ORGANIC MOLECULES Organic compound a carbon containing compound

ORGANIC MOLECULES
Organic compound -a carbon containing compound in which one or more atoms of carbon are covalently bonded to atoms of
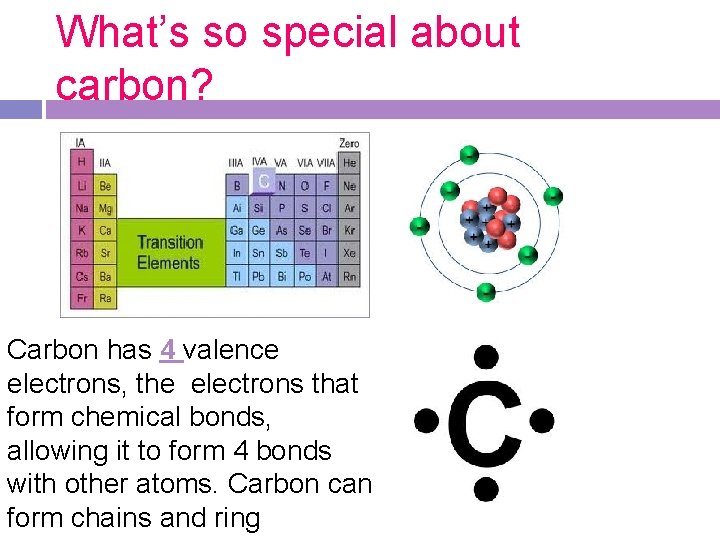
What’s so special about carbon? Carbon has 4 valence electrons, the electrons that form chemical bonds, allowing it to form 4 bonds with other atoms. Carbon can form chains and ring

The Chemistry of Carbon -Carbon can bond with itself and many elements, including hydrogen, oxygen, phosphorus, sulfur, and nitrogen to form the molecules of life. -Living organisms are made up of molecules that consist of carbon and these other elements.

Macromolecules
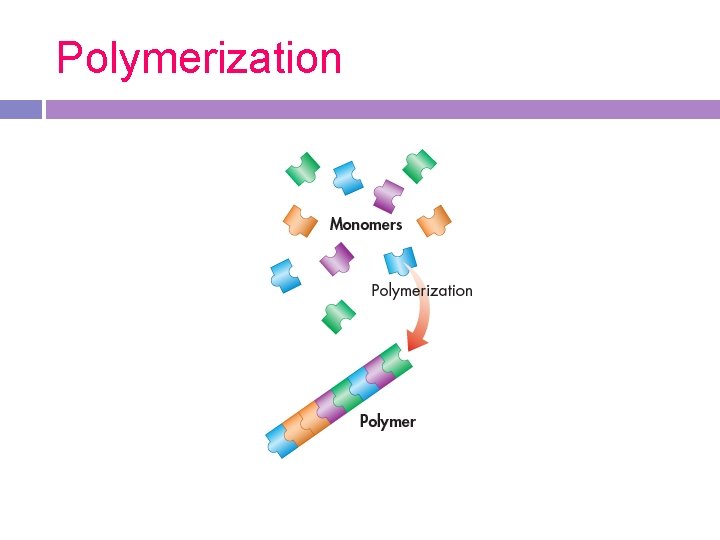
Polymerization

Monomers build polymers Polymer Monomer -“Small” units -“Many” units -mono means -complex organic molecules made one of monomers -poly means many

Ways to make or break polymers 1. dehydration synthesis (sometimes referred to as a condensation reaction) 2. hydrolysis

Dehydration synthesis -anabolic process by which two molecules are chemically bonded through a loss of water -a way to make polymers
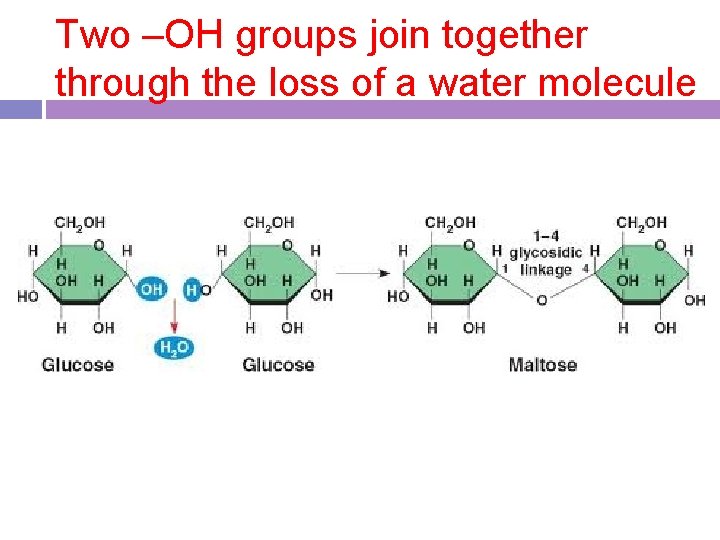
Two –OH groups join together through the loss of a water molecule
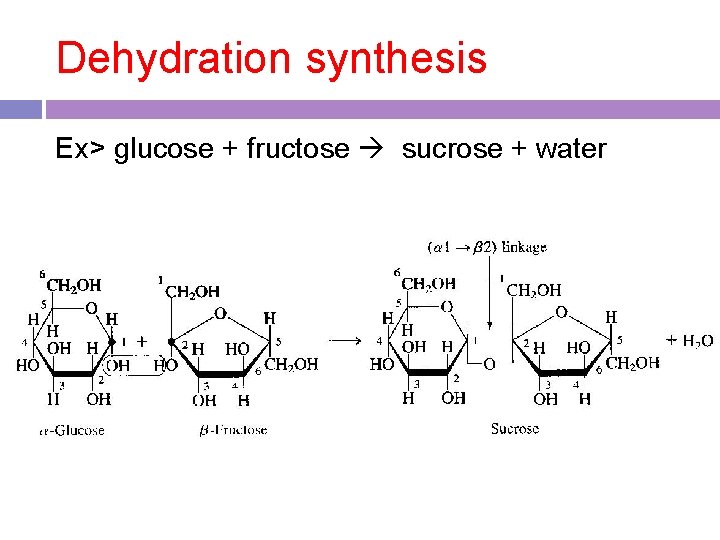
Dehydration synthesis Ex> glucose + fructose sucrose + water

