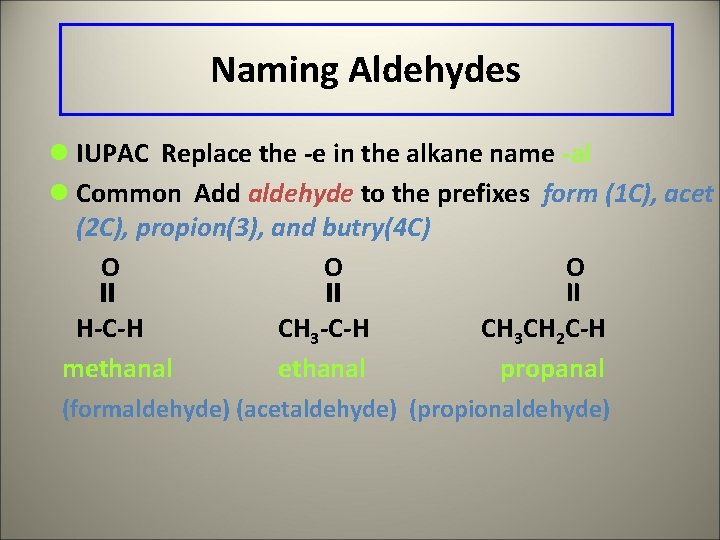
Naming Aldehydes l IUPAC Replace the -e in the alkane name -al l Common Add aldehyde to the prefixes form (1 C), acet (2 C), propion(3), and butry(4 C) O O O II H-C-H CH 3 CH 2 C-H methanal propanal (formaldehyde) (acetaldehyde) (propionaldehyde)

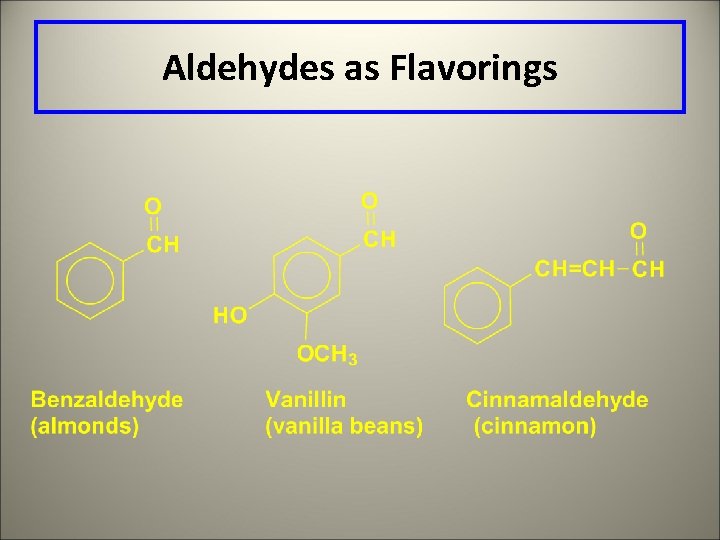
Aldehydes as Flavorings

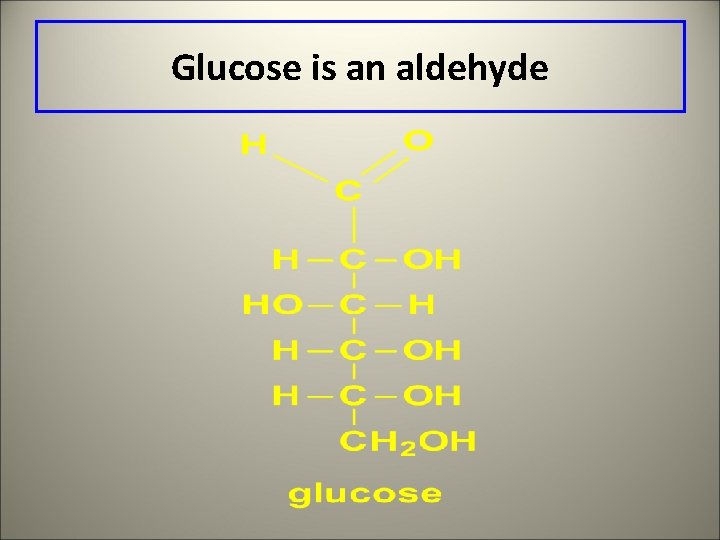
Glucose is an aldehyde

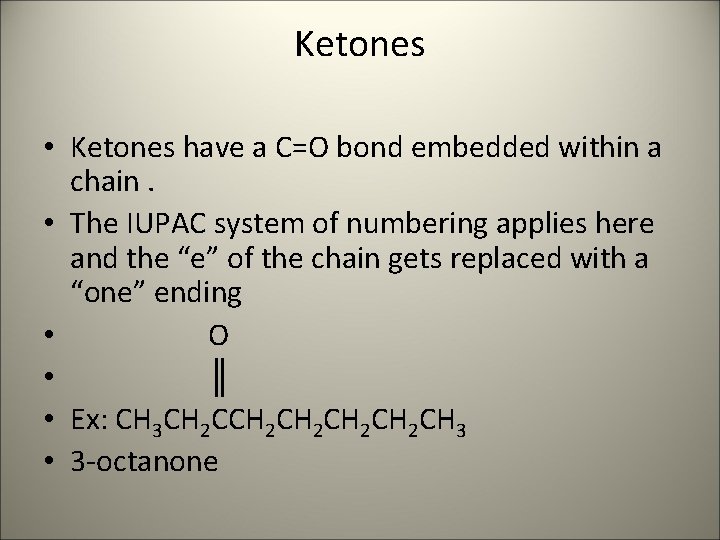
Ketones • Ketones have a C=O bond embedded within a chain. • The IUPAC system of numbering applies here and the “e” of the chain gets replaced with a “one” ending • O • ║ • Ex: CH 3 CH 2 CH 2 CH 2 CH 3 • 3 -octanone

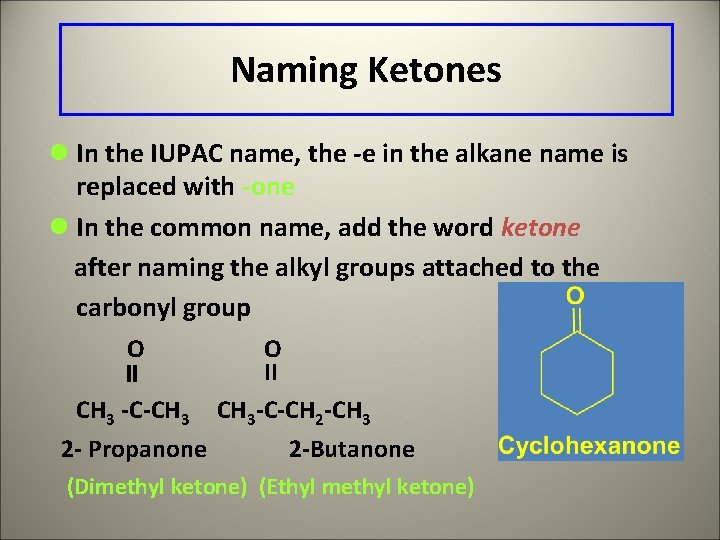
Naming Ketones l In the IUPAC name, the -e in the alkane name is replaced with -one l In the common name, add the word ketone after naming the alkyl groups attached to the carbonyl group O O II CH 3 -C-CH 3 -C-CH 2 -CH 3 2 - Propanone 2 -Butanone (Dimethyl ketone) (Ethyl methyl ketone)

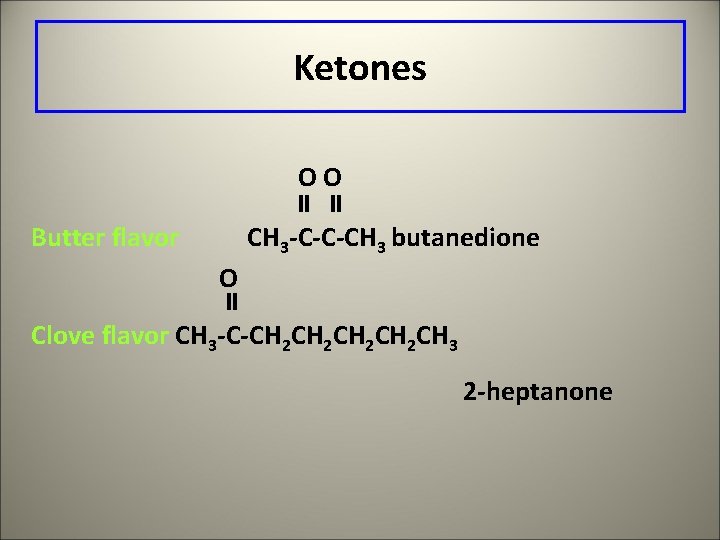
Ketones OO Butter flavor CH 3 -C-C-CH 3 butanedione O Clove flavor CH 3 -C-CH 2 CH 2 CH 3 2 -heptanone

