Organic Macromolecules Carbohydrates Lipids Proteins Enzymes Nucleic Acids

Organic Macromolecules Carbohydrates, Lipids, Proteins, Enzymes, Nucleic Acids, ATP and Micronutrients

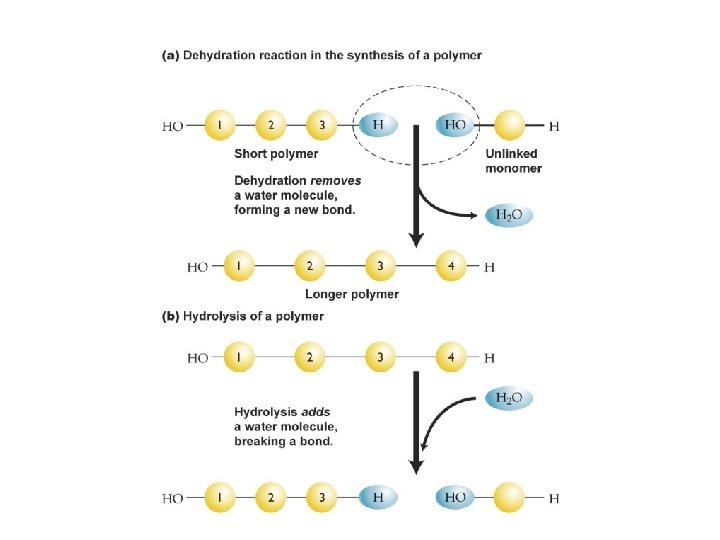
Organic Macromolecules § Organic compounds are comprised of C, H and usu. O § Organic chemicals found in living organisms come in four categories: § Carbohydrates – sugars in humans/ starches in plants § Lipids – fats, phospholipids and steroids § Proteins – amino acids § Nucleic Acids – RNA and DNA § These molecules are called macromolecules. § They are composed of subunits called monomers, which join together to form polymers.

I. Carbohydrates (sugars) § Major source of energy for cellular metabolism; body uses carbohydrates first for fuel § The energy is stored in the bonds between the 6 carbon, 12 hydrogen, and 6 oxygen atoms in the glucose sugar molecule. § Chemical formula: C 6 H 12 O 6 § Glucose is a monosaccharide, consisting of a single ring structure.
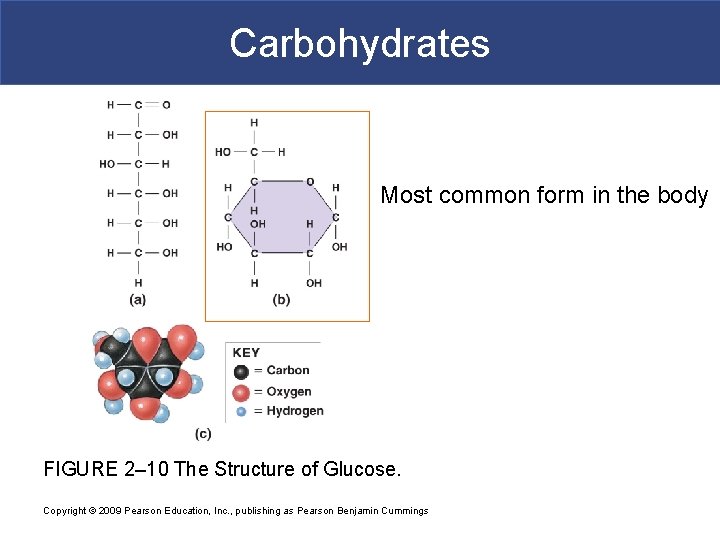
Carbohydrates Most common form in the body FIGURE 2– 10 The Structure of Glucose. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
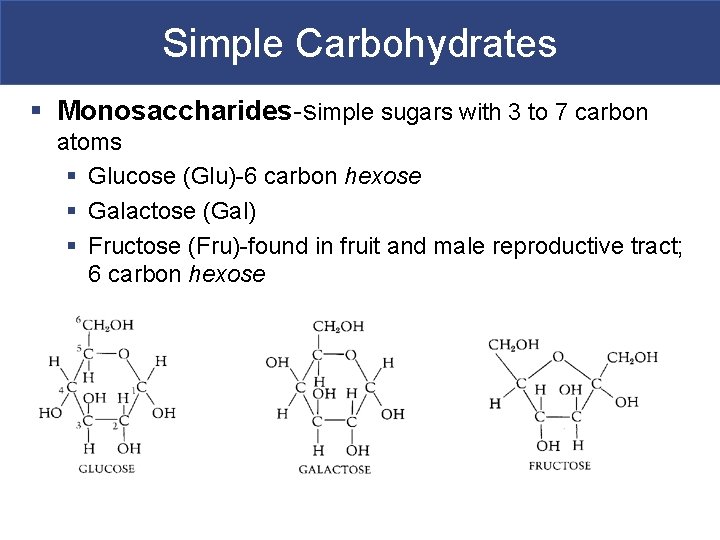
Simple Carbohydrates § Monosaccharides-simple sugars with 3 to 7 carbon atoms § Glucose (Glu)-6 carbon hexose § Galactose (Gal) § Fructose (Fru)-found in fruit and male reproductive tract; 6 carbon hexose
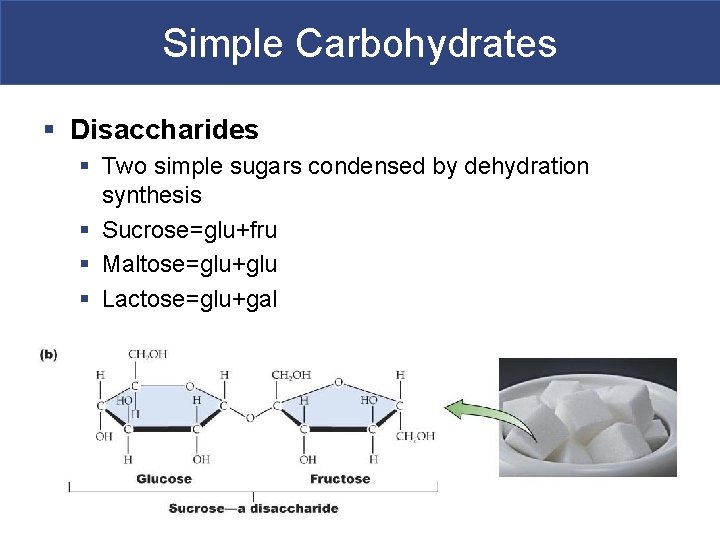
Simple Carbohydrates § Disaccharides § Two simple sugars condensed by dehydration synthesis § Sucrose=glu+fru § Maltose=glu+glu § Lactose=glu+gal
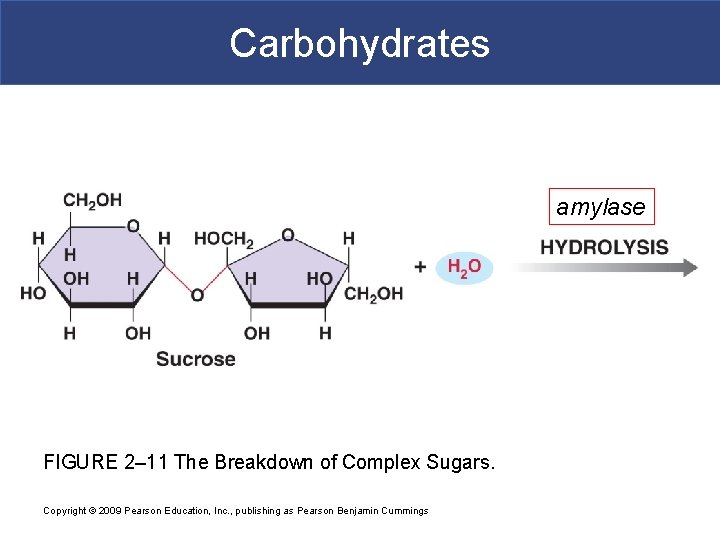
Carbohydrates amylase FIGURE 2– 11 The Breakdown of Complex Sugars. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
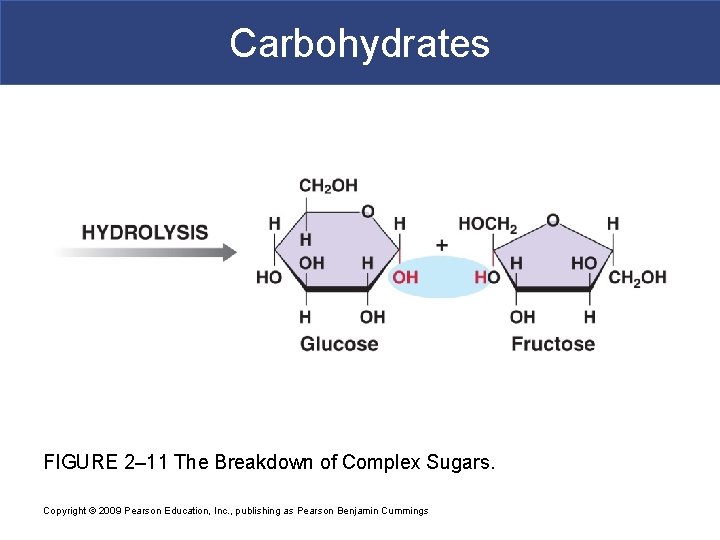
Carbohydrates FIGURE 2– 11 The Breakdown of Complex Sugars. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

