ORGANIC MOLECULES Honors Anatomy Physiology 4 Categories 1








































- Slides: 73

ORGANIC MOLECULES Honors Anatomy & Physiology

4 Categories 1. 2. 3. 4. Carbohydrates Lipids Proteins Nucleic Acids

Carbohydrates Simple Carbohydrates � Sugars � Monosaccharides � Disaccharides Complex Carbohydrates � Polysaccharides

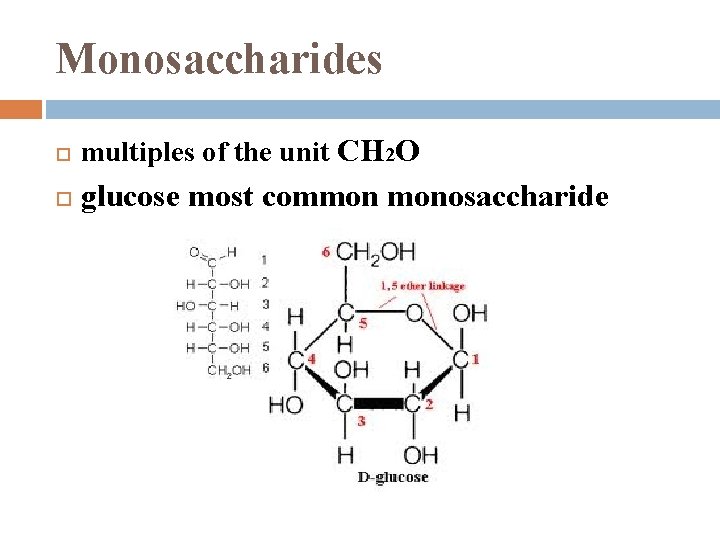
Monosaccharides multiples of the unit CH 2 O glucose most common monosaccharide

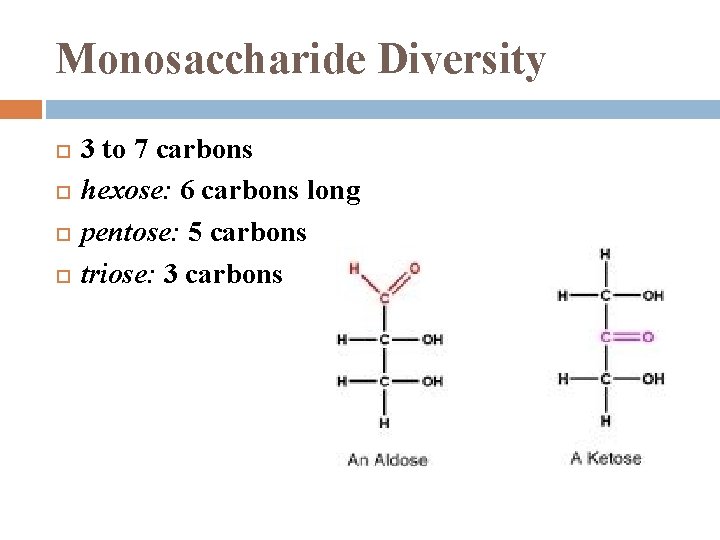
Monosaccharide Diversity 3 to 7 carbons hexose: 6 carbons long pentose: 5 carbons triose: 3 carbons

Monosaccharide Diversity most hexoses and pentoses form rings in aqueous solutions used in cellular respiration (especially glucose) serve as raw materials for synthesis of amino acids and fatty acids � if not immediately used in these ways used to build disaccharides or polysaccharides

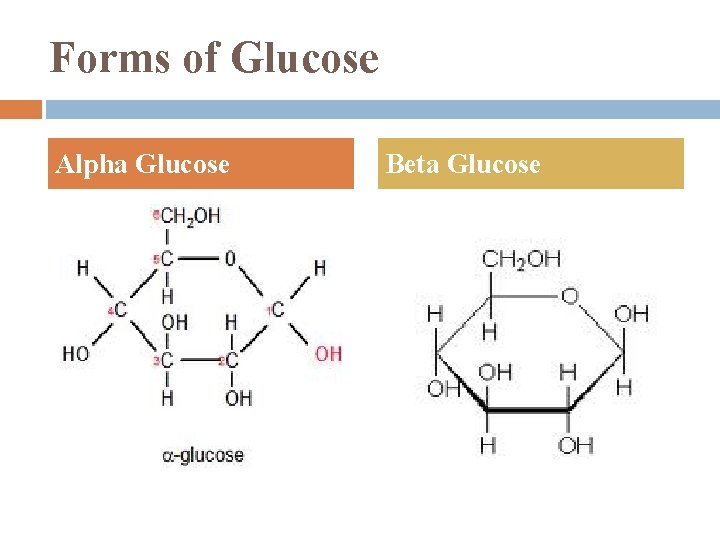
Forms of Glucose Alpha Glucose Beta Glucose

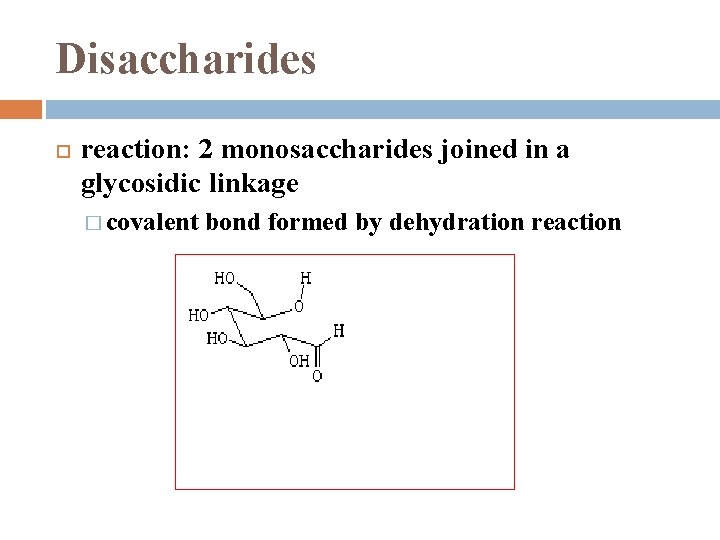
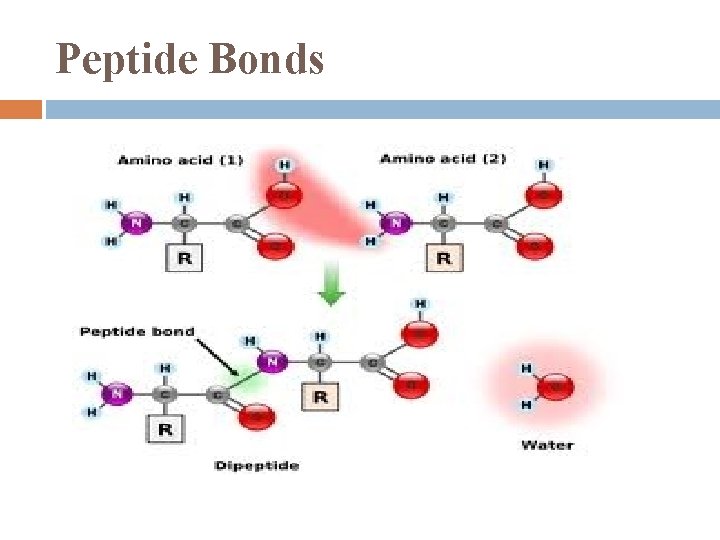
Disaccharides reaction: 2 monosaccharides joined in a glycosidic linkage � covalent bond formed by dehydration reaction

