Organic Introduction Quiz Dr Deborah Bunker Topic Organic

Organic Introduction Quiz Dr Deborah Bunker

Topic Organic Chemistry Quiz Aims Ø This resource has been developed to support learners to develop their Organic Chemistry subject specialist knowledge whilst having a little fun, gaming for learning. The resource can be used to introduce an element of competition between individuals and/or groups which learners appear to enjoy. Level 2, Level 3 Method Power. Point Presentation. This can be used as an Ice Breaker activity to assess prior knowledge. A the start of a lesson provide the Connect to previous sessions learning, providing a fun interactive activity whilst providing checks on learning. This can also be used at the end of a lesson to assess that learning had taken place. Equipment Ø Laptop/Projector Duration >30 Minutes

What’s the Isomerism function? What’s in a name? Types of formula Testing, testing 1 pt 1 pt 2 pt 2 pt 3 pt 3 pt 4 pt 4 pt 5 pt 5 pt

Structural Isomers

What have the same molecular formula but different structures?

A chiral carbon

What feature is required for optical isomerism? or What do you call a carbon with four different groups attached?

C=C with each carbon having two different groups attached

What two features are needed for cis/trans isomerism?

butane and methylpropane

Name two structural isomers with the molecular formula C 4 H 10

They rotate plane polarised light in different directions

How do you tell the difference between 2 different optical isomers?
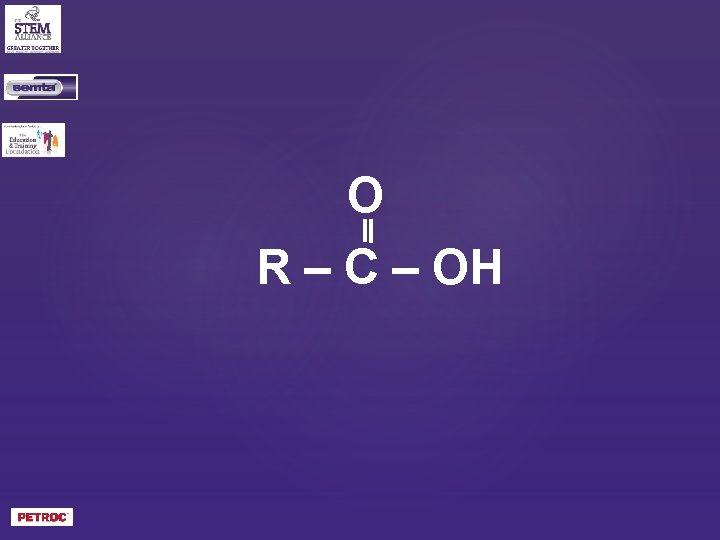

What is the general formula of a carboxylic acid?
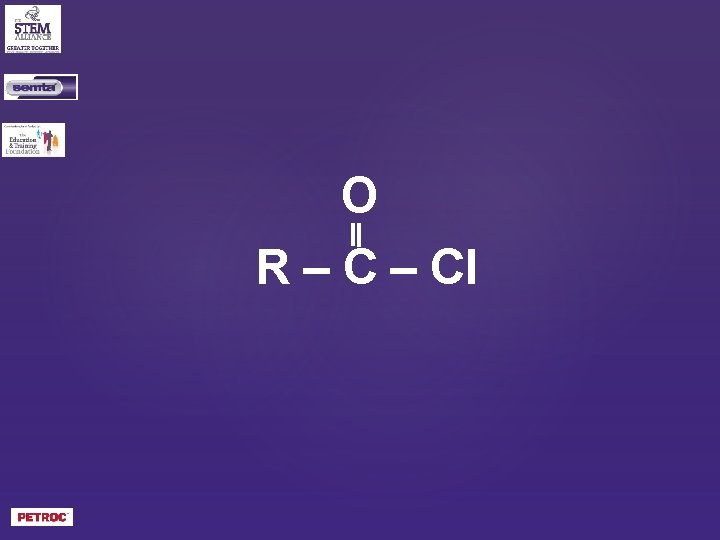
O R – Cl