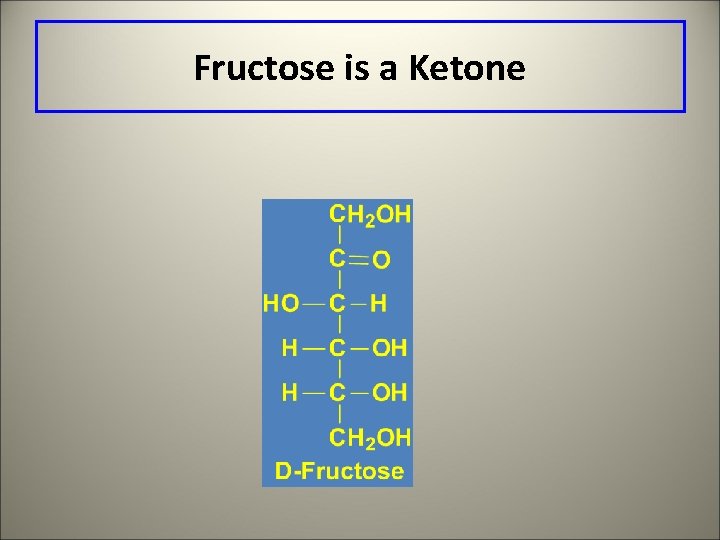
Fructose is a Ketone

Ketones as Hormones

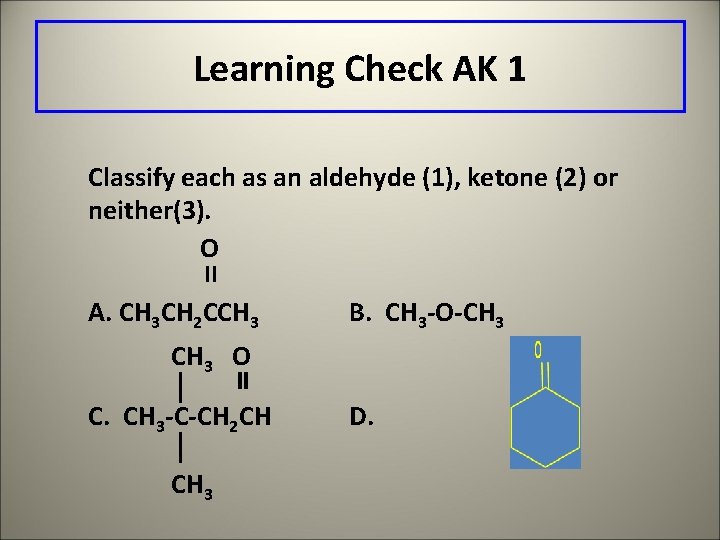
Learning Check AK 1 Classify each as an aldehyde (1), ketone (2) or neither(3). O II A. CH 3 CH 2 CCH 3 B. CH 3 -O-CH 3 O C. CH 3 -C-CH 2 CH CH 3 D.

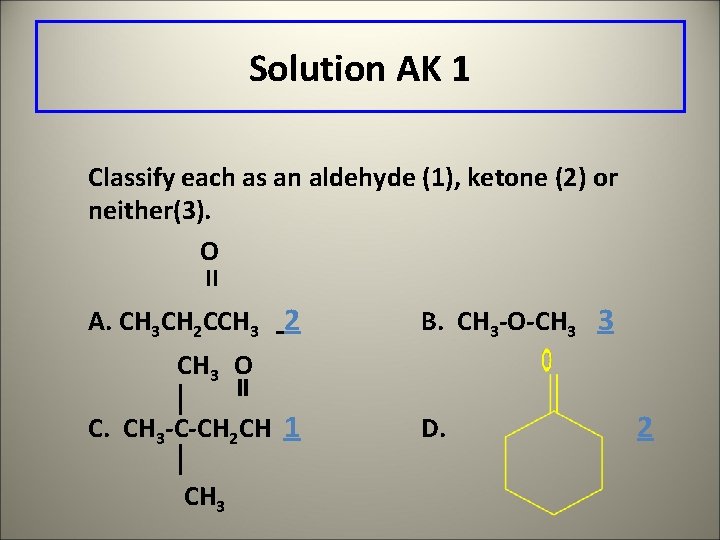
Solution AK 1 Classify each as an aldehyde (1), ketone (2) or neither(3). O II A. CH 3 CH 2 CCH 3 2 B. CH 3 -O-CH 3 3 CH 3 O C. CH 3 -C-CH 2 CH 1 CH 3 D. 2

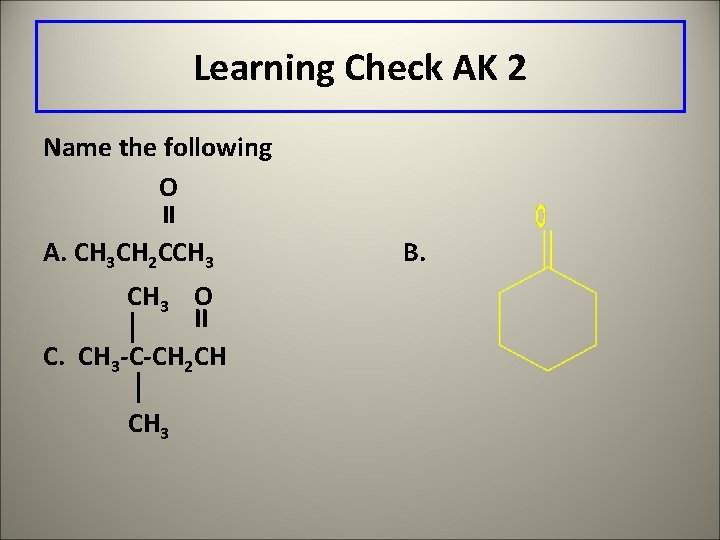
Learning Check AK 2 Name the following O A. CH 3 CH 2 CCH 3 O II C. CH 3 -C-CH 2 CH CH 3 B.

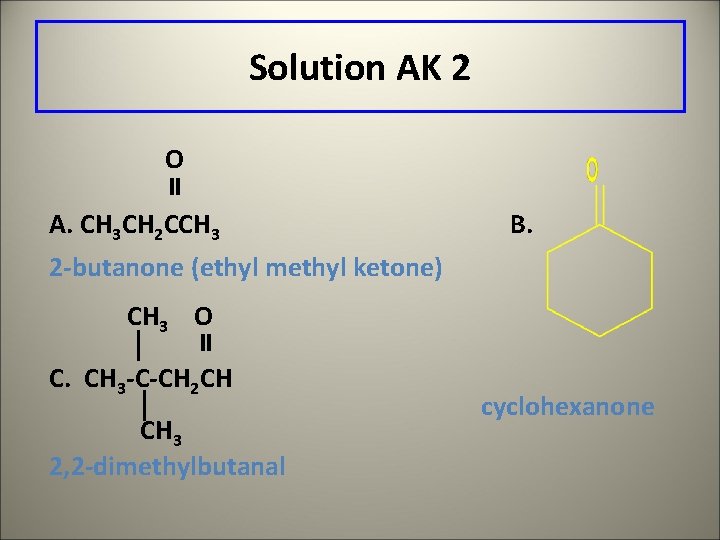
Solution AK 2 O A. CH 3 CH 2 CCH 3 B. 2 -butanone (ethyl methyl ketone) CH 3 O C. CH 3 -C-CH 2 CH CH 3 2, 2 -dimethylbutanal cyclohexanone

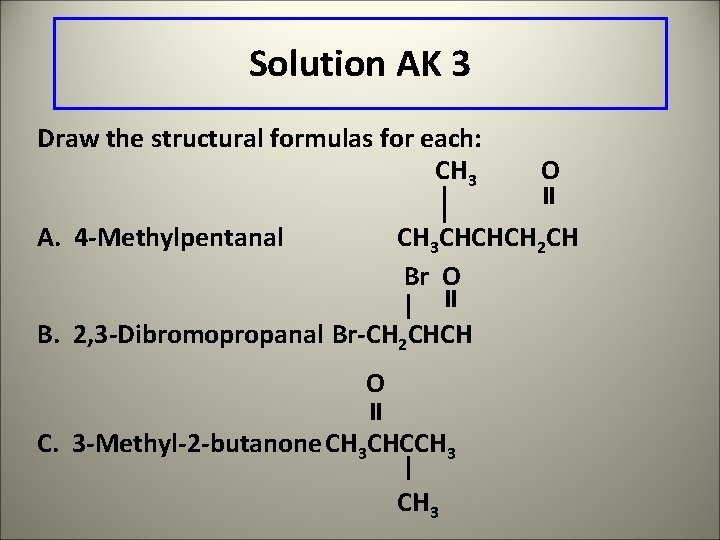
Learning Check AK 3 Draw the structural formulas for each: A. 3 -Methylpentanal B. 2, 3 -Dichloropropanal C. 3 -Methyl-2 -butanone

Solution AK 3 Draw the structural formulas for each: CH 3 A. 4 -Methylpentanal O CH 3 CHCHCH 2 CH Br O B. 2, 3 -Dibromopropanal Br-CH 2 CHCH O C. 3 -Methyl-2 -butanone CH 3 CHCCH 3

ethers • Have an O atom within the carbon chain itself. In other words C-O-C. • Components of the chain on both sides of the O have to be named. • Ethers have the general formula ROR′. • No numbering system is needed since the chain is broken. • The name remains the same just add the ending ether

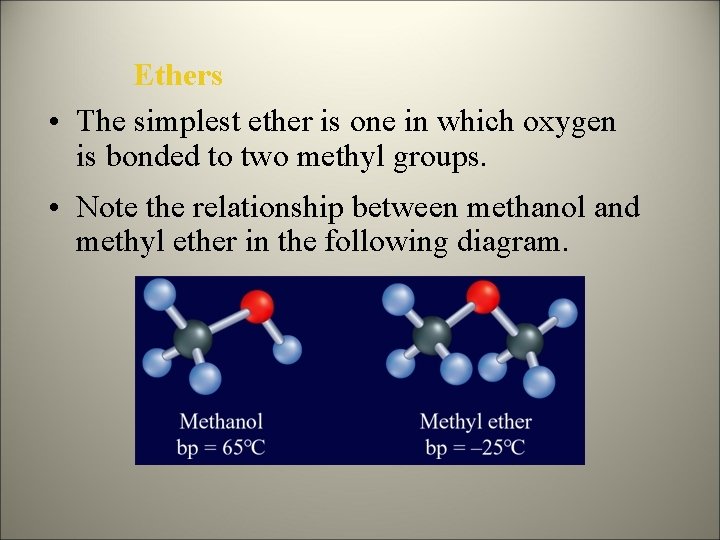
Ethers • The simplest ether is one in which oxygen is bonded to two methyl groups. • Note the relationship between methanol and methyl ether in the following diagram.