Disaccharides 2 glucose = maltose (malt sugar) glucose + galactose glucose + fructose = sucrose (table sugar)

Polysaccharides 1. 2. polymers of hundreds to thousands of monosaccharides joined by glycosidic linkages function determined by its sugar monomers & positions of glycosidic linkages 2 types: storage of monosaccharides to be used for energy when needed building material

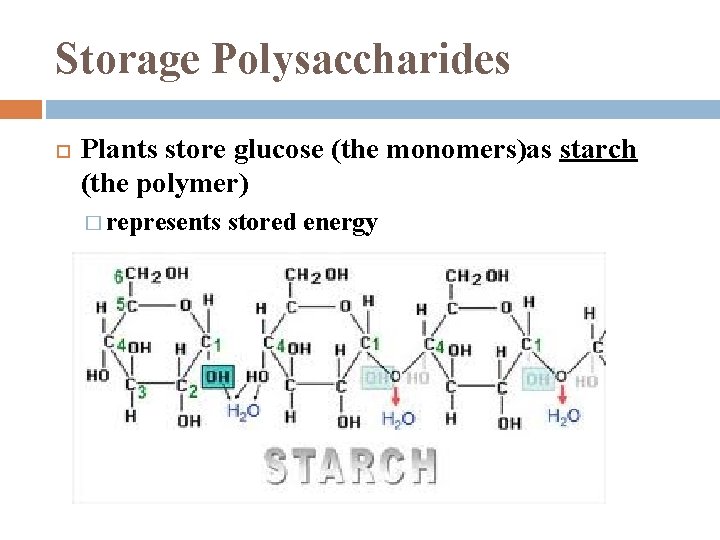
Storage Polysaccharides Plants store glucose (the monomers)as starch (the polymer) � represents stored energy

Starch most is made of α glucose monomers joined in 14 linkages � simplest form of starch (amylose) is unbranched complex starch, amylopectin, has 1 -6 linkage

Storage Polysaccharides Animals: store glucose (the monomers) as glycogen (the polymer) in 1 -4 & 1 -6 linkages � stored mainly in liver & muscle cells � humans store about 1 days supply of glucose this way

Cellulose digested by very few organisms (don’t have enzymes to do it) in humans: passes thru GI tract abrading walls & stimulating mucus secretion along the way smoother passage of food thru not technically a nutrient but is important

“Insoluble Fiber” = Cellulose

Lipids large group of hydrophobic molecules do not have true monomers Includes: � Waxes � Steroids � Some Pigments � Oils, Fats � Phospholipids

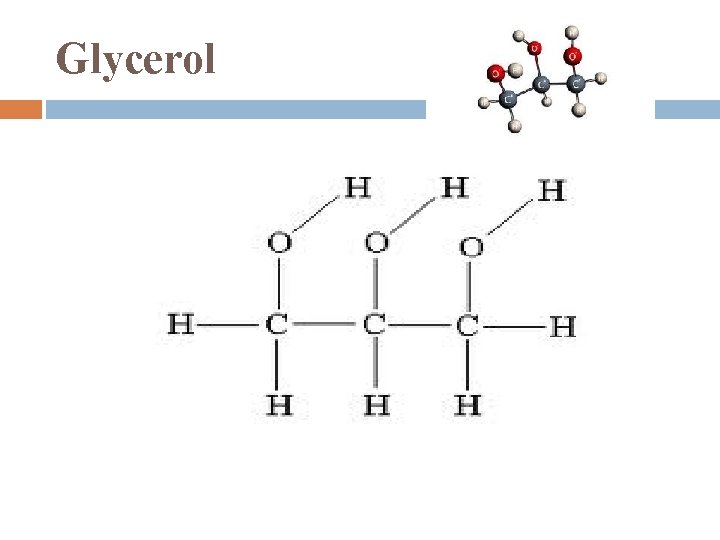
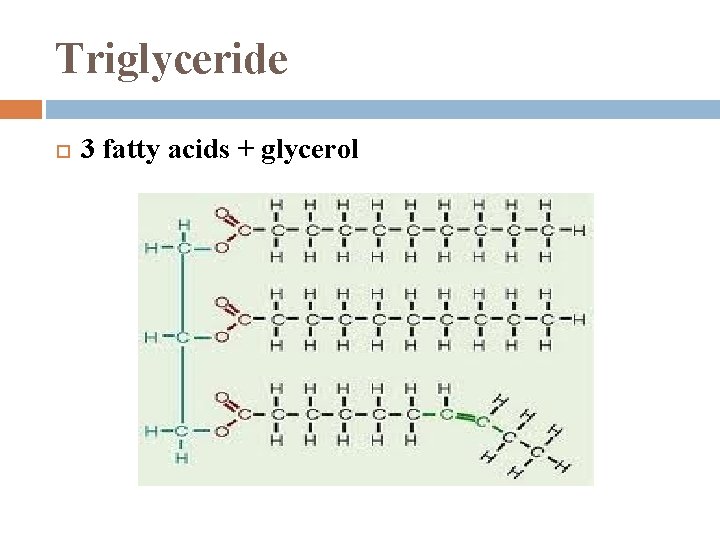
Fats 1. 2. large molecules assembled from smaller molecules by a dehydration reaction 2 parts: Glycerol Fatty Acid

