Organic Compounds OBJ Define an Organic Compound What

Organic Compounds

OBJ: Define an Organic Compound

What does Organic Mean? • Organic Chemistry- The study of all compounds that contain bonds between carbon atoms. • Carbon can bond with many elements, including itself. • The most versatile element

OBJ: Compare and contrast the structure and formation of monomers and polymers

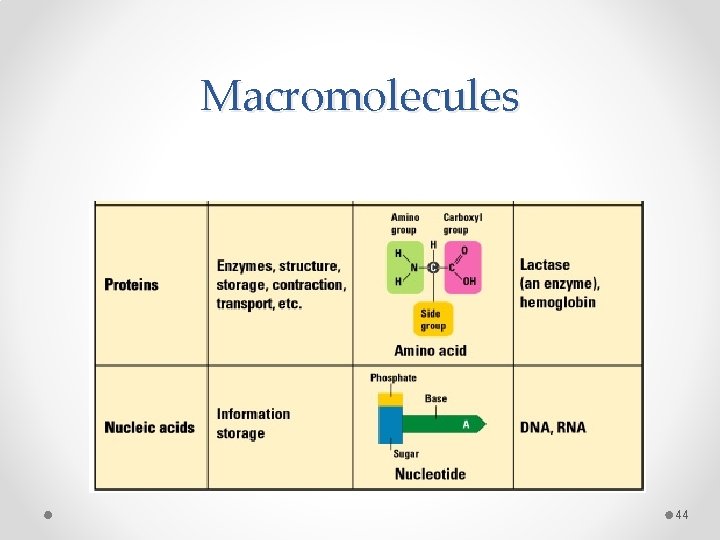
Macromolecules • “Giant Molecules” • Made from smaller molecules • Polymerization: process in which large molecules are built together by joining smaller ones together
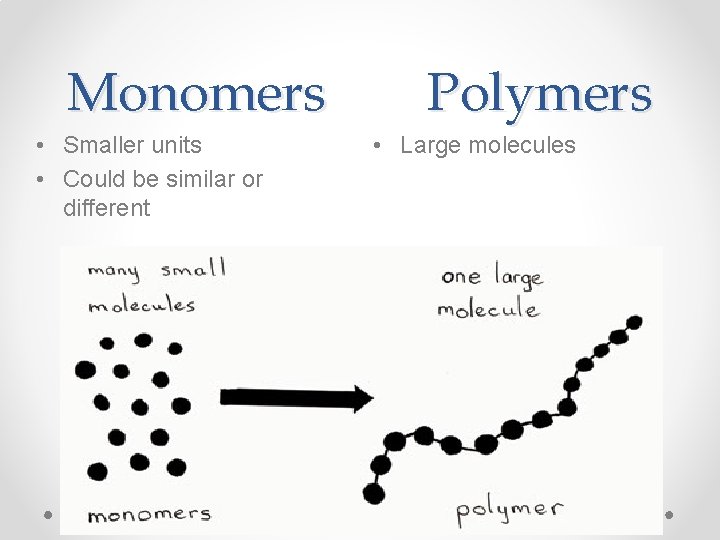
Monomers • Smaller units • Could be similar or different Polymers • Large molecules
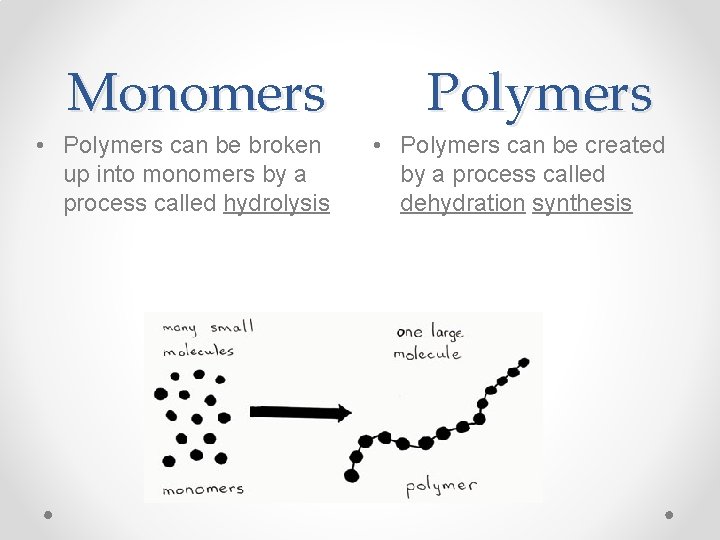
Monomers • Polymers can be broken up into monomers by a process called hydrolysis Polymers • Polymers can be created by a process called dehydration synthesis