Ethers • Contain an -O- between two carbon groups • Simple ethers named from -yl names of the attached groups and adding ether. CH 3 -O-CH 3 dimethyl ether CH 3 -O-CH 2 CH 3 ethyl methyl ether

• Ex: CH 3 OCH 2 CH 2 CH 3 • Methyl-n-pentyl ether The “n” signifies that the carbons are in a straight chain with no branches.

Ethers as Anesthetics l Anesthetics inhibit pain signals to the brain l CH 3 CH 2 -O-CH 2 CH 3 used for over a century (Morton, 1846) l Causes nausea and is highly flammable l 1960 s developed nonflammable anesthetics Cl F F Cl F H H-C-C-O-C-H F F F Ethane(enflurane) H-C-C-O-C-H HF H Penthrane

MTBE l Methyl tert-butyl ether CH 3 -O-C-CH 3 l Second in production or organic chemicals l Additive to improve gasoline performance

Carboxylic acids • These compounds end with a COOH group where the C has a double bonded O and a single bonded OH • The ending of the parent chain changes from “ane” etc. to “oic” and add the word acid • Ex: CH 3 CH 2 COOH • Butanoic acid

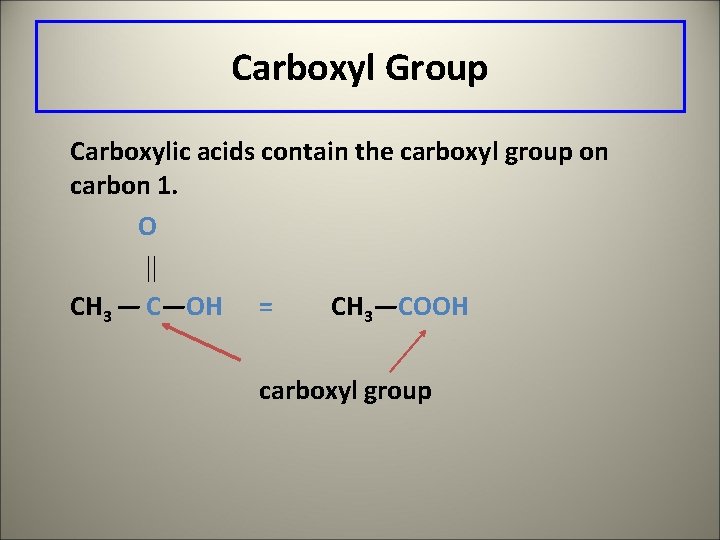
Carboxyl Group Carboxylic acids contain the carboxyl group on carbon 1. O CH 3 — C—OH = CH 3—COOH carboxyl group

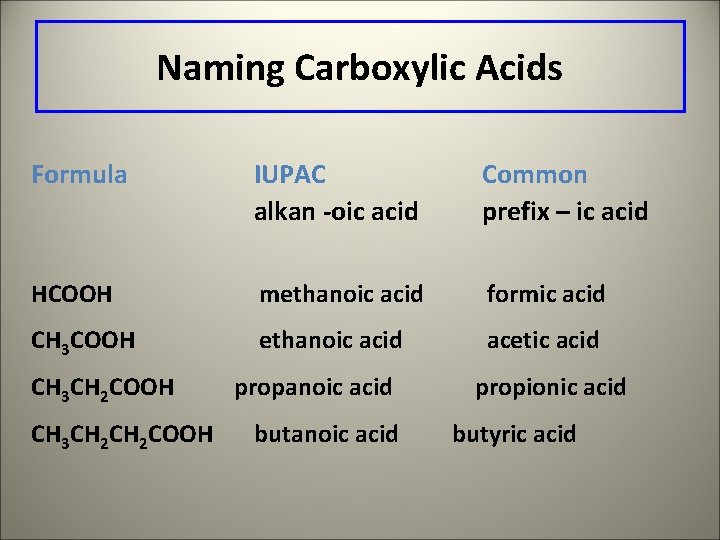
Naming Carboxylic Acids Formula IUPAC alkan -oic acid Common prefix – ic acid HCOOH methanoic acid formic acid CH 3 COOH ethanoic acid acetic acid CH 3 CH 2 COOH propanoic acid butanoic acid propionic acid butyric acid

Naming Rules • Identify longest chain • (IUPAC) Number carboxyl carbon as 1 • • CH 3 | CH 3 — CH—CH 2 —COOH IUPAC 3 -methylbutanoic acid

Learning Check CA 1 Give IUPAC and common names: A. CH 3 COOH CH 3 | B. CH 3 CHCOOH

Solution CA 1 A. CH 3 COOH ethanoic acid; acetic acid CH 3 | B. CH 3 CHCOOH 2 -methylpropanoic acid;

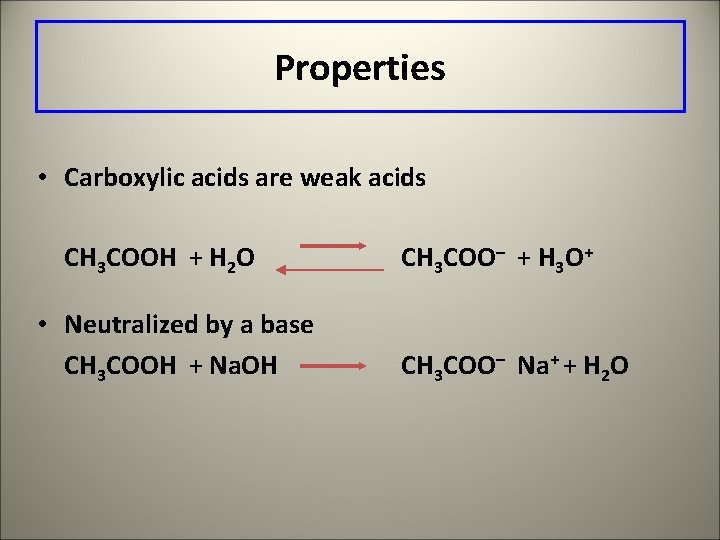
Properties • Carboxylic acids are weak acids CH 3 COOH + H 2 O • Neutralized by a base CH 3 COOH + Na. OH CH 3 COO– + H 3 O+ CH 3 COO– Na+ + H 2 O

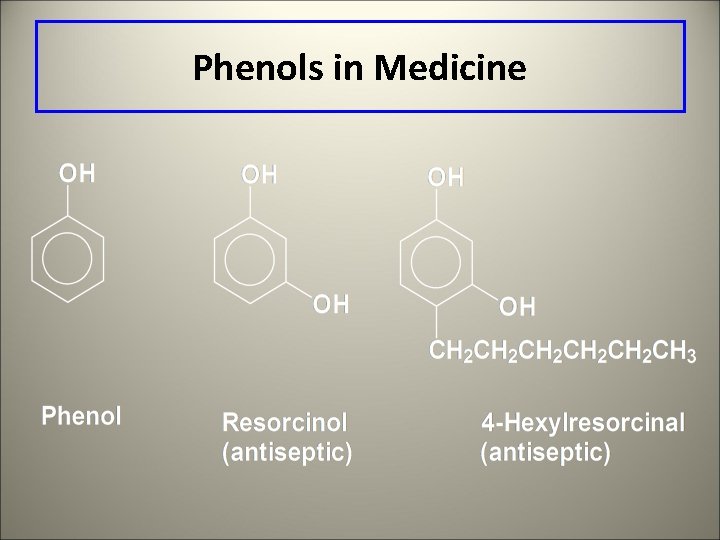
Phenols l IUPAC name for benzene with a hydroxyl group l Many are used as antiseptics and disinfectants

Phenols in Medicine

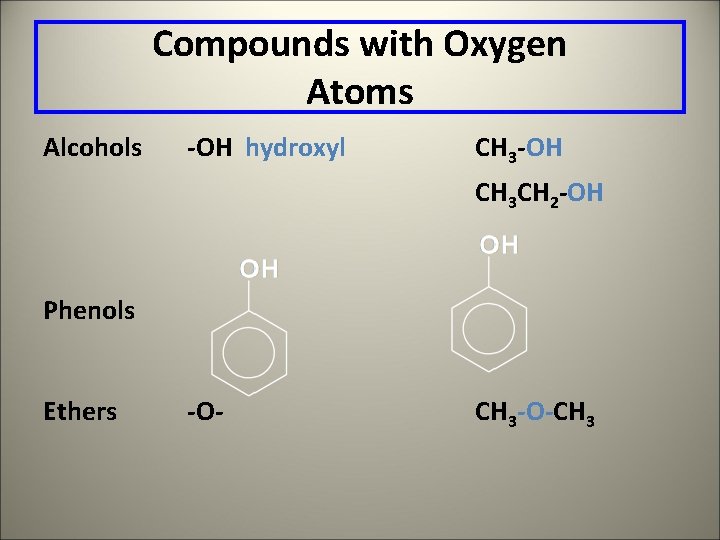
Compounds with Oxygen Atoms Alcohols -OH hydroxyl CH 3 -OH CH 3 CH 2 -OH Phenols Ethers -O- CH 3 -O-CH 3