Glycerol

Fatty Acids long (16 -18) chain of carbons (hydrophobic) @ one end carboxyl group (hence fatty acid)

Triglyceride 3 fatty acids + glycerol

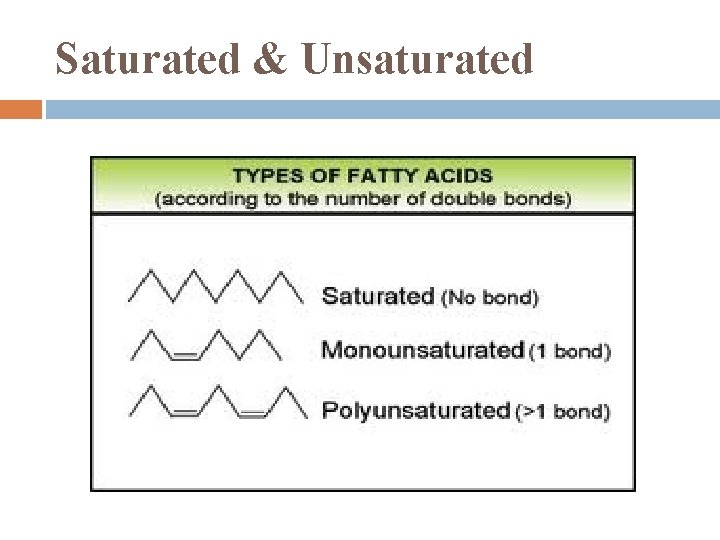
Saturated & Unsaturated

Saturated Fats include most animal fats most are solids @ room temperatures

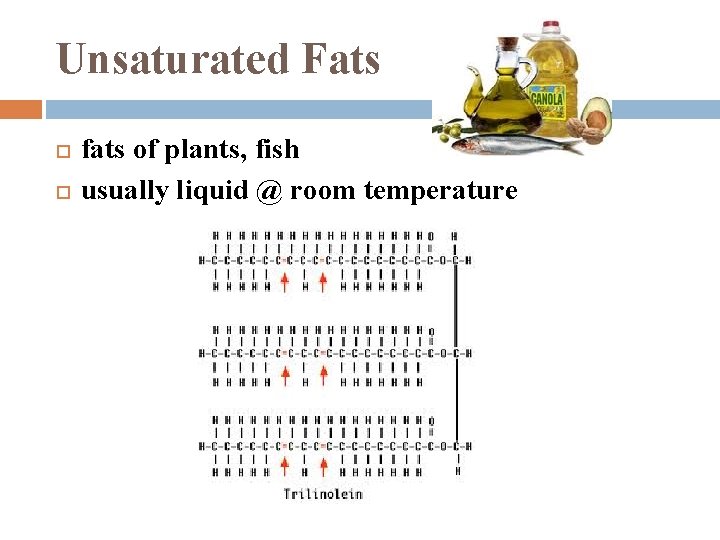
Unsaturated Fats fats of plants, fish usually liquid @ room temperature

Hydrogenated Vegetable Oil seen on some food labels means that unsaturated fats have been synthetically converted to saturated fats to keep from separating

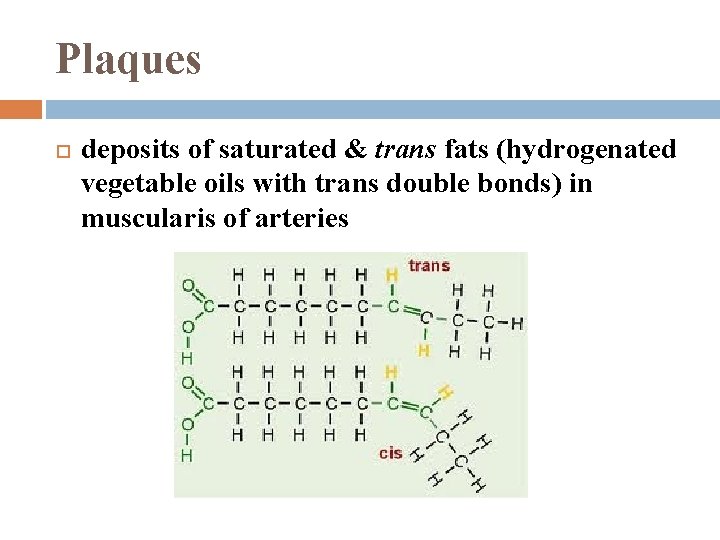
Plaques deposits of saturated & trans fats (hydrogenated vegetable oils with trans double bonds) in muscularis of arteries

Trans Fats USDA now requires nutritional labels to include amount of trans fats some cities & Denmark ban restaurants from using trans fats

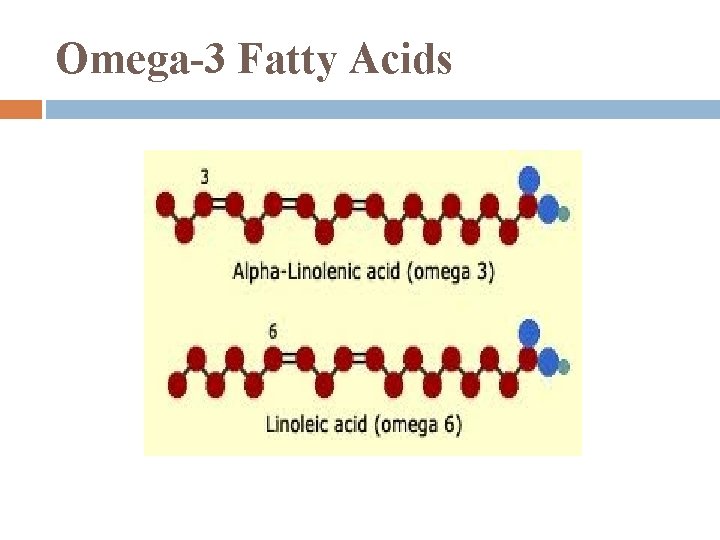
Essential Fatty Acids cannot be synthesized in body so must be included in diet include: omega-3 fatty acids: required for normal growth in children probably protect against cardiovascular disease in adults

Omega-3 Fatty Acids

Functions of Fat 1. 2. 3. Plants: storage of energy Animals: storage of energy protect organs insulation


