Organic Macromolecules Macromolecules Smaller organic molecules join together












![§ § § c. Polysaccharides: (C 6 H 10 O 5)n [molecular formula] N= § § § c. Polysaccharides: (C 6 H 10 O 5)n [molecular formula] N=](https://slidetodoc.com/presentation_image_h2/4a31eed8abb13e4e31a916e16ff55d96/image-21.jpg)








- Slides: 35

Organic Macromolecules

Macromolecules Smaller organic molecules join together to form larger molecules macromolecules 4 major classes of macromolecules: carbohydrates lipids proteins nucleic acids

Polymers Long molecules covalently bonded by linking chains of repeating smaller units polymers monomers = repeated small units each cell has millions of diff macromolecules Think: 26 letters of alphabet for all possible combinations

How to break down a polymer Hydrolysis use H 2 O to break apart monomers reverse of condensation reaction H 2 O is split into H and OH H & OH group attach where the covalent bond used to be ex: digestion is hydrolysis

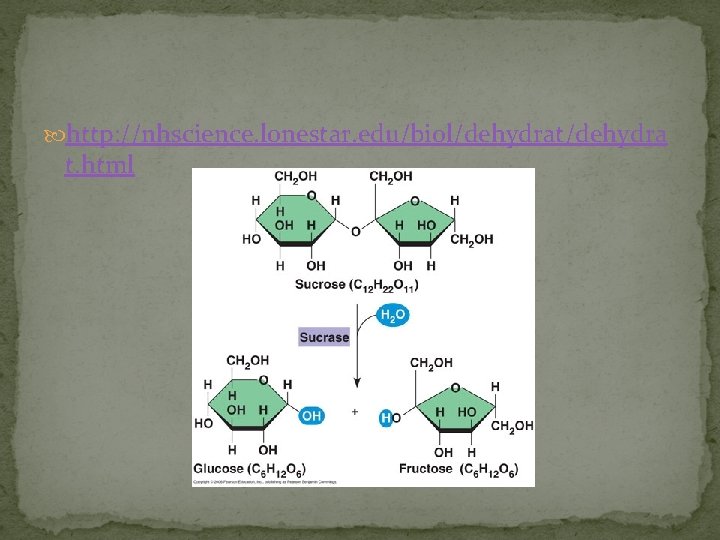
http: //nhscience. lonestar. edu/biol/dehydrat/dehydra t. html

Carbohydrates So what’s all this talk about carbs? ? Atkin’s Diet & South Beach Diet

1. contain C, H, O 2. 1: 2: 1 ratio 3. provide energy or storage 4. many end in “ose” 5. three main groups – monosaccharides, disaccharides, polysaccharides

Carbohydrates are composed of C, H, O carbo - hydr - ate CH 2 O (CH 2 O)x C 6 H 12 O 6 Function: energy raw materials energy storage u structural materials u Monomer: single sugar molecules (monosaccharides) ex: sugars & starches

Sugars Most names for sugars end in -ose Classified by number of carbons 6 C = hexose (glucose) 5 C = pentose (fructose, ribose) 3 C = triose (glyceraldehyde): imp intermediate in metabolic process of cell respiration (burning glucose for energy)

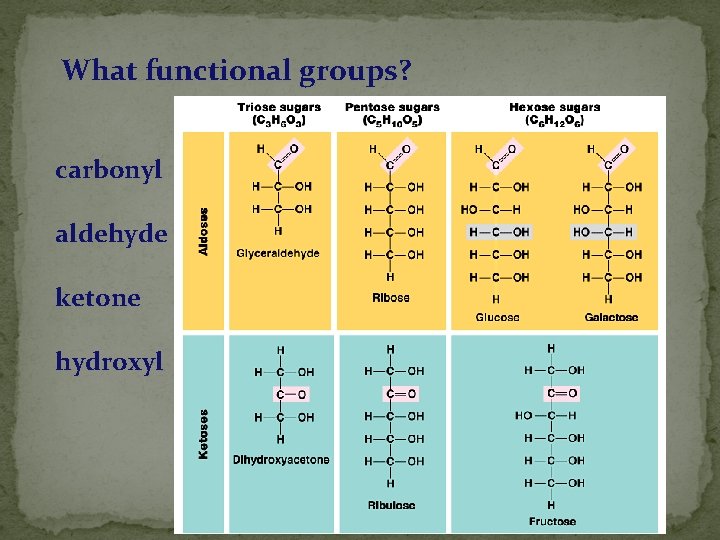
What functional groups? carbonyl aldehyde ketone hydroxyl 2005 -2006