Composite Compound • When monomer that make up a polymer have multiple parts

OBJ: Compare and Contrast the structures and roles of carbohydrates, proteins, fats, and nucleic acids in the functioning of a cell
Carbohydrates (Sugar) Compounds Made up of: Carbon, Hydrogen, Oxygen Atoms
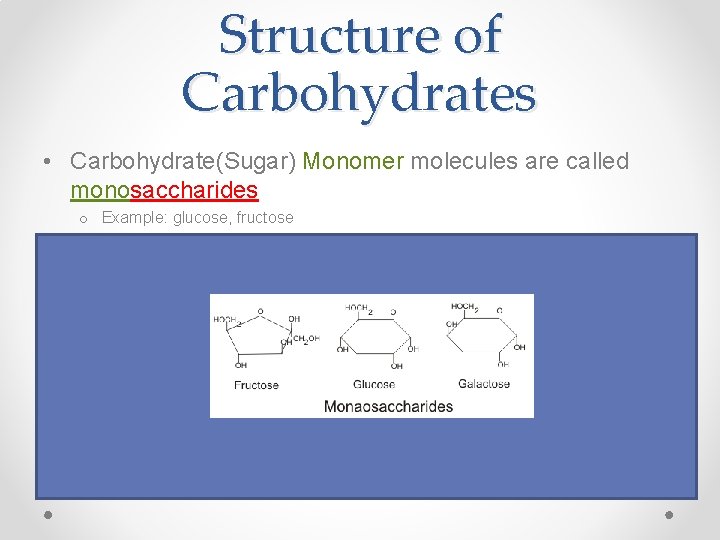
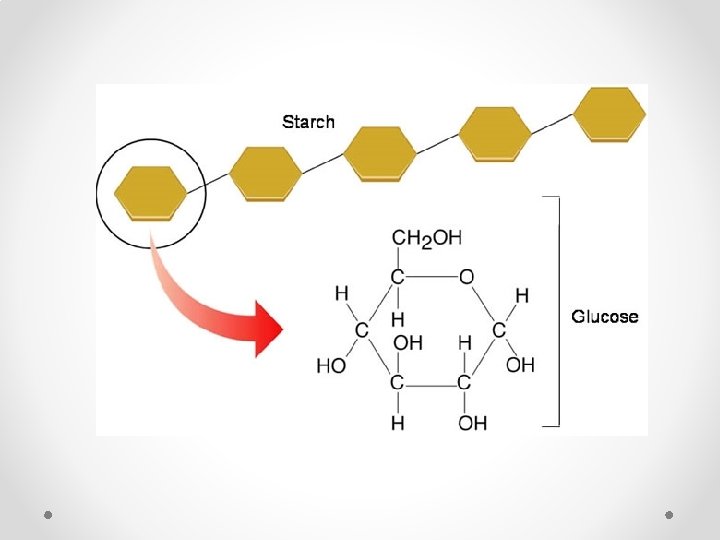
Structure of Carbohydrates • Carbohydrate(Sugar) Monomer molecules are called monosaccharides o Example: glucose, fructose • Carbohydrate (Sugar) “Pair” molecules are called disaccharides o Example: sucrose, lactose • Carbohydrate(Sugar) Polymer (molecule chains) are called polysaccharides o Example: starch (plants), glycogen (animals)