What is the general formula of an acyl chloride?
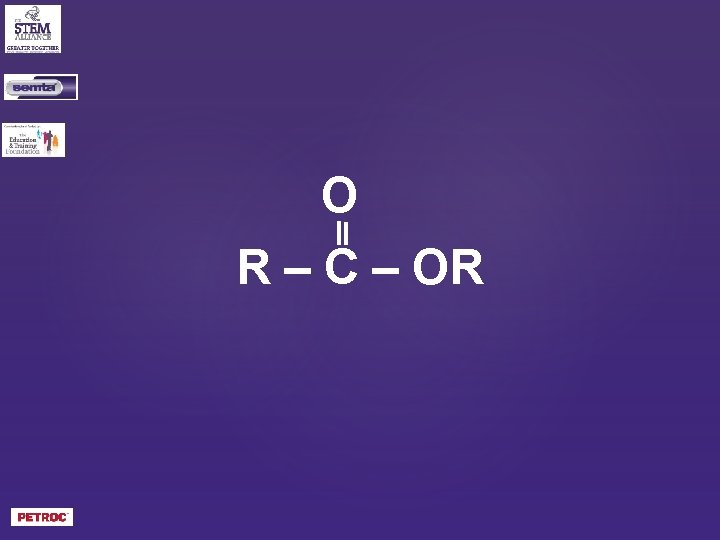

What is the general formula of an ester?
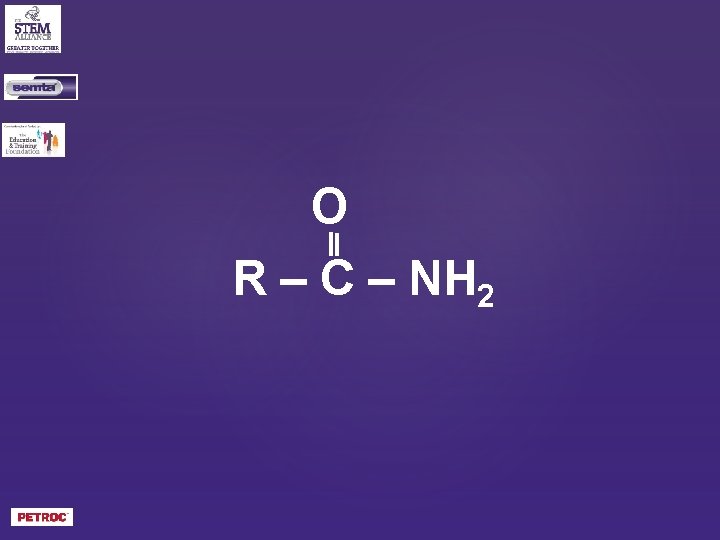
O R – C – NH 2

What is the general formula of an amide?
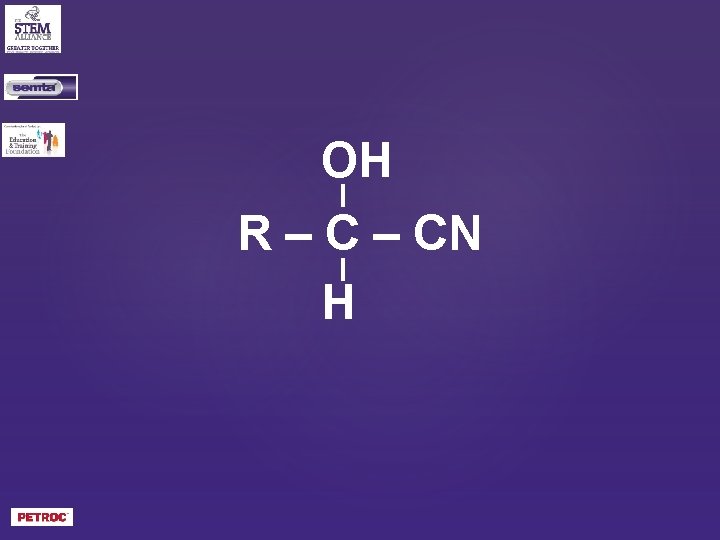
OH R – CN H

What is the general formula of a hydroxynitrile?