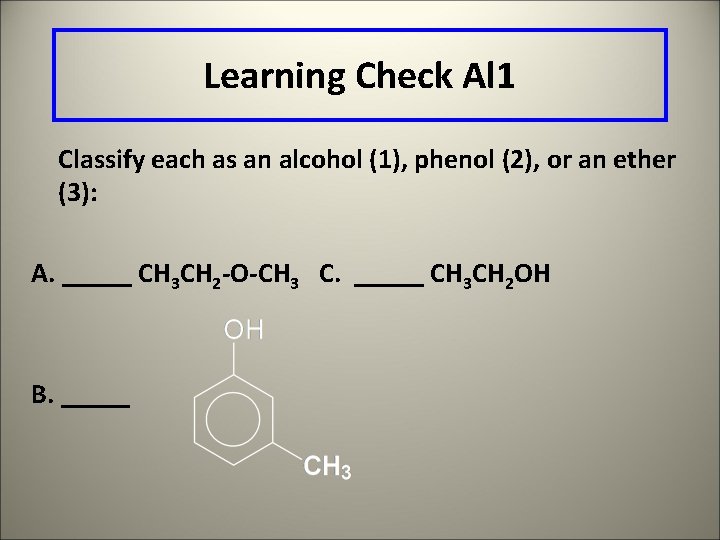
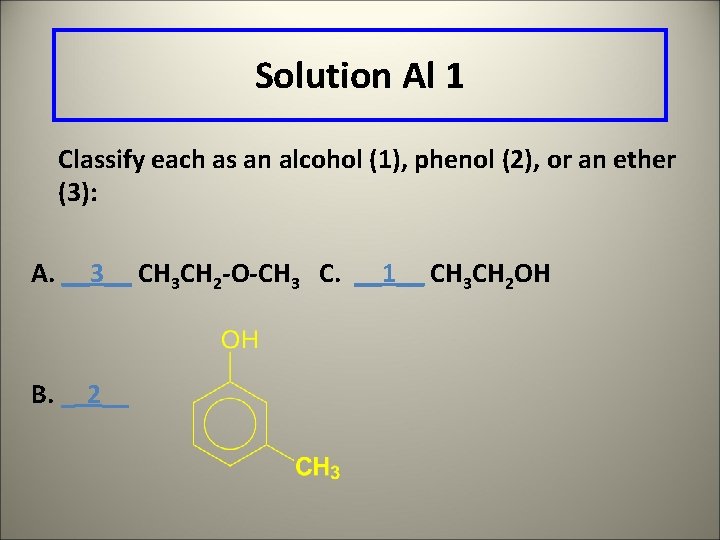
Learning Check Al 1 Classify each as an alcohol (1), phenol (2), or an ether (3): A. _____ CH 3 CH 2 -O-CH 3 C. _____ CH 3 CH 2 OH B. _____

Solution Al 1 Classify each as an alcohol (1), phenol (2), or an ether (3): A. __3__ CH 3 CH 2 -O-CH 3 C. __1__ CH 3 CH 2 OH B. _ 2__

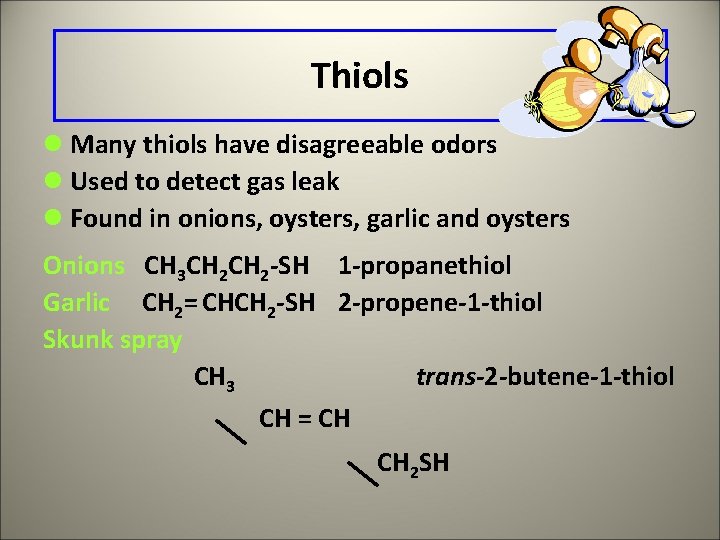
Thiols l Contain the functional group -SH l Named by adding thiol to the name of the longest carbon chain l Number the -SH group in longer chains CH 3 -SH methanethiol CH 3 -CH 2 SH ethanethiol SH CH 3 -CH-CH 3 2 -propanethiol

Thiols l Many thiols have disagreeable odors l Used to detect gas leak l Found in onions, oysters, garlic and oysters Onions CH 3 CH 2 -SH 1 -propanethiol Garlic CH 2= CHCH 2 -SH 2 -propene-1 -thiol Skunk spray CH 3 trans-2 -butene-1 -thiol CH = CH CH 2 SH

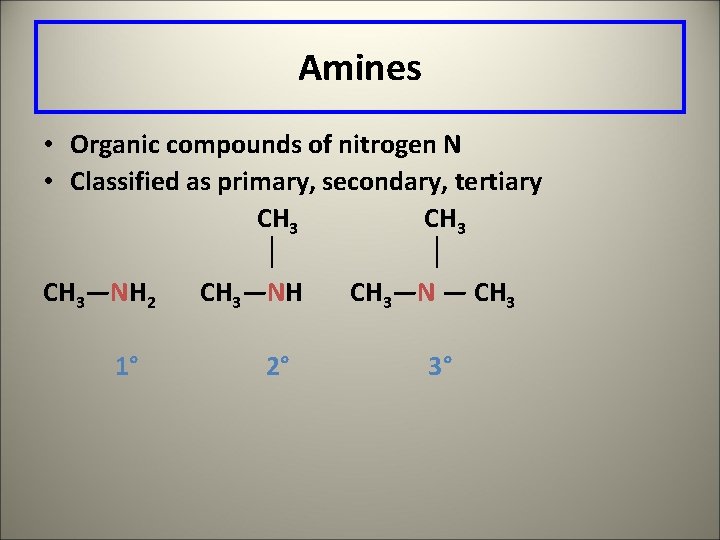
Amines • Organic compounds of nitrogen N • Classified as primary, secondary, tertiary CH 3—NH 2 CH 3—NH CH 3—N — CH 3 1° 2° 3°

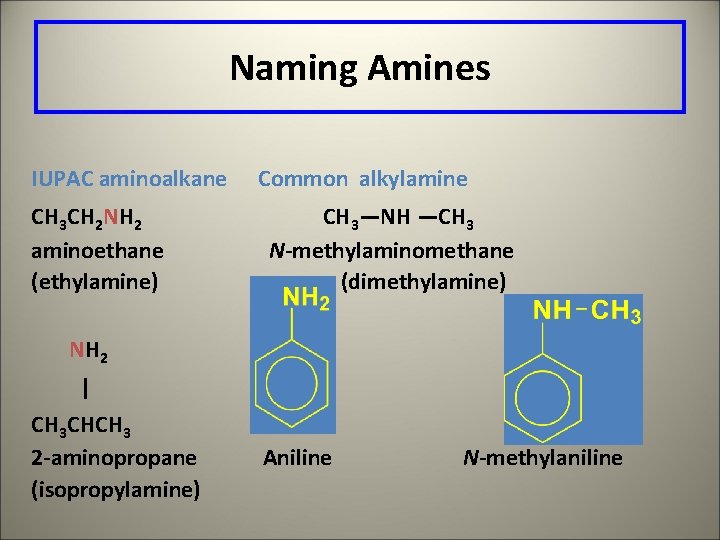
Naming Amines IUPAC aminoalkane CH 3 CH 2 NH 2 aminoethane (ethylamine) Common alkylamine CH 3—NH —CH 3 N-methylaminomethane (dimethylamine) NH 2 | CH 3 CHCH 3 2 -aminopropane (isopropylamine) Aniline N-methylaniline

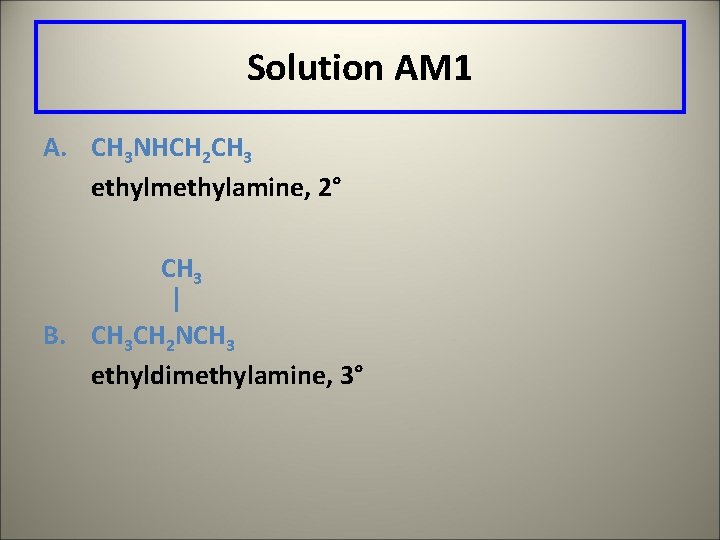
Learning Check AM 1 Give the common name and classify: A. CH 3 NHCH 2 CH 3 | B. CH 3 CH 2 NCH 3

