Organic Molecules n Molecules that contain carbon and

Organic Molecules n. Molecules that contain carbon and hydrogen n 4 major classes: carbohydrates, lipids, proteins and nucleic acids

Inorganic versus Organic Molecules Inorganic n Usually contain +, - ions always contain carbon and hydrogen n Usually ionic bonding always covalent bonding n Always contain a small # of atoms Often quite large, many atoms n Often associated with nonliving matter usually associated with living organisms

Carbon atom n n Needs 4 electrons to complete its outer shell Can share with as many as four other elements Often shares electrons with another carbon atom Hydrocarbons – carbon atoms bonded exclusively with hydrogen atoms

Functional Groups n n n Specific combination of bonded atoms that always reacts in the same way Connected to the carbon backbone of the organic molecule Figure 3. 2 Hydrophobic – not soluble in water Hydrophilic – soluble in water Determines the polarity of an organic molecule and the types of reactions that will occur

isomers n n Organic molecules that have identical molecular formulas but a different arrangement of atoms. Ex. Glyceraldehyde and dihydroxyacetone q n C 3 H 6 O 3 Glucose, fructose, galactose

Macromolecules n n n Polymers – largest macromolecules, made up of monomers Monomers – linked together, make up polymers Amino acids = proteins Nucleotides = nucleic acids Sugars = carbohydrates Fats = glycerol and fatty acids (not a polymer)

Synthesis and degradation of polymers n Dehydration reaction – the equivalent of a water molecule (-OH hydroxyl group) and (-H hydrogen atom) is removed as the reaction occurs to synthesize a macromolecule. n Hydrolysis reaction – used to degrade a macromolecule, -OH group from water attaches to one subunit and an –H from water attaches to the other subunit. Water is used to break the bond holding the subunits together.
Carbohydrates n Energy source and structural component 1: 2: 1 ratio of carbon to hydrogen to oxygen n Monosaccharides – single sugar n q Glucose, 6 carbon sugar n q Major source of cellular fuel Ribose, deoxyribose, 5 carbon sugars, DNA, RNA

Disaccharides n 2 monosaccharides n q q q Dehydration reaction Glucose + glucose = maltose Glucose + fructose = sucrose (table sugar) Glucose + galactose = lactose (milk sugar)

Polysaccharides Short term energy storage n n n Much larger than sugars Helical in shape Not as soluble in water Cannot easily pass through the cell membrane Plants q n Store glucose as starch Animals q Store glucose as glycogen in the liver

Polysaccharides: structural molecules n Plants q Cellulose n n Animals and fungus q n Most abundant organic molecule on earth! Chitin Bacteria q peptidoglycan


Lipids – long term energy storage n n n Insoluble in water Used for insulation and long term energy Includes fats, oils, phospholipids, steroids and waxes

Triglycerides: fats and oils n Glycerol – contains 3 –OH groups, polar q q n 3 fatty acids (hence triglyceride) Fatty acids prevent fats and oils from mixing with water Fatty Acids q Saturated – no double bonds between carbons n q Solid at room temp, animal origin Unsaturated – double bonds, liquid at room temp, usually of plant origin

Phospholipids, steroids, waxes n phospholipids q q n Glycerol and phosphate group = polar head 2 fatty acid chains = nonpolar tails Steroids q Skeletons of four fused carbon rings (different lipid structure) n n n Cholesterol – animal plasma membrane Estrogen, progesterone, testosterone Waxes q q q Long chain of fatty acid and alcohol Hydrophobic, waterproof Skin and fur maintenance, repels insects, protection

Proteins n Functions q q q Support – keratin, collagen Enzymes – speed chemical rxns Transport – channel and carrier, hemoglobin Defense - antibodies Hormones – insulin, growth Motion – actin and myosin