CH 3 CH 2 CH(CH 3)2

2 -methylbutane?
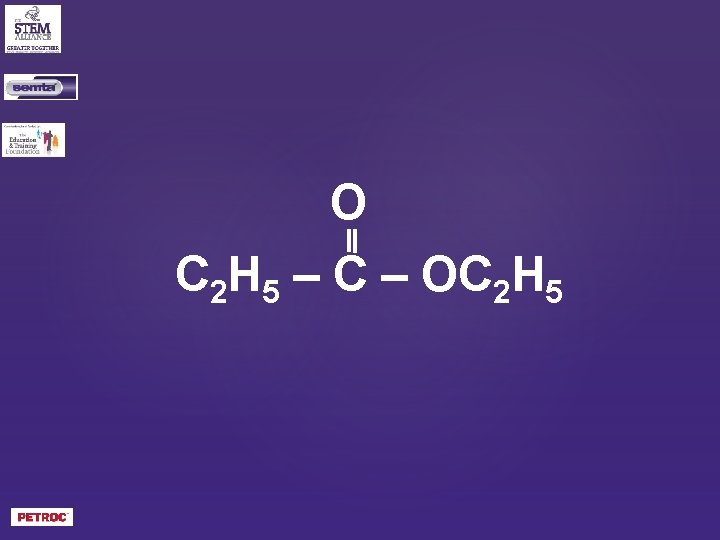
O C 2 H 5 – C – OC 2 H 5

ethylpropanoate?

CH 3 CHOHCH(CH 3)2

3 -methylbutan-2 -ol?
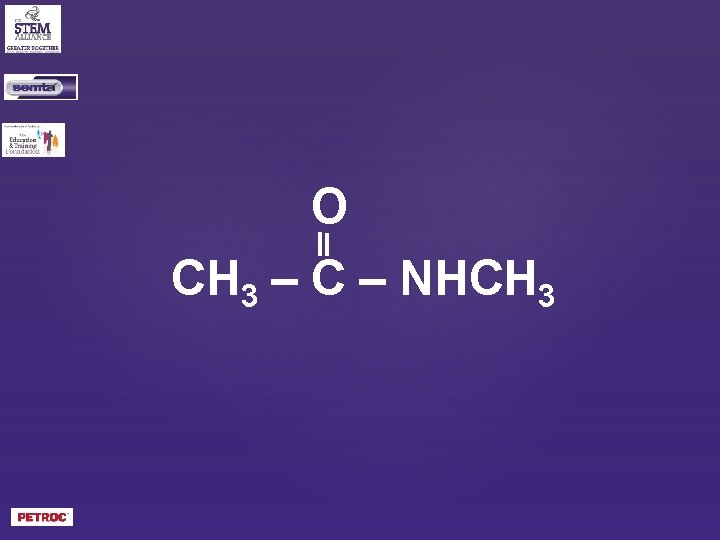
O CH 3 – C – NHCH 3

N-methylethanamide?
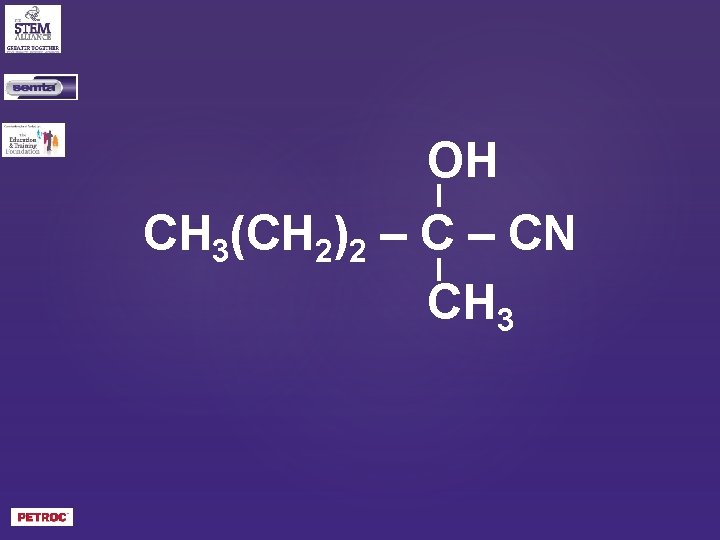
OH CH 3(CH 2)2 – CN CH 3

2 -hydroxy-2 -methyl pentanenitrile?