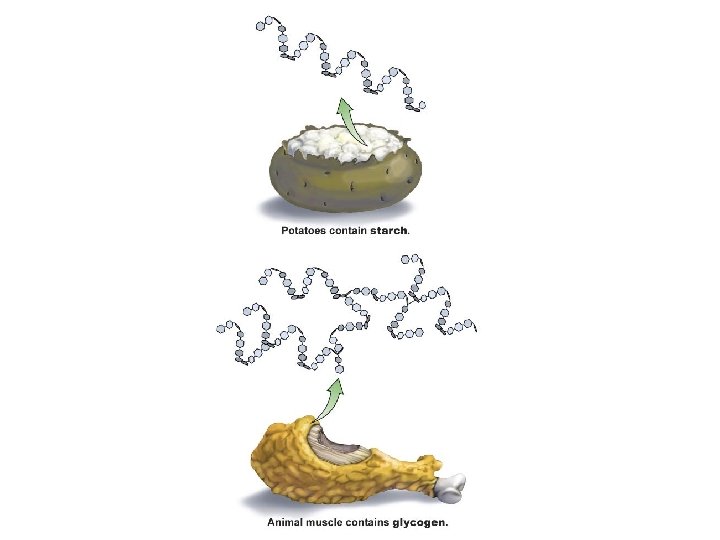
Complex Carbohydrates § Polysaccharides § Many monosaccharides condensed by dehydration synthesis to form a polymer § Form polysaccharides by dehydration synthesis in body § Take more time to digest § Glycogen-storage form of glc in animals that is stored in liver and skeletal muscle § Starch-storage form of glc in plants that is stored in roots (most we can digest) § Cellulose-component of plant cell wall that we cannot digest
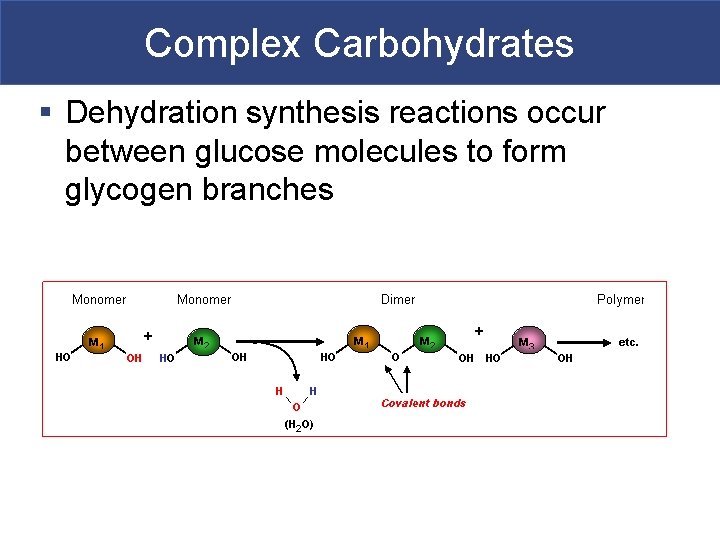
Complex Carbohydrates § Dehydration synthesis reactions occur between glucose molecules to form glycogen branches
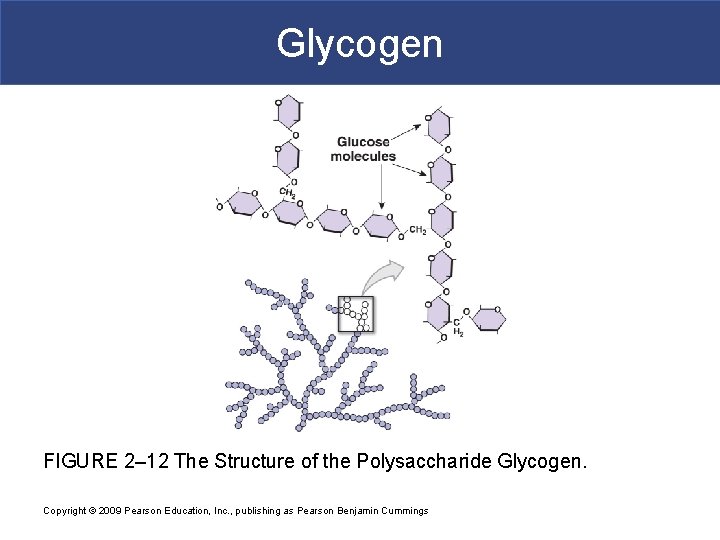
Glycogen FIGURE 2– 12 The Structure of the Polysaccharide Glycogen. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Complex Carbohydrates § Cellulose-insoluble dietary fiber that keeps your digestive tract moving. § Soluble dietary fiber (oat bran, legumes, fruits and veggies) slows down digestion, protects blood vessels, decreases cholesterol and the risk of some cancers. § What is soluble fiber?

Simple vs. Complex Carbs
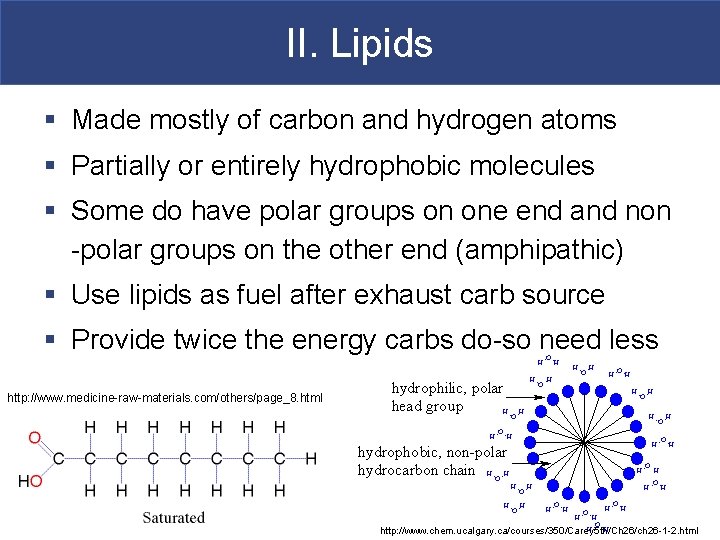
II. Lipids § Made mostly of carbon and hydrogen atoms § Partially or entirely hydrophobic molecules § Some do have polar groups on one end and non -polar groups on the other end (amphipathic) § Use lipids as fuel after exhaust carb source § Provide twice the energy carbs do-so need less http: //www. medicine-raw-materials. com/others/page_8. html http: //www. chem. ucalgary. ca/courses/350/Carey 5 th/Ch 26/ch 26 -1 -2. html

Lipids § Lipids need transport proteins to go into blood because they are not water soluble § Most lipids are transported by some type of lipoproteins (LDL and HDL) § Free fatty acids are transported by albumin (made by liver) § Examples of lipids: triglycerides, phospholipids, eicosanoids, and steroids.

IIA. Triglycerides § Triglycerides= glycerol backbone + 3 fatty acid tails § Non polar, so they have transport proteins called very low density lipoproteins (VLDL) to transport them in blood § What we typically call “fats” – We eat them, they are stored in adipocytes as energy reserves; fatty acid tails are broken down for fuel § Have four important functions: – Used as fuel – Protect internal organs – Use as insulation so you lose less heat to environment – Attract and store lipid-soluble vitamins A, D, E & K, drugs, and toxins (Hg & DDT)**
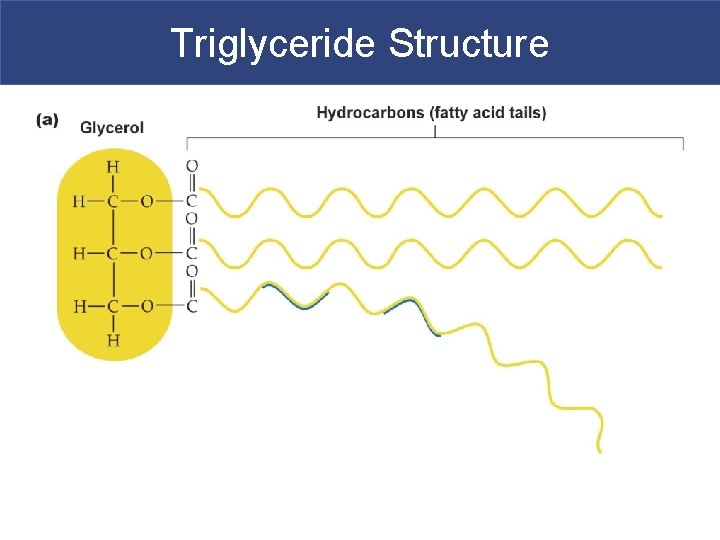
Triglyceride Structure
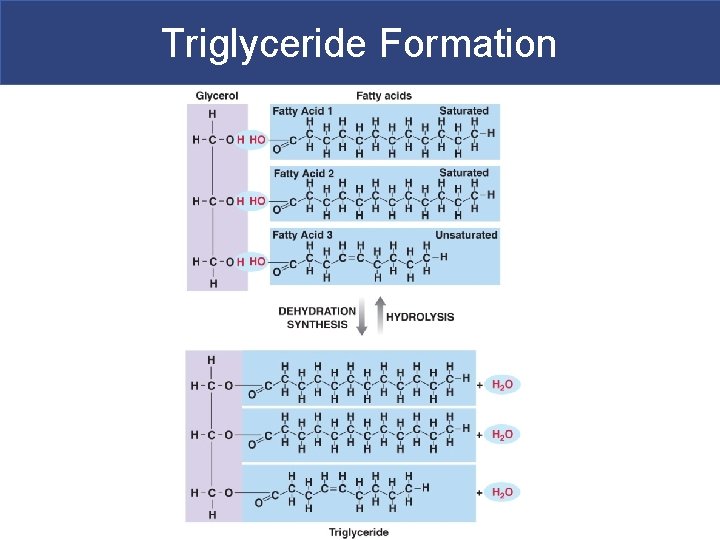
Triglyceride Formation

Fatty Acids § Fatty Acids/ Fatty Acid Tails § Long chains of carbon and hydrogen with a carboxylic acid group (COOH) at one end § Hydrocarbon chain is nonpolar while the carboxylic acid group is polar
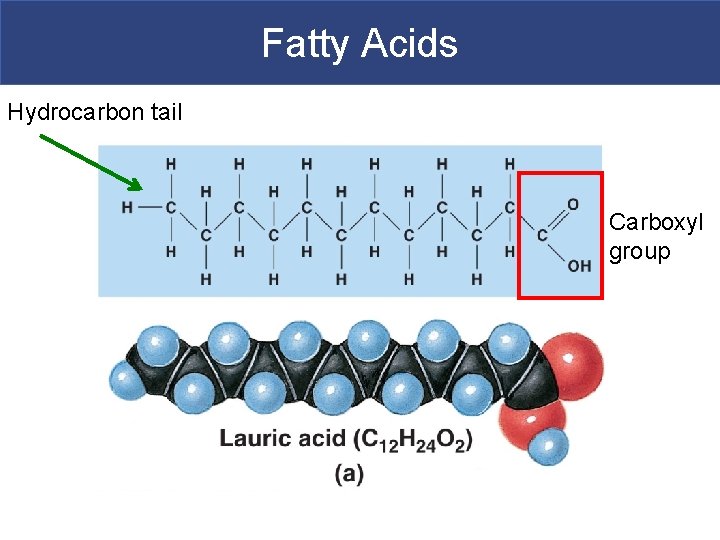
Fatty Acids Hydrocarbon tail Carboxyl group

Fatty Acids § Breaking the C-C bond yields energy; since there are more bond than in carbs you get more energy § Most we can synthesize but some we must get from our diet § Characterized by chain length (short, medium or long-chain fatty acids) & number of double bonds (saturated, unsaturated, or polyunsaturated fatty acids)

Fatty Acids § Omega-3 and omega-6 are essential fatty acids. They must be obtained from the diet. § Omega-3 fatty acids are associated with healthy brain and nerve function and decreased body inflammation. § Eicosapentaenoic, docasahexaenoic, and alpha linolenic acids § Found in salmon, sardines, flax seed, and walnuts

Essential Fatty Acids § Omega-6 fatty acids are associated with inflammation. We can produce small amounts of this f. a. § Omega-6 f. a. are found in eggs, animal meat, and vegetable oil. § We get enough omega-6 from our diet; we need more omega-3 in our diet.