Structure of Carbohydrates • Carbohydrate(Sugar) Monomer molecules are called monosaccharides o Example: glucose, fructose • Carbohydrate (Sugar) “Pair” molecules are called disaccharides o Example: sucrose, lactose Dehydration synthesis: http: //vimeo. com/4419365 • Carbohydrate(Sugar) Polymer (molecule chains) are called polysaccharides o Example: starch (plants), glycogen (animals)
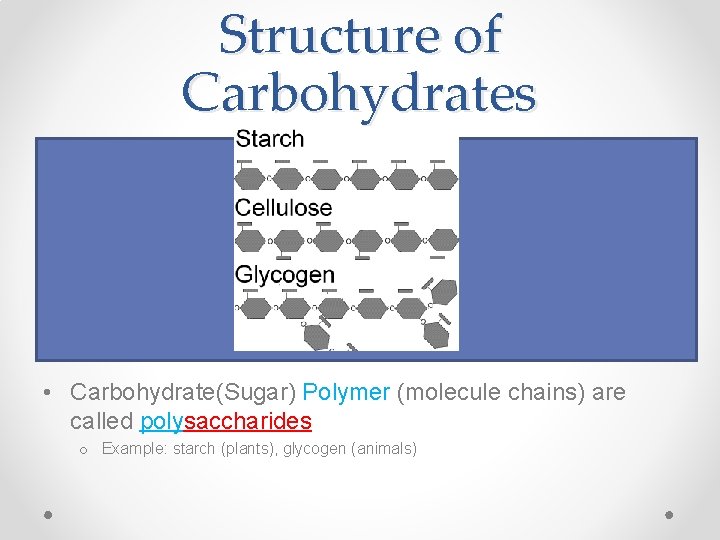
Structure of Carbohydrates • Carbohydrate(Sugar) Monomer molecules are called monosaccharides o Example: glucose, fructose • Carbohydrate (Sugar) “Pair” molecules are called disaccharides o Example: sucrose, lactose • Carbohydrate(Sugar) Polymer (molecule chains) are called polysaccharides o Example: starch (plants), glycogen (animals)

Structure of Carbohydrates • Carbohydrate(Sugar) Monomer molecules are called monosaccharides o Example: glucose, fructose • Carbohydrate (Sugar) “Pair” molecules are called disaccharides o Example: sucrose, lactose • Carbohydrate(Sugar) Polymer (molecule chains) are called polysaccharides o Example: starch (plants), glycogen (animals)

Roles of Carbohydrates • Main energy source • Plants and some animals use carbohydrates a structural support o Cellulose- cell walls of plant cells • Store excess sugar

Artificial Carbohydrates • Sweeteners: http: //www. youtube. com/watch? v=Ah. Uyw 2 lcio • White Breads: http: //youtu. be/Jq. UR 0 I 68 Hw 4

Testing for the Presence of Carbohydrates • Part A: Testing for simple sugar (monosaccharide's) o Benedicts Solution and heat • Positive: yellow-Orange • Negative: no color change • Part B: Testing for complex sugar (polysaccharide) o Iodine • Positive: Blue-Black • Negative: no color change

Lipids (Fats) Compounds Made up mostly of: Carbon, Hydrogen. Few Oxygen.

Structure of Lipids • Compound o 3 Fatty Acid o 1 Glycerol
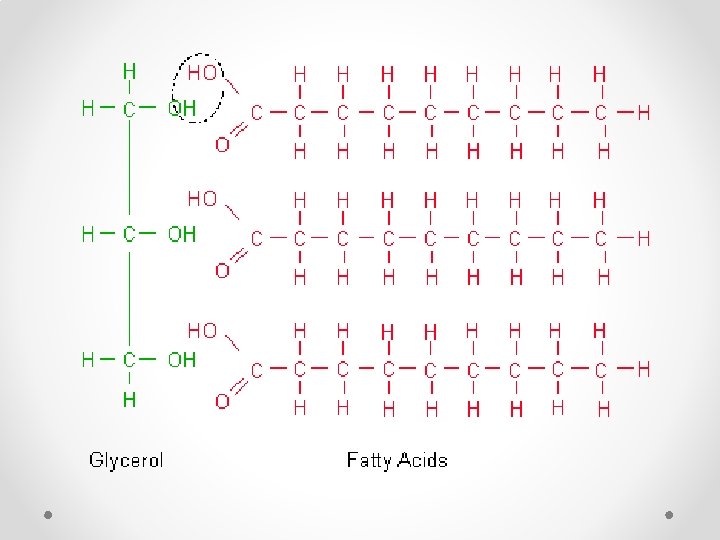

Lipids Saturated Fats Unsaturated Fats • Fatty acid chain is saturated with hydrogen atoms • Solid at room temperature (i. e. butter) • Fatty acid chain is composed of carbon molecules not completely surrounded by hydrogen molecules o Carbons are double bonded together • Liquid at room temperature (I. e. olive oil)

