Organic Molecules Carbon building block of life Carbon
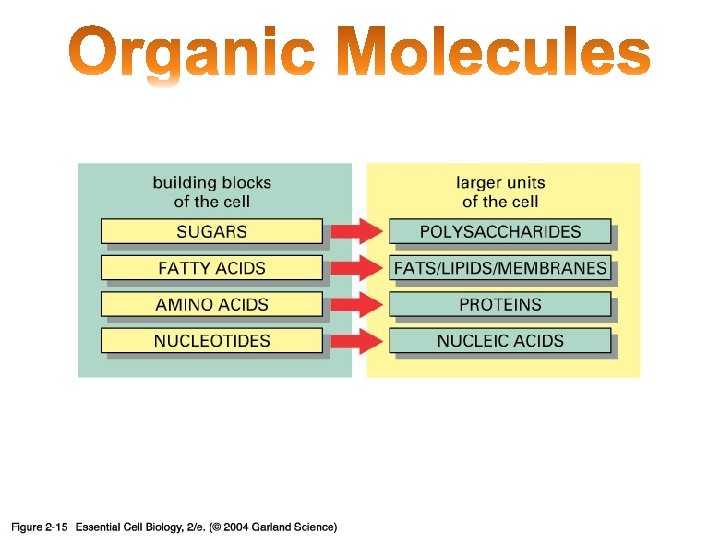
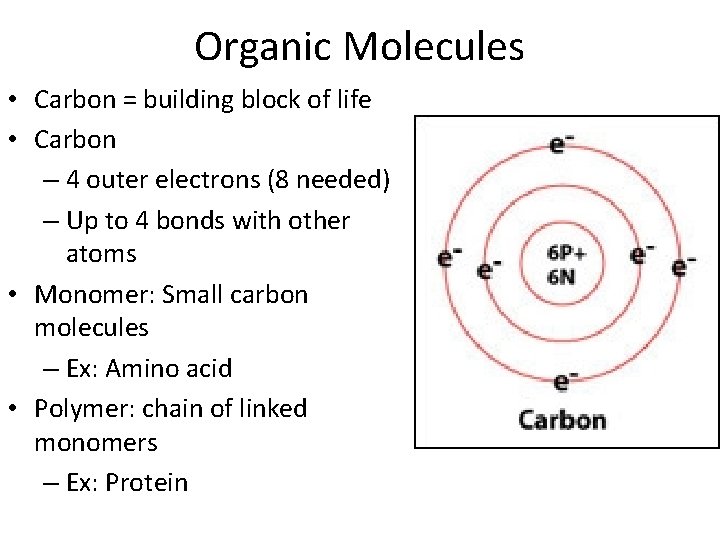
Organic Molecules • Carbon = building block of life • Carbon – 4 outer electrons (8 needed) – Up to 4 bonds with other atoms • Monomer: Small carbon molecules – Ex: Amino acid • Polymer: chain of linked monomers – Ex: Protein

Carbon creates 4 bonds to be stable
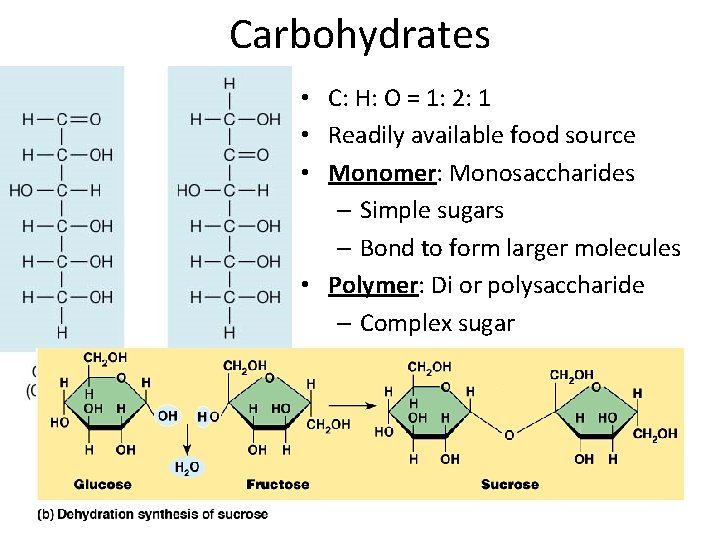
Carbohydrates • C: H: O = 1: 2: 1 • Readily available food source • Monomer: Monosaccharides – Simple sugars – Bond to form larger molecules • Polymer: Di or polysaccharide – Complex sugar
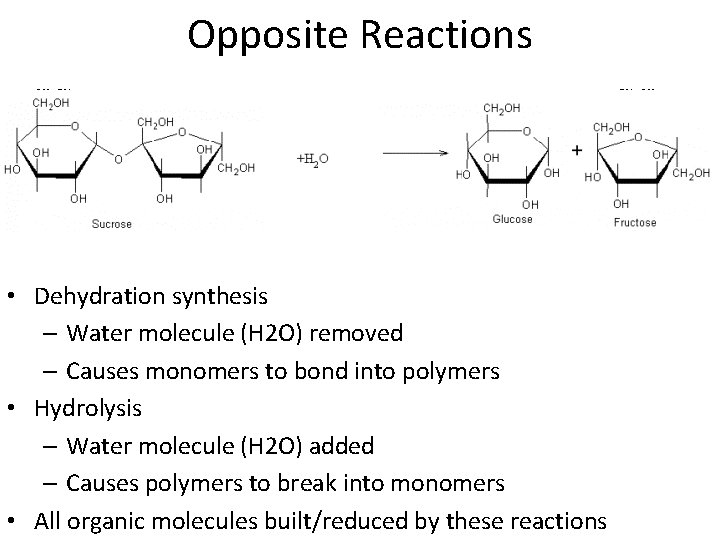
Opposite Reactions • Dehydration synthesis – Water molecule (H 2 O) removed – Causes monomers to bond into polymers • Hydrolysis – Water molecule (H 2 O) added – Causes polymers to break into monomers • All organic molecules built/reduced by these reactions