Saturated vs. Unsaturated Fatty Acids § Fatty acids/fatty acid tails can be: § Saturated (no double bonds between carbons) § All carbons have the max amount of H’s bonded § Unsaturated (one or more double bonds in the fatty acid chain): § monounsaturated = one double bond § polyunsaturated = two or more double bonds
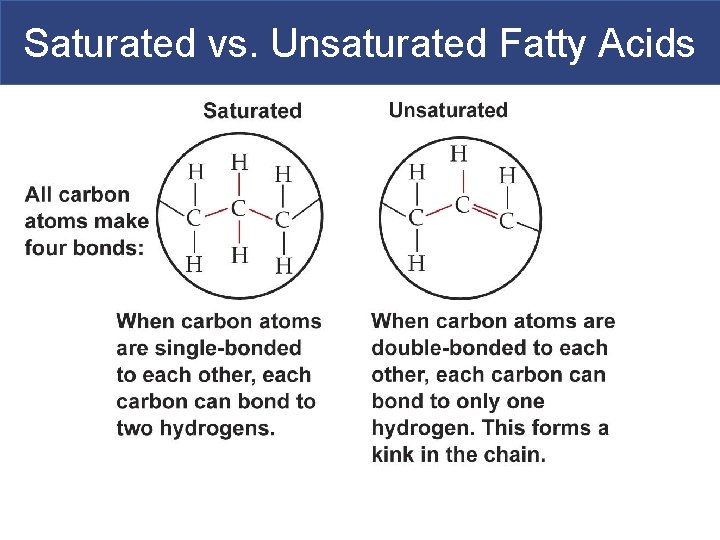
Saturated vs. Unsaturated Fatty Acids

Saturated vs. Unsaturated Fatty Acids § The single bonds in saturated fats/saturated fatty acid tails allow the fats to pack together tightly. That is why they are solid at room temperature. § The double bonds in unsaturated fats make the fatty acid tails kink. Thus, they are liquid at room temperature. § Vegetable oils are unsaturated fats. § Plants tend to be richer in unsaturated fats than animals.

Saturated vs. Unsaturated Fatty Acids § Excess saturated fat intake is associated with several diseases § Nutritionists recommend that the amount of fat in our diet be limited to 30% § Fewer negative health effects are associated with unsaturated fats § Unsaturated fats require more energy to break down
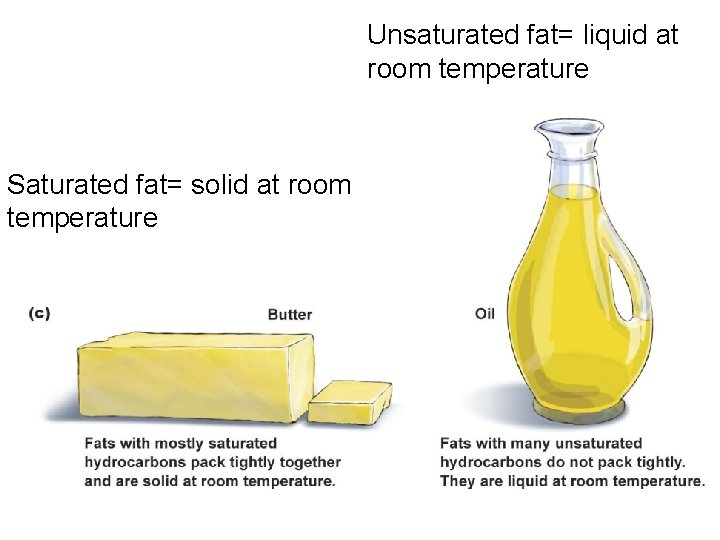
Unsaturated fat= liquid at room temperature Saturated fat= solid at room temperature

Cis and Trans Unsaturated Fats § cis configuration – when the hydrogen atoms are on the same side of the carbon to carbon double bond. These occur in nature. § trans configuration – when the hydrogen atoms are on opposite sides of the double bond. § These are not found in nature. § These are the source of potential health risks. § The conformation of the trans fat helps it adhere to artery walls and form plaque.
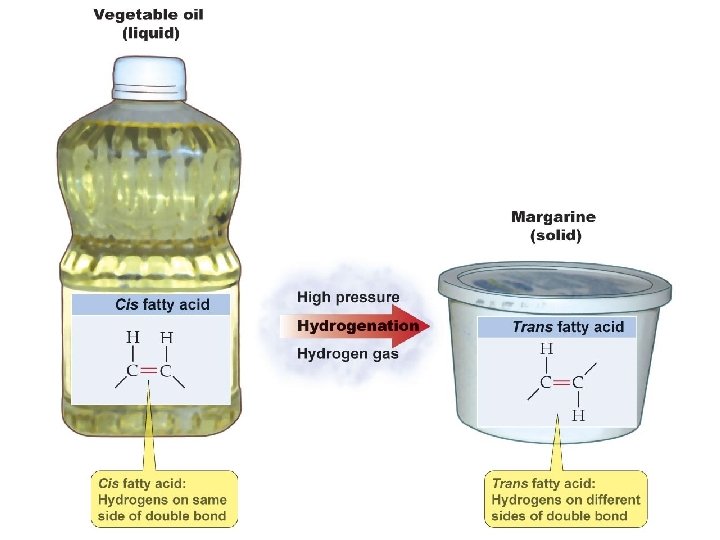
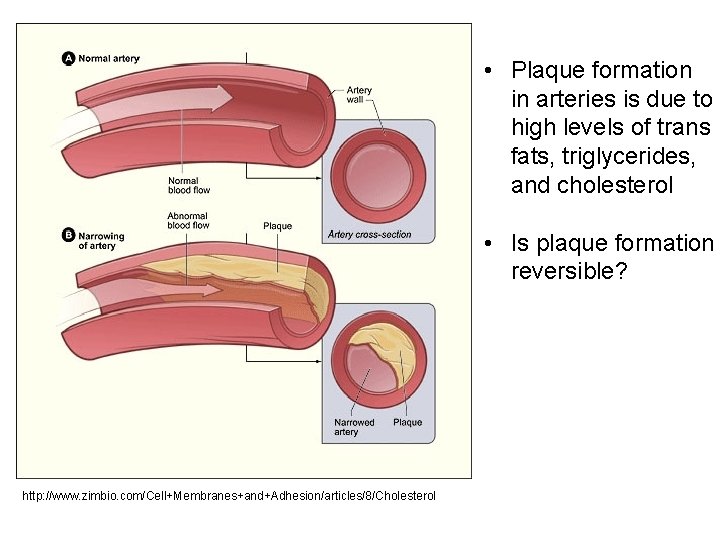
• Plaque formation in arteries is due to high levels of trans fats, triglycerides, and cholesterol • Is plaque formation reversible? http: //www. zimbio. com/Cell+Membranes+and+Adhesion/articles/8/Cholesterol

IIB. Phospholipids § Phospholipids= phosphate group + 2 fatty acid tails § It is a diglyceride (has 2 fatty acid tails) § It has a hydrophilic head and hydrophobic tail § Main component of cellular membrane
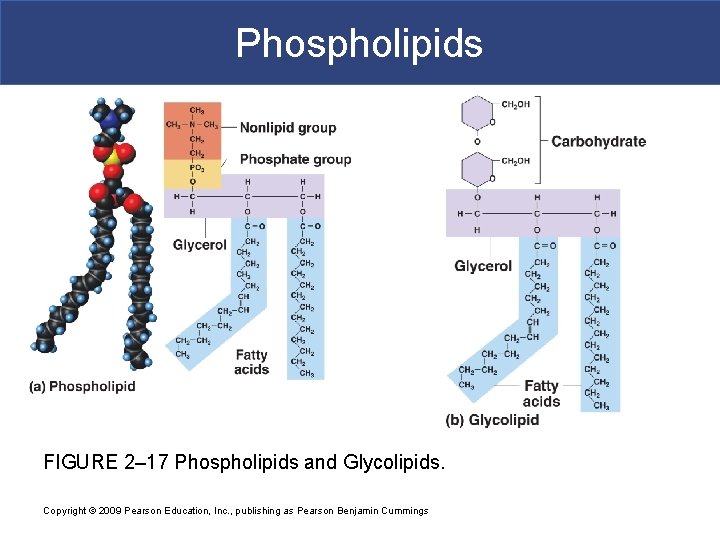
Phospholipids FIGURE 2– 17 Phospholipids and Glycolipids. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
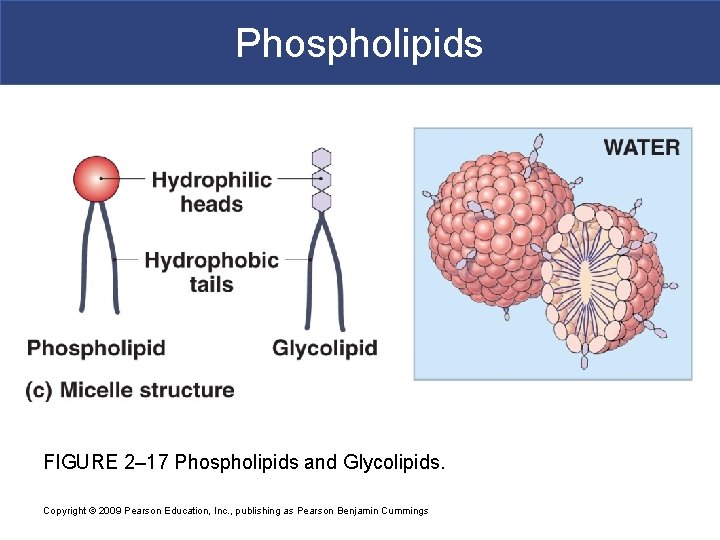
Phospholipids FIGURE 2– 17 Phospholipids and Glycolipids. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
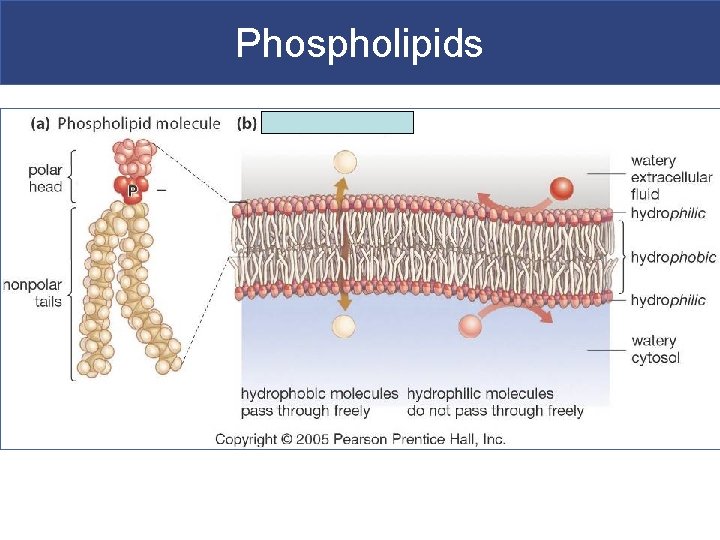
Phospholipids