Lipids Saturated Fats Unsaturated Fats
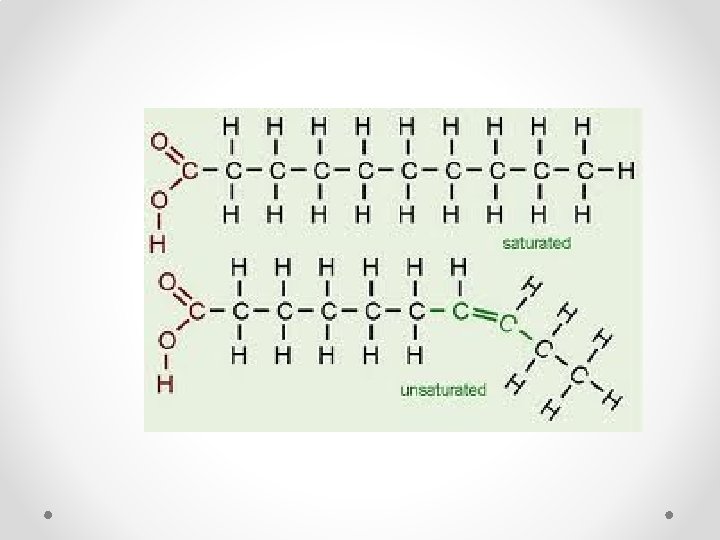

Unsaturated Lipids Monounsaturated • 1 double bond between carbons o Example: Olive oil Polyunsaturated • More than 1 double bond between carbons o Example: Nuts

Trans Fats? • Chemically altered by adding hydrogen's to unsaturated fats- lengthens self-life • Partially hydrogenated oils
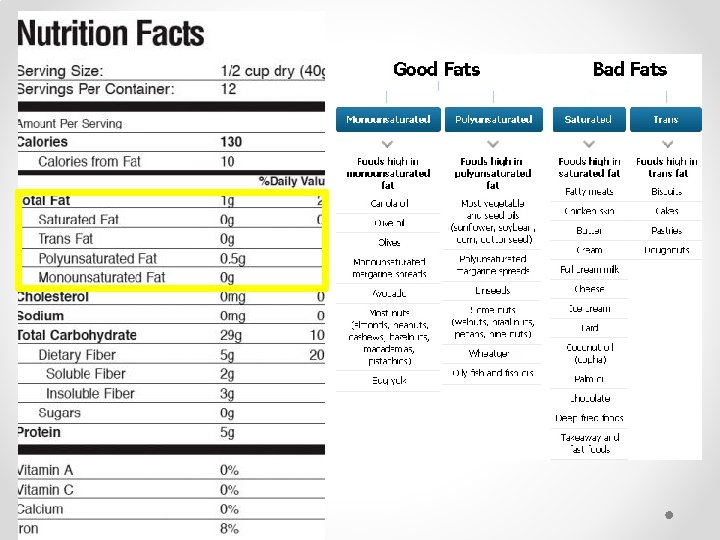

Roles of Lipids • Generally not soluble in waterhydrophobic • Fats, Oils, Waxes and Steroids (chemical messenger) • Store energy • Part of protective membranes / waterproof coverings