Solution AM 1 A. CH 3 NHCH 2 CH 3 ethylmethylamine, 2° CH 3 | B. CH 3 CH 2 NCH 3 ethyldimethylamine, 3°

Alkaloids • Physiologically active nitrogen-containing compounds • Obtained from plants • Used as anesthetics, antidepressants, and stimulants • Many are addictive

Nicotine

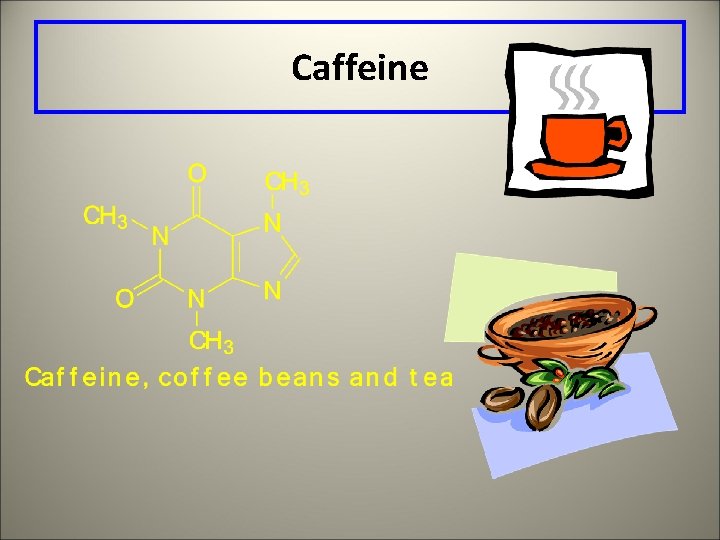
Caffeine

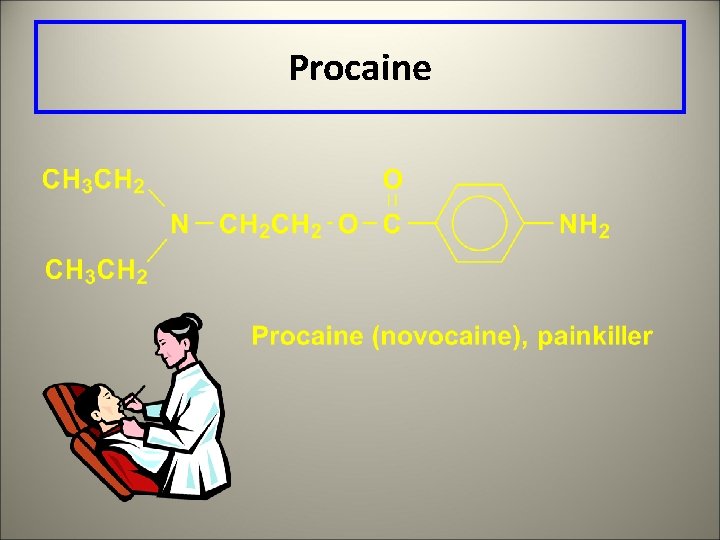
Procaine

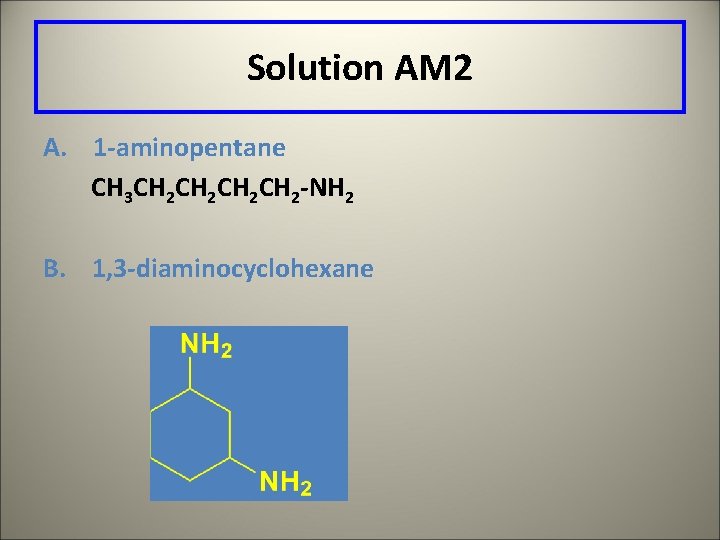
Leaning Check AM 2 Write a structural formula for A. 2 -aminopentane B. 1, 3 -diaminocyclohexane

Solution AM 2 A. 1 -aminopentane CH 3 CH 2 CH 2 -NH 2 B. 1, 3 -diaminocyclohexane

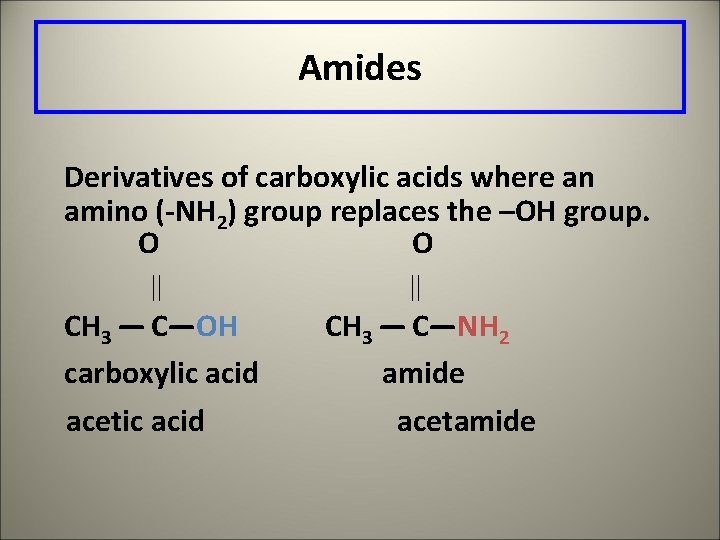
Amides Derivatives of carboxylic acids where an amino (-NH 2) group replaces the –OH group. O O CH 3 — C—OH carboxylic acid CH 3 — C—NH 2 amide acetic acid acetamide

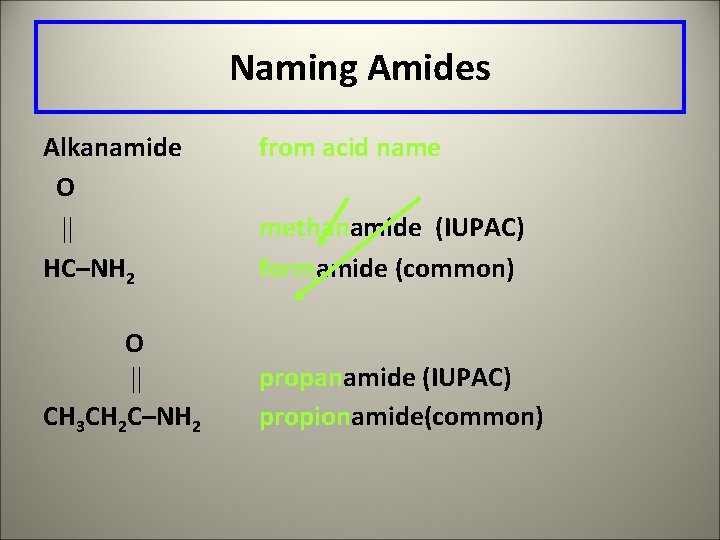
Naming Amides Alkanamide O HC–NH 2 O CH 3 CH 2 C–NH 2 from acid name methanamide (IUPAC) formamide (common) propanamide (IUPAC) propionamide(common)

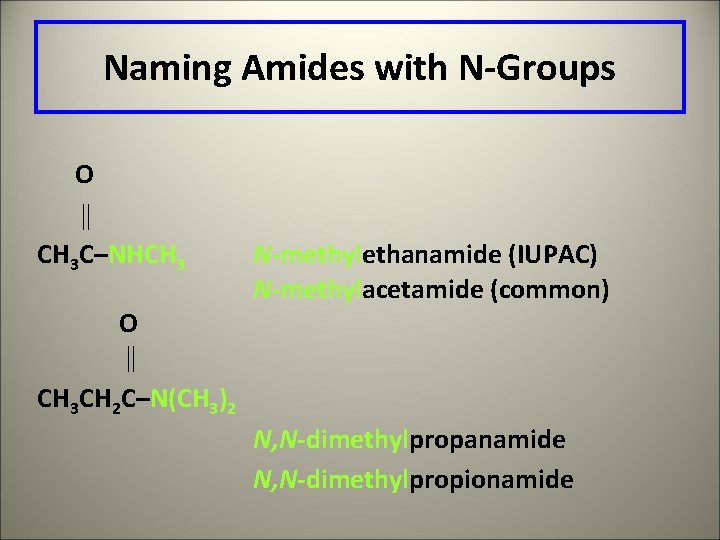
Naming Amides with N-Groups O CH 3 C–NHCH 3 O CH 3 CH 2 C–N(CH 3)2 N-methylethanamide (IUPAC) N-methylacetamide (common) N, N-dimethylpropanamide N, N-dimethylpropionamide