IID. Steroids § Made up of four fused carbon-containing rings with variable side groups § Cholesterol: § Precursor to other steroids § component of plasma (cell) membranes; aids with fluidity § obtain from diet or synthesize in body § Estrogens and testosterone: § sex hormones § Corticosteroids and calcitriol (vitamin D): § metabolic regulation and calcium absorption § Bile salts: § derived from steroids
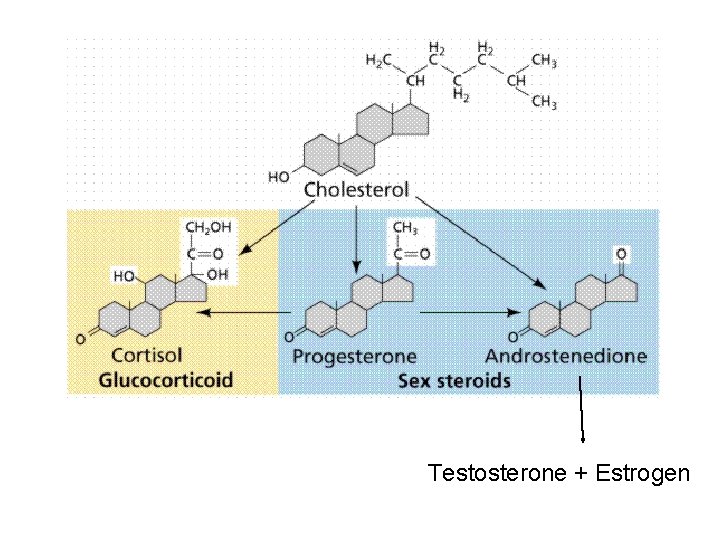
Testosterone + Estrogen

Cholesterol § Cholesterol is insoluble in water. It is carried throughout the body attached to proteins called lipoproteins. § Lipoproteins have three parts: § Core composed of cholesterol esters and triacylglycerols § Surface monolayer composed of phospholipids and cholesterol § On top of surface layer is an apoprotein
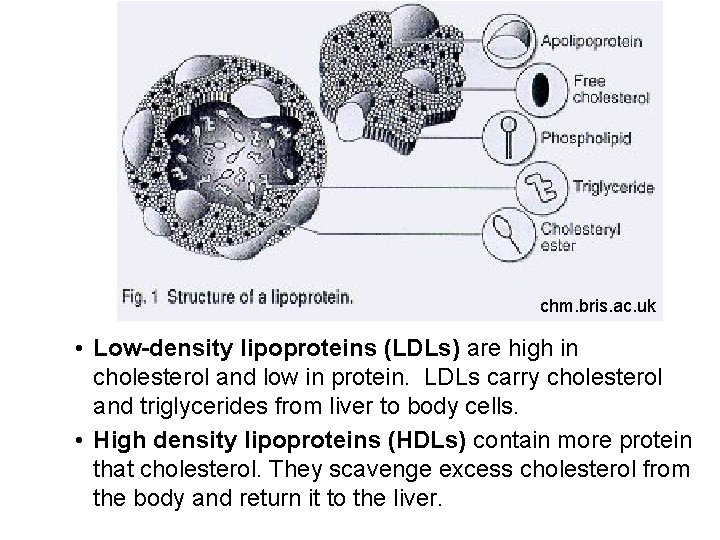
chm. bris. ac. uk • Low-density lipoproteins (LDLs) are high in cholesterol and low in protein. LDLs carry cholesterol and triglycerides from liver to body cells. • High density lipoproteins (HDLs) contain more protein that cholesterol. They scavenge excess cholesterol from the body and return it to the liver.

Cholesterol § The LDL/HDL ratio is an index of the rate at which cholesterol leaves cells and returns to the liver. § A low ratio is good, indicating that less cholesterol is in the bloodstream. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
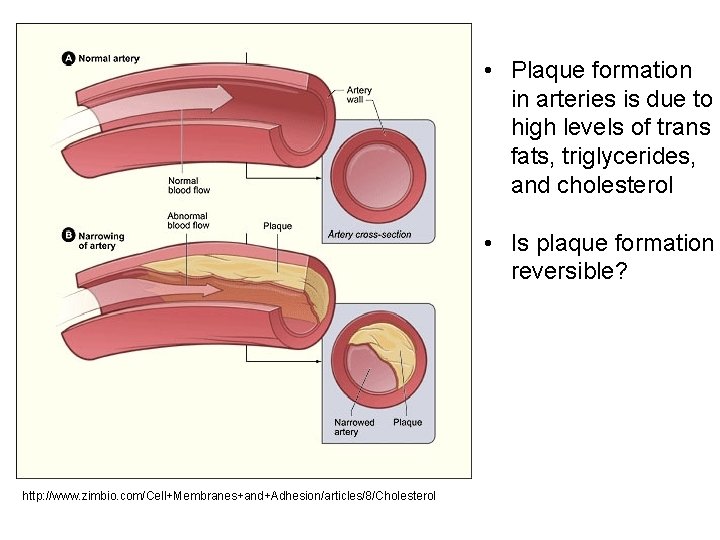
• Plaque formation in arteries is due to high levels of trans fats, triglycerides, and cholesterol • Is plaque formation reversible? http: //www. zimbio. com/Cell+Membranes+and+Adhesion/articles/8/Cholesterol
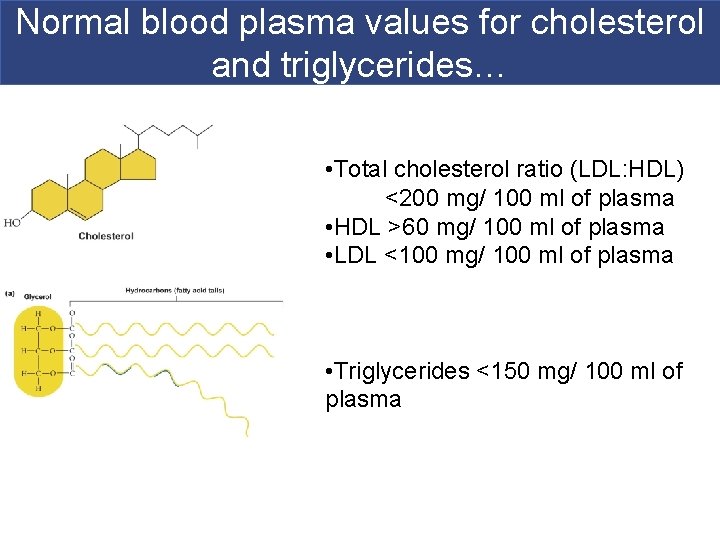
Normal blood plasma values for cholesterol and triglycerides… • Total cholesterol ratio (LDL: HDL) <200 mg/ 100 ml of plasma • HDL >60 mg/ 100 ml of plasma • LDL <100 mg/ 100 ml of plasma • Triglycerides <150 mg/ 100 ml of plasma

III. Proteins § Proteins are the most abundant and important organic molecules § Contain basic elements § Carbon (C), hydrogen (H), oxygen (O), and nitrogen (N) Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Proteins § Seven major protein functions § Support § Structural proteins § Movement § Contractile proteins § Transport (carrier) proteins § Buffering § Regulation of p. H § Metabolic regulation § Enzymes § Coordination and control § Hormones § Defense § Antibodies Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

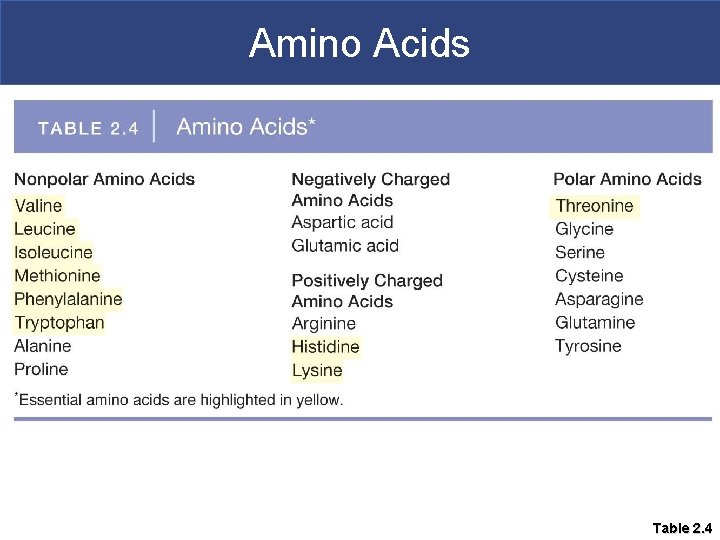
Proteins § Composed of amino acids § There are 20 different amino acids; most can be synthesized by the body § Those the body cannot synthesize are called essential amino acids; these must come from the diet § Protein-rich foods include beef, poultry, fish, beans, eggs, nuts, and dairy products such as milk, yogurt, and cheese.
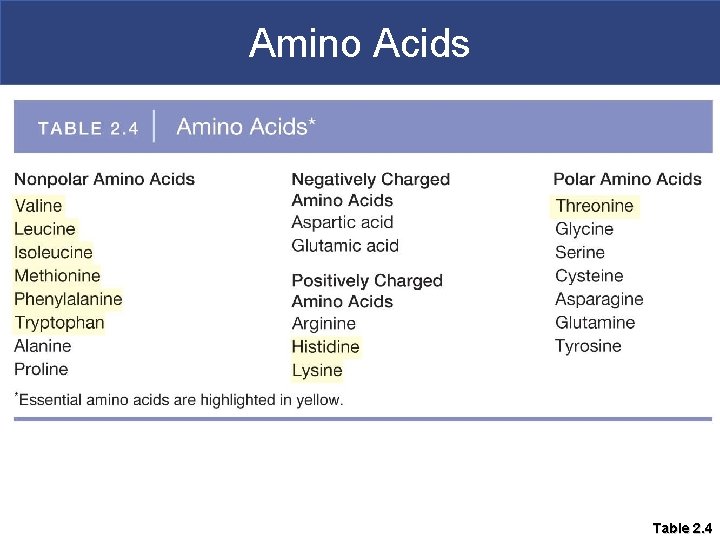
Amino Acids Table 2. 4

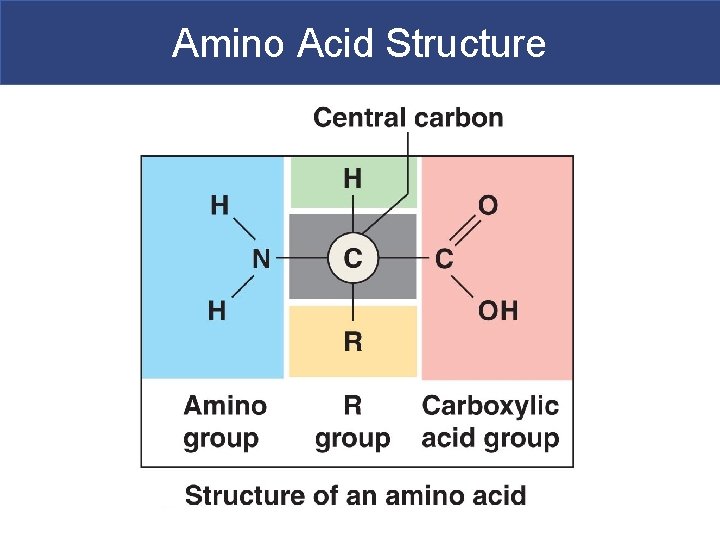
Amino Acids § Amino acid structure § All amino acids have the same: § Central carbon atom § Hydrogen atom § Amino group (—NH 2) § Carboxylic acid group (—COOH) § The variable side chain or R group is different for each amino acid
Amino Acid Structure