Hydrolysis -a catabolic process by which the bond between monomers is broken by the addition of water -lysis=Greek for “to separate” -a way to break polymers down
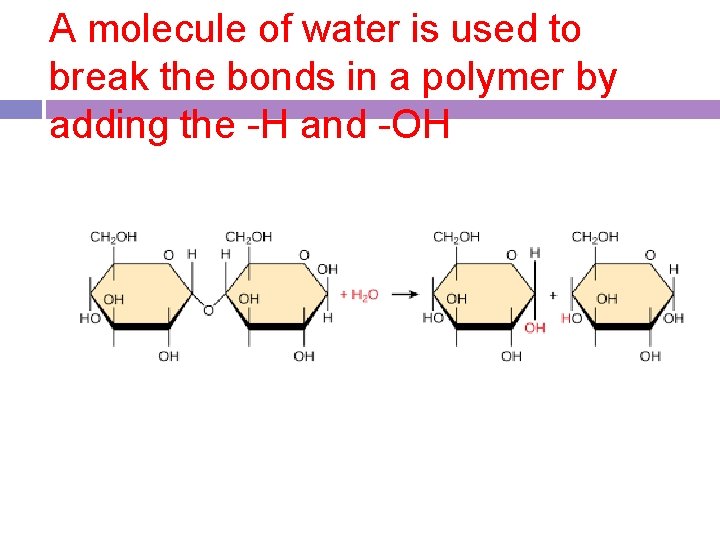
A molecule of water is used to break the bonds in a polymer by adding the -H and -OH

Macromolecules -Biochemists sort the macromolecules found in living things into groups based on their chemical composition.

1. Carbohydrates - building block (monomer) is a monosaccharide such as glucose -carbs provide energy for cell activities and have structural purposes in both plants and animals
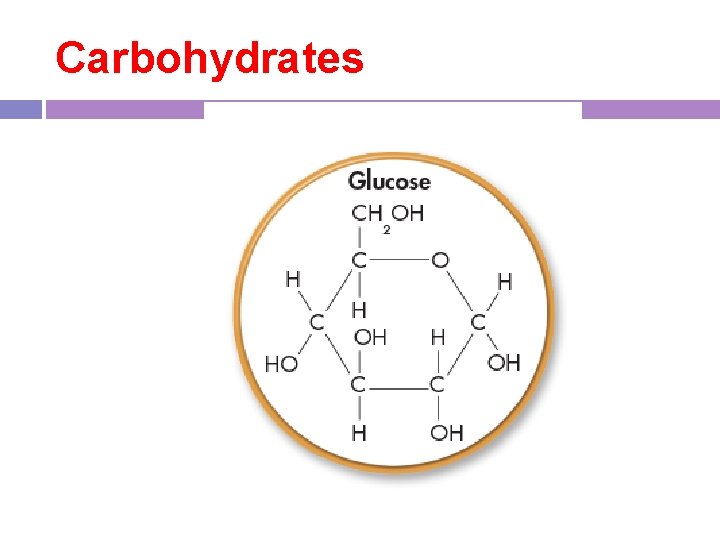
Carbohydrates

Carbohydrates -bonds are easily made and broken to store or release energy -known as the sugars ( look for suffix “-ose”) -ose -Composition: C, H, O
TYPES OF CARBOHYDRATES

1. monosaccharides -single or simple sugars -all have the same chemical formula (C 6 H 12 O 6), but a different arrangement of atoms -C, H, and O in a 1: 2: 1 ratio - a form of sugar plants and animals can immediately use
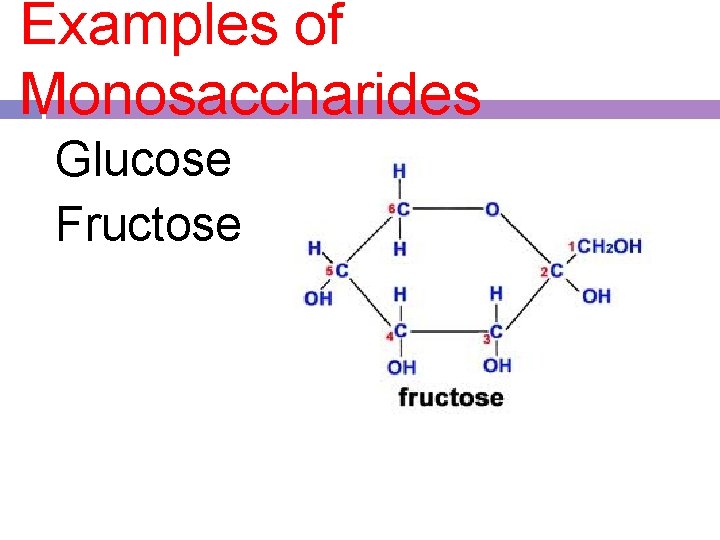
Examples of Monosaccharides Glucose Fructose