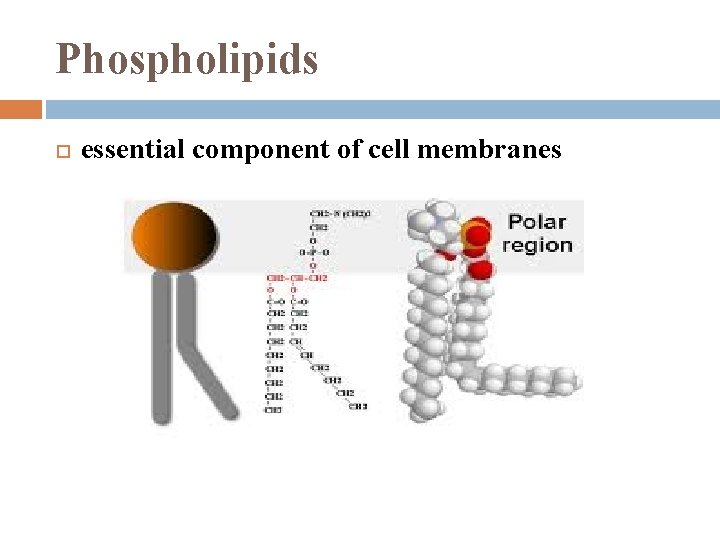
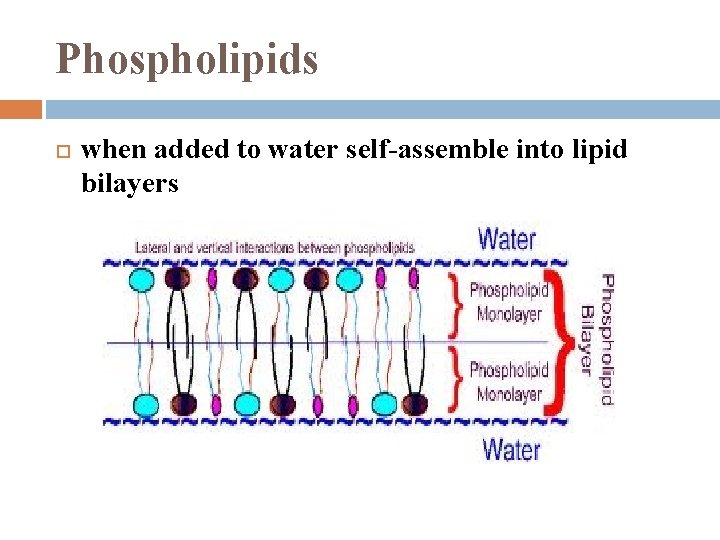
Phospholipids essential component of cell membranes

Phospholipids when added to water self-assemble into lipid bilayers

Steroids lipids characterized by a carbon skeleton made of 4 fused rings cholesterol & sex hormones have functional groups attached to these fused rings

Cholesterol in Humans part of cell membranes precursor for other steroids vertebrates make it in liver + dietary intake saturated fats & trans fats increase cholesterol levels which is ass’c with atherosclerotic disease

Proteins word in Greek from “primary” account for >50% of dry mass of most cells instrumental in almost everything organisms do

Proteins are Worker Molecules

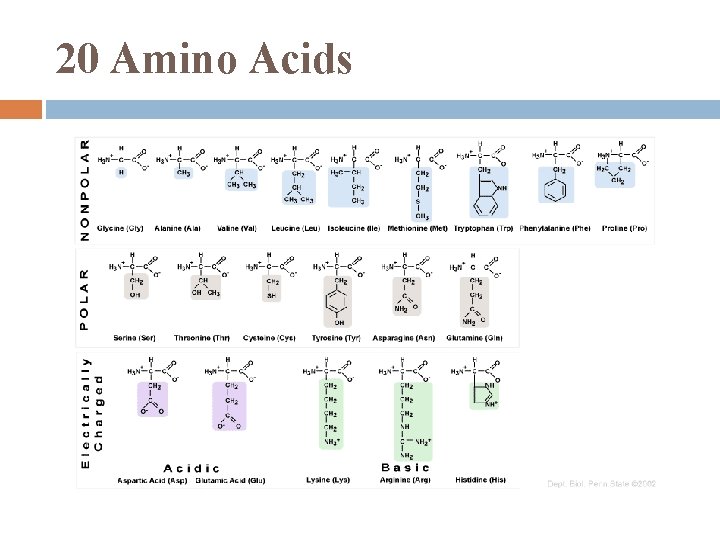
Proteins humans have tens of thousands of proteins, each with specific structure & function all made from 20 amino acids (a. a. ) Proteins are biologically functional molecules made of 1 or more polypeptides, each folded & coiled into a specific 3 -D structure

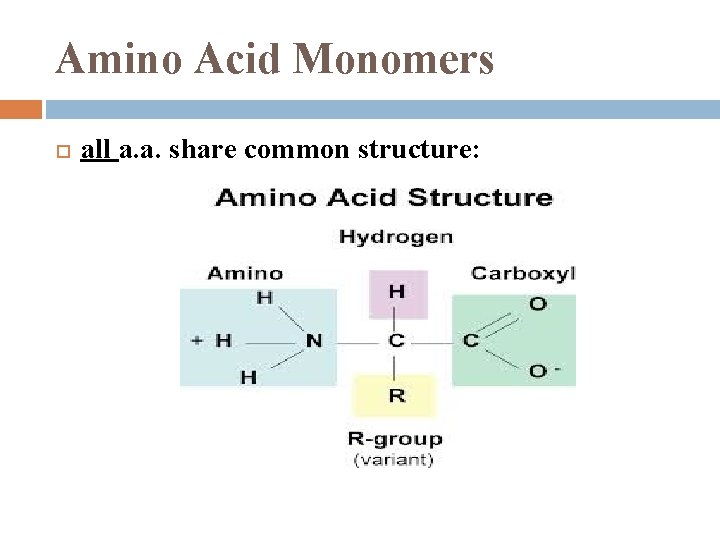
Amino Acid Monomers all a. a. share common structure:

20 Amino Acids

R Groups its physical & chemical properties determine the unique characteristics of a. a. so affect the physical & chemical properties of the polypeptide chain