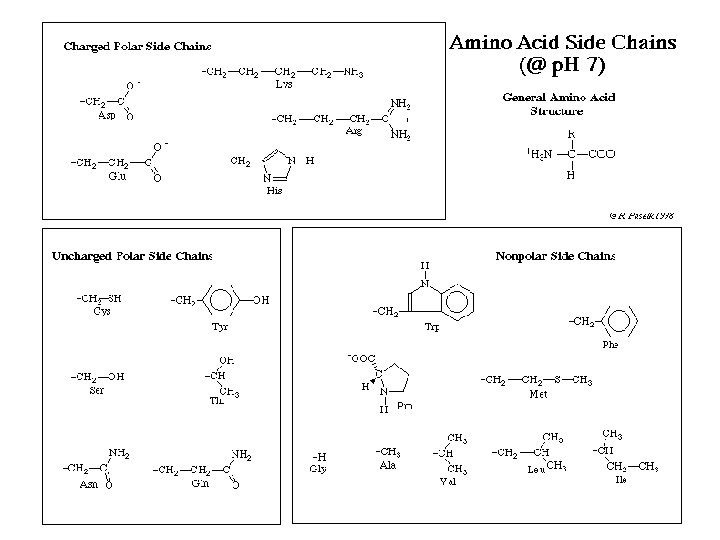
Amino Acids § Each particular amino acid has its own variable side chain (R-group) § The side group gives the amino acid its chemical properties; they can be polar, non-polar, positively charged, or negatively charged
Amino Acids Table 2. 4

Proteins § Hooking amino acids together requires § A dehydration synthesis between § The amino group of one amino acid § And the carboxylic acid group of another amino acid § This produces a peptide Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
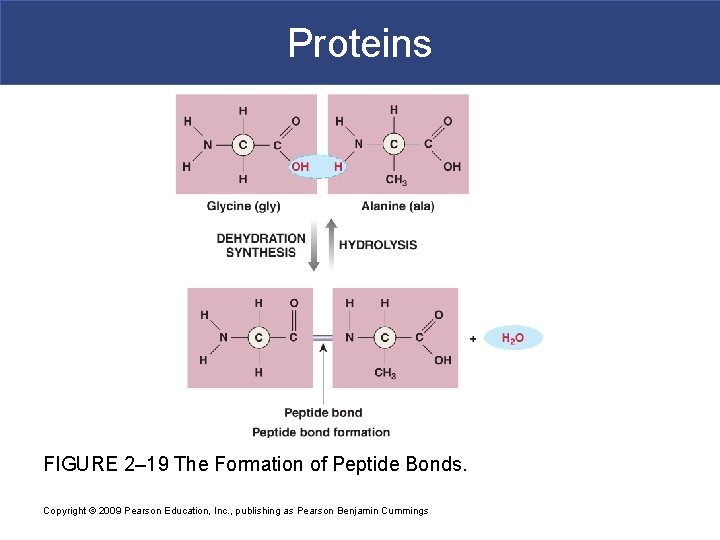
Proteins FIGURE 2– 19 The Formation of Peptide Bonds. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

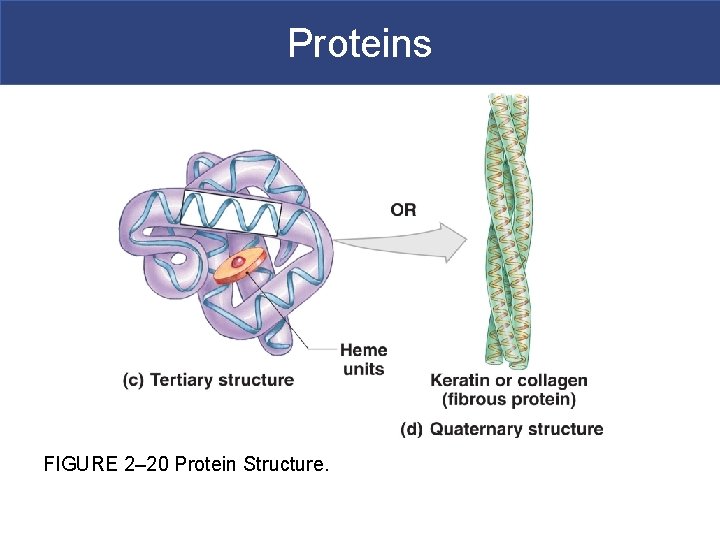
Protein Folding Sequence § Protein folding sequence: § Primary structure § The sequence of amino acids along a polypeptide § Secondary structure § Hydrogen bonds form a-helix or b-pleated sheet § Tertiary structure § Secondary structure folds into a unique shape § Quaternary structure § Final protein shape: – several tertiary structures together Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
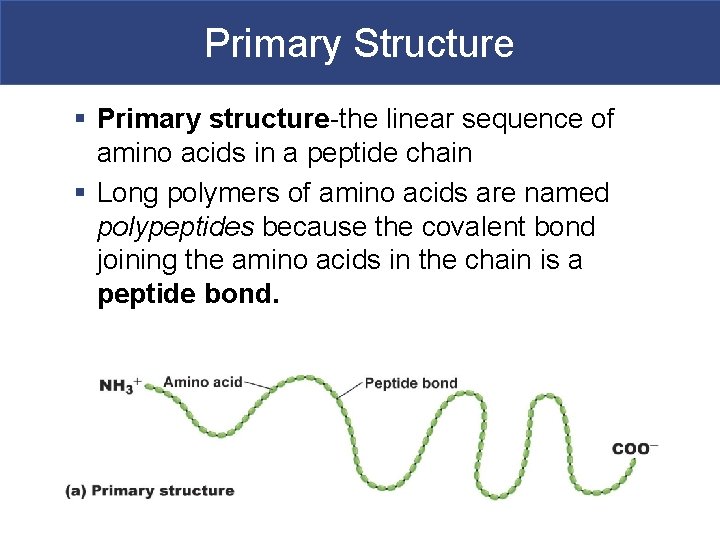
Primary Structure § Primary structure-the linear sequence of amino acids in a peptide chain § Long polymers of amino acids are named polypeptides because the covalent bond joining the amino acids in the chain is a peptide bond.

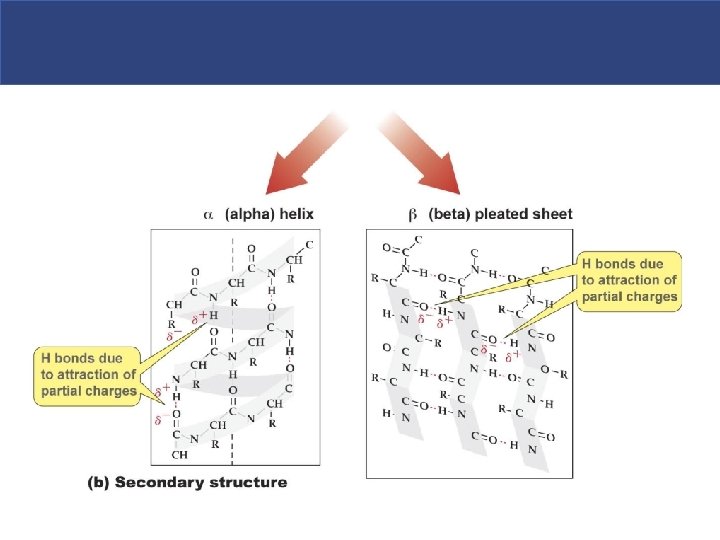
Secondary Structure § Secondary protein structure – the polypeptide folds on itself in a regular pleated fashion. § Folding produce helical structures (ahelices) or accordion-like pleats (b-pleated sheets). § Secondary structures result from hydrogen bonding between the amino group of one amino acid and the carboxyl group of another.
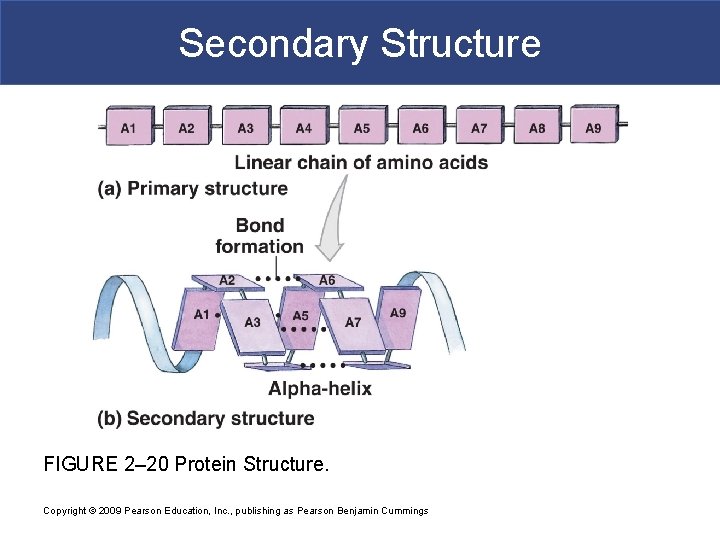
Secondary Structure FIGURE 2– 20 Protein Structure. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

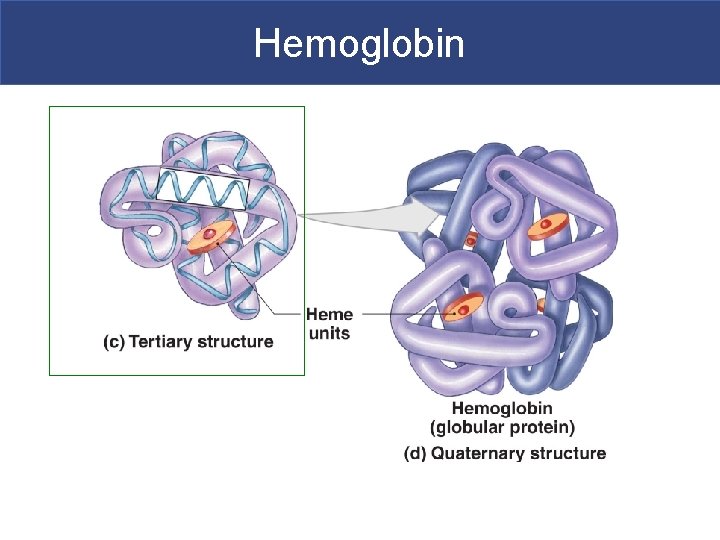
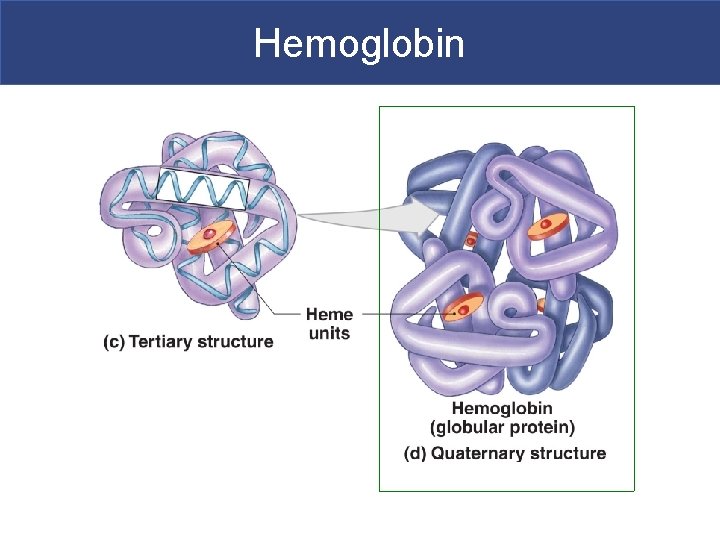
Tertiary Structure § Tertiary structure-comes from the interaction of the amino acid side groups § These interactions involve hydrogen, ionic and covalent bonds § Side groups interact with one another to affect the three-dimensional structure of the protein
Hemoglobin

Quaternary Structure § Quaternary structure-two or more tertiary structures bound together § Gives protein it’s final shape § Structure determines function
Hemoglobin

Proteins § Two structural classes of proteins: § Fibrous proteins (keratin)-structural sheets or strands (STRONG!) § Globular proteins (hemoglobin) § Soluble spheres with active functions § Protein function is based on shape § Shape is based on sequence of amino acids
Proteins FIGURE 2– 20 Protein Structure.