C 6 H 14

What is the molecular formula of hexane?

C 3 H 7

What is the empirical formula of hexane or What is the empirical formula of any structural isomer of hexane?

CH 3 CH 2 CH(CH 3)2

What is the structural formula of 2 -methylpentane?
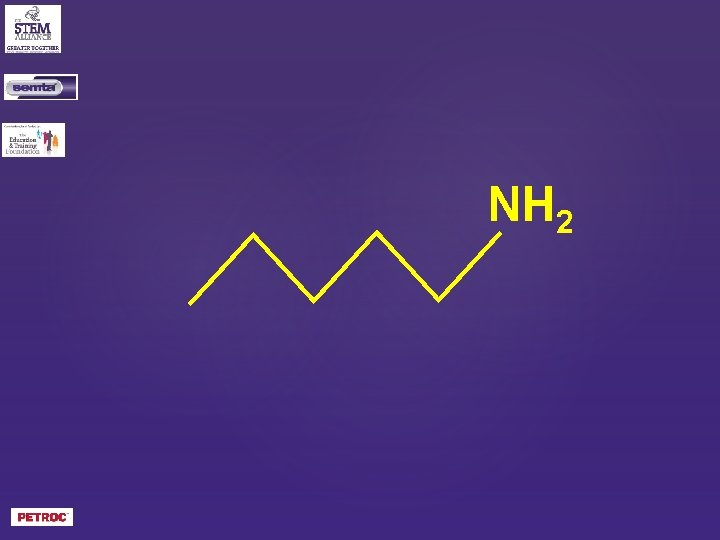
NH 2

What is the skeletal formula of pentylamine or what is the skeletal formula of 1 -aminopentane?
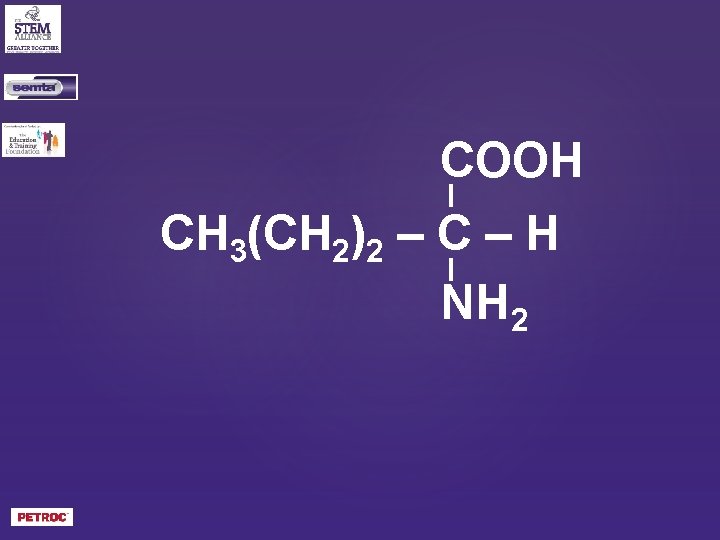
COOH CH 3(CH 2)2 – C – H NH 2

What is the structural formula of 2 -aminopentanoic acid?

Bromine water when added dropwise goes from orange to colourless

What is the test for an alkene?

Na. HCO 3 fizzes

What is the test for a carboxylic acid?

Forms a silver mirror on the wall of the test tube

What do you see if you warm an aldehyde with Tollen’s reagent?

Forms orangeyellow crystals with a clearly defined melting point

What do you see if you add excess 2, 4 DNPH to an aldehyde or ketone?

A white precipitate with an antiseptic smell

Apart from a colour change of orange to colourless what else do you see if you add bromine water to a phenol?

For further information please contact The STEM Alliance enquiries@STEMalliance. uk or visit www. STEMalliance. uk
- Slides: 54