Organic Molecules Atoms Identify an Element Atom smallest












- Slides: 29

Organic Molecules

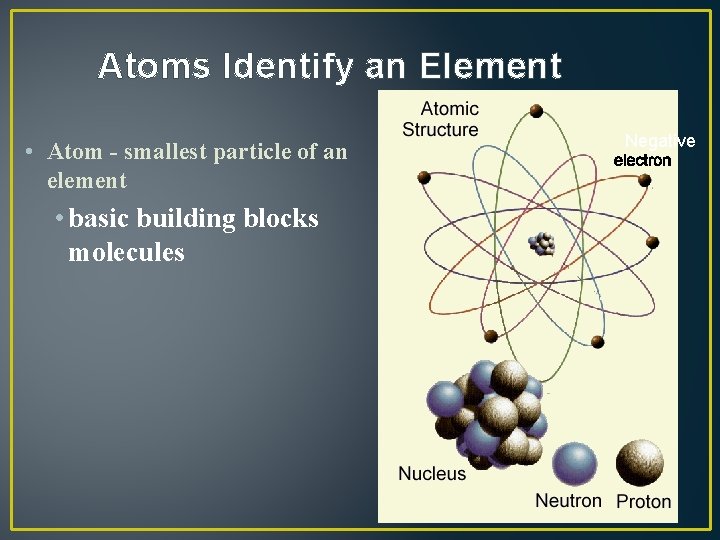
Atoms Identify an Element • Atom - smallest particle of an element • basic building blocks molecules Negative

What’s an organic molecule? ? • Molecules made up of carbon atoms !!!!!

Carbon – The Atom of All Organisms • Carbon atoms are unique in that they can bond together and create large polymers or macromolecules Single Bond- Double Bond Triple Bond

What’s a polymer? Polymers – Many Monomers A monomer is a single unit such as this link in the entire chain.

Now, we will look at a few organic molecules and their functions • • Carbohydrates Lipids Nucleic Acids Proteins

Carbohydrates – C, H and O

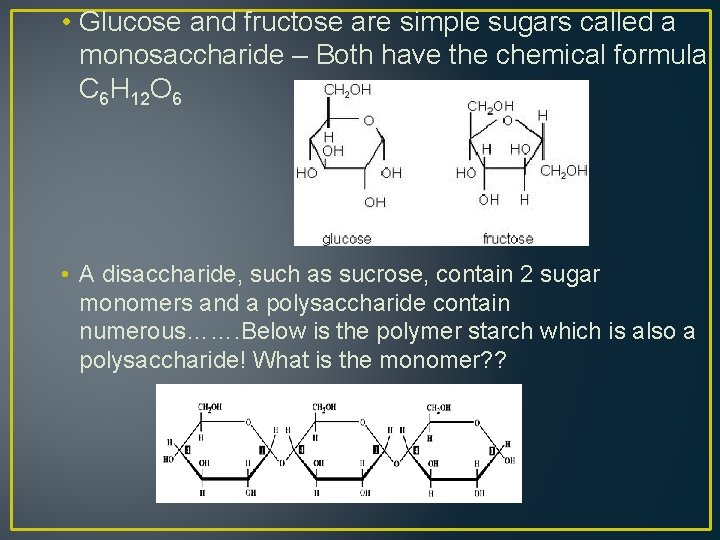
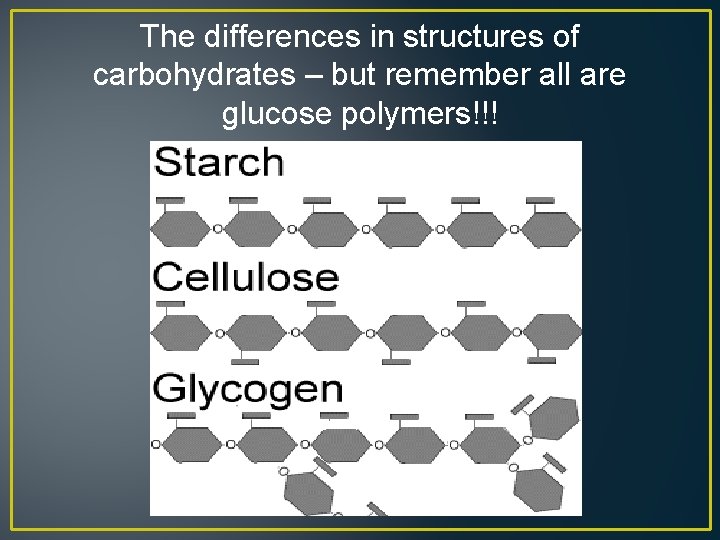
• Glucose and fructose are simple sugars called a monosaccharide – Both have the chemical formula C 6 H 12 O 6 • A disaccharide, such as sucrose, contain 2 sugar monomers and a polysaccharide contain numerous……. Below is the polymer starch which is also a polysaccharide! What is the monomer? ?