Isomers- compounds that have the same molecular formula but different molecular structures
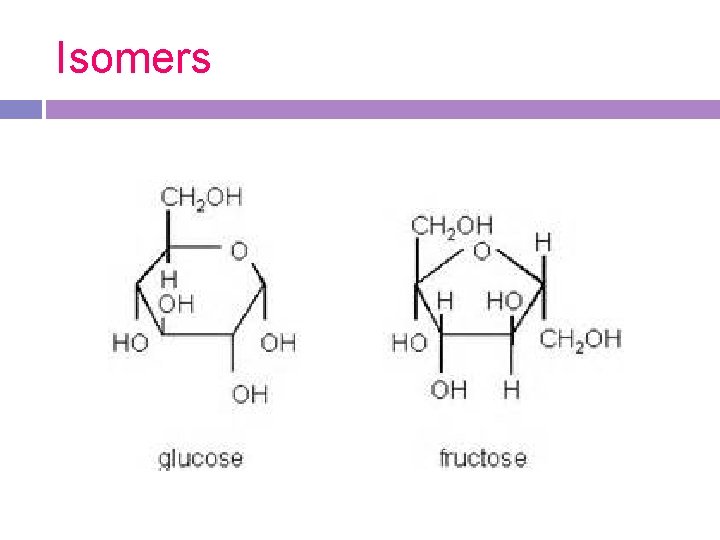
Isomers
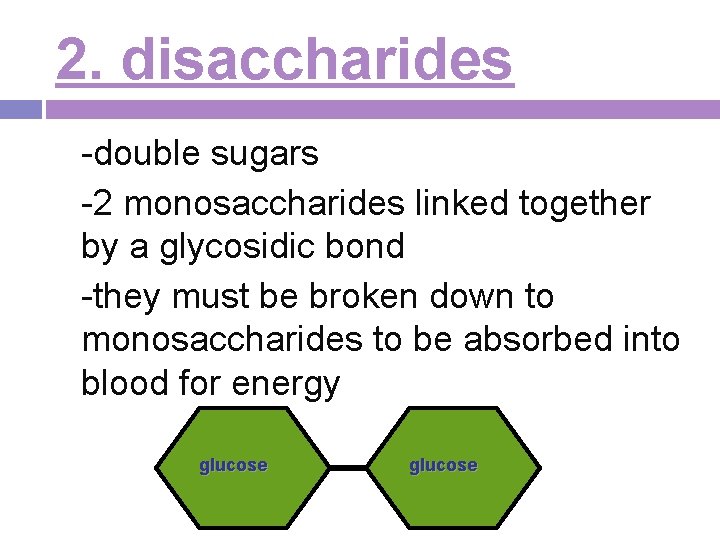
2. disaccharides -double sugars -2 monosaccharides linked together by a glycosidic bond -they must be broken down to monosaccharides to be absorbed into blood for energy glucose
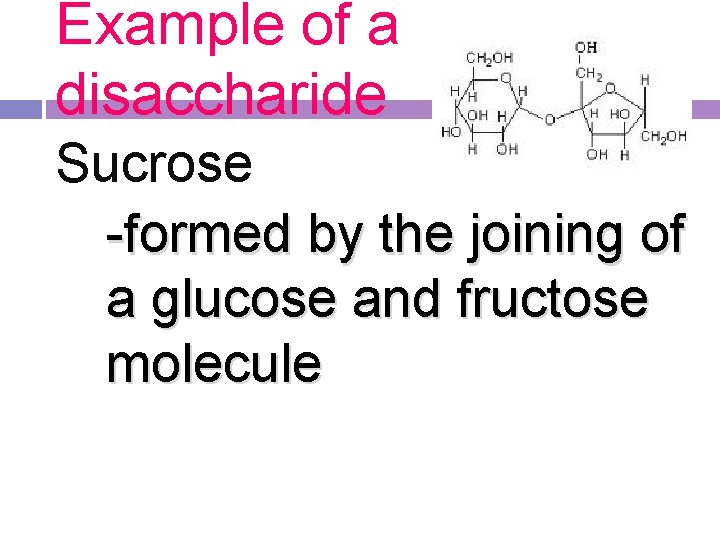
Example of a disaccharide Sucrose -formed by the joining of a glucose and fructose molecule

3. polysaccharides -complex sugars -consist of a large number(10+) of monosaccharides joined together
Examples starches, glycogen, cellulose, chitin
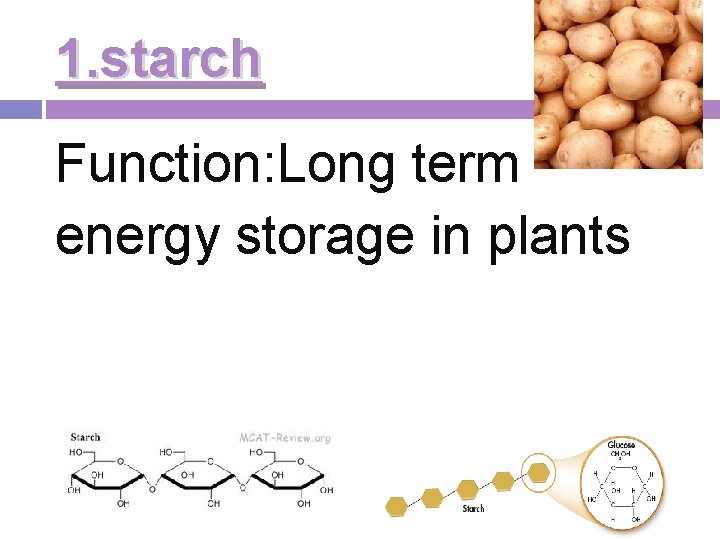
1. starch Function: Long term energy storage in plants