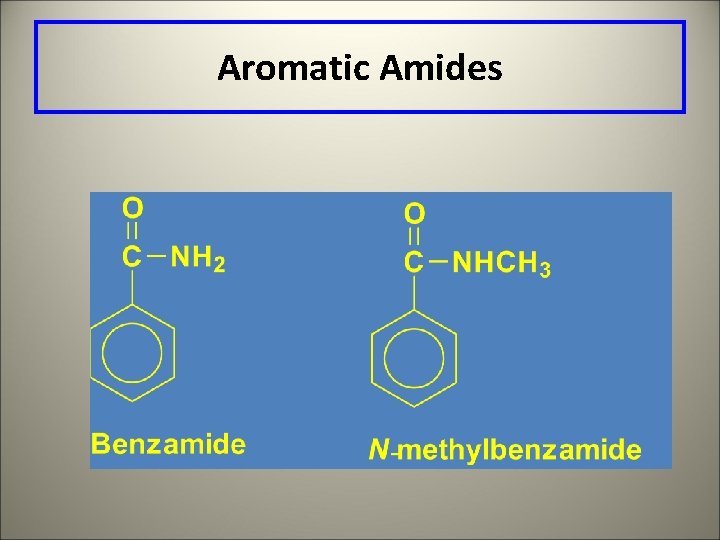
Aromatic Amides

Learning Check AM 3 Name the following amides: O A. CH 3 CH 2 C–NH 2 B. O CH 3 C–N(CH 2 CH 3)2

Solution AM 3 O A. CH 3 CH 2 C–NH 2 butanamide; butryamide (common) O B. CH 3 C–N(CH 2 CH 3)2 N, N-diethylethanamide; N, N-diethylacetamide

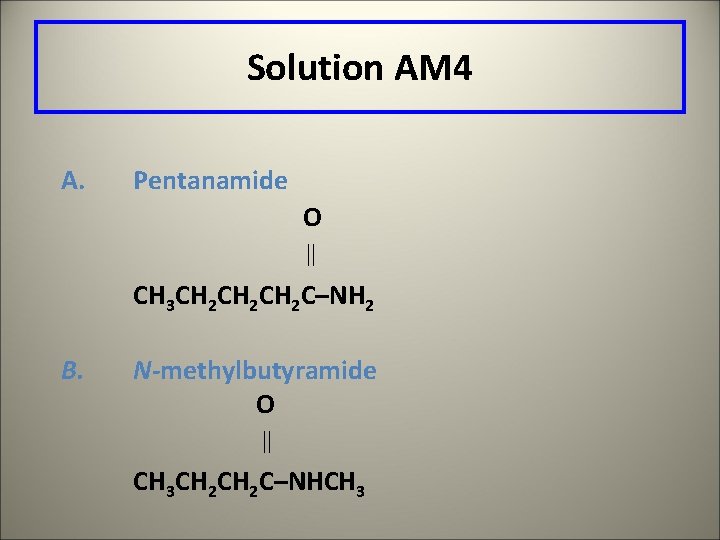
Learning Check AM 4 Draw the structures of A. Pentanamide B. N-methylbutyramide

Solution AM 4 A. Pentanamide O CH 3 CH 2 CH 2 C–NH 2 B. N-methylbutyramide O CH 3 CH 2 C–NHCH 3

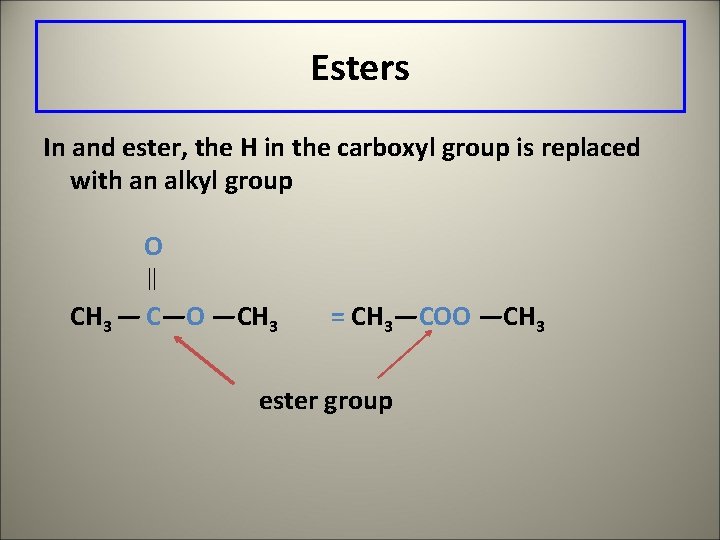
Esters In and ester, the H in the carboxyl group is replaced with an alkyl group O CH 3 — C—O —CH 3 = CH 3—COO —CH 3 ester group

Esters in Plants Esters give flowers and fruits their pleasant fragances and flavors.

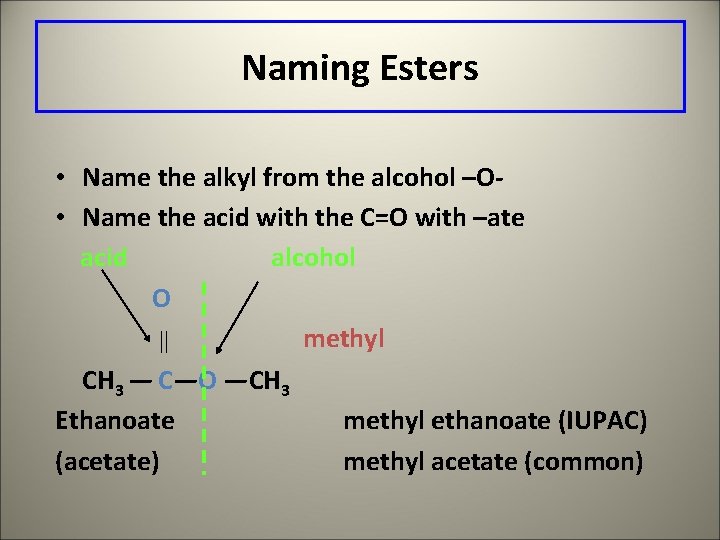
Naming Esters • Name the alkyl from the alcohol –O • Name the acid with the C=O with –ate acid alcohol O methyl CH 3 — C—O —CH 3 Ethanoate methyl ethanoate (IUPAC) (acetate) methyl acetate (common)

Some Esters and Their Names Flavor/Odor Raspberries HCOOCH 2 CH 3 ethyl methanoate (IUPAC) ethyl formate (common) Pineapples CH 3 CH 2 COOCH 2 CH 3 ethyl butanoate (IUPAC) ethyl butyrate (common)

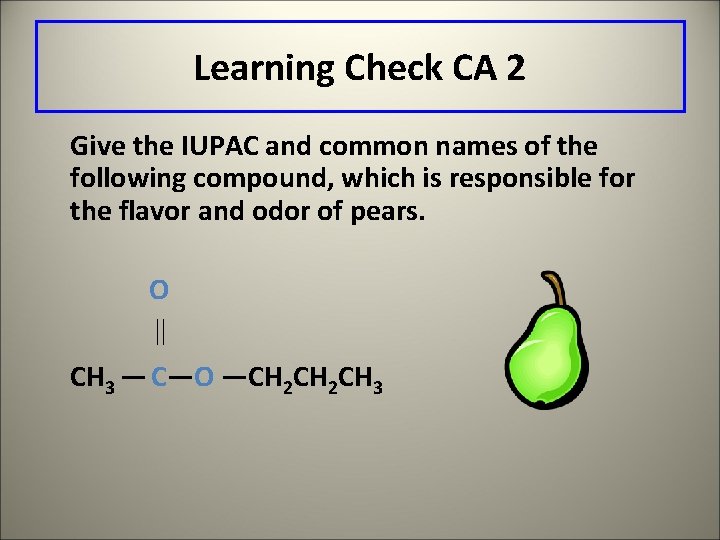
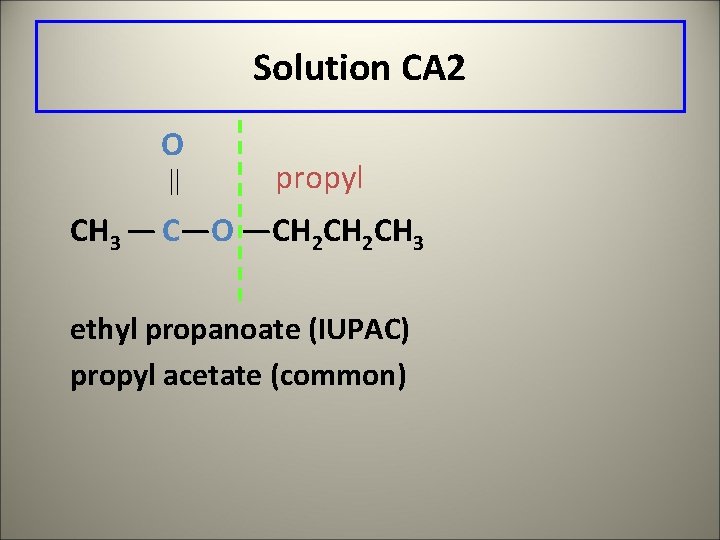
Learning Check CA 2 Give the IUPAC and common names of the following compound, which is responsible for the flavor and odor of pears. O CH 3 — C—O —CH 2 CH 3

Solution CA 2 O propyl CH 3 — C—O —CH 2 CH 3 ethyl propanoate (IUPAC) propyl acetate (common)

Learning Check CA 3 Draw the structure of the following compounds: A. 3 -bromobutanoic acid B. Ethyl propionoate

Isomers =Same molecular formula =Same number and types of atoms =Different arrangement of atoms

isomers • Isomers- al the possible spatial arrangements for a given hydrocarbon. • Follow these steps= • 1. start with the longest straight chain. • 2. shorten the chain by one carbon, and add that extra carbon as a methyl group in all the possible positions starting from the left and moving right.