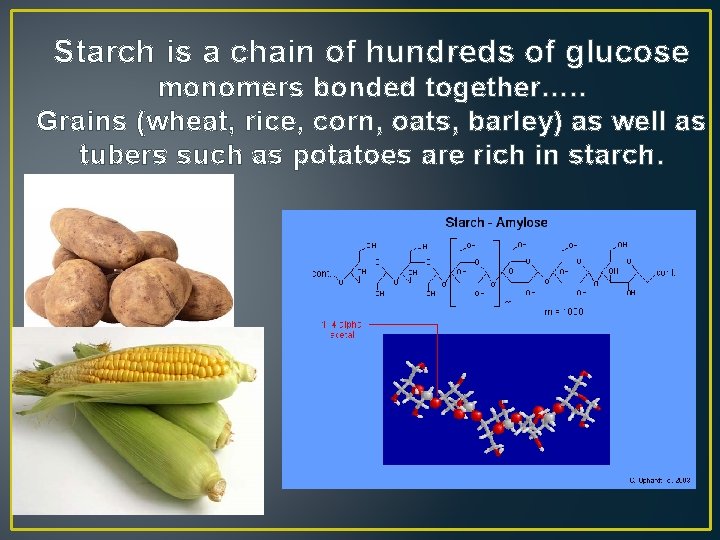
Starch is a chain of hundreds of glucose monomers bonded together…. . Grains (wheat, rice, corn, oats, barley) as well as tubers such as potatoes are rich in starch.

Glycogen in Animals **A branched polymer made up of numerous glucose monomers ** Stored in the Liver and muscle of mammals **Long-term energy storage ** Quickly broken down into glucose for immediate energy

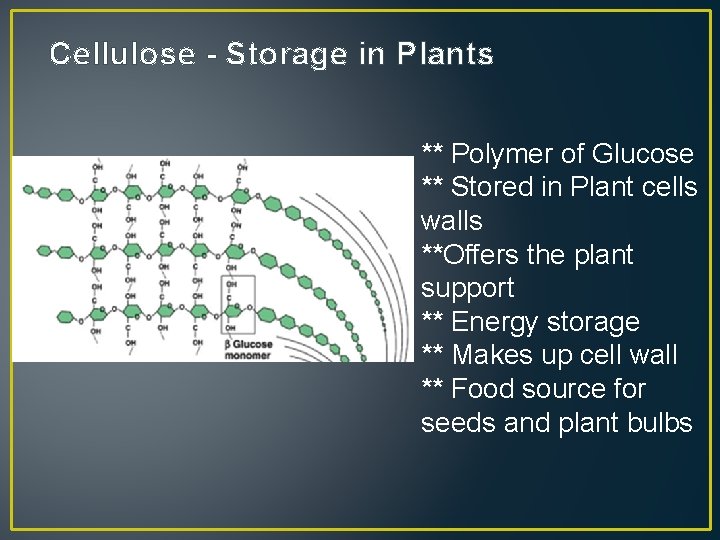
Cellulose - Storage in Plants ** Polymer of Glucose ** Stored in Plant cells walls **Offers the plant support ** Energy storage ** Makes up cell wall ** Food source for seeds and plant bulbs

The differences in structures of carbohydrates – but remember all are glucose polymers!!!