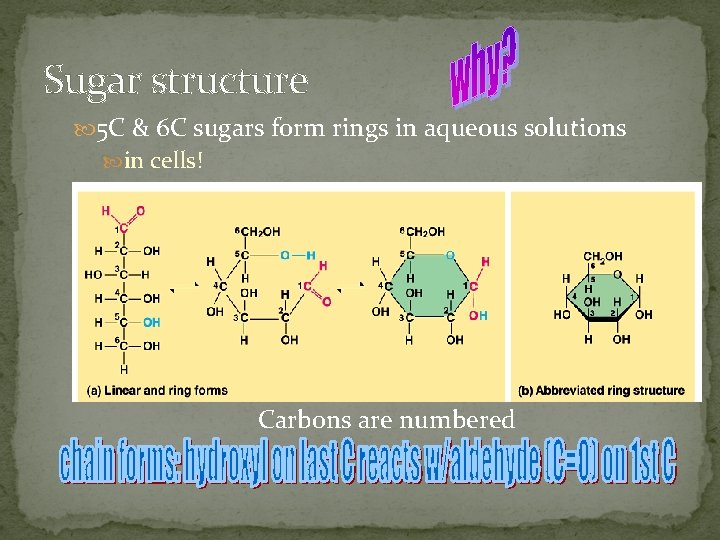
Sugar structure 5 C & 6 C sugars form rings in aqueous solutions in cells! Carbons are numbered

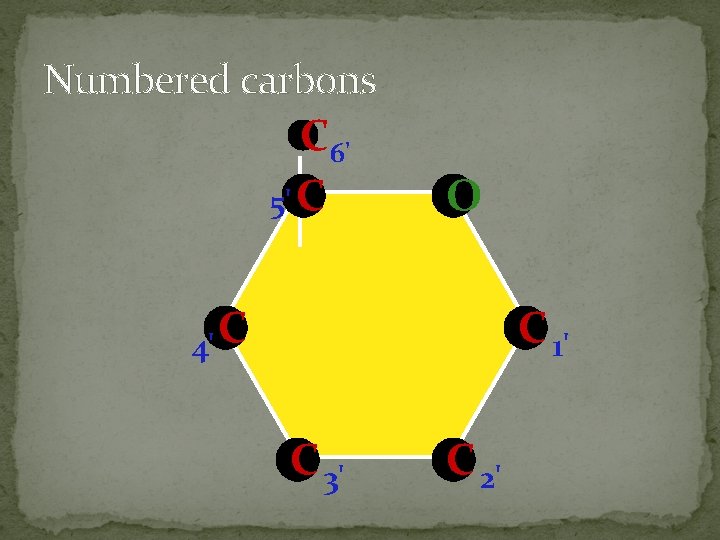
Numbered carbons C 6' 5' C O 4' C C 1' C 3' C 2'

Simple & complex sugars Monosaccharides simple 1 monomer sugars glucose Disaccharides 2 monomers sucrose Polysaccharides large polymers starch