Peptide Bonds

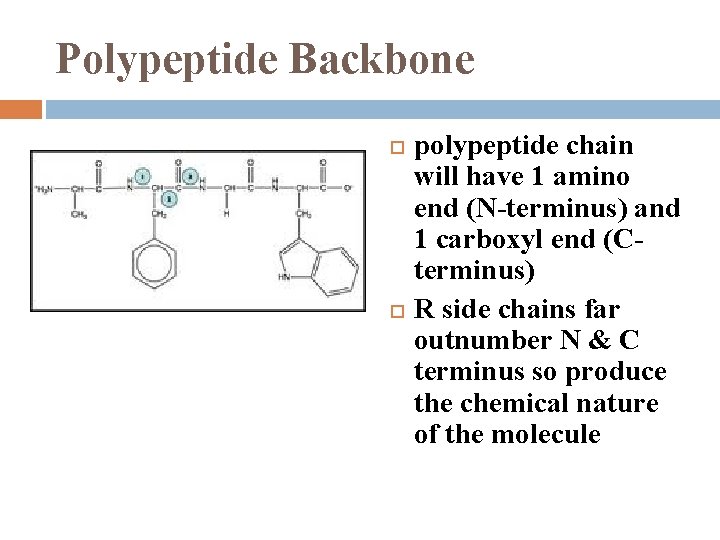
Polypeptide Backbone polypeptide chain will have 1 amino end (N-terminus) and 1 carboxyl end (Cterminus) R side chains far outnumber N & C terminus so produce the chemical nature of the molecule

Protein Structure & Function polypeptide ≠ protein

Functional Protein is not just a polypeptide chain but 1 or more polypeptides precisely twisted, folded, & coiled into a uniquely shaped molecule

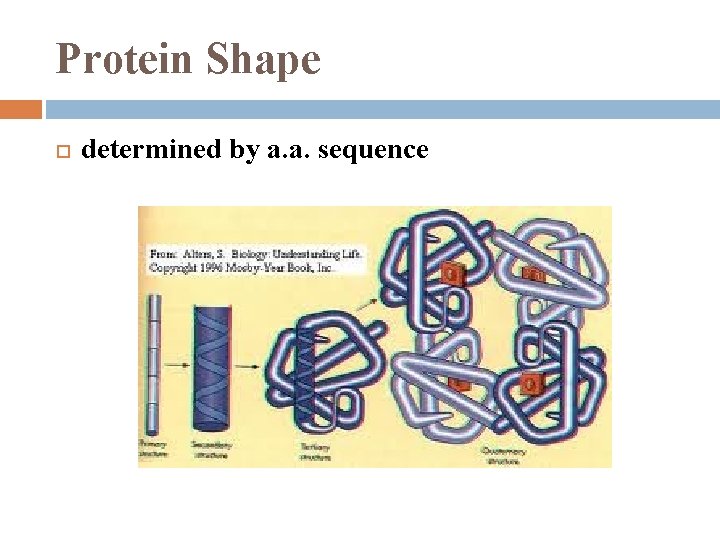
Protein Shape determined by a. a. sequence

Protein Shape 1. Globular Protein � 2. roughly spherical Fibrous Protein � long fibers when polypeptide released from ribosome it will automatically assume the functional shape for that protein’s (due to its primary structure)

Name that Shape

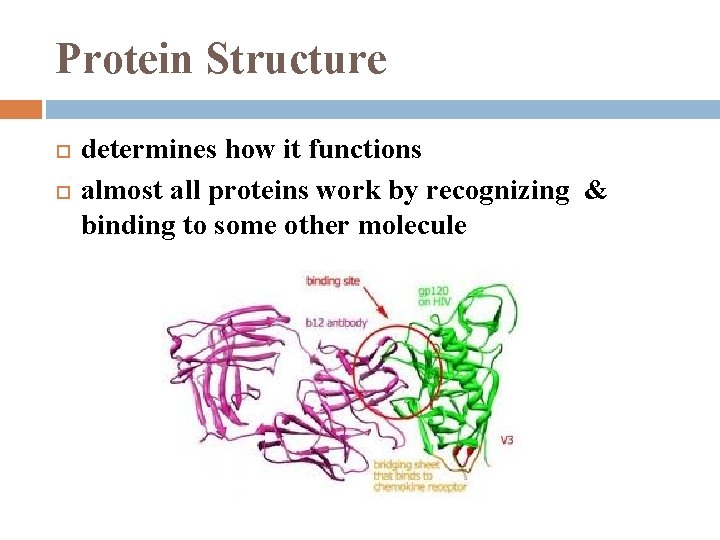
Protein Structure determines how it functions almost all proteins work by recognizing & binding to some other molecule

Protein Structure http: //www. stolaf. edu/people/giannini/flashan imat/proteins/protein%20 structure. swf

Collagen fibrous protein: 40% of all protein in human body 3 identical polypeptides “braided” into triple helix gives collagen its great strength

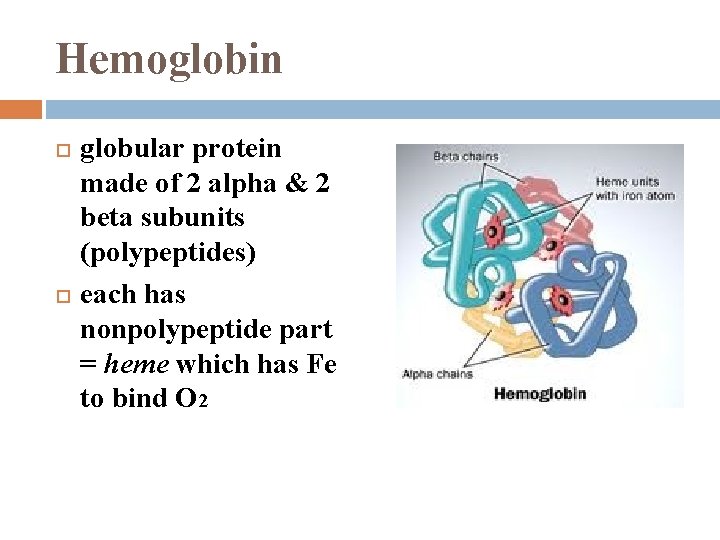
Hemoglobin globular protein made of 2 alpha & 2 beta subunits (polypeptides) each has nonpolypeptide part = heme which has Fe to bind O 2