2. glycogen Function: temporary animal energy storage

Complex Carbohydrates - when the level of glucose in your blood runs low, glycogen can be broken down and released into the blood, serving as the fuel for cellular respiration -glycogen stored in your muscles supplies the energy for muscle contraction.
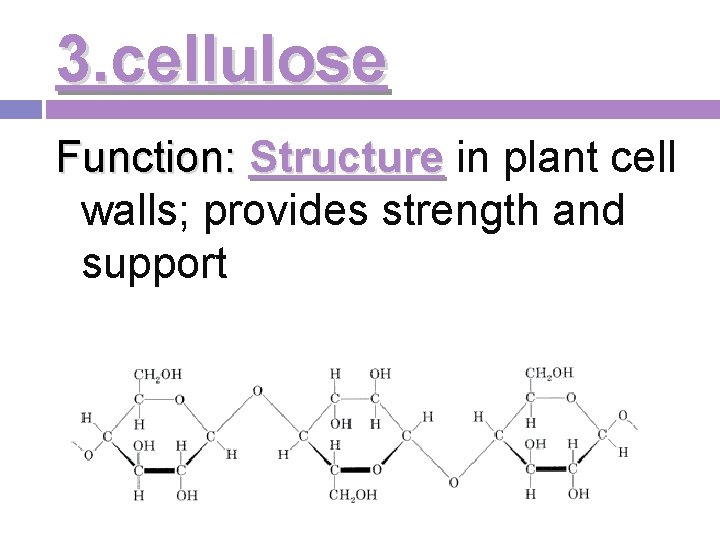
3. cellulose Function: Structure in plant cell walls; provides strength and support
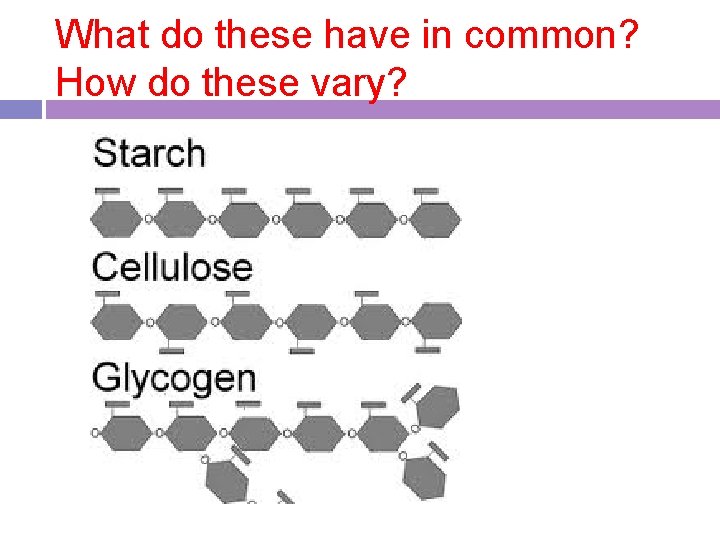
What do these have in common? How do these vary?

Cellulose “digestion” -cellulose is difficult to digest -animals such as cows, koalas, and horses all “digest” cellulose, but these animals do not have the enzyme that digests cellulose; instead, these animals have microbes in their digestive tract that can digest it for them -for other animals, it is a good source of indigestible fiber in the diet

4. chitin Function: structure in some animals that have exoskeletons
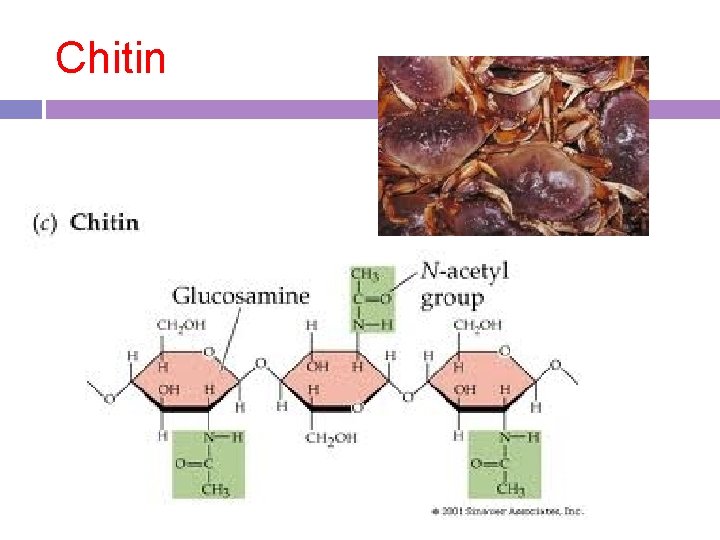
Chitin

2. Lipids Examples: Fats and oils Phospholipids Sterols (steroids) Waxes

Lipids -Lipids are a varied group of biological molecules grouped together because of the common feature that they do not dissolve in water. Composition: carbon, hydrogen, and a small amount of oxygen

Lipids -Because lipids are not composed of repeating subunits, such as a carbohydrate is, they do not have monomers and are therefore not considered to be polymers.