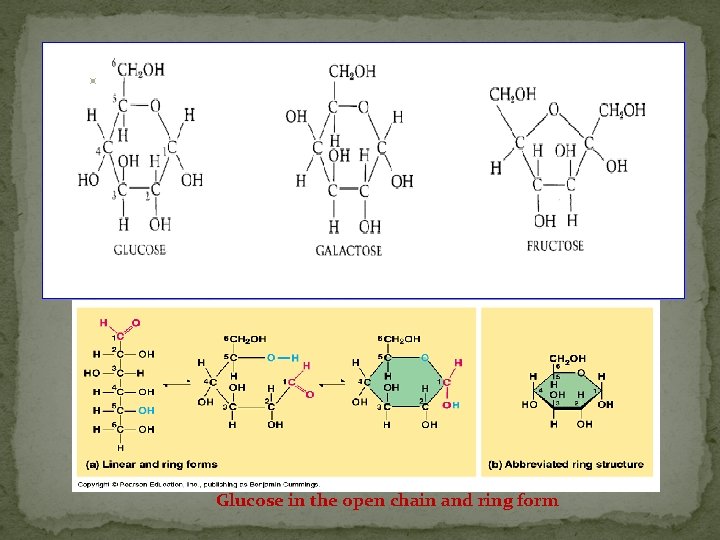
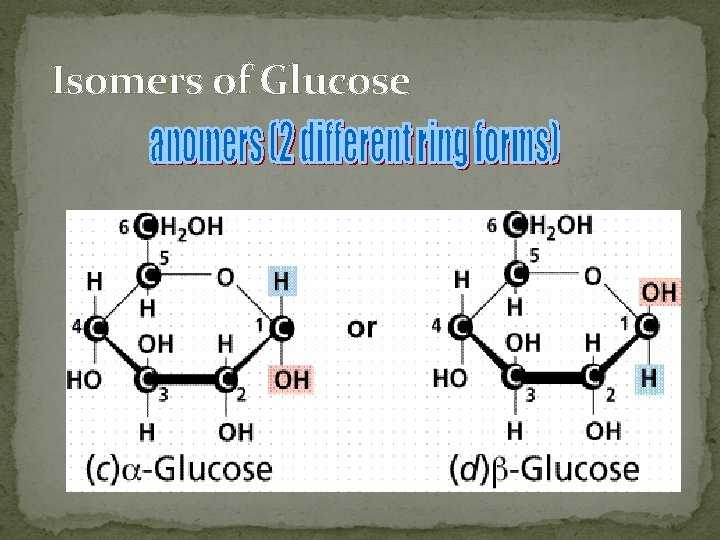
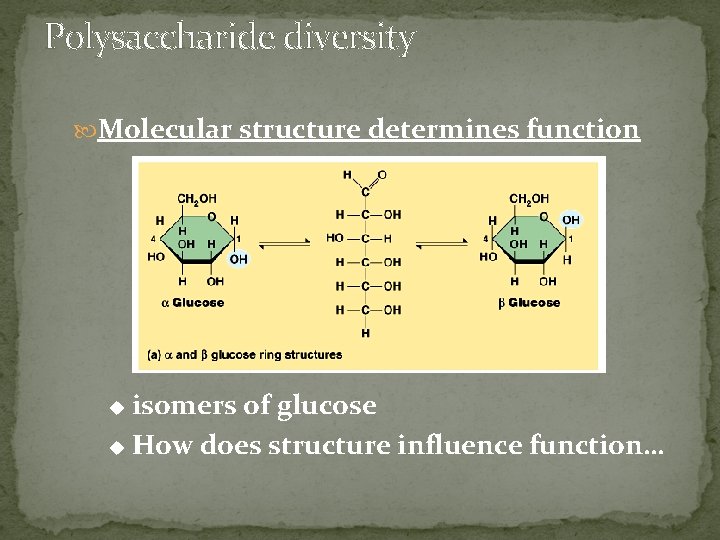
§ § § § a. Monosaccharides: simplest form of carbohydrates building blocks for larger carbohydrates (also called monomers) usually contain molecular formula C 6 H 12 O 6 isomers: glucose, fructose, galactose glucose (blood sugar), fructose & galactose (fruit sugars) can occur as straight (open) chains or as rings numbering of carbon atoms in ring depends on the straight chain (always # the carbon atoms so that the carbon attached to the functional group is the smallest number possible)