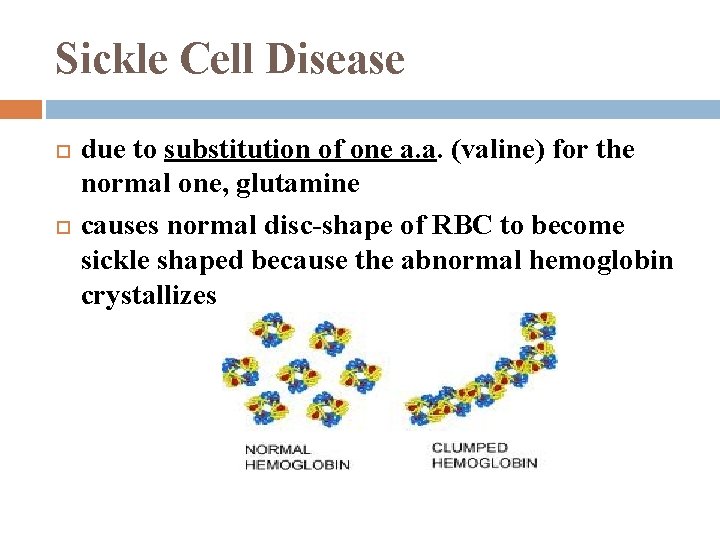
Sickle Cell Disease due to substitution of one a. a. (valine) for the normal one, glutamine causes normal disc-shape of RBC to become sickle shaped because the abnormal hemoglobin crystallizes

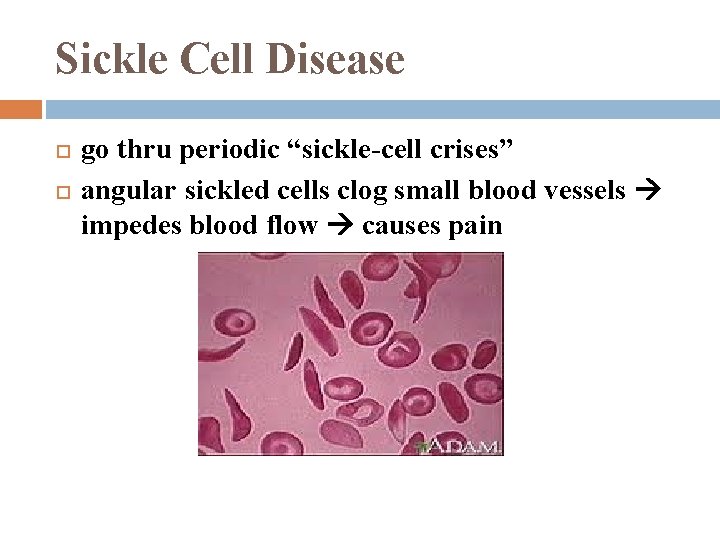
Sickle Cell Disease go thru periodic “sickle-cell crises” angular sickled cells clog small blood vessels impedes blood flow causes pain

Protein Structure 1. 2. 3. also depends on physical & chemical environment protein is in: p. H salt concentration temperature all of the above can change weak bonds & forces holding protein together

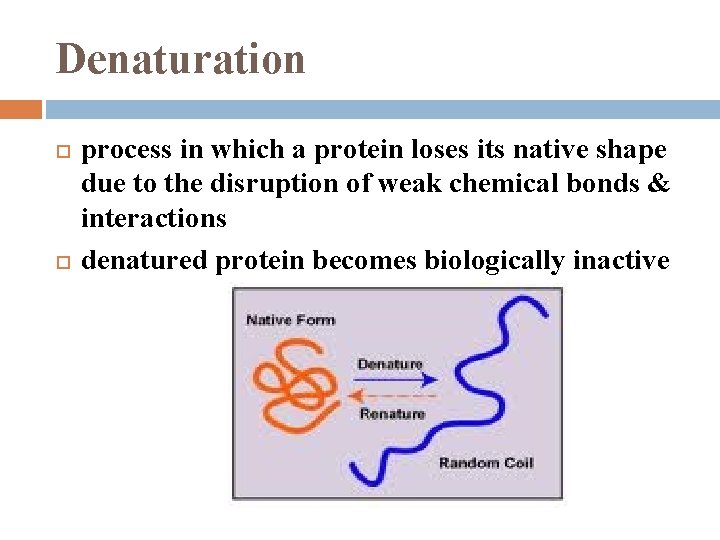
Denaturation process in which a protein loses its native shape due to the disruption of weak chemical bonds & interactions denatured protein becomes biologically inactive

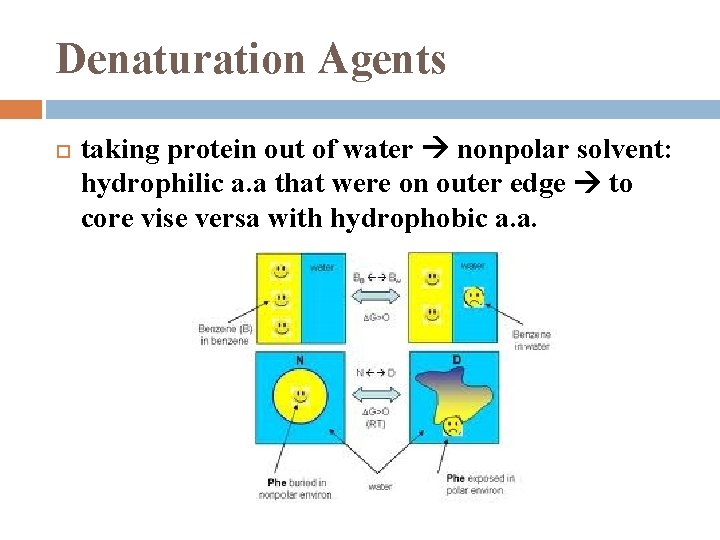
Denaturation Agents taking protein out of water nonpolar solvent: hydrophilic a. a that were on outer edge to core vise versa with hydrophobic a. a.