Lipids – C, H very little O

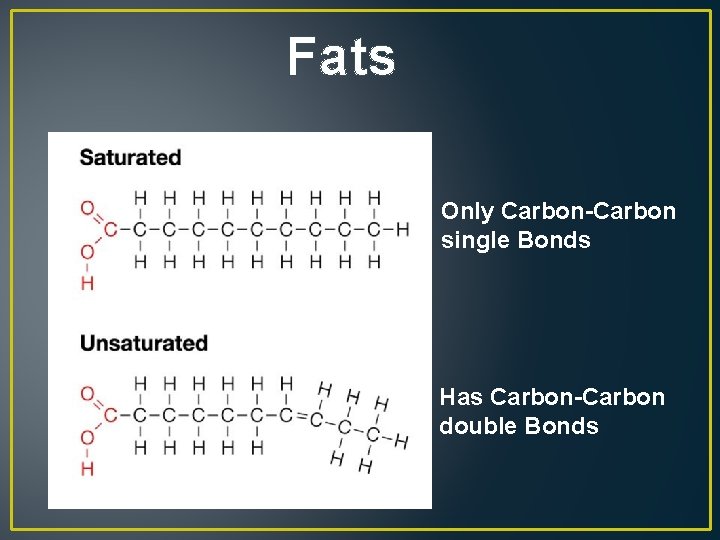
Fats Only Carbon-Carbon single Bonds Has Carbon-Carbon double Bonds

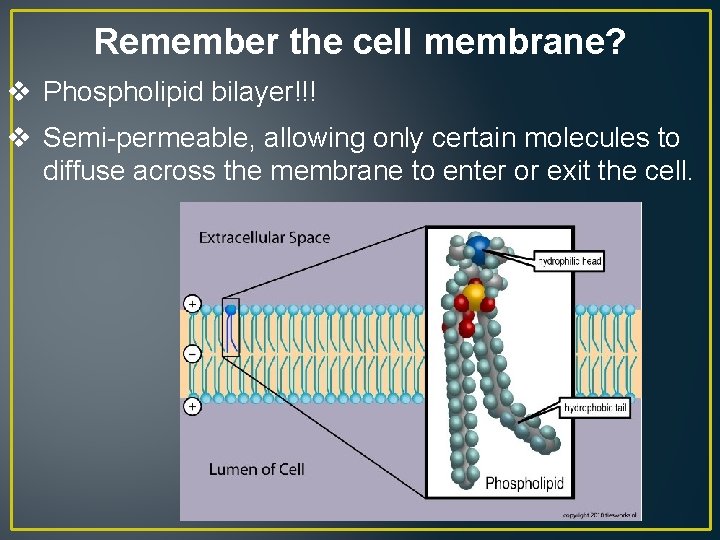
Remember the cell membrane? v Phospholipid bilayer!!! v Semi-permeable, allowing only certain molecules to diffuse across the membrane to enter or exit the cell.

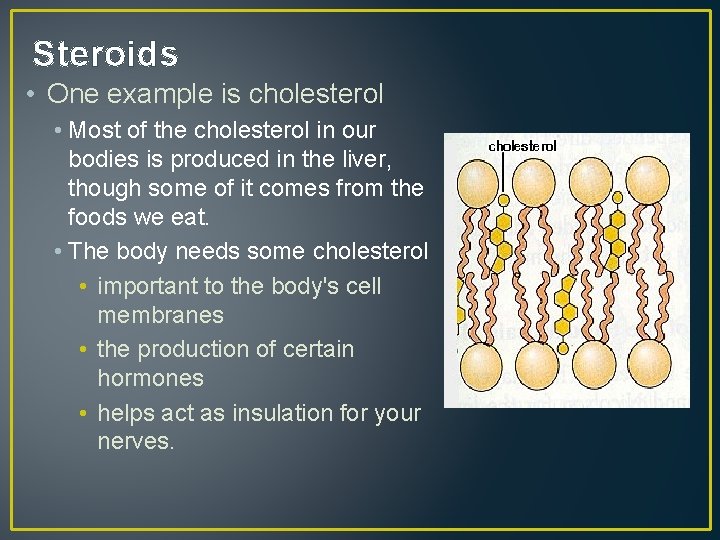
Steroids • One example is cholesterol • Most of the cholesterol in our bodies is produced in the liver, though some of it comes from the foods we eat. • The body needs some cholesterol • important to the body's cell membranes • the production of certain hormones • helps act as insulation for your nerves.