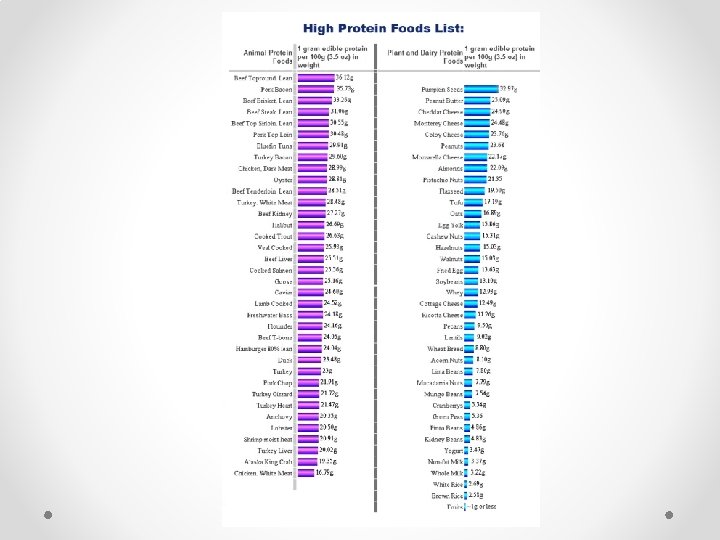
Proteins Made up mostly of: Nitrogen Carbon, Hydrogen, Oxygen.
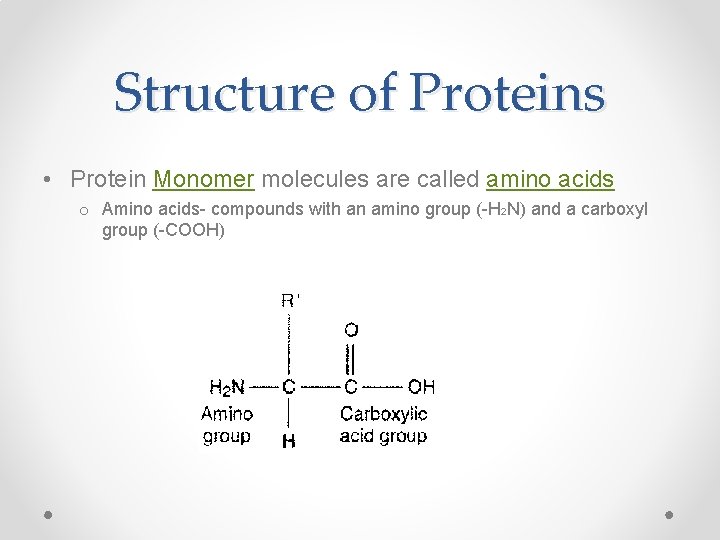
Structure of Proteins • Protein Monomer molecules are called amino acids o Amino acids- compounds with an amino group (-H 2 N) and a carboxyl group (-COOH)
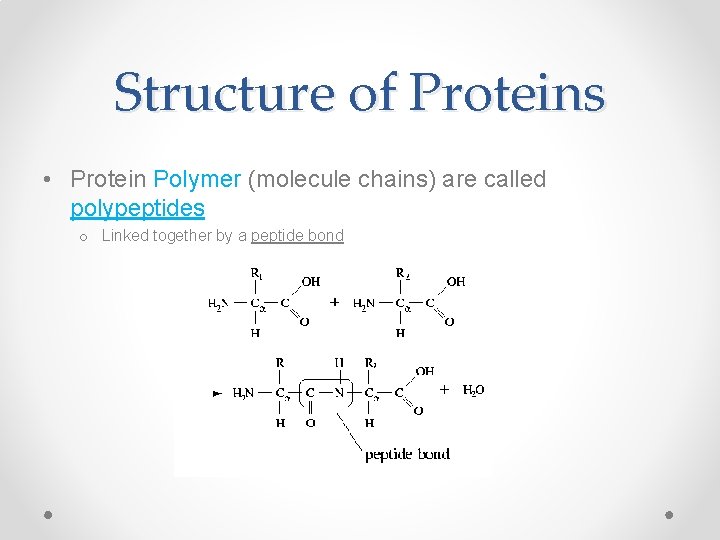
Structure of Proteins • Protein Polymer (molecule chains) are called polypeptides o Linked together by a peptide bond
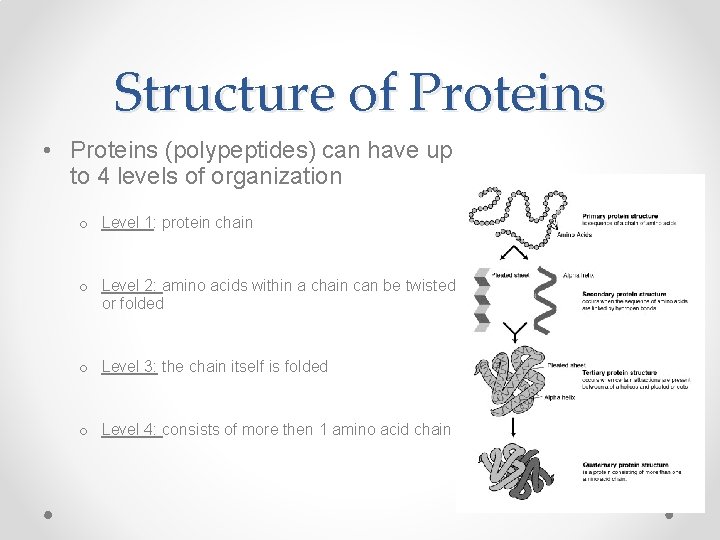
Structure of Proteins • Proteins (polypeptides) can have up to 4 levels of organization o Level 1: protein chain o