Protein Function § Proteins serve many important functions: § Proteins called enzymes speed up chemical reactions. Enzymes are also components of cell membranes. § Proteins regulate gene expression in cells § Proteins are important structural components of cells § Proteins (hormones) function as chemical messengers.

How long would it take to digest this?

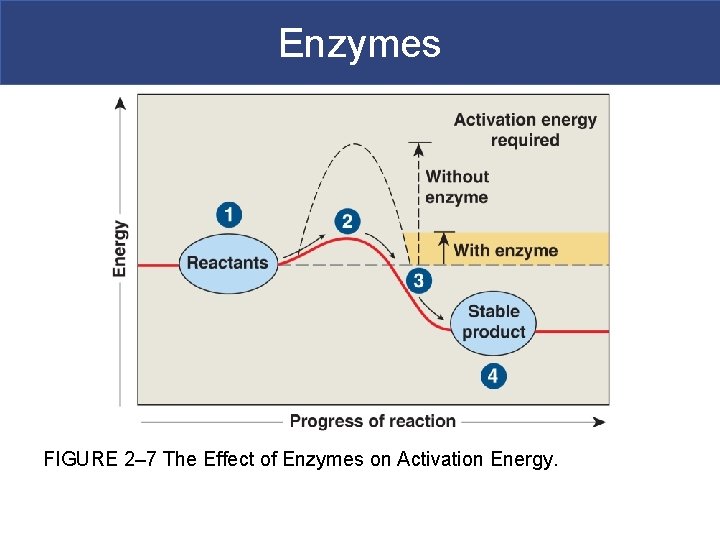
Enzymes § Enzymes – proteins that speed up (catalyze) the rate of chemical reactions, thus liberating the energy stored in the bonds of food. § The speed and efficiency of the enzyme affects the rate a person can metabolize food. § Enzymes lower the ENERGY requirements for chemical reactions to occur, so that the reactions can occur more quickly.

Enzymes Are Used In: § Decomposition reaction (catabolism) § Breaks chemical bonds § AB A + B § Hydrolysis: ABCDE + H 2 O ABC—H + HO—DE § Synthesis reaction (anabolism) § Forms chemical bonds § A + B AB § Dehydration synthesis (condensation) ABC—H + HO—DE ABCDE + H 2 O

Enzymes § How do enzymes work? They decrease the activation energy of the reaction § Activation energy – the energy required to start a chemical reaction. § Enzymes catalyze metabolic reactions by decreasing this activation energy barrier.
Enzymes FIGURE 2– 7 The Effect of Enzymes on Activation Energy.

Enzymes § Substrates – chemicals metabolized by enzyme-catalyzed reactions § Enzymes decrease the activation energy barrier by binding to their substrate and stressing its bonds to decrease the energy required to break the bonds. § Active site – location on the enzyme where the substrate binds.
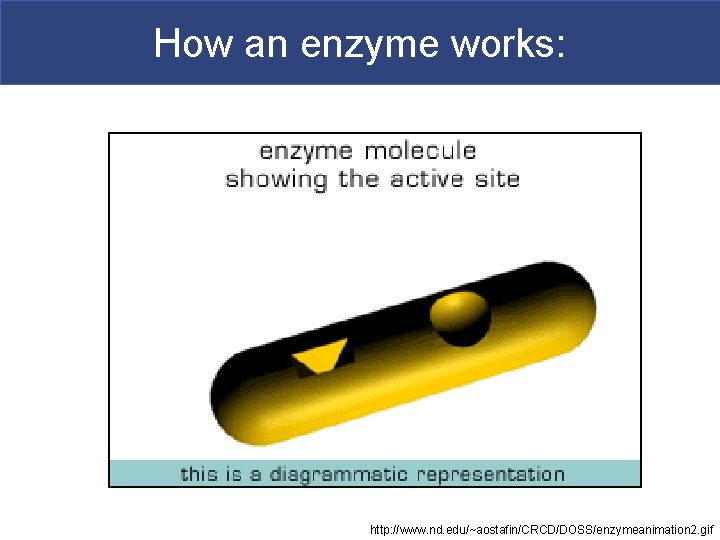
How an enzyme works: http: //www. nd. edu/~aostafin/CRCD/DOSS/enzymeanimation 2. gif
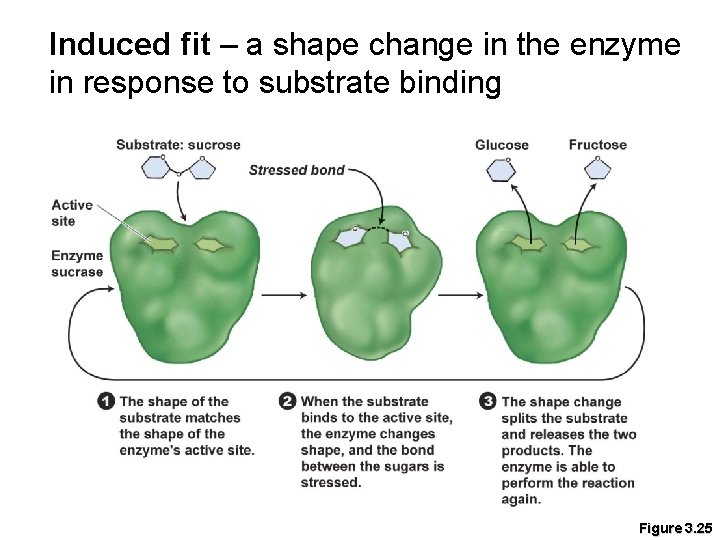
Induced fit – a shape change in the enzyme in response to substrate binding Figure 3. 25

Enzymes § Enzymes show specificity in the reactions they can catalyze. § Specificity is the result of the unique shape of different enzymes because of their amino acid sequences. § Enzymes can be regulated
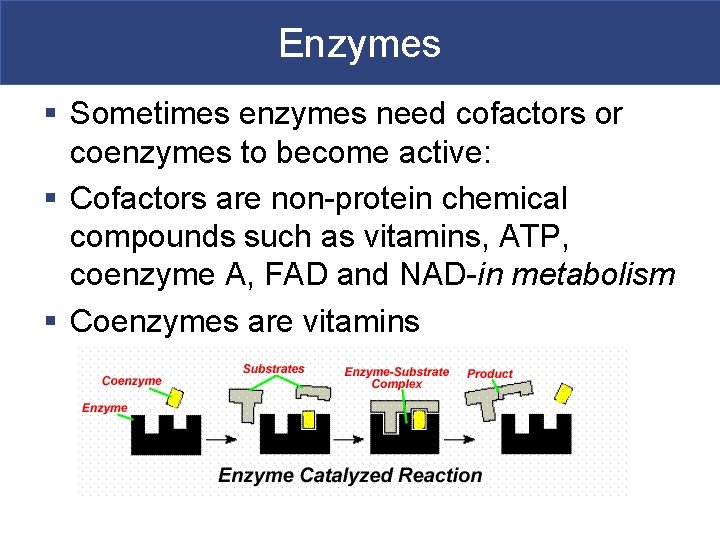
Enzymes § Sometimes enzymes need cofactors or coenzymes to become active: § Cofactors are non-protein chemical compounds such as vitamins, ATP, coenzyme A, FAD and NAD-in metabolism § Coenzymes are vitamins

Enzymes § Enzymes only work as fast as they can… § § You can saturate an enzyme v= velocity of reactants products Vmax=maximum velocity of enzyme [S]=substrate(s)/ reactant(s) concentration(s)/level(s)

Enzymes § Enzymes can be inhibited by: § Heat-causes denaturation § Altering the surrounding p. H-each enzyme has a specific p. H it prefers
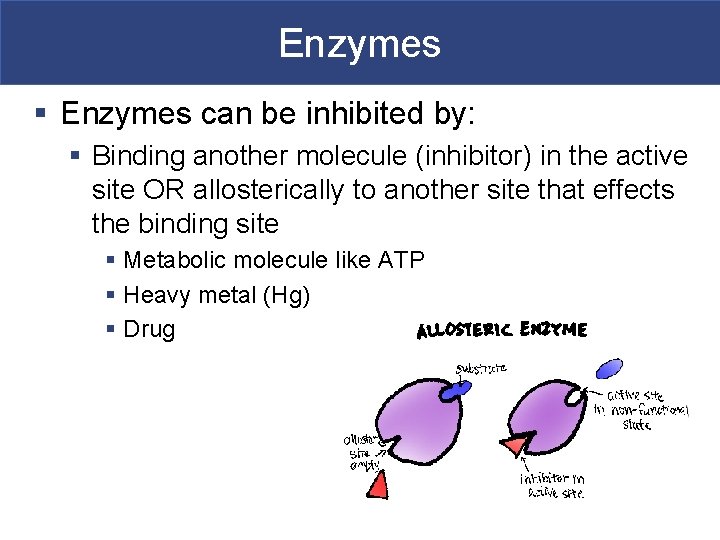
Enzymes § Enzymes can be inhibited by: § Binding another molecule (inhibitor) in the active site OR allosterically to another site that effects the binding site § Metabolic molecule like ATP § Heavy metal (Hg) § Drug