Misfolded Proteins ass‘c with: � Alzheimer’s � Mad Cow disease � Parkinson’s � Senile Dementia


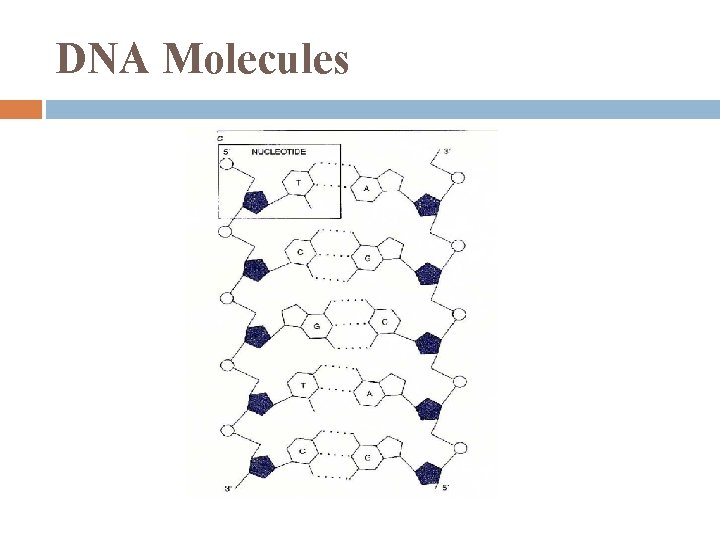
NUCLEIC ACIDS 1. are polymers made of monomers called nucleotides genes code for a. a. sequences in proteins DNA deoxyribonucleic acid RNA ribonucleic acid

Nucleic Acid Roles DNA: 1. self-replication 2. reproduction of organism 3. flow of genetic information: DNA RNA synthesis protein synthesis

Nucleic Acid Roles RNA: 1. m. RNA � � � conveys genetic instructions for building proteins from DNA ribosomes in eukaryotic cells means from nucleus cytoplasm prokaryotic cells also use m. RNA

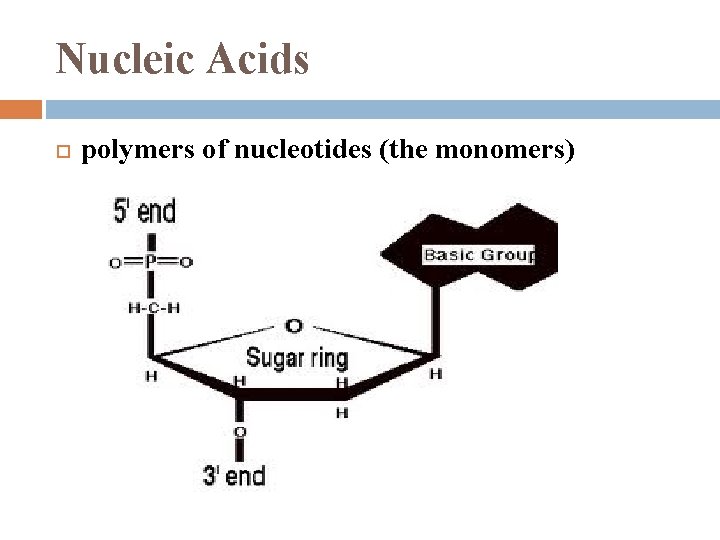
Nucleic Acids polymers of nucleotides (the monomers)

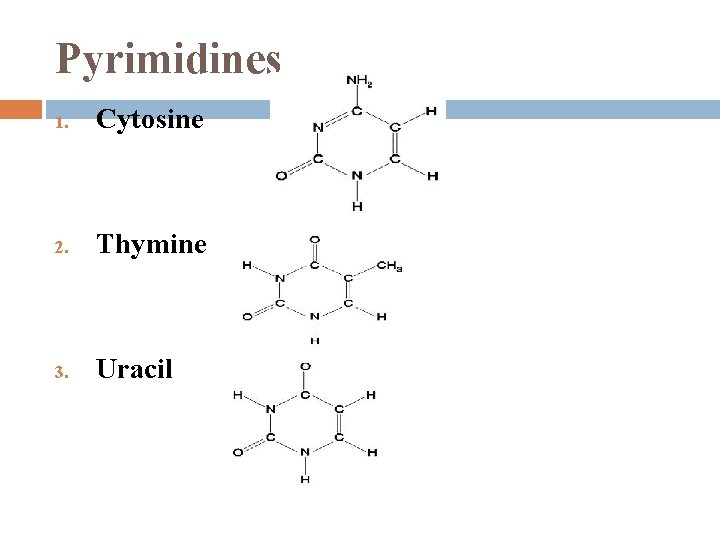
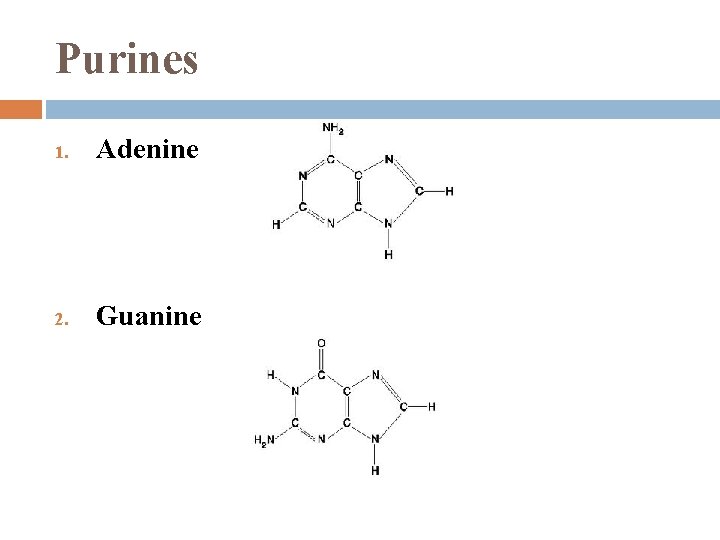
Nitrogenous Bases 1. each has 1 or 2 rings that include N are bases because the N atoms can take up H+ 2 families: Pyrimidines � 2. (1) 6 -sided ring made of C & N Purines � (1) 6 -sided ring fused to a 5 -sided ring

Pyrimidines 1. Cytosine 2. Thymine 3. Uracil

Purines 1. Adenine 2. Guanine

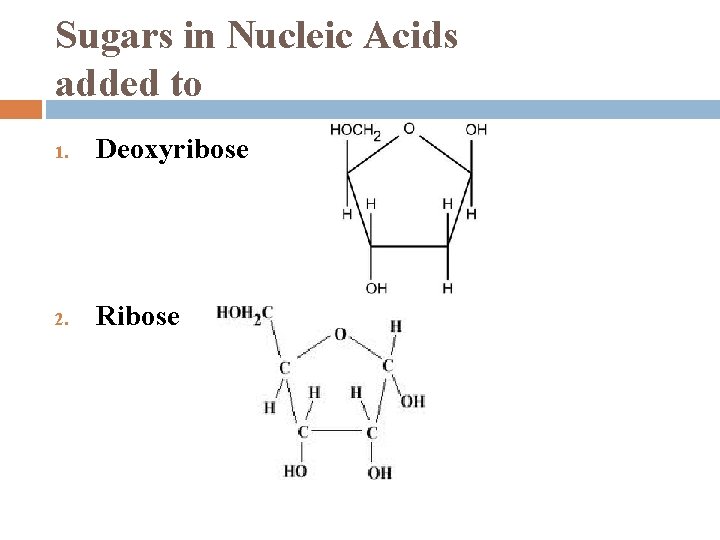
Sugars in Nucleic Acids added to 1. Deoxyribose 2. Ribose