Level 2: amino acids within a chain can be twisted or folded o Level 3: the chain itself is folded o Level 4: consists of more then 1 amino acid chain
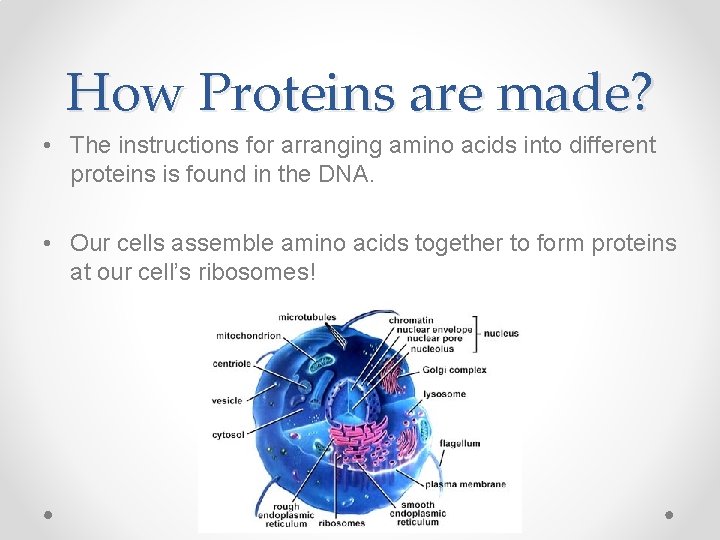
How Proteins are made? • The instructions for arranging amino acids into different proteins is found in the DNA. • Our cells assemble amino acids together to form proteins at our cell’s ribosomes!
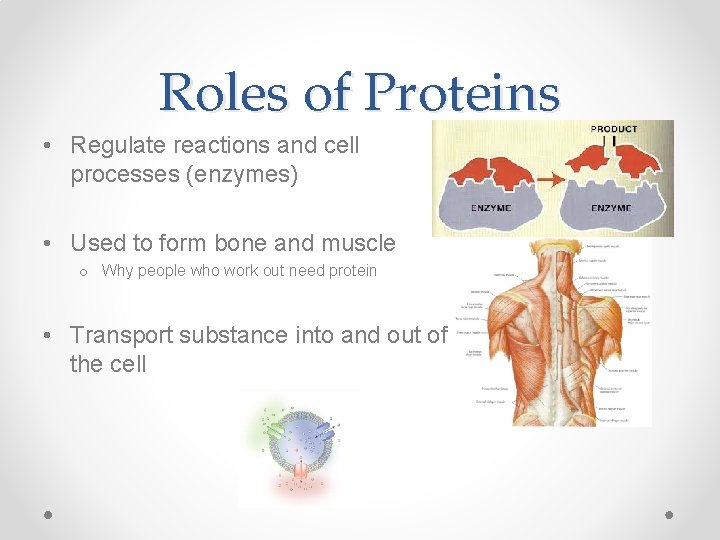
Roles of Proteins • Regulate reactions and cell processes (enzymes) • Used to form bone and muscle o Why people who work out need protein • Transport substance into and out of the cell