Glucose in the open chain and ring form

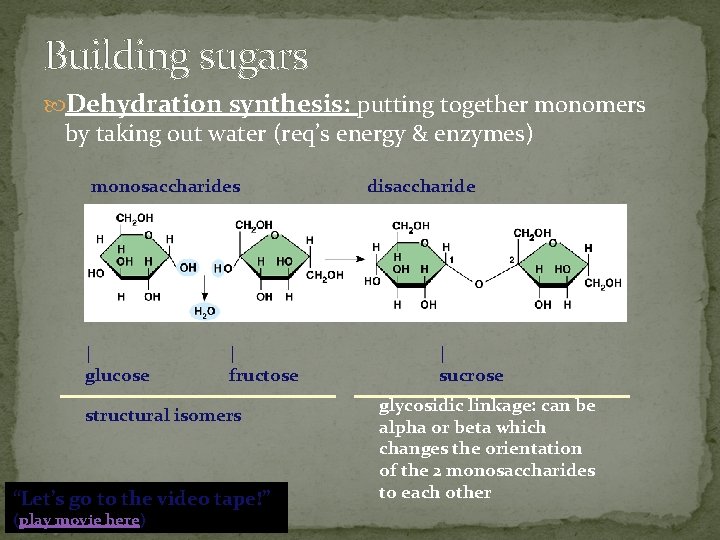
Building sugars Dehydration synthesis: putting together monomers by taking out water (req’s energy & enzymes) monosaccharides | glucose | fructose structural isomers “Let’s go to the video tape!” (play movie here) disaccharide | sucrose glycosidic linkage: can be alpha or beta which changes the orientation of the 2 monosaccharides to each other

Isomers of Glucose

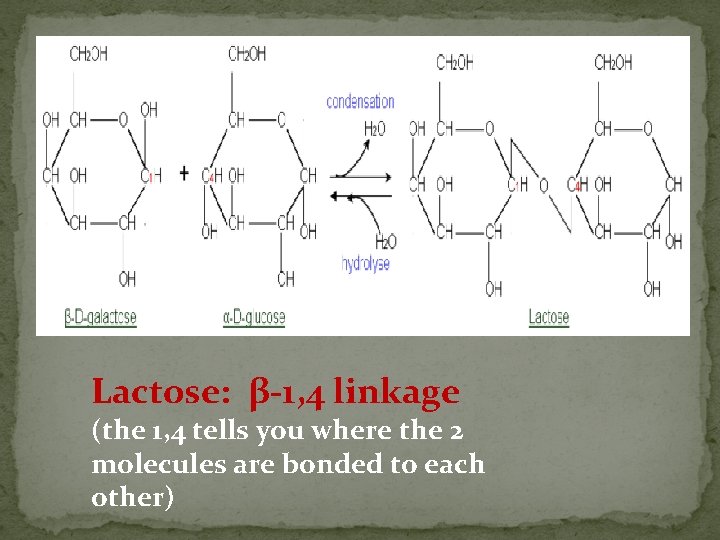
Lactose: β-1, 4 linkage (the 1, 4 tells you where the 2 molecules are bonded to each other)

§ § § § § b. Disaccharides: C 12 H 22 O 11 (molecular formula) 2 monosaccharides joined twice the energy sucrose (table sugar) = 1 glucose + 1 fructose maltose (found in sugar cane) = 1 glucose + 1 glucose lactose (milk sugar) = 1 glucose + 1 galactose maltose has alpha linkage (bottom to bottom) sucrose has alpha linkage (bottom to bottom) lactose has beta linkage (top to bottom) 1, 4 or 1, 2 numbering tells you what carbon numbers the linkage is being formed at

Polysaccharides (polymers of hundreds to thousands of monosaccharides) Polymers of sugars costs little energy to build easily reversible = release energy Function: energy storage starch (plants) glycogen (animals) building materials = structure cellulose (plants) chitin (arthropods & fungi)
![c Polysaccharides C 6 H 10 O 5n molecular formula N § § § c. Polysaccharides: (C 6 H 10 O 5)n [molecular formula] N=](https://slidetodoc.com/presentation_image_h2/4a31eed8abb13e4e31a916e16ff55d96/image-21.jpg)
§ § § c. Polysaccharides: (C 6 H 10 O 5)n [molecular formula] N= 5 -5, 000 monosaccharides Form chains of polymers (repeating monomer units) 2 main functions: food storage and structure Food storage Structure cellulose plant starches animal starches amylose amylopectin glycogen chitin

Branched v linear polysaccharides (made of repeating glucose monomers) Q: can you see the difference between starch & glycogen? Which is easier to digest? 2005 -2006

Glycogen α -1, 4 linkage on the straight chain α -1, 6 linkage at the branch points (every 10 glucose molecules)

Polysaccharide diversity Molecular structure determines function isomers of glucose u How does structure influence function… u