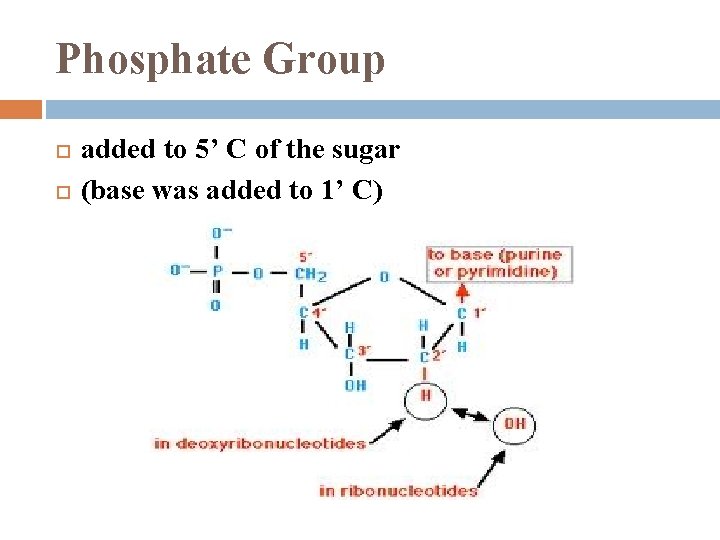
Phosphate Group added to 5’ C of the sugar (base was added to 1’ C)

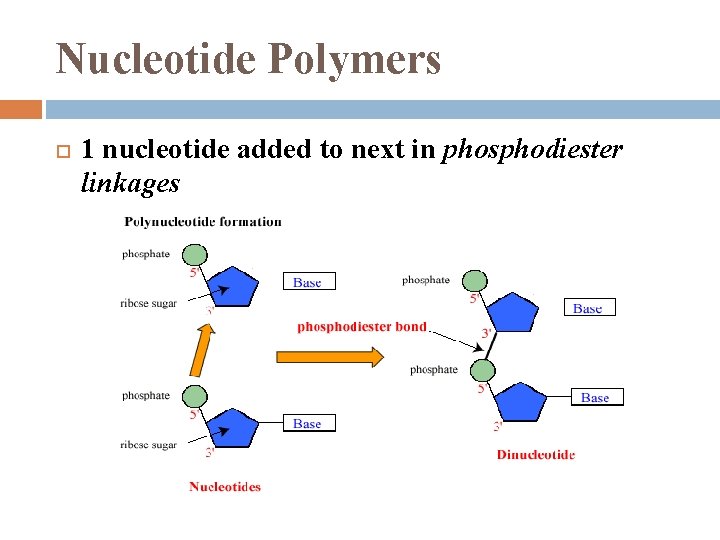
Nucleotide Polymers 1 nucleotide added to next in phosphodiester linkages

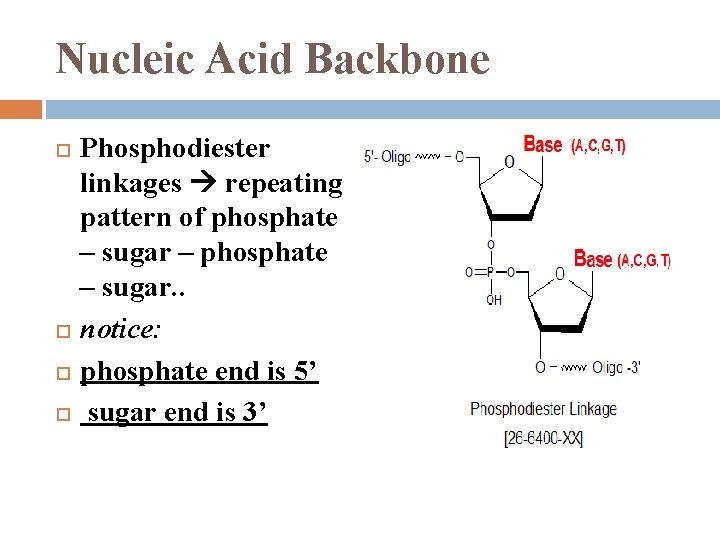
Nucleic Acid Backbone Phosphodiester linkages repeating pattern of phosphate – sugar – phosphate – sugar. . notice: phosphate end is 5’ sugar end is 3’

Linear Order of Bases specifies start, stop of transcription/translation and codons determine primary structure of proteins (which determines the 3 -D structure of a protein which in turn determines the function of the protein)

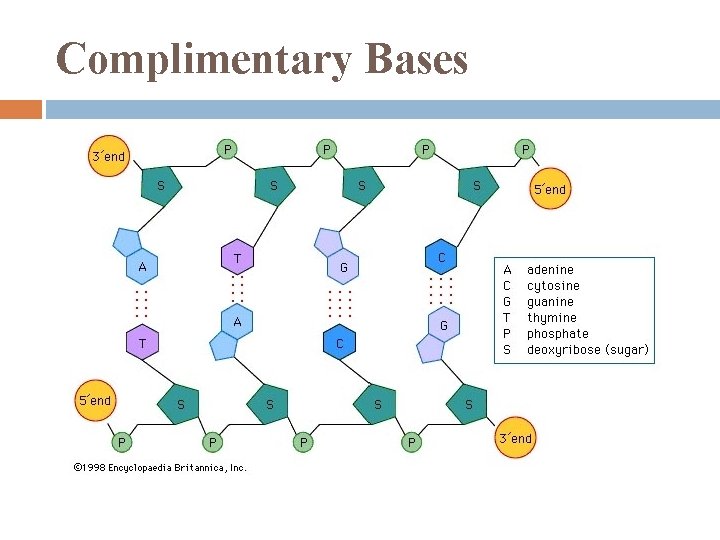
Complimentary Bases

DNA Molecules