Lipids -Lipids are non-polar. They do not experience an attraction to water molecules and are therefore non soluble in water. Hydrophobic-“water fearing”

1. Fats and oils -large molecules made of glycerol bonded to 3 fatty acid chains

Fats -fatty acids are chains of carbon atoms connected by single or double bonds
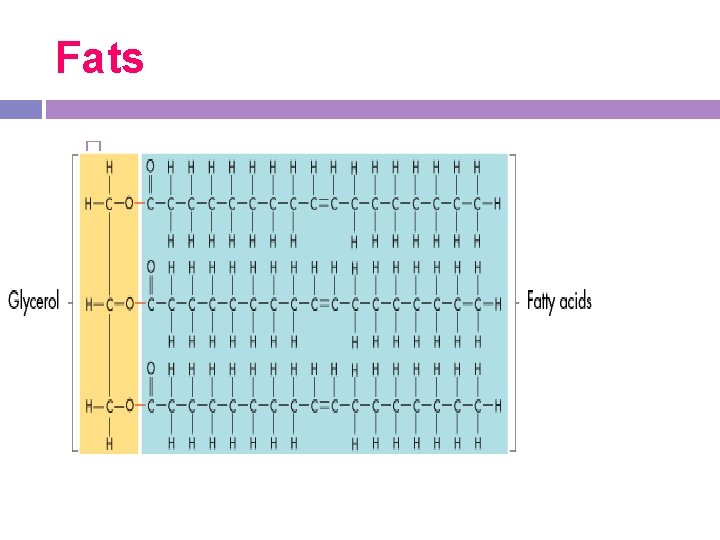
Fats �

Function of fats: 1. Good long term energy storage molecules in animals, who can convert organic compounds, such as glycogen, into

How do we store fat? Adipose tissue- tissue composed of cells that have lipid storage compartments

Functions of fats 2. when stored, acts as insulation in higher vertebrates

Functions of fats 3. stored fat acts as a shock absorber (cushions) for bones and internal organs

Types of Fats and oils: -Fatty acid chains can be saturated or unsaturated. When saturated, carbon atoms have only single bonds, and therefore the most hydrogen atoms bonded. They are solids. -Unsaturated molecules develop a kink or bend where the double bond is, making the fat more likely to melt at room temperature. They are liquids (oils).
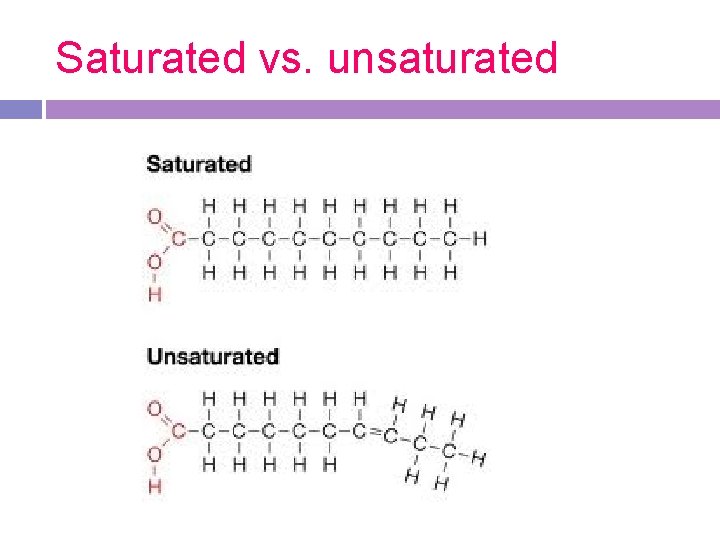
Saturated vs. unsaturated

Saturated Fats -solid at room temperature -found in animals (animal fat) -fatty acids contain no double bonds between carbon atoms Ex. Butter -known as “bad fats”

Unsaturated fats (oils) -liquid at room temperature -found mostly in plants (vegetable oils) -fatty acids contain double bonds between carbon atoms

2. Phospholipids -fat derivatives in which one fatty acid chain has been replaced by a polar phosphate group and one of several nitrogen-containing molecules

Two fatty acid “tails” and a phosphate group “head”

Function of phospholipids Function- main components of the cell membrane as well as other organelle membranes in the cell

Phospholipids -The “head” region is polar. It will be attracted to water and is called hydrophilic. The “tail” region is nonpolar, which helps the cell membrane be impermeable to water, meaning water cannot cross the cell membrane directly.

Cell membrane structure -The interior and exterior of the cell are water solutions. To prevent water from entering and exiting the cell uncontrollably , there is a double layer of phospholipids in the cell membrane.

3. Sterols (steroids) -best known & most abundant steroid is cholesterol, which is the molecule from which testosterone & estrogen (sex hormones) are made -characterized by a carbon skeleton with 4 fused rings
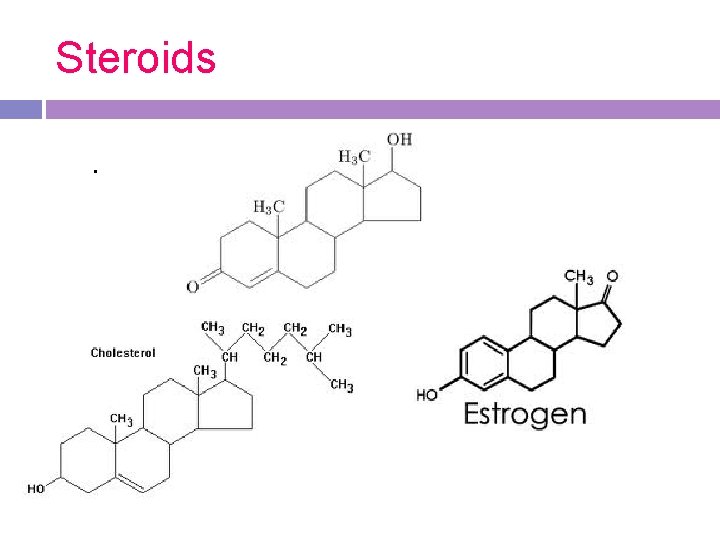
Steroids.