IV. Nucleotides § Nucleotides large organic molecules that form the genet material of the cell and are used in energy transfer for th cell § Nucleotides comprise: § Deoxyribonucleic acid (DNA) § Ribonucleic acid (RNA) § Adenosine monophosphate (AMP) § Examples of related molecules: § Adenosine triphosphate (ATP); cyclic adenosine monophosphate (c. AMP) § Nicotinamide adenine dinucleotide (NAD) and flavin adenine dinucleotide (FAD)
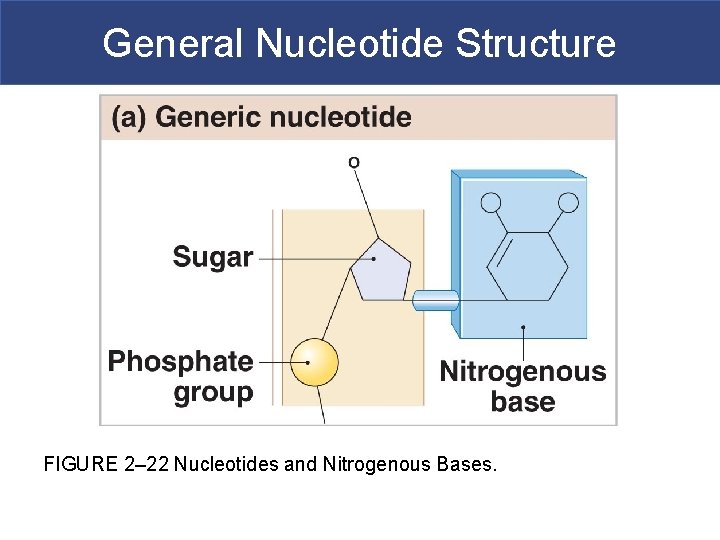
General Nucleotide Structure FIGURE 2– 22 Nucleotides and Nitrogenous Bases.
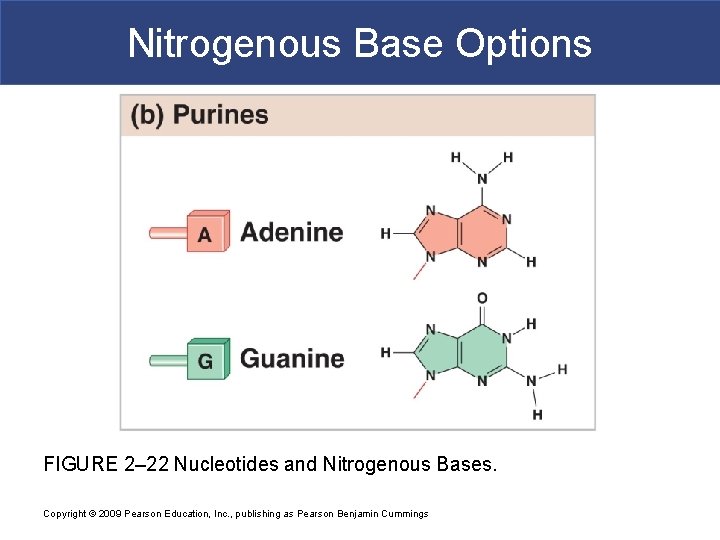
Nitrogenous Base Options FIGURE 2– 22 Nucleotides and Nitrogenous Bases. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
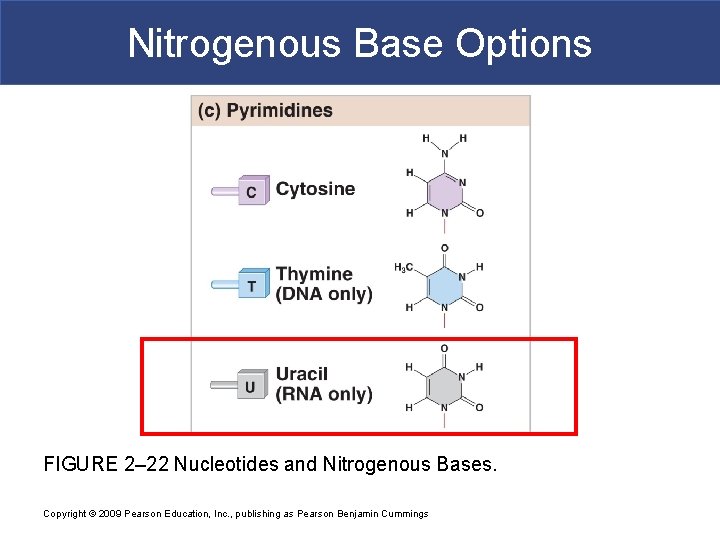
Nitrogenous Base Options FIGURE 2– 22 Nucleotides and Nitrogenous Bases. Copyright © 2009 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
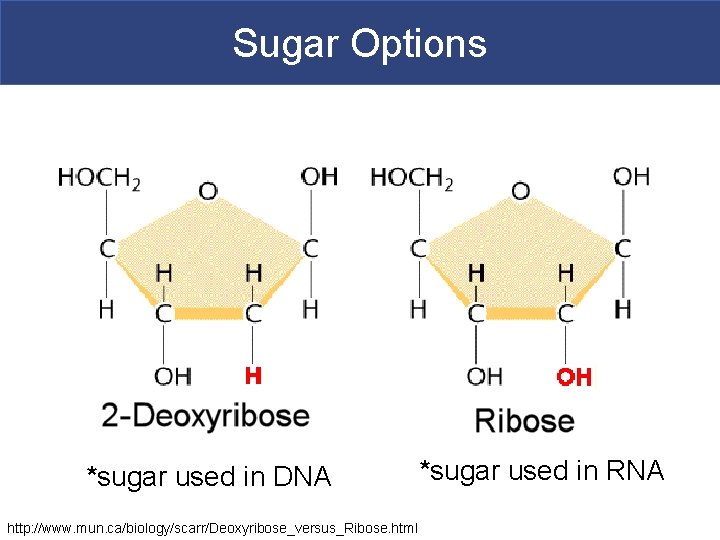
Sugar Options *sugar used in DNA http: //www. mun. ca/biology/scarr/Deoxyribose_versus_Ribose. html *sugar used in RNA

Nucleic Acids § Deoxyribonucleic Acid (DNA) § Comprised of two strands of complementary chains of nucleotides § Nucleotide=phosphate + deoxyribose sugar + nitrogenous base
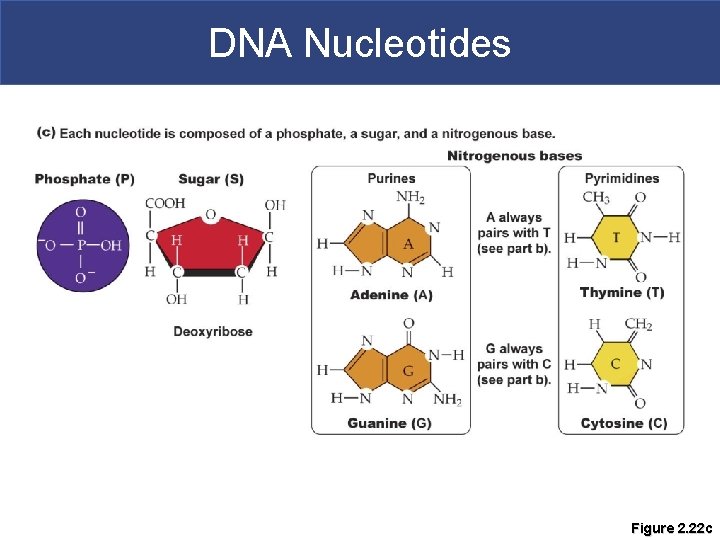
DNA Nucleotides Figure 2. 22 c
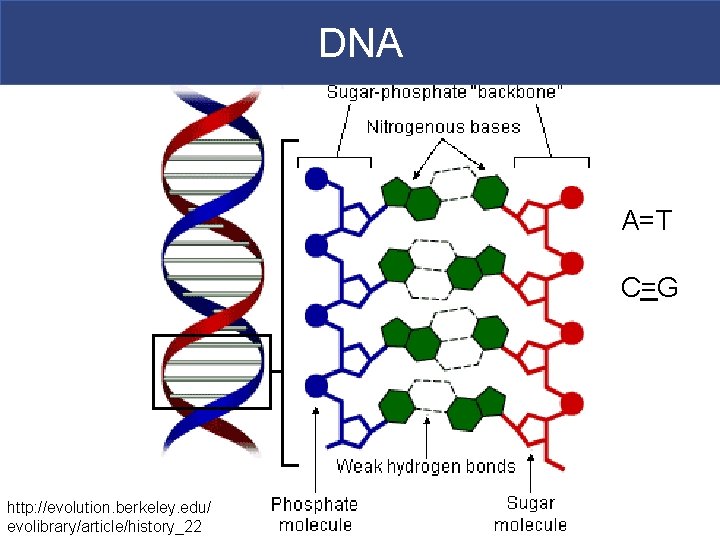
DNA A=T C=G http: //evolution. berkeley. edu/ evolibrary/article/history_22

DNA § Deoxyribonucleic Acid (DNA) § Stores genetic information in the form of genes § DNA genes chromosomes § Function: § Duplicate DNA for cell division OR § Use as a “template” for protein synthesis § Carries inherited characteristics, and controls enzyme production and metabolism

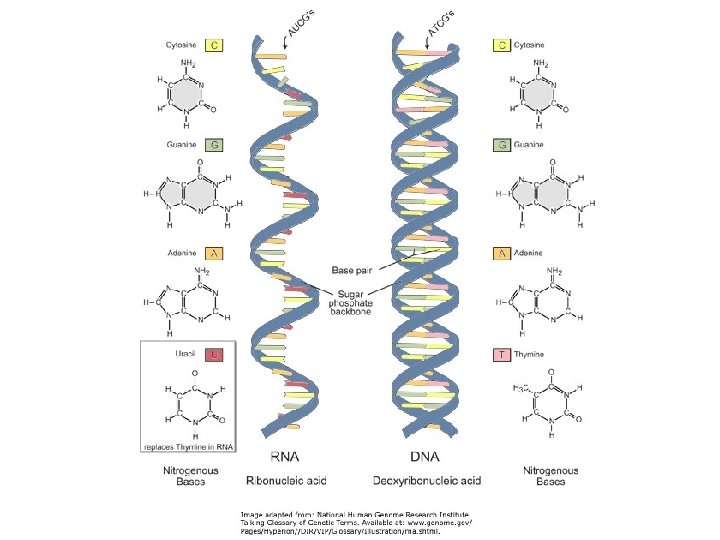
RNA § Ribonucleic Acid (RNA) § RNA takes the codes from DNA and translates it into proteins. § RNA is single stranded, has a ribose sugar, and contains uracil (U) instead of thymine. § m. RNA takes the codes from DNA and translates it into proteins. § Messenger RNA § transfer RNA § ribosomal RNA

Nucleosides § Nucleosides can be used to store energy (ATP), to transfer energy (FAD/ NAD), or as a messenger (c. AMP): § Adenosine monophosphate (AMP) has one phosphate group attached § Adenosine diphosphate (ADP) has two phosphate groups; di= 2 § Adenosine triphosphate (ATP) has three phosphate groups
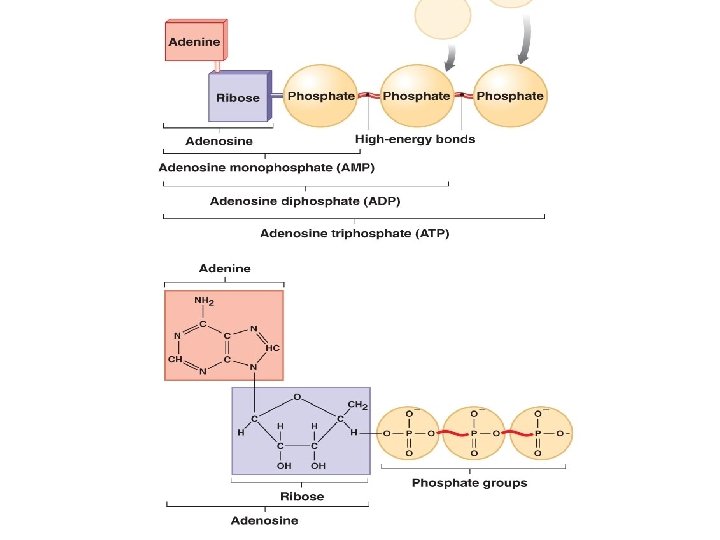