Proteins – C, H, O, N sometimes sulfur

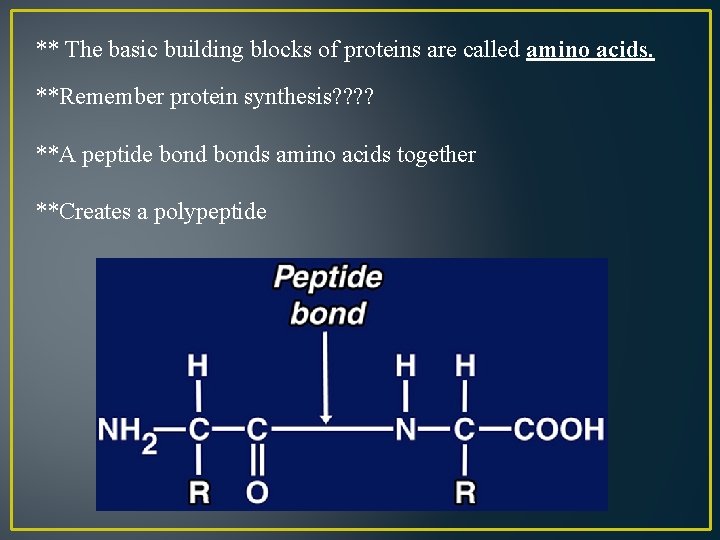
** The basic building blocks of proteins are called amino acids. **Remember protein synthesis? ? **A peptide bonds amino acids together **Creates a polypeptide

Insulin • Chemical signaler protein produced in the pancreas • Causes cells in the liver, muscle, and fat tissue to take up glucose from blood and convert it to glycogen that can be stored in the liver and muscles • Diabetes is a condition when a person has high blood glucose (blood sugar), either because insulin production is inadequate, or because the body's cells do not respond properly to insulin, or both.

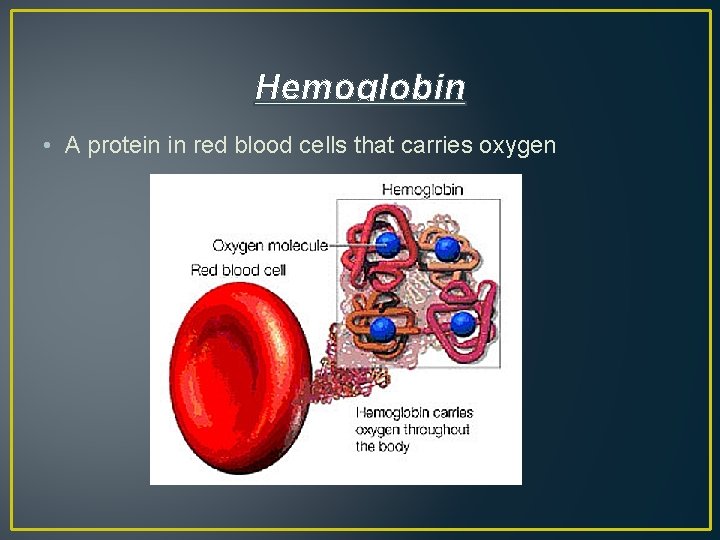
Hemoglobin • A protein in red blood cells that carries oxygen

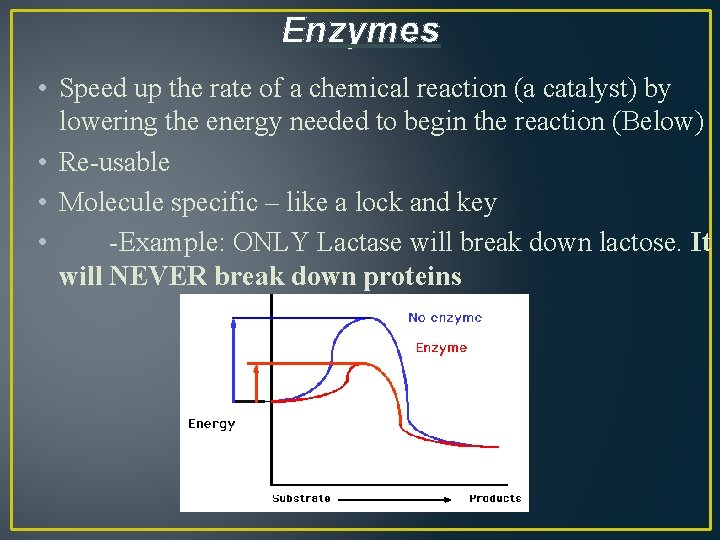
Enzymes • Speed up the rate of a chemical reaction (a catalyst) by lowering the energy needed to begin the reaction (Below) • Re-usable • Molecule specific – like a lock and key • -Example: ONLY Lactase will break down lactose. It will NEVER break down proteins