Proteins n Amino acids dehydration reaction protein q q q Bonded by covalent bond called peptide bond Peptide – 2 or more amino acids bonded Polypeptide – chain of many amino acids
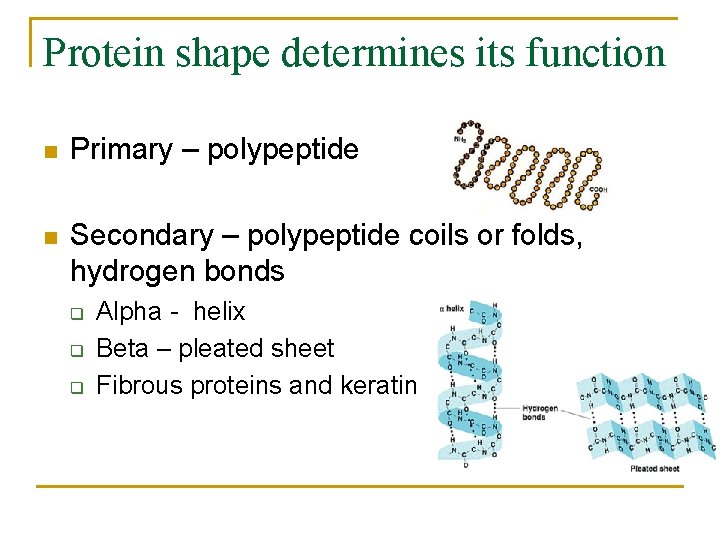
Protein shape determines its function n Primary – polypeptide n Secondary – polypeptide coils or folds, hydrogen bonds q q q Alpha - helix Beta – pleated sheet Fibrous proteins and keratin
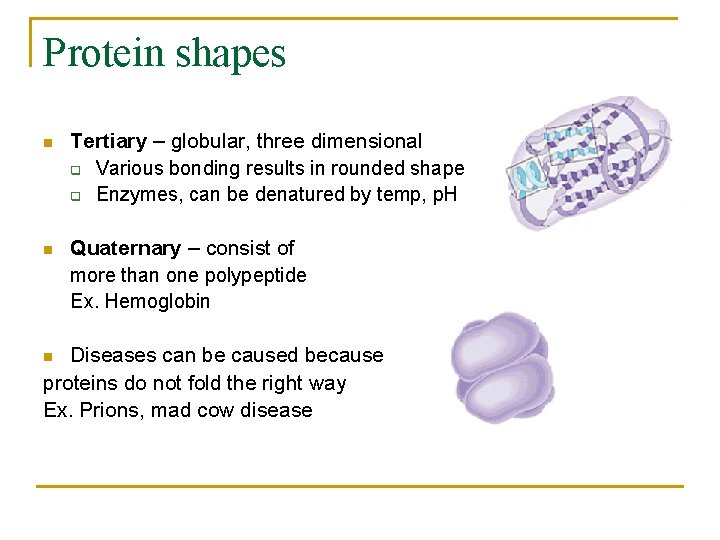
Protein shapes n Tertiary – globular, three dimensional q Various bonding results in rounded shape q Enzymes, can be denatured by temp, p. H n Quaternary – consist of more than one polypeptide Ex. Hemoglobin Diseases can be caused because proteins do not fold the right way Ex. Prions, mad cow disease n
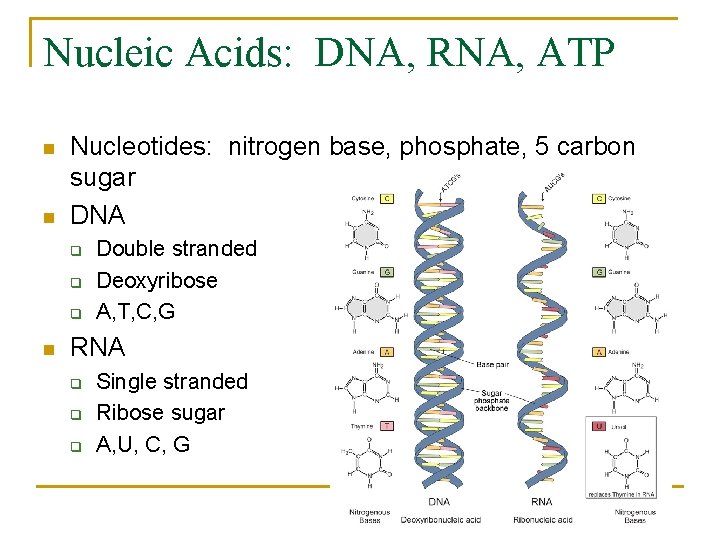
Nucleic Acids: DNA, RNA, ATP n n Nucleotides: nitrogen base, phosphate, 5 carbon sugar DNA q q q n Double stranded Deoxyribose A, T, C, G RNA q q q Single stranded Ribose sugar A, U, C, G
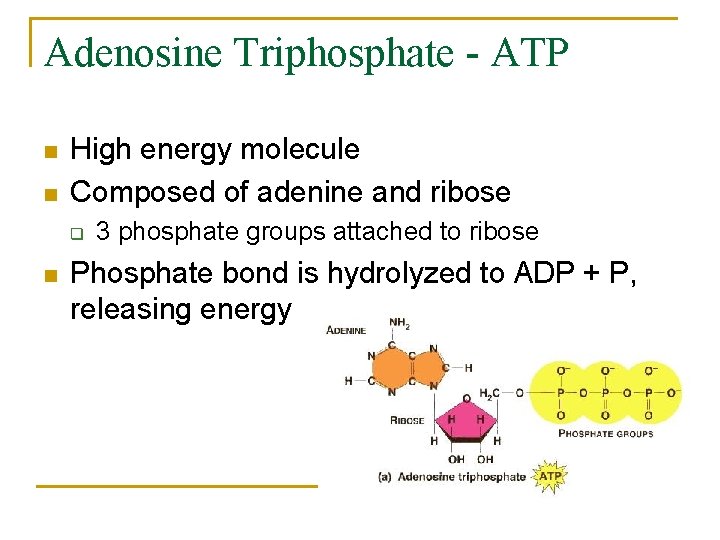
Adenosine Triphosphate - ATP n n High energy molecule Composed of adenine and ribose q n 3 phosphate groups attached to ribose Phosphate bond is hydrolyzed to ADP + P, releasing energy
- Slides: 21