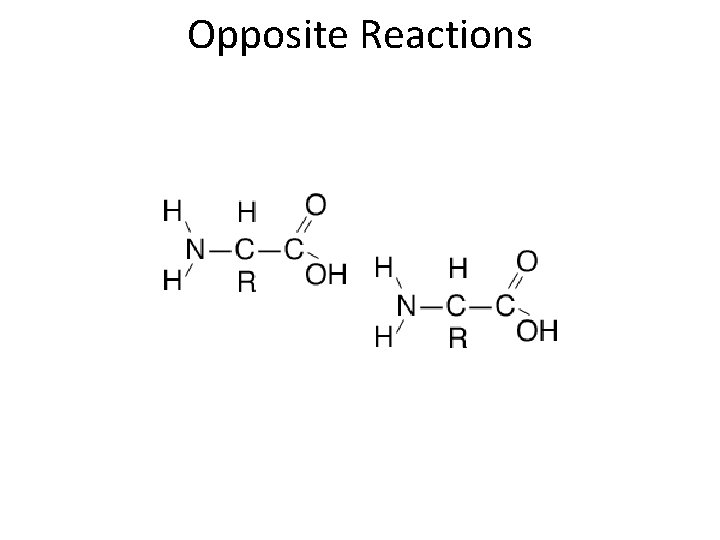
Opposite Reactions
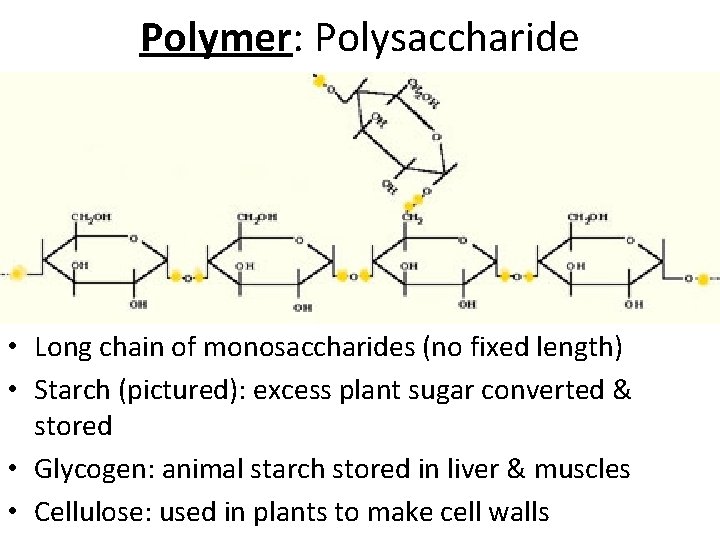
Polymer: Polysaccharide • Long chain of monosaccharides (no fixed length) • Starch (pictured): excess plant sugar converted & stored • Glycogen: animal starch stored in liver & muscles • Cellulose: used in plants to make cell walls

Chapter 2 Free Response Question Version 1: Diagram the process of hydrolysis using a sugar as an example. Be sure to indicate the following: a. Original molecules b. End molecules c. Label all parts, including the bond atoms Version 2: Diagram the process of dehydration synthesis using a sugar as an example. Be sure to indicate the following: a. Original molecules b. End molecules c. Label all parts, including the bond atoms
- Slides: 8