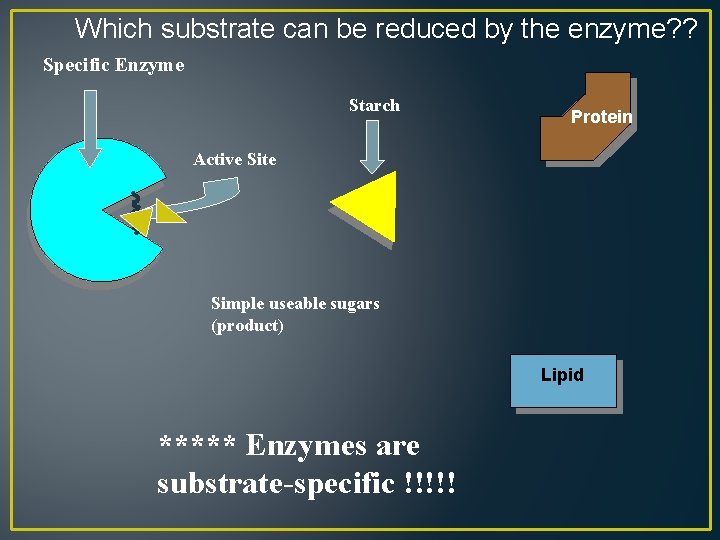
Which substrate can be reduced by the enzyme? ? Specific Enzyme Starch Protein Active Site Simple useable sugars (product) Lipid ***** Enzymes are substrate-specific !!!!!

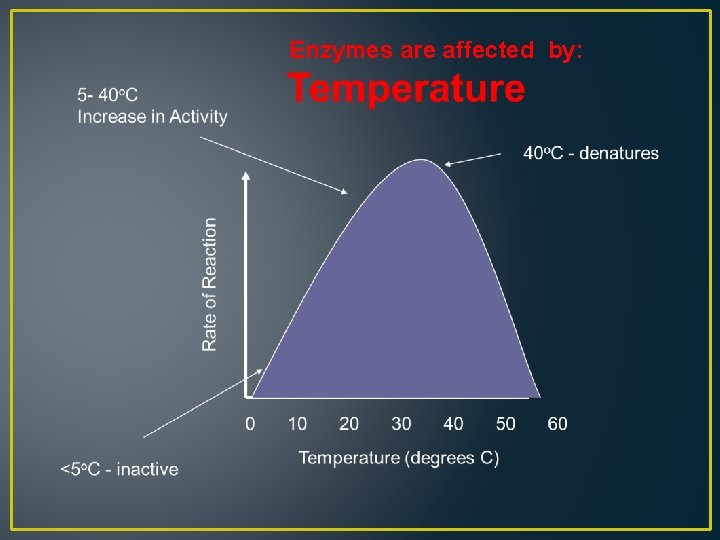
Enzymes are affected by:

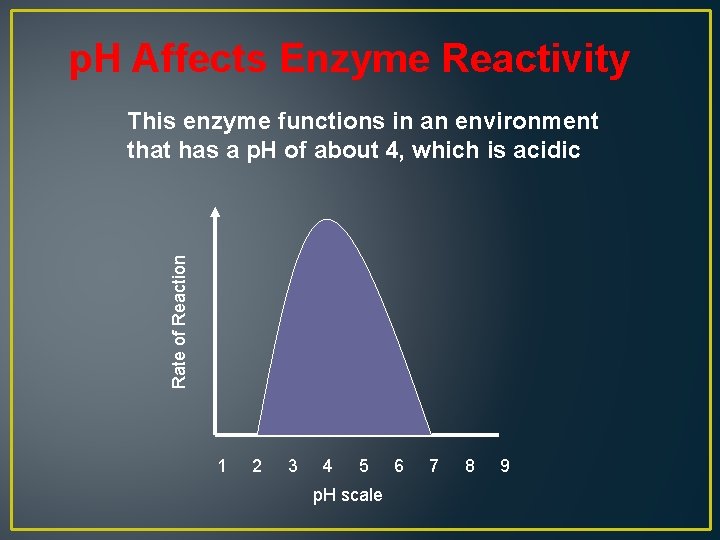
p. H Affects Enzyme Reactivity Rate of Reaction This enzyme functions in an environment that has a p. H of about 4, which is acidic 1 2 3 4 5 p. H scale 6 7 8 9