• 3. check each time to make sure the names don’t repeat. • 4. shorten the chain by 2 carbons. Add each carbon separately as a methyl to all the possible places. Keep your numbering system correct. Then add the 2 carbons as an ethyl group following the same pattern. • Ex: Try heptane and octane

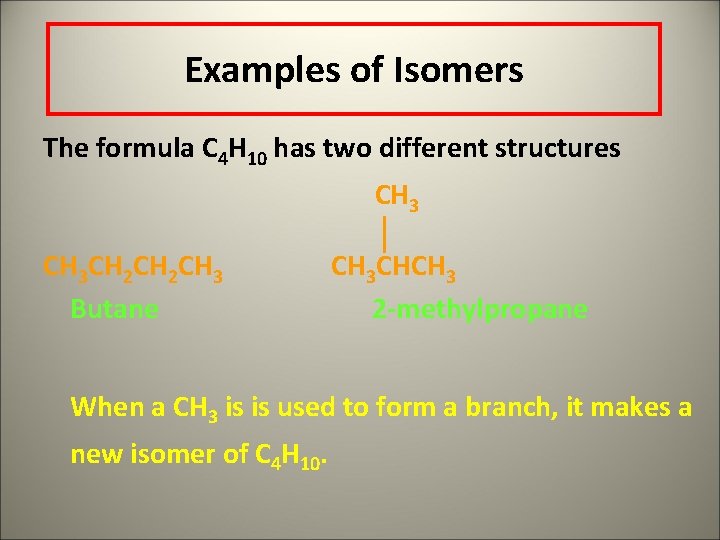
Examples of Isomers The formula C 4 H 10 has two different structures CH 3 CH 2 CH 3 Butane CH 3 CHCH 3 2 -methylpropane When a CH 3 is is used to form a branch, it makes a new isomer of C 4 H 10.

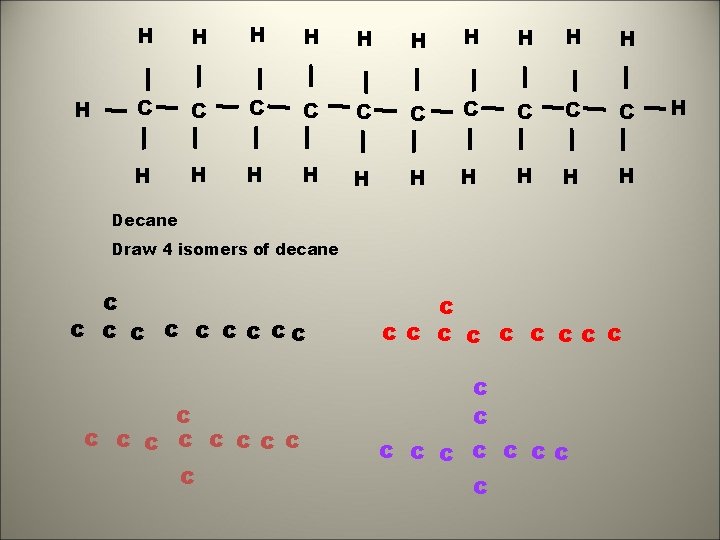
H H H C C C C C H H H H H Decane Draw 4 isomers of decane c c c c ccc c ccc c c c cc c H

Learning Check Alk 6 Write 3 isomers of C 5 H 12 and name each.

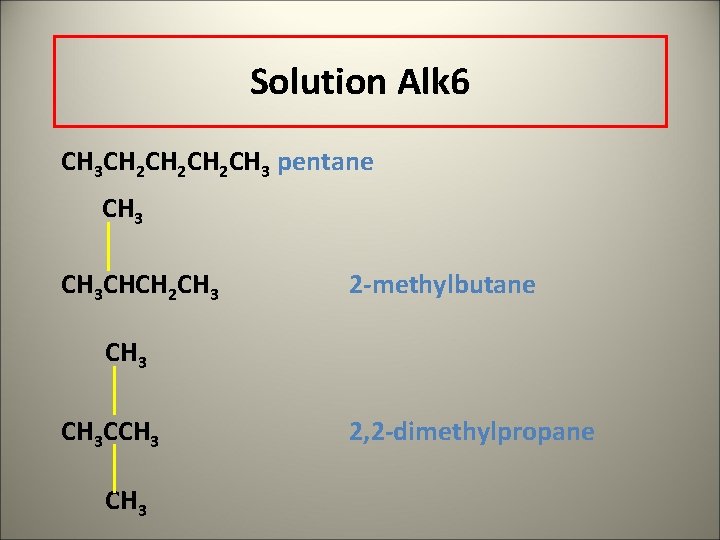
Solution Alk 6 CH 3 CH 2 CH 2 CH 3 pentane CH 3 CHCH 2 CH 3 2 -methylbutane CH 3 CCH 3 2, 2 -dimethylpropane

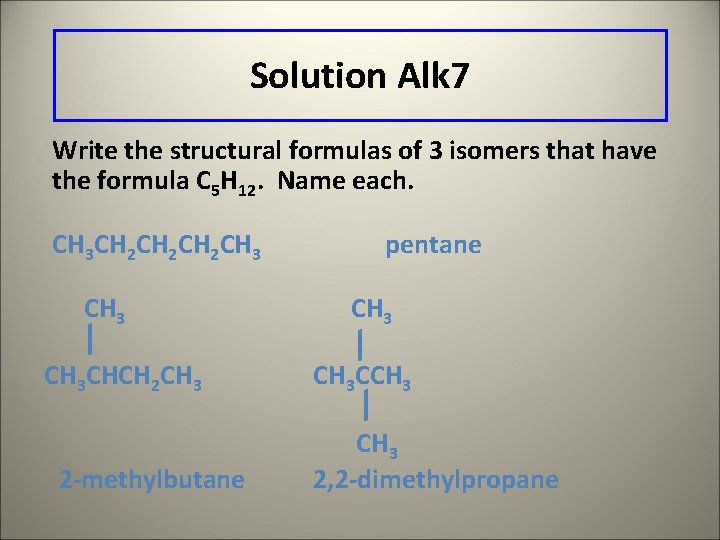
Learning Check Alk 7 Write the structural formulas of 3 isomers that have the formula C 5 H 12. Name each.

Solution Alk 7 Write the structural formulas of 3 isomers that have the formula C 5 H 12. Name each. CH 3 CH 2 CH 2 CH 3 CHCH 2 CH 3 2 -methylbutane pentane CH 3 CCH 3 2, 2 -dimethylpropane

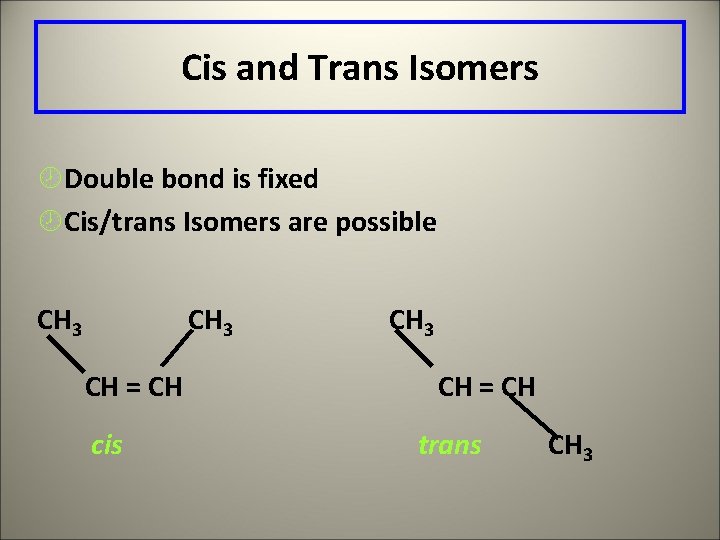
Cis trans isomers • This also occurs around the double or triple bonds in straight chains. • Ex: C 4 H 8 has 4 isomers

Cis and Trans Isomers ¾Double bond is fixed ¾Cis/trans Isomers are possible CH 3 CH = CH cis CH 3 CH = CH trans CH 3

1 -butene Cis-2 -butene Trans-2 -butene 2 -methylpropene ( isobutene) Cis is also called the Z isomer for the German word zusammen = together • Trans are also called the E isomer for the word entegen = opposite. • • •

Draw all the isomers • For – C 5 H 10 and C 6 H 12 and name them include all cis /trans isomers

conformations • 3 dimensional molecular structures interrelated by rotations about the bonds. • Ex. Ethane C 2 H 6 • Can be described as eclipsed and staggered. • See handout