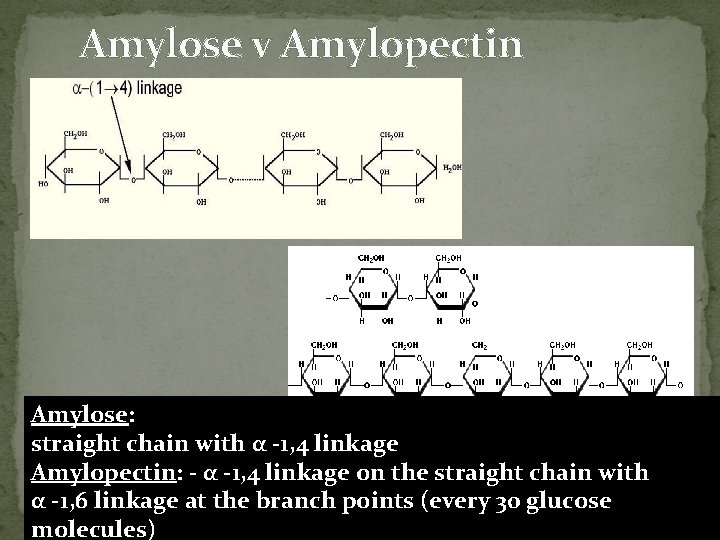
Amylose v Amylopectin Amylose: straight chain with α -1, 4 linkage Amylopectin: - α -1, 4 linkage on the straight chain with α -1, 6 linkage at the branch points (every 30 glucose molecules)

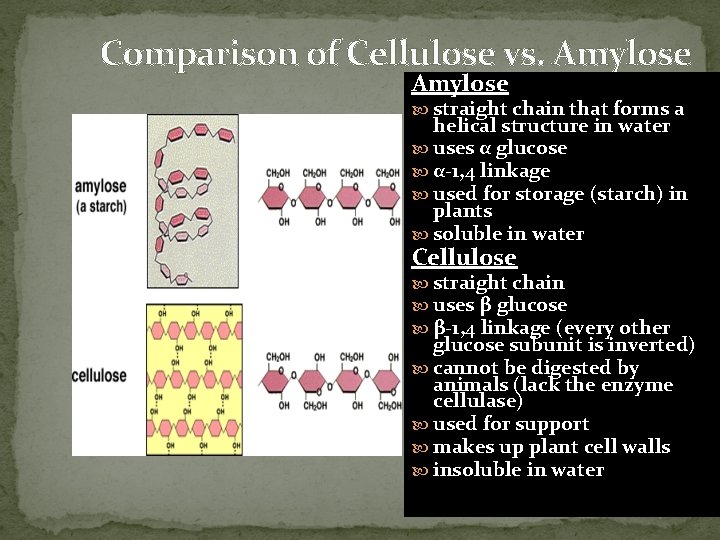
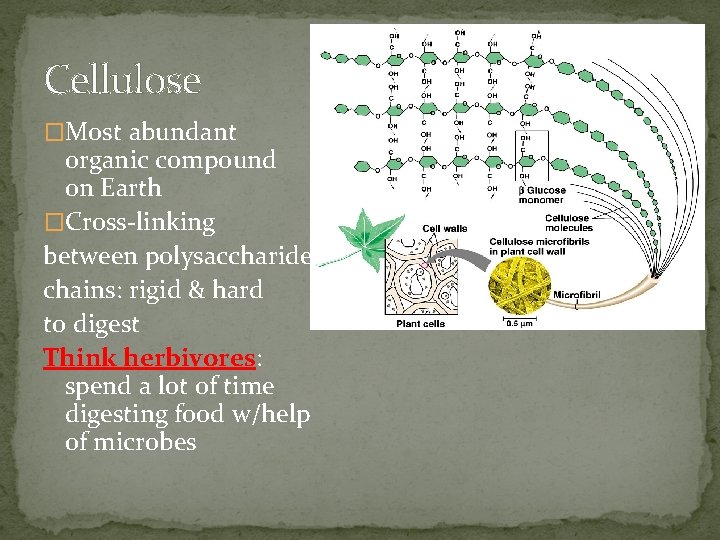
Comparison of Cellulose vs. Amylose straight chain that forms a helical structure in water uses α glucose α-1, 4 linkage used for storage (starch) in plants soluble in water Cellulose straight chain uses β glucose β-1, 4 linkage (every other glucose subunit is inverted) cannot be digested by animals (lack the enzyme cellulase) used for support makes up plant cell walls insoluble in water