Nucleosides § Nucleosides can be used to store energy (ATP), to transfer energy (FAD/ NAD), or as a messenger (c. AMP): § An ATPase is an enzyme that catalyzes phosphorylation (the addition of a high-energy phosphate group to a molecule) or needs ATP to do work § FAD and NAD are electron carriers in metabolism § Cyclic AMP is used in the cell as a secondary messenger system for outside communication inside the cell
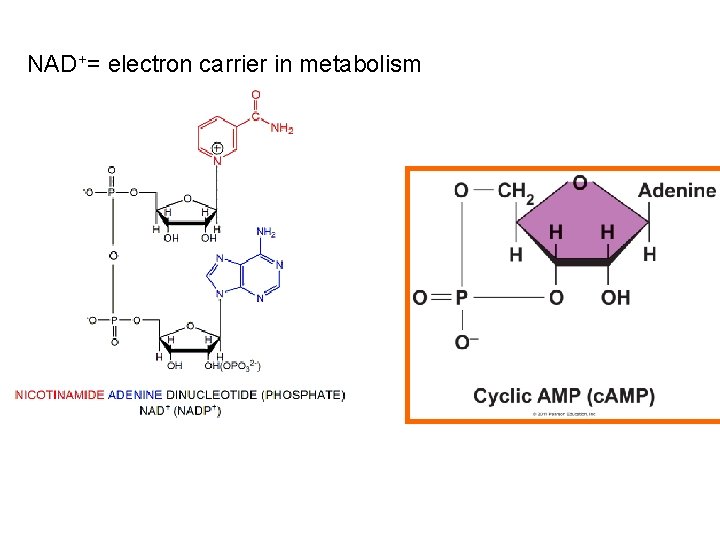
NAD+= electron carrier in metabolism

Micronutrients § Substances such as vitamins and minerals are required in small amounts by the body.

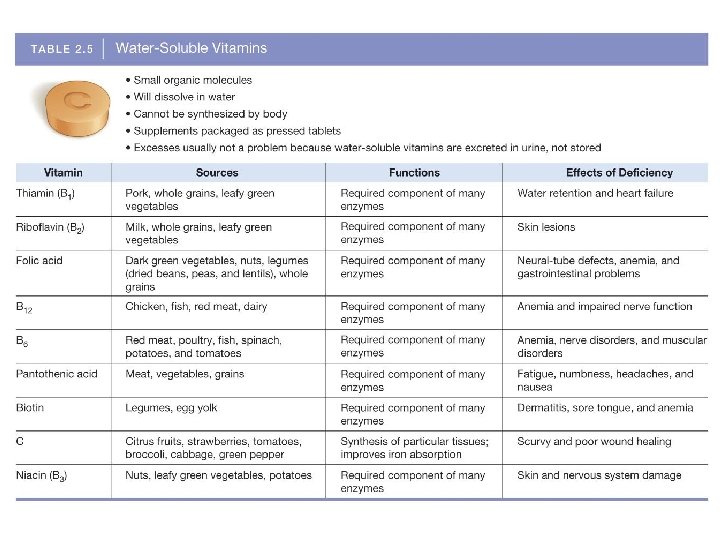
V. Micronutrients § Vitamins § 13 essential vitamins § Most can’t be synthesized by the body (D & K are the exceptions) § Function as coenzymes to help speed up chemical reactions § Vitamin deficiencies can affect every cell in the body

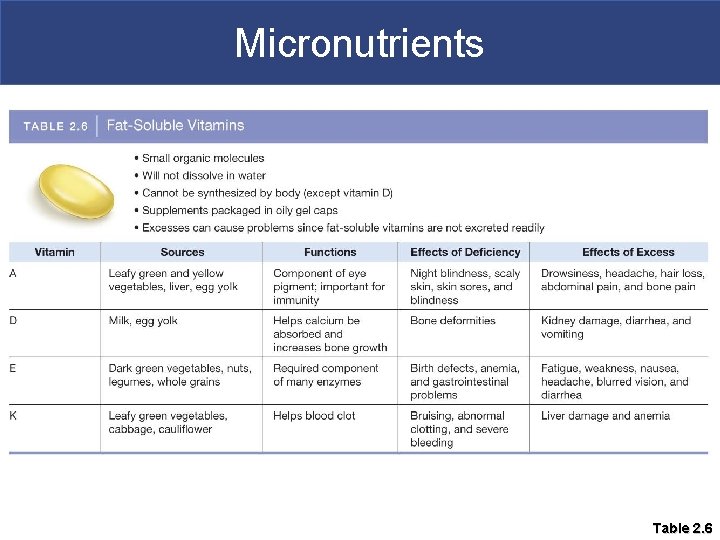
Micronutrients § Vitamin D is the only vitamin the body can make, but sunlight is required § Vitamin D deficiency disrupts Ca+2 absorption in intestine and bone growth § You can get vitamin D in dairy products
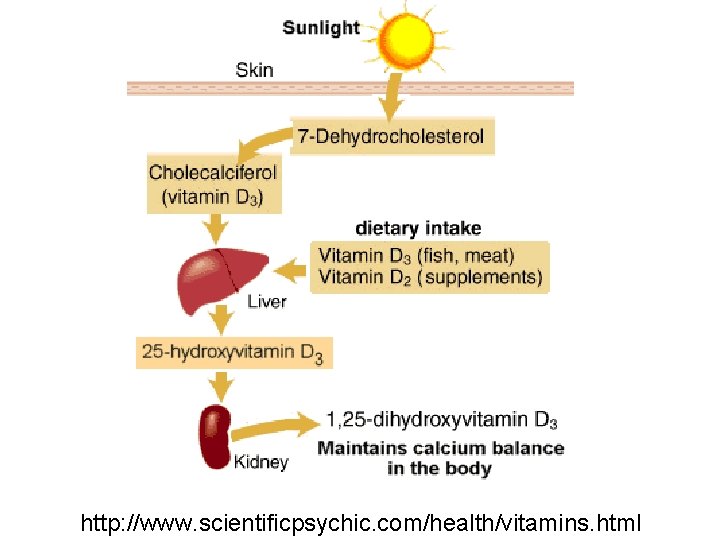
http: //www. scientificpsychic. com/health/vitamins. html

Micronutrients § Vitamin K is required for proper blood clotting. § It is not produced by human cells but comes from bacteria living in our large intestine and green-leafy vegetables § The drug coumadin prevents blood clots by inhibiting vitamin K synthesis § If patients are on coumadin they should not eat green-leafy vegetables, why?

Micronutrients § Hydrophilic vitamins can be dissolved in plasma § Therefore they are not stored in the body; they are excreted in the urine § Hydrophobic vitamins are not soluble in water § Hydrophobic vitamins (A, D, E and K) are stored in our body fat § It is possible to have an excess of these fatsoluble vitamins in body tissues; this can result in toxicity
Micronutrients Table 2. 6

Micronutrients § Minerals § Do not contain carbon (inorganic), but are essential for normal cell functions. § Necessary for proper fluid balance, muscle and nerve function, and building bones and teeth § Examples are calcium, chloride, sulfur, magnesium, phosphorous, and potassium. § Must be supplied through the diet
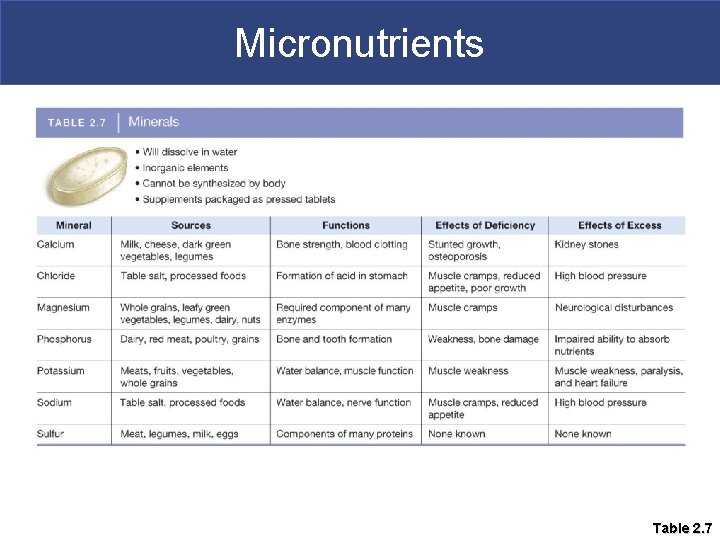
Micronutrients Table 2. 7

Micronutrients § Antioxidants § Protect cells and tissues from damage caused by highly reactive substances called free radicals, which have an incomplete outer electron shell and can thus oxidize (remove electrons from) other molecules. § Antioxidants bind free radicals and prevent them from doing damage. § Several vitamins and minerals are antioxidants.
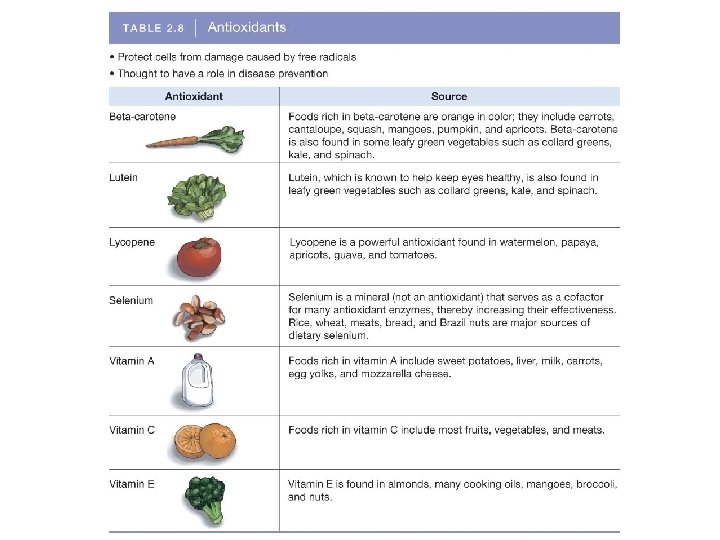

Micronutrients § Is it possible to compensate for a poor diet with diet and sports drinks? § In some cases these drinks can fill a gap in nutrition. § Eating fruits, vegetables, and grains is a better option because they contain micronutrients.

Micronutrients § Sports drinks may cause health issues § Many are high in calories § Some are hypertonic and draw water from blood and body tissues, increasing the risk of dehydration § Health claims about sports drinks may be misleading § Try a natural electrolyte drink, coconut water!
- Slides: 109