Function of steroids Function: act as chemical messengers or hormones within the body -cholesterol can be found throughout the cell membrane

Waxes Function- some organisms, such as insects, are coated with waxes for protection from water, and others, such as some plants, are coated (cuticle) to prevent water loss

3. Nucleic Acids Monomer-a nucleotide Composition: carbon, hydrogen, oxygen, nitrogen, and phosphorus.

Nucleic Acids Nucleotides are composed of: 1. 5 Carbon Sugar 2. Phosphate Group 3. A Nitrogenous Base
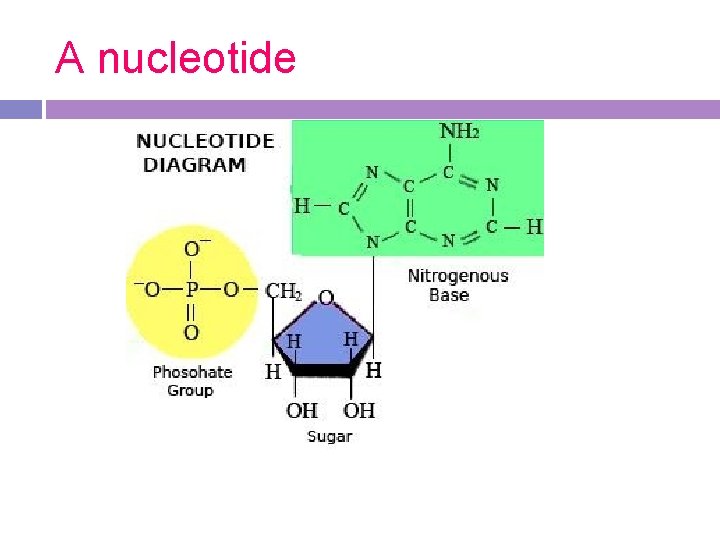
A nucleotide
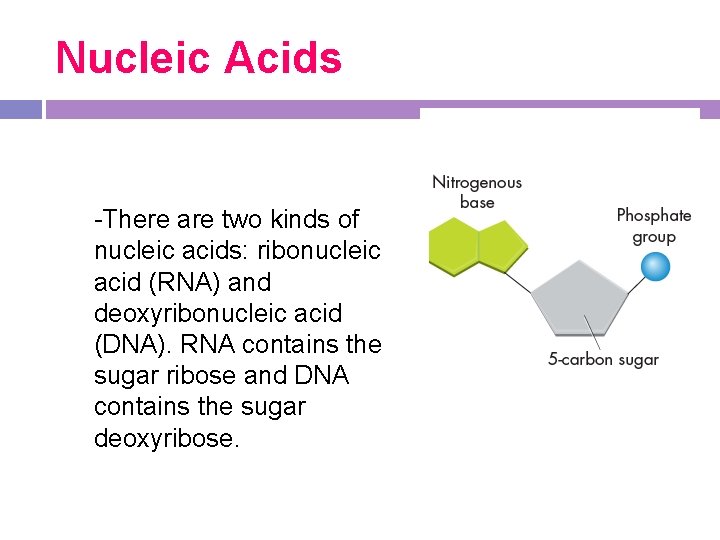
Nucleic Acids -There are two kinds of nucleic acids: ribonucleic acid (RNA) and deoxyribonucleic acid (DNA). RNA contains the sugar ribose and DNA contains the sugar deoxyribose.
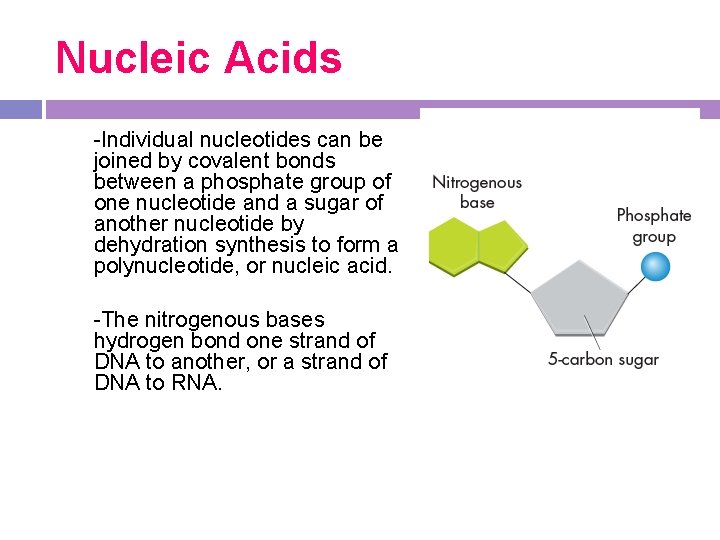
Nucleic Acids -Individual nucleotides can be joined by covalent bonds between a phosphate group of one nucleotide and a sugar of another nucleotide by dehydration synthesis to form a polynucleotide, or nucleic acid. -The nitrogenous bases hydrogen bond one strand of DNA to another, or a strand of DNA to RNA.
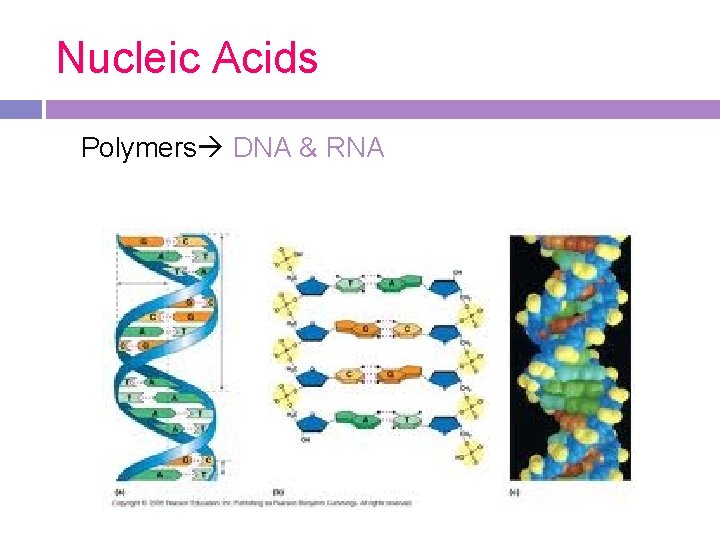
Nucleic Acids Polymers DNA & RNA