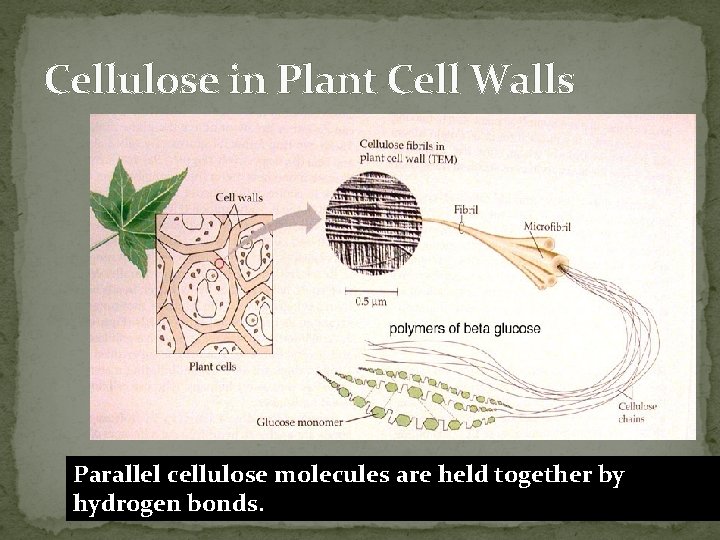
Cellulose in Plant Cell Walls Parallel cellulose molecules are held together by hydrogen bonds.

Cellulose �Most abundant organic compound on Earth �Cross-linking between polysaccharide chains: rigid & hard to digest Think herbivores: spend a lot of time digesting food w/help of microbes

Digesting starch vs. cellulose (starch: all the glycosidic linkages are on same side: molecule lies flat)

Cow can digest cellulose well; no need to eat supplemental sugars Gorilla can’t digest cellulose well; must supplement with sugar source, like fruit 2005 -2006

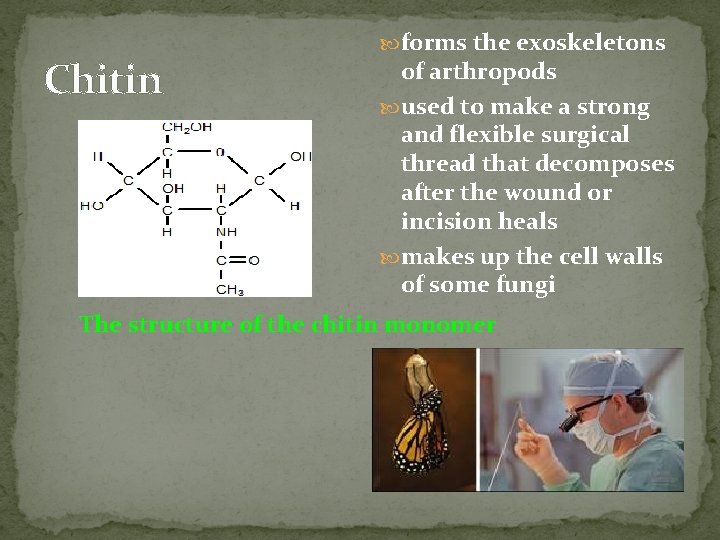
Chitin forms the exoskeletons of arthropods used to make a strong and flexible surgical thread that decomposes after the wound or incision heals makes up the cell walls of some fungi The structure of the chitin monomer

Glycemic index Which food will get into your blood more quickly? apple rice cakes corn flakes bagel peanut M&M 2005 -2006

Glycemic index Ranking of carbohydrates based on their immediate effect on blood glucose (blood sugar) levels Carbohydrate foods that breakdown quickly during digestion have the highest glycemic indices. Their blood sugar response is fast & high. 2005 -2006

Glycemic index Which food will get into your blood more quickly? apple rice cakes corn flakes bagel peanut M&M 36 82 84 72 33 2005 -2006

Let’s build some Carbohydrates!