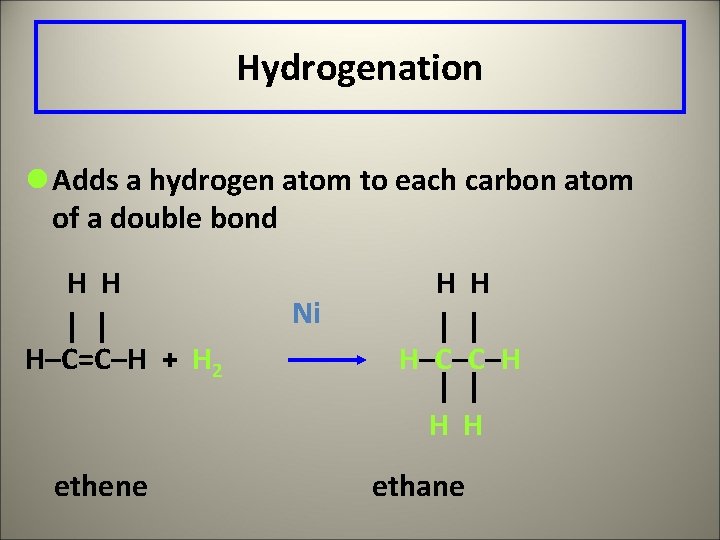
Hydrogenation l Adds a hydrogen atom to each carbon atom of a double bond H H H–C=C–H + H 2 Ni H H H–C–C–H H H ethene ethane

Products of Hydrogenation Adding H 2 to vegetable oils produces compounds with higher melting points l Margarines l Soft margarines l Shortenings (solid)

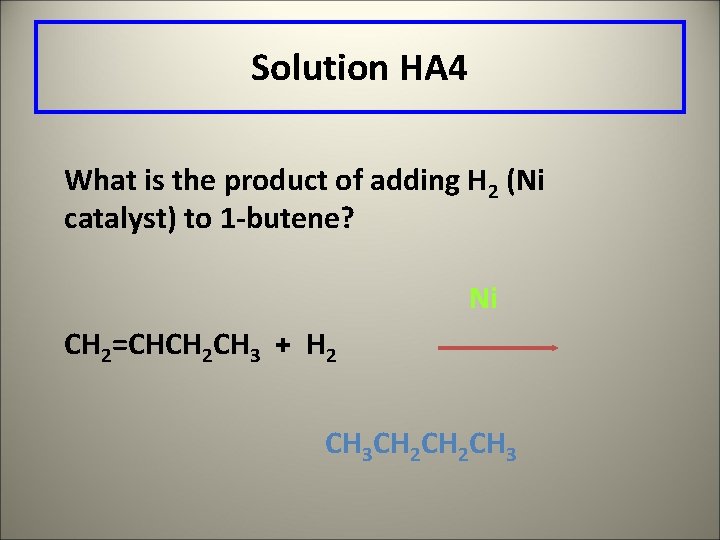
Learning Check HA 4 What is the product of adding H 2 (Ni catalyst) to 1 -butene?

Solution HA 4 What is the product of adding H 2 (Ni catalyst) to 1 -butene? Ni CH 2=CHCH 2 CH 3 + H 2 CH 3 CH 2 CH 3

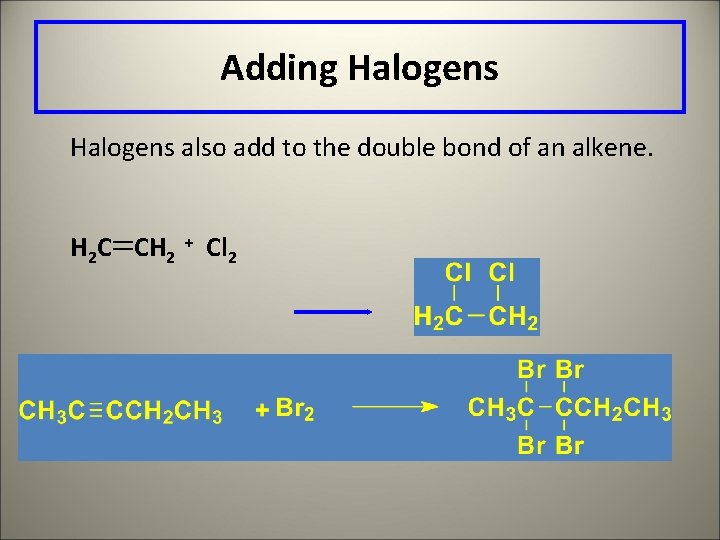
Adding Halogens also add to the double bond of an alkene. H 2 C CH 2 + Cl 2

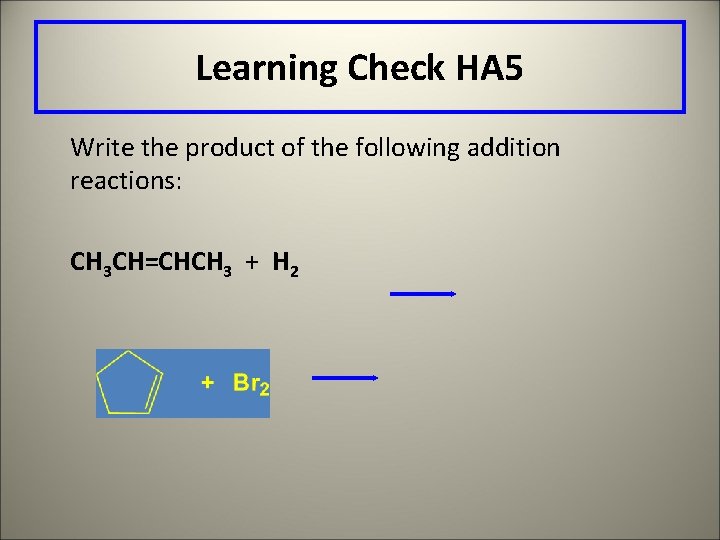
Learning Check HA 5 Write the product of the following addition reactions: CH 3 CH=CHCH 3 + H 2

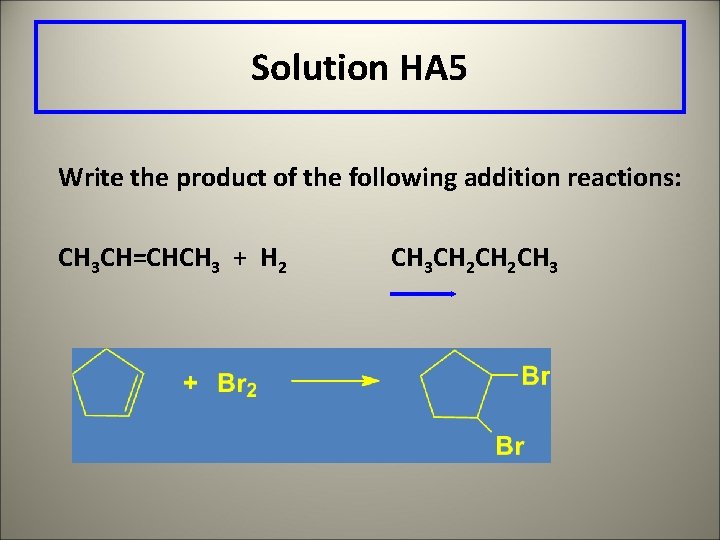
Solution HA 5 Write the product of the following addition reactions: CH 3 CH=CHCH 3 + H 2 CH 3 CH 2 CH 3

Unsaturated Fatty Acids l Fatty acids in vegetable oils are omega-6 acids (the first double bond occurs at carbon 6 counting from the methyl group) l A common omega-6 acid is linoleic acid CH 3 CH 2 CH 2 CH=CHCH 2 CH=CH(CH 2)7 COOH 6 linoleic acid, a fatty acid

Trans Fats u. In vegetable oils, the unsaturated fats usually contain cis double bonds. u. During hydrogenation, some cis double bonds are converted to trans double bonds (more stable) causing a change in the fatty acid structure u. If a label states “partially” or “fully hydrogenated”, the fats contain trans fatty acids.

Trans Fats u. In the US, it is estimated that 2 -4% of our total Calories is in the form of trans fatty acid. utrans fatty acids behave like saturated fatty acids in the body. u. Several studies reported that trans fatty acids raise LDL -cholesterol. Some studies also report that trans fatty acid lower HDL-cholesterol u. The trans fatty acids controversy will continue to be debated.

Fats and Atheroschlerosis l Inuit people of Alaska have a high fat diet and high blood cholesterol levels, but a very low occurrence of atherosclerosis and heart attacks. l Fat in the Intuit diet was primarily from fish such as salmon, tuna and herring rather than from land animals (as in the American diet).

Omega-3 Fatty Acids l Fatty acids in the fish oils are mostly the omega-3 type (first double bond occurs at the third carbon counting from the methyl group). l linolenic acid 18 carbon atoms CH 3 CH 2 CH=CHCH 2 CH=CH(CH 2)7 COOH l eicosapentaenoic acid (EPA) 20 carbon atoms CH 3 CH 2(CH=CHCH 2)5(CH 2)2 COOH

Learning Check HA 6 (1) Ture or (2) False A. ____ There are more unsaturated fats in vegetable oils. B. ____ Vegetable oils have more omega-3 oils than found in fish. C. ____ Hydrogenation of oils converts some cis-double bonds to trans- double bonds. D. ____ Animal fats have more saturated fats.