Function of nucleic acids Function- Encode & transmit genetic information -DNA & RNA contain the genetic instructions used in the development and functioning of all living organisms
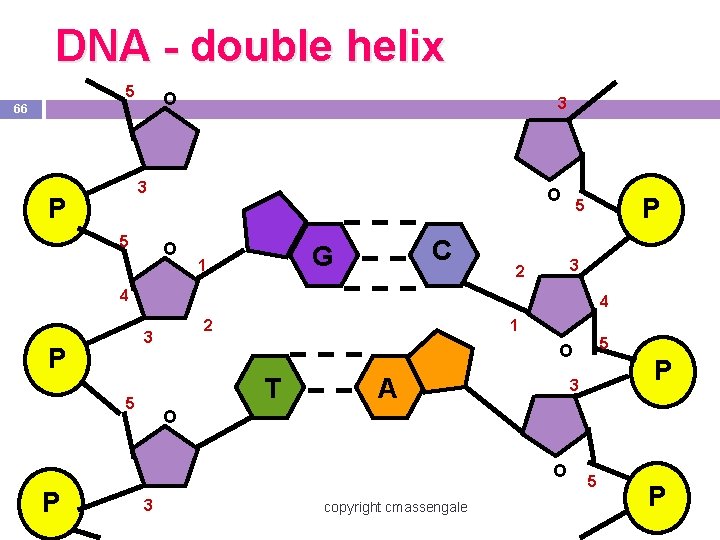
DNA - double helix 5 O 66 3 3 P 5 O O C G 1 P 5 3 2 4 4 2 3 P 1 T 5 A P 3 O O P 5 O 3 copyright cmassengale 5 P

Encoding and transmitting -Carbs, such as starches, have identically repeating monomer subunits. -Because nucleotides can be composed of 4 varying nitrogenous bases, there are different varieties of nucleotides (monomers) bonded in differing sequences that can build the polymers (DNA or RNA).

Encoding and transmitting -This variation is what makes nucleic acids special- the nucleotide variations, like letters of an alphabet making up a story, allow DNA and RNA to encode genetic information.
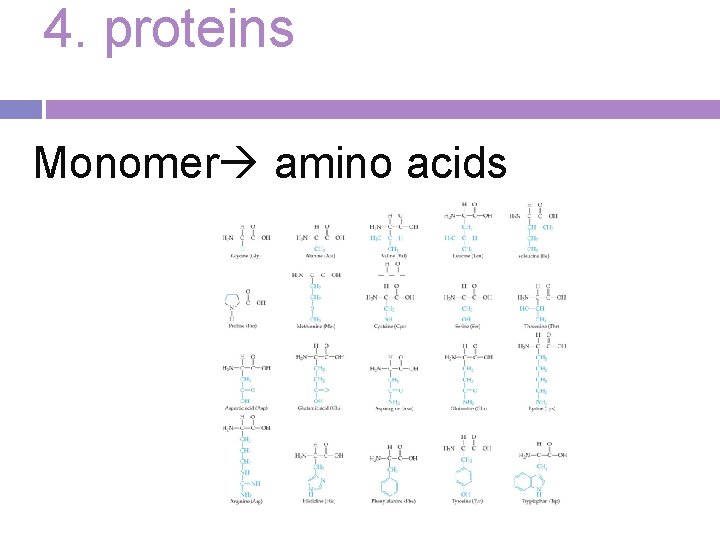
4. proteins Monomer amino acids