Nucleic Acids – A Polymer of Nucleotides

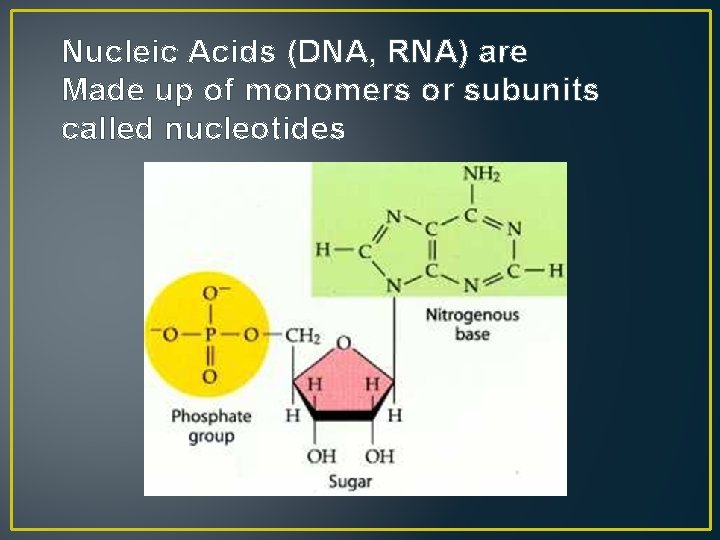
Nucleic Acids (DNA, RNA) are Made up of monomers or subunits called nucleotides

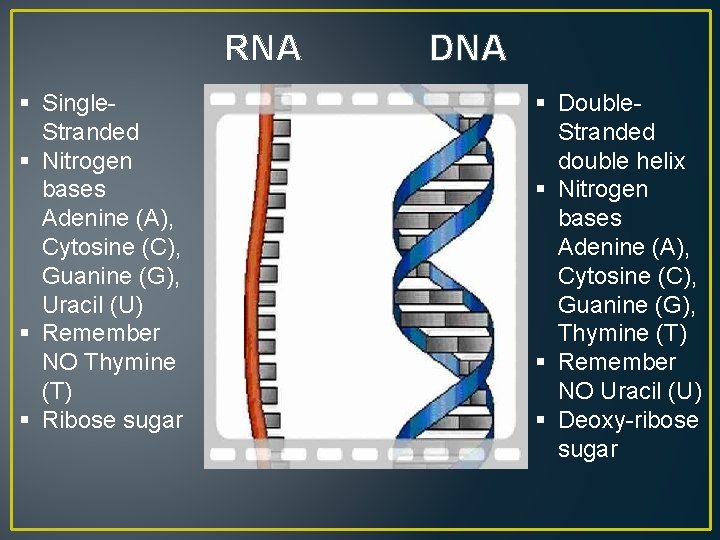
RNA § Single. Stranded § Nitrogen bases Adenine (A), Cytosine (C), Guanine (G), Uracil (U) § Remember NO Thymine (T) § Ribose sugar DNA § Double. Stranded double helix § Nitrogen bases Adenine (A), Cytosine (C), Guanine (G), Thymine (T) § Remember NO Uracil (U) § Deoxy-ribose sugar

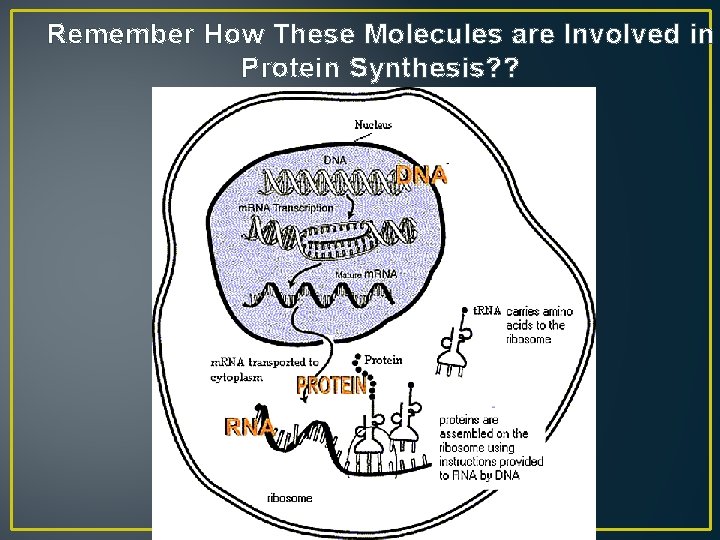
Remember How These Molecules are Involved in Protein Synthesis? ?

Your Turn…. • Complete the table of organic molecules • Use the handout and this Power. Point to guide you • Cut out the illustrations and paste them in the appropriate place!