Types of Proteins • Structural Contractile • Transport Storage

Nucleic Acids Made up mostly of: Nitrogen, Carbon, Hydrogen, Oxygen, Phosphorus.
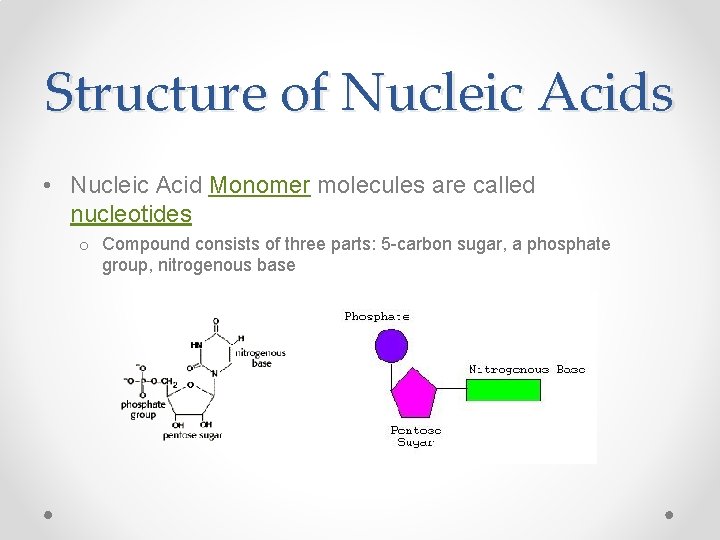
Structure of Nucleic Acids • Nucleic Acid Monomer molecules are called nucleotides o Compound consists of three parts: 5 -carbon sugar, a phosphate group, nitrogenous base
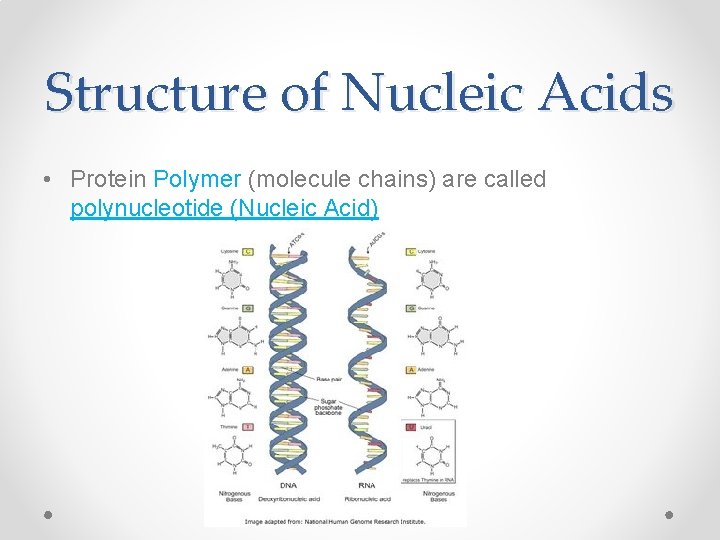
Structure of Nucleic Acids • Protein Polymer (molecule chains) are called polynucleotide (Nucleic Acid)
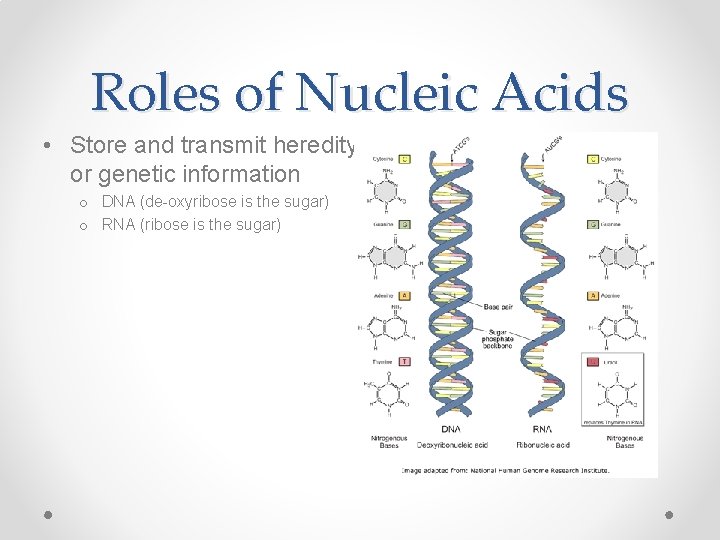
Roles of Nucleic Acids • Store and transmit heredity, or genetic information o DNA (de-oxyribose is the sugar) o RNA (ribose is the sugar)

Macromolecules 43
Macromolecules 44

Formal Lab Write Up

Unknown Organic Substance • 1. Make a prediction which organic substance is in the unknown sample. • 2. Design the test for that organic molecule on the unknown substance. • 3. Perform the each test until you have positively identified the unknown substance. • 4. Write up your lab report.

Lab Report • • • Title- Unknown Organic Substance Introduction. Purpose/Hypothesis- Prediction Materials- The difference substances you have used Procedures- Your plan Data/Results- What the tests showed(positive/negative) • Discussion- What the results of the tests mean • Conclusion- Was your hypothesis correct or not?
- Slides: 47