Protein Composition: contain carbon, hydrogen, oxygen, and nitrogen

Proteins Polymer one or more polypeptide chains (long straight chains made up of amino acids held together by peptide bonds) bent and folded around each other in a specific manner Peptide bonds form a bond between two amino acids via dehydration synthesis; when peptide bonds are broken by hydrolysis, amino acids are released
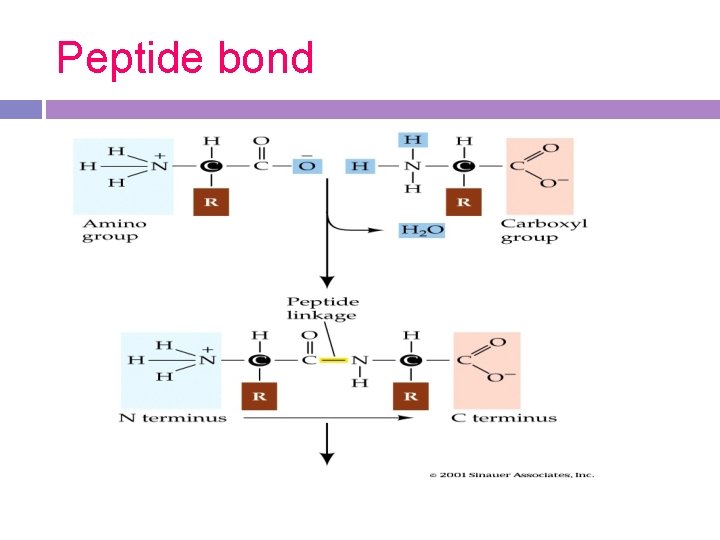
Peptide bond
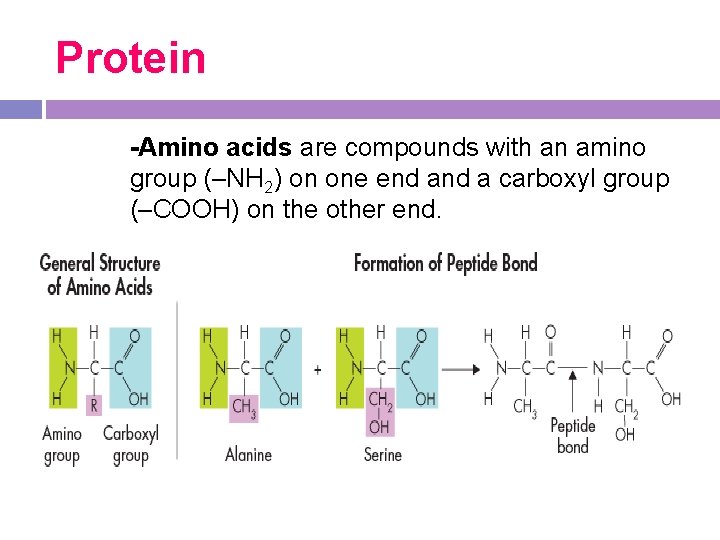
Protein -Amino acids are compounds with an amino group (–NH 2) on one end a carboxyl group (–COOH) on the other end. �
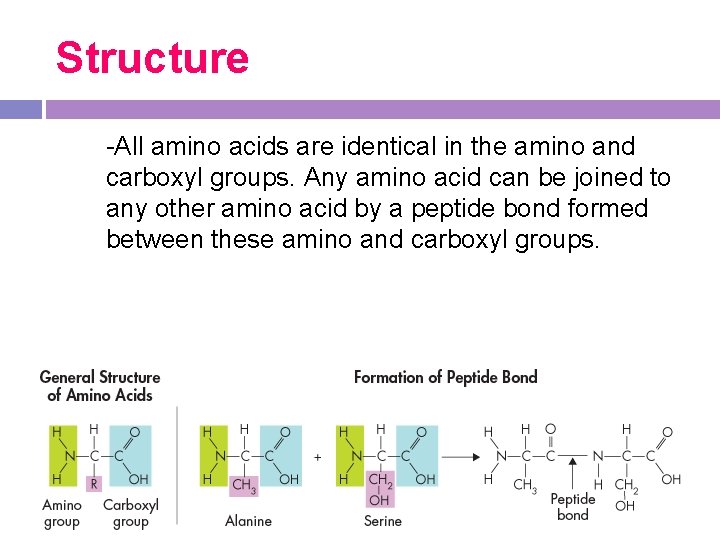
Structure -All amino acids are identical in the amino and carboxyl groups. Any amino acid can be joined to any other amino acid by a peptide bond formed between these amino and carboxyl groups.
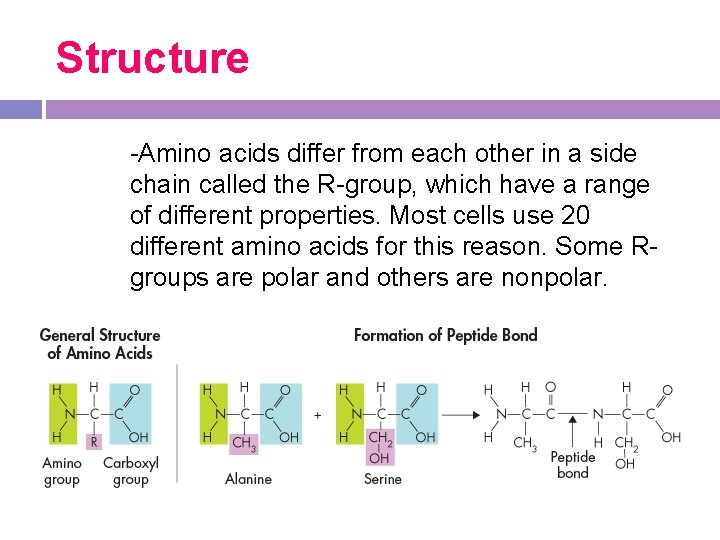
Structure -Amino acids differ from each other in a side chain called the R-group, which have a range of different properties. Most cells use 20 different amino acids for this reason. Some Rgroups are polar and others are nonpolar.

Functions of proteins 1. function as enzymes in chemical reactions by acting as catalysts, enzymes that carry out specific cell reactions; without these enzymes, chemical reactions will progress too slowly to

2. Transport—moves other substances; ex. Hemoglobin carries oxygen in our blood 3. Copying and synthesizing DNA 4. Communication between cells ex>hormonal-coordinates body activities; ex. Insulin

5. Cell structure- much of the cell is composed of protein (cytoskeleton) 6. In animals, protein composes our hair, nails, and muscles 7. Proteins in cell membrane regulate what enters or exits the cell

More than 20 different amino acids are found in nature. This variety results in proteins being among the most diverse macromolecules.